Facile synthesis of reduced graphene oxide/Pt–Ni nanocatalysts: their magnetic and catalytic properties†
Abstract
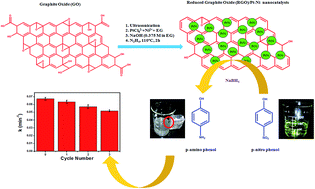
The catalytic performance of metals can be enhanced by intimately alloying different metals with Reduced Graphene Oxide (RGO). In this work, we have demonstrated a simplistic in situ one-step reduction approach for the synthesis of RGO/Pt–Ni nanocatalysts with different atomic ratios of Pt and Ni, without using any capping agent. The physical properties of the as-synthesized nanocatalysts have been systematically investigated by XRD, FTIR, Raman spectroscopy, XPS, EDX, ICP-AES, and TEM. The composition dependent magnetic properties of the RGO/Pt–Ni nanocatalysts were investigated at 5 and 300 K, respectively. The results confirm that the RGO/Pt–Ni nanocatalysts show a super-paramagnetic nature at room temperature in all compositions. Furthermore, the catalytic activities of the RGO/Pt–Ni nanocatalysts were investigated by analyzing the reduction of p-nitrophenol, and the reduction rate was found to be susceptible to the composition of Pt and Ni. Moreover, it has been found that RGO/Pt–Ni nanocatalysts show superior catalytic activity compared with the bare Pt–Ni of the same composition. Interestingly, the nanocatalysts can be readily recycled by a strong magnet and reused for the next reactions.

 Please wait while we load your content...
Please wait while we load your content...