Preparation and characterization of surface modified boron nitride epoxy composites with enhanced thermal conductivity
Abstract
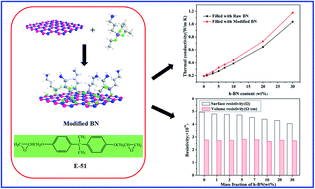
Hexagonal boron nitride (h-BN) microparticles, modified by surface coupling agent 3-aminopropyl triethoxy silane (APTES), were used to fabricate thermally conductive epoxy/BN composites, and the effects of modified-BN content on the thermal and insulating properties were investigated. It was found that incorporation of h-BN particles in the epoxy matrix significantly enhanced the thermal conductivity of the composites. With 30 wt% modified-BN loading, the thermal conductivity of the composites was 1.178 W m−1 K−1, 6.14 times higher than that of the neat epoxy. Fabricated epoxy/BN composites exhibited improved thermal stability, storage modulus, and glass transition temperature with increased BN content. The composites also possessed excellent electrical insulation properties. These results revealed that epoxy/BN composites are promising as efficient heat-releasing materials for thermal management and microelectronic encapsulation.

 Please wait while we load your content...
Please wait while we load your content...