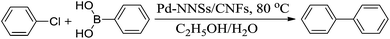Facile synthesis, characterization and application of highly active palladium nano-network structures supported on electrospun carbon nanofibers†
Xinwen Peng,
Wan Ye,
Yichun Ding,
Shaohua Jiang*,
Muddasir Hanif,
Xiaojian Liao and
Haoqing Hou*
Chemistry and Chemical Engineering College, Jiangxi Normal University, Nanchang, Jiangxi 330022, P.R. China. E-mail: s.jiang19830913@gmail.com; haoqing@jxnu.edu.cn; Tel: +86 791 88120740
First published on 29th August 2014
Abstract
Palladium nano-network structures supported on electrospun carbon nanofibers (Pd-NNSs-ECNFs) were successfully prepared through a novel K2PdIICl4/K4FeII(CN)6 cyanogel method. The Pd-NNSs have the features and properties of small particle size, low dimensionality, quantum effects and high stability. SEM, TEM and XPS were used to characterize the Pd-NNSs-ECNFs. The Pd-NNSs-ECNFs were used as a catalyst in different types of Suzuki coupling reactions to evaluate the catalytic abilities. The results showed that the heterogeneous catalyst (Pd-NNSs-ECNFs) had a high catalytic activity (high yields to the products) to the Suzuki coupling reactions in an environmentally friendly solvent system (ethanol/H2O). The catalyst can be recycled by filtration, and reused seven times without losing the catalytic activity. This research opens new applications of Pd-NNSs-ECNFs in the area of green chemistry.
1. Introduction
Palladium (Pd) nanoparticles have received intense interest because of their fascinating properties and potential applications in diverse fields such as hydrogen storage,1 organic synthesis,2,3 fuel cells,4 sensors5 and electrochemical detection, etc.6 Due to the excellent catalytic activities, Pd nanoparticles have been widely used in organic synthesis and electrocatalysis.7–13 Pd nanoparticles could be supported on a number of materials such as carbon materials,14,15 polymers,16–18 and mesoporous inorganic materials.19,20 The heterogeneous Pd nanoparticles show good catalytic abilities and the supported structure is benefit for the recovery and recycle use. Compared with the traditional Pd nanoparticles, palladium nano-network structures (Pd-NNSs) have features and properties of small particle size, low dimensionality, quantum effect and high stability.21–23 Pd-NNSs could be produced through a hydrogel method.24–27 The hydrogels were prepared by mixing the K2RCl4 (R = Pd/Pt) and Kx[M(CN)n] (n = 4, 6; M = Co, Fe, Ru, Os, Ni, Cr) in polymer solutions and the mixture of the coordination polymers was also called cyanogels. Cyanogels were a special class of three-dimensional double-metal cyanides for preparing the Pd-NNSs and other metal or alloys nanostructures. Zhang et al. had fabricated Pd-NNSs by using NaBH4 to reduce the K2PdIICl4/K4FeII(CN)6 cyanogel.24 The prepared Pd-NNSs had high electrocatalytic activity and good electrochemical self-stability.24In this work, we highlight the novel Pd nano-network structures supported on electrospun carbon nanofibers (Pd-NNSs-ECNFs) as catalyst for Suzuki coupling reactions. Cyanogels method for the preparation of the Pd-NNSs-ECNFs catalyst is emphasized. The structures of Pd-NNSs-ECNFs were characterized by SEM, TEM and XPS. The obtained Pd-NNSs-ECNFs catalyst showed good activity, highly efficient and cyclic utilization as heterogeneous catalysts for the Suzuki coupling under the environmentally friendly solvents.
2. Experimental
2.1. Materials
Polyacrylonitrile (PAN, Mw = 150![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000) and anhydrous N,N-dimethylformamide (DMF, 99.8%) were purchased from Sigma-Aldrich. Pd/C catalysts (wetted with ca. 55% water) containing 10 wt% of palladium, K2PdCl4 (99%), K4Fe(CN)6 (98.5%) and NaBH4 (98%) were purchased from Alfa Aesar. Potassium carbonate (K2CO3, 99%), ethanol (99.5%), phenylboronic acid (99%), 4-nitrobenzene, 4-chlorobenzene (99%), 4-iodobenzene (97%), 4-bromobenzene (99%), 4-nitrophenylboronic acid (98%), 4-methoxyphenylboronic acid (98%) and 4-methoxybenzene (98%) were obtained from J&K chemical Ltd.
000) and anhydrous N,N-dimethylformamide (DMF, 99.8%) were purchased from Sigma-Aldrich. Pd/C catalysts (wetted with ca. 55% water) containing 10 wt% of palladium, K2PdCl4 (99%), K4Fe(CN)6 (98.5%) and NaBH4 (98%) were purchased from Alfa Aesar. Potassium carbonate (K2CO3, 99%), ethanol (99.5%), phenylboronic acid (99%), 4-nitrobenzene, 4-chlorobenzene (99%), 4-iodobenzene (97%), 4-bromobenzene (99%), 4-nitrophenylboronic acid (98%), 4-methoxyphenylboronic acid (98%) and 4-methoxybenzene (98%) were obtained from J&K chemical Ltd.
2.2. Preparation of electrospun carbon nanofibers (ECNFs)
ECNFs were prepared by carbonizing the electrospun PAN nanofibers. PAN nanofibers were prepared according to previous report in our group.12 10 wt% PAN/DMF solution was electrospun to nanofibers in an electric field of 100 kV m−1, by applying a 25 kV voltage to a 25 cm gap between the spinneret and the collector. The carbonized electrospun nanofibers were prepared in high temperature furnace with following steps: (1) PAN nanofibers were heated to 230 °C (1 °C min−1) and annealing for 2 h in air for the pre-oxidation; (2) heating up to 800 °C at a heating rate of 5 °C min−1 and annealing for 30 min for the carbonization.In order to improve the hydrophilicity, the ECNFs were activated by mixing the ECNFs and nitric acid (40 vol%) in a round bottom flask under ultrasonic for 5 min and then heated at 90 °C for 2 h.28
2.3. Synthesis of Pd-NNSs-ECNFs
10 mL of 50 mM K2PdIICl4 aqueous solutions, 1 mL of PVP aqueous solution (10 wt% in water) and 5 mL of 50 mM K4FeII(CN)6 aqueous solutions were mixed at 10–15 °C. Then, a piece of activated ECNFs was immersed into the above mixture solution and pumped by vacuum pump for 10 min until the ECNFs were completely wetted. After stood for 20 min, the mixture was further treated by vacuum pumping for 10–110 min, then the orange-red K2PdIICl4/K4FeII(CN)6 cyanogel was formed. After that, 25 mL of 0.5 M NaBH4 solution was added to the orange-red K2PdIICl4/K4FeII(CN)6 cyanogel and the resulting cyanogel was stood for an additional 1 h. Finally, the mixture was filtered, washed and dried in a vacuum oven at 50 °C for 12 h.2.4. Catalysis of Pd-NNSs-ECNFs for Suzuki coupling
The obtained Pd-NNSs-ECNFs were used as catalysts for Suzuki coupling reactions. The reaction procedure is described as follows, and all reactions were carried out under nitrogen atmosphere. 7 mL of the ethanol, 7 mL of the deionized water, 1.5 mmol NaOH, 1 mmol of the aryl halide, 1.25 mmol phenylboronic acid, 0.5 mol% Pd-NNSs/ECNFs were placed in a 50 mL flask and heated at 80 °C for 10 h with stirring (500 rpm). And then the above mixture was filtered at room temperature. The filtrate was extracted with ethyl acetate for three times and the organic phase was added anhydrous Na2SO4 to absorb residual water. Finally, the solution was filtered and concentrated in vacuum. The products were obtained by column chromatography using ethyl acetate/petroleum ether. The filter cake (Pd-NNSs-ECNFs) was washed with distilled water for recycle use.2.5. Characterizations
X-ray photoelectron spectra (XPS) were recorded on an Escalab-MKII spectrometer with a MoKα X-ray as excitation source. Scanning electron microscope (SEM) was performed on SEM, TESCAN vega 3. The transmission electron microscope (TEM) was conducted by a JEOL 2000 transmission electron microscope operating at 200 kV. The samples for TEM characterization were prepared by placing a small piece of ultrathin Pd-NNSs-ECNFs mat on the copper grid and dropping a drop of ethanol on the sample for a good “adhesive” between the mat and the grid. Proton and carbon nuclear magnetic resonance spectra (1H and 13C-NMR) were measured at 400 MHz and 100 Hz respectively on a Bruker Avance-400 spectrometers. FT-IR spectra were recorded by Perkin-Elmer SP one FT-IR. Elemental analyses were taken by a Perkin-Elmer model 2400 CHN analyses. Inductively coupled plasma atomic emission spectroscopy (ICP-AES) (Optima 7300DV, Perkin-Elmer) was employed to measure the concentrations of Palladium. The quantity of Pd-NNSs-ECNFs was measured by the way of ICP (Varian, 725-ES).3. Results and discussion
3.1. Preparation of Pd-NNSs-ECNFs
The activation of ECNFs by nitric acid solution (40 vol%) for 2 h at 90 °C improved their hydrophilicity29,30 and so that the aqueous solution could easily permeate into the carbon nanofiber mat. The amount of Pd-NNSs-ECNFs could be tuned by controlling the pumping cyanogel time (Fig. 1). The amount of Pd-NNSs inside of electrospun carbon nanofiber mat was in proportional to the pumping time. The quantity of Pd-NNSs supported on ECNFs were measured by the Inductively Coupled Plasma (ICP) method. 0.5 g Pd-NNSs-ECNFs were put in 60 mL aqua regia, then vigorously stirring for 60 min at room temperature. Then the solution was diluted, and the quantity of palladium was measured by ICP.3.2. Structural characterization
The structure and morphology of the Pd-NNSs-ECNFs were characterized by the scanning electron microscopy (SEM, Fig. 2) and transmission electron microscopy (TEM, Fig. 3). The Pd-NNSs were located inside of the ECNFs so that they were difficult to drop out. Pd-NNSs were seen to be dispersed in the carbon nanofibers in Fig. 3. The transmission electron microscopic (TEM) images (Fig. 3) showed Pd-NNSs were composed of small spherical Pd-NPs (about 5 nm). These small primary NPs are interconnected to one another to form larger secondary 3D networks nanostructures with abundant pores.3.3. XPS spectra of Pd-NNSs
X-ray photoelectron spectroscopy (XPS) measurements were shown in Fig. 4a and deconvoluted XPS spectra of the Pd-NNSs were plotted in the Fig. 4b. XPS spectra showed a big peak at 285 eV for the Cls and two small peaks at 334 eV and 342 eV, for the Pd 3d regions.24 The spectra also indicated that the PdII was successfully reduced to the metallic Pd0. XPS spectra further indicated that very little Pd-NNSs are loaded on the surface of the carbon nanofibers and mainly embedded in the carbon nanofibers.24 This happens because the K2PdIICl4/K4FeII(CN)6 cyanogel could intrude inner fibers and after reduction Pd-NNSs on the outer surface could easily wash away leaving behind the firmly embedded Pd-NNSs inside the fibers. | ||
| Fig. 4 XPS spectra of Pd-NNSs: total spectrum (a) and enlarged Pd 3d spectrum (b) supported on the ECNFs. | ||
3.4. Catalytic characteristics of Pd-NNSs-ECNFs
Suzuki coupling reaction catalysed by Pd is an important and powerful strategy for the formation of carbon–carbon (C–C) bonds.31,32 In this study, in order to evaluate the catalytic activity, the Pd-NNSs-ECNFs were used as catalysts in the Suzuki (C–C coupling) reaction between aryl halide and phenylboronic acid, as shown in the Scheme 1. | ||
| Scheme 1 Suzuki coupling reactions of aryl halide with phenylboronic acid, with sodium hydroxide in ethanol/water, catalysed by Pd-NNSs-ECNFs respectively. | ||
In Suzuki coupling reaction, the various bases have big influence on the efficiency.11 Therefore, we firstly optimized the experimental conditions of using different bases. The high yields were obtained when using NaOH, KOH, or NaOMe as base (Table 1, entry 5–7) and NaOH was the most efficient (high yield and cost effective) base in Suzuki coupling reaction with ethanol/water used as the solvent.
| Entry | Base | Temperature | Time | Yieldb |
|---|---|---|---|---|
| a Reaction conditions: 4-chlorobenzene (1 mmol), phenylboronic acid (1.5 mmol), base (3.0 mmol), deionized water (8 mL), ethanol (8 mL) and 0.5 mol% Pd-NNSs-ECNFs, 80 °C, under N2.b Determined by GC analysis with an internal standard. | ||||
| 1 | K2CO3 | 80 °C | 10 h | 65% |
| 2 | K3PO4 | 80 °C | 10 h | 45% |
| 3 | Cs2CO3 | 80 °C | 10 h | 56% |
| 4 | Na2CO3 | 80 °C | 10 h | 41% |
| 5 | NaOH | 80 °C | 10 h | 91% |
| 6 | KOH | 80 °C | 10 h | 92% |
| 7 | NaOMe | 80 °C | 10 h | 93% |
Secondly, Pd-NNSs-ECNFs were used as catalysts for Suzuki–Miyaura reactions of various aryl halides (Table 2). The Pd-NNSs-ECNFs showed good catalytic activity to different aryl halides. It should be noted that the low-active 4-chlorobenzene also could be coupled in good yield (Table 2, entry 1). For the high-active aryl bromides and aryl iodides, higher yields were obtained (Table 2, entry 2–5).
| Entry | Time | Yieldb (%) | Product | Catalyst recycled | |
|---|---|---|---|---|---|
| a Reaction conditions: aryl halide (1.0 mmol), phenylboronic acid (1.5 mmol), NaOMe (3.0 mmol), ethanol (8 mL), water (8 mL), and 0.5 mol% Pd-NNSs-ECNFs, 80 °C, under N2.b Determined by the GC-analysis with an internal standard.c Pd(OAc)2 (0.5 mol% Pd) as catalyst.d Pd/C (0.5 mol% Pd) as catalyst.e 4-Nitrophenylboronic acid instead of phenylboronic acid.f 4-Methoxyphenylboronic acid instead of phenylboronic acid. | |||||
| 1 |  |
10 h | 91–86% |  |
7 times |
| 2 |  |
10 h | 97–92% | 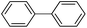 |
7 times |
| 3 | 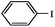 |
10 h | 99–96% | 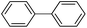 |
7 times |
| 4 | 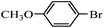 |
10 h | 96–91% | 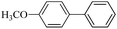 |
7 times |
| 5 | 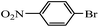 |
10 h | 98–93% | 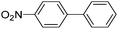 |
7 times |
| 6c | 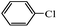 |
10 h | <3% | 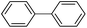 |
— |
| 7d | 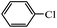 |
10 h | 32% | 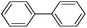 |
— |
| 8e | 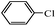 |
10 h | 95–90% | 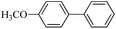 |
7 times |
| 9f | 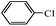 |
10 h | 97–92% | 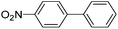 |
7 times |
The unreduced PdII (Pd(OAc)2) was also used as catalyst for the coupling reaction (Table 2, entry 6). However, C–C bond formation was not observed, indicating that palladium in the reduced form is the active site for the Suzuki–Miyaura coupling reaction.
A commercial Pd/C catalyst (Alfa Aesar, 0.5 mol% Pd) was also applied for the comparison purpose for the same coupling reaction (Table 2, entry 7). The yield was only 32% of the cross-coupled product by using Pd/C in the coupling of 4-chlorobenezene with phenylboronic acid. The high catalytic activity of Pd-NNSs-ECNFs may be ascribed to the large active surface area, which originated from the particular 3D networks structure24 and plentiful pores of electrospun carbon nanofibers mat. Such a structure ensured the high dispersion of Pd active sites and facilitates the diffusion of the reactants in the pores.
Generally, heterogeneous catalysts easily lose their catalytic activity due to extensive leaching of the active metal species. To our surprise, the Pd-NNSs-ECNFs can be recycled and reused seven times without losing its catalytic activity (Table 2, entry 1–5). Simple filtering could retrieve the catalyst from the reaction pot. Obviously, the excellent self-stability of Pd-NNSs enhanced the catalytic stability.
4. Conclusions
Palladium nano-network structures supported on the electrospun carbon nanofibers (Pd-NNSs-ECNFs) were successfully prepared by a novel K2PdIICl4/K4FeII(CN)6 cyanogel method. The structures of Pd-NNSs-ECNFs were characterized by the SEM, TEM and XPS. The Pd-NNSs-ECNFs catalyst was successful applied in Suzuki coupling reaction and showed high catalytic activity in an environmentally friendly solvent system (C2H5OH/H2O). The catalyst could be recycled and reused many times without losing its catalytic activity. We anticipate that the Pd-NNSs-ECNFs would be applied in various fields and provide new opportunities in green chemistry area.Acknowledgements
This Work is supported by the National Natural Science Foundation of China (Grant no. 21374044), the Academic and Technical Leader Plan of Jiangxi Provincial Main Disciplines (Grant no. 050008), The Major Special Projects of Jiangxi Provincial Department of Science and Technology (Grant no. 20114ABF05100) and the Technology Plan Landing Project of Jiangxi Provincial Department of Education.Notes and references
- V. A. Vonsa, H. Leegwaterb, W. J. Legersteeb, S. W. H. Eijtb and A. Schmidt-Ott, Int. J. Hydrogen Energy, 2010, 35, 5479–5489 CrossRef PubMed.
- M. A. Khalily, O. Ustahuseyin, R. Garifullin, R. Genc and M. O. Guler, Chem. Commun., 2012, 48, 11358–11360 RSC.
- M. Mondal and U. Bora, Green Chem., 2012, 14, 1873–1876 RSC.
- X. G. Wang, W. M. Wang, Z. Qi, C. Zhao, H. Ji and Z. H. Zhang, J. Power Sources, 2010, 195, 6740–6747 CrossRef CAS PubMed.
- X. Q. Zeng, M. L. Latimer, Z. L. Xiao, S. Panuganti, U. Welp, W. K. Kwok and T. Xu, Nano Lett., 2011, 11, 262–268 CrossRef CAS PubMed.
- V. Kumar, S. Srivastava, S. Umrao, R. Kumar, G. Nath, G. Sumana, P. S. Saxena and A. Srivastava, RSC Adv., 2014, 4, 2267–2273 RSC.
- B. Cornelio, G. A. Rance, M. Laronze-Cochard, A. Fontana, J. Sapi and A. N. Khlobystov, J. Mater. Chem. A, 2013, 1, 8737–8744 CAS.
- S. Cacchi, E. Caponetti, M. A. Casadei, A. D. Giulio, G. Fabrizi, G. Forte, A. Goggiamani, S. Moreno, P. Paolicelli, F. Petrucci, A. Prastaro and M. L. Saladino, Green Chem., 2012, 14, 317–320 RSC.
- P. W. Liu, Z. M. Dong, Z. B. Ye, W.-J. Wang and B.-G. Li, J. Mater. Chem. A, 2013, 1, 15469–15478 CAS.
- J. Liu, Y. Deng, H.-B. Wang, H. Zhang, G. X. Yu, B. B. Wu, H. Zhang, Q. Li, T. B. Marder, Z. Yang and A. W. Lei, Org. Lett., 2008, 10, 2661–2664 CrossRef CAS PubMed.
- B. Z. Yuan, Y. Y. Pan, Y. W. Li, B. L. Yin and H. F. Jiang, Angew. Chem., 2010, 49, 4054–4058 CrossRef CAS PubMed.
- G. Duan, H. Zhang, S. Jiang, M. Xie, S. Chen, M. Hanif and H. Hou, Adv. Mater. Lett., 2014, 122, 178–181 CrossRef CAS PubMed.
- G. Z. Hua, F. Nitzea, H. R. Barzegara, T. Sharifia, A. Mikolajczukb, C.-W. Taic, A. Borodzinskib and T. Waberga, J. Power Sources, 2012, 209, 236–242 CrossRef PubMed.
- C. Pham-Huu, N. Keller, L. J. Charbonniere, R. Ziessel and M. J. Ledoux, Chem. Commun., 2000, 19, 1871–1872 RSC.
- S.-Y. Liu, H.-Y. Li, M.-M. Shi, H. Jiang, X.-L. Hu, W.-Q. Li, L. Fu and H.-Z. Chen, Macromolecules, 2012, 45, 9004–9009 CrossRef CAS.
- E. Weiss, B. Dutta, Y. Schnell and R. A. Reziq, J. Mater. Chem. A, 2014, 2, 3971–3977 CAS.
- S. Ogasawara and S. J. Kato, J. Am. Chem. Soc., 2010, 132, 4608–4646 CrossRef CAS PubMed.
- B. J. Gallon, R. W. Kojima, R. B. Kaner and P. L. Diaconescu, Angew. Chem., Int. Ed., 2007, 46, 7251–7254 CrossRef CAS PubMed.
- G. W. Kabalka, R. M. Pagni and C. M. Hair, Org. Lett., 1999, 1, 1423–1425 CrossRef CAS.
- Z. H. Zhang and Z. Y. Wang, J. Org. Chem., 2006, 71, 7485–7487 CrossRef CAS PubMed.
- Y. J. Song, R. M. Garcia, R. M. Dorin, H. R. Wang, Y. Qiu, E. N. Coker, W. A. Steen, J. E. Miller and J. A. Shelnutt, Nano Lett., 2007, 7, 3650–3655 CrossRef CAS PubMed.
- S. Takenaka, N. Susuki, H. Miyamoto, E. Tanabe, H. Matsune and M. Kishida, J. Catal., 2011, 279, 381–388 CrossRef CAS PubMed.
- A. Mohanty, N. Garg and R. C. Jin, Angew. Chem., 2010, 122, 5082–5086 CrossRef PubMed.
- G. J. Zhang, L. Zhang, L. P. Shen, Y. Chen, Y. M. Zhou, Y. W. Tang and T. H. Lu, ChemPlusChem, 2012, 77, 936–940 CrossRef CAS PubMed.
- B. W. Pfennig, A. B. Bocarsly and R. K. Prudhomme, J. Am. Chem. Soc., 1993, 115, 2661–2665 CrossRef CAS.
- M. Heibel, G. Kumar, C. Wyse, P. Bukovec and A. B. Bocarsly, Chem. Mater., 1996, 8, 1504–1511 CrossRef CAS.
- R. S. Deshpande, S. L. Sharp-Goldman, J. L. Willson and A. B. Bocarsly, Chem. Mater., 2003, 15, 4239–4246 CrossRef CAS.
- G. Marban and A. B. Fuertes, Appl. Catal., B, 2001, 34, 43–53 CrossRef CAS.
- V. Gómez-Serrano, M. Acedo-Ramos, A. J. López-Peinado and C. Valenzuela-Calahorro, Thermochim. Acta, 1997, 291, 109–115 CrossRef.
- C.-M. Yang and K. Kaneko, J. Colloid Interface Sci., 2002, 246, 34–39 CrossRef CAS PubMed.
- H. Firouzabadi, N. Iranpoor, M. Gholinejad, S. Akbari and N. Jeddi, RSC Adv., 2014, 4, 17060–17070 RSC.
- N. Miyaura and A. Suzuki, Chem. Rev., 1995, 95, 2457–2483 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra07632j |
| This journal is © The Royal Society of Chemistry 2014 |