Extraction of alpha-fetoprotein (AFP) with magnetic albuminoid-imprinted poly(ethylene-co-vinyl alcohol) nanoparticles from human hepatocellular carcinoma HepG2 cellular culture medium
Mei-Hwa Leea,
Arti Ahluwaliab,
Ke-Ming Hsuc,
Wei-Ti China and
Hung-Yin Lin*c
aDepartment of Materials Science and Engineering, I-Shou University, Kaohsiung 840, Taiwan
bCNR Institute of Clinical Physiology, Pisa and Research Center “E. Piaggio, University of Pisa, 56125 Pisa, Italy
cDepartment of Chemical and Materials Engineering, National University of Kaohsiung, Kaohsiung 81148, Taiwan. E-mail: linhy@ntu.edu.tw; linhy@caa.columbia.edu
First published on 13th August 2014
Abstract
Selective adsorption by magnetic nanoparticles is an effective method for obtaining valuable molecules from biological fluids. The synthesis of molecularly imprinted polymeric composite particles may be utilized routinely to remove a target from a complex mixture (e.g. biological fluids). Nowadays alpha-fetoprotein (AFP) is used as an important biomarker in the screening of human hepatocellular carcinoma. The imprinting of albumin (ALB) on poly(ethylene-co-vinyl alcohol) based nanoparticles may be adopted for the selective adsorption of AFP, because the two molecules have similar amino acid sequences. In this work, we studied the adsorption of ALB and AFP onto magnetic albuminoid-imprinted poly(ethylene-co-vinyl alcohol) nanoparticles (i.e. ALB or AFP MMIPs). The binding capacity of the MMIPs increases threefold with imprinting protein concentration (from 0.1 to 1.0 mg mL−1). MMIPs imprinted with 0.5 mg mL−1 of albumin or AFP adsorbed high levels of alpha-fetoprotein (60 ± 6 and 185 ± 17 μg g−1 respectively) from a HepG2 culturing medium diluted by 50×.
Introduction
Human hepatocellular carcinoma cell lines are found to secrete major plasma proteins and the hepatitis B surface antigen.1 The proteins may provide a major tool for the investigation of the control of biosynthesis. Almost half of the people of the world have suffered from a chronic infection by hepatitis B virus (HBV), and 5–10% of the infections convert to liver cancer.2 In Taiwan, the last two decades have seen a large increase in the number of liver cancer patients, and the death rate has doubled from 17 to 34 per hundred thousand. The diagnosis of liver cancer and the discovery of cancer biomarkers are very important to healthcare. Current methods of screening for liver cancer in at-risk patients involve measuring the serum level of alpha-fetoprotein (AFP).3 Many studies have indicated that AFP is a member of the albuminoid gene superfamily4,5 which includes albumin, AFP, afamin and vitamin D-binding proteins. Alpha-fetoprotein and albumin comprise approximately 600 amino acids and have a 40.2% sequence identity and a 59.1% structural similarity.4The recognition of proteins using molecularly imprinted polymers has recently become attractive because of its use in the development of biomedical applications. Whitcombe et al. recently reviewed the methods, applications and problems of protein imprinting.6 Approaches to synthesizing polymers for molecular imprinting include epitope peptide imprinting or the use of complete protein molecules as template. The most common protein is albumin, because it is highly abundant in biological fluid – and especially in blood. Hence, the concentrations of albumin in plasma and urine are routinely evaluated in clinical practice.7 The polymeric materials used for the synthesis of albumin-imprinted polymer are acrylamide,8 agarose gel,9 alginate,10,11 m-aminophenylboronic acid (APBA),12–14 chitosan,10 3-dimethylaminopropyl methacrylate (DMAPM),15,16 N-isopropylacrylamide (NIPA),17,18 methacrylic acid (MAA)19 and methyl methacrylate (MMA).20 The mean sizes of the protein-imprinted polymers vary from sub-millimeter for natural polymers, to as small as micrometers to tenths of nanometers for most synthesized polymers. Smaller particles have higher specific area and are less affected by gravity; therefore, they have greater potential for active adsorption in aqueous solution. Larger particles are generally packed in an high-performance liquid chromatography (HPLC) column to undergo chromatography.18 The authors' earlier works have involved albumin as a target protein in urinalysis21–23 and some characterization, such as zeta potential, reusability and selectivity.23
Magnetic molecularly imprinted polymers (MMIPs) may potentially be useful in sample preparation, which has been reviewed recently by Xie et al.24 For adsorbing specific proteins from biological fluids, hemoglobin,25,26 lysozyme23,27,28 and albumin18,23,29,30 are employed as the model proteins. Functionalized magnetic nanoparticles (e.g. MMIPs) have several advantages, including smaller amounts of sorbent, shorter extraction and separation duration, better dispersibility and higher selectivity compared with traditional solid phase extraction (SPE) sorbents.24
In this study we investigate the feasibility of extracting AFP from the human hepatocellular carcinoma HepG2 cellular culture medium using albuminoid-imprinted MMIPs (Scheme 1). We focused our analysis on the morphology and binding mechanism of magnetic albuminoid-imprinted poly(ethylene-co-vinyl alcohol) (EVAL) particles. The effect of the albumin imprinting concentration on the adsorption capacities of human albumin and alpha-fetoprotein was also assessed. Finally the recovery of alpha-fetoprotein in a HepG2 culture medium from ALB- or AFP-imprinted EVAL composite particles (i.e. ALB MMIPs or AFP MMIPs) was realized using different imprinting concentrations and adsorption times using a commercial serum analyzer.
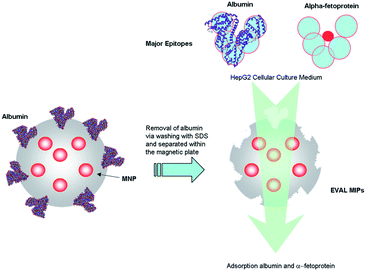 | ||
| Scheme 1 The selective adsorption of alpha-fetoprotein using albuminoid MMIP nanoparticles from the human hepatocellular carcinoma HepG2 cellular culture medium. | ||
Experimental section
Reagents and chemicals
Human purified AFP (P109-3, from cord serum, >95%) was purchased from Acris Antibodies GmbH (Herford, Germany). Albumin (from bovine serum, minimum 98%) and poly(ethylene-co-vinyl alcohol), EVAL, with ethylene 27, 32, 38 and 44 mol% (product no. 414077, 414093, 414085, 414107) were from Sigma-Aldrich Co. (St. Louis, MO). Dimethyl sulfoxide (DMSO, product # 161954) was purchased from Panreac (Barcelona, Spain) and used as the solvent to dissolve EVAL polymer particles in the concentration of 1 wt%. Iron(III) chloride 6-hydrate (97%) and iron(II) sulphate 7-hydrate (99.0%) were also from Panreac. Absolute ethyl alcohol was from J. T. Baker (ACS grade, NJ). Sodium dodecyl sulphate (SDS) was purchased from Sigma-Aldrich Co. (St. Louis, MO) and used for the removal of target molecules. All chemicals were used as received unless otherwise indicated. HepG2 cell line (#60025) was purchased from Bioresource Collection and Research Center (BCRC), Hsinchu, Taiwan. The culture medium for HepG2 cell lines contains Eagle's Minimum Essential Medium (EMEM including salts, L-glutamine and non-essential amino acids), sodium bicarbonate (NaHCO3) 1.5 g L−1, sodium pyruvate 1 mM (ca. 0.11 g L−1), 10% v/v of fetal bovine serum (FBS) and 1% v/v of antibiotics (penicillin and streptomycin).Formation of composite molecularly imprinted poly(ethylene-co-ethylene alcohol) nanoparticles
Synthesis of magnetic nanoparticles was by the Massart method, which is simply co-precipitation of a mixture of iron(III) chloride 6-hydrate and iron(II) sulphate 7-hydrate by sodium hydroxide. This magnetite was mixed with oleic acid for better dispersion and repeatedly washed while adsorbed on a magnetic plate, and then freeze-dried overnight.The synthesis of magnetic albumin-imprinted and non-imprinted EVAL nanoparticles included three steps: (1) magnetic nanoparticles were added to the EVAL solution (EVAL–DMSO = 1.0 wt%) to a concentration of 20 mg mL−1. The EVAL/magnetic particle solution was mixed with 0.1–1.0 mg mL−1 of albumin or 0.1, 0.5 mg mL−1 of AFP; (2) dispersion of the EVAL/magnetic particle solution in 10 mL non-solvent solution31 (deionized water–isopropanol = 2/3 in weight) for EVAL at 5 °C; and then (3) removal of the template molecules and EVAL particles without encapsulated magnetic nanoparticles using a magnetic plate and washing with 2 mL 1% w/v SDS solution 10 min for two times and then deionized water 10 min for four times. All composite nanoparticles were equilibrated with deionized water overnight before use. The non-imprinted polymers (NIPs) were prepared identically, except that the template was omitted. AFP imprinted magnetic nanoparticles were prepared using the same method, but only at two concentrations.
Binding kinetic measurement of the ALB MMIPs and MNIPs
The rebinding measurements of the template molecules to the molecularly imprinted (MIP) or non-imprinted (NIP) polymers were performed with 1 mL 0.05 mg mL−1 of albumin solution, which was dissolved in the phosphate buffer saline (PBS). These solutions were added to 1 mL of MMIP particles (15 mg) in deionized water on a magnetic plate for 1 minute. A UV/Vis spectrophotometer (Lambda 40, PerkinElmer, Wellesley MA) was then used to measure the concentration decrease in the stock solution, determined by absorption at 280 nm for albumin.Size distribution and morphology of magnetic albumin-imprinted EVAL composite particles
Magnetic and albumin-imprinted polymers composite nanoparticles were monitored by a dynamic light scattering (DLS) particle sizer (90Plus, Brookhaven Instrument Co., New York). The measurement of the particle size distribution was set at 25 °C with 3 minutes duration data collection at 90° detection angle. The average count rate of the background was 15 kcps and that of each measurement was between 20 and 500 kcps.The morphology of the magnetic molecularly imprinted polymers was also monitored by atomic force microscopy (NT-MDT Solver P47H-PRO, Moscow, Russia). Scanning was performed in air (room temperature (ca. 27 °C) and 87% relative humidity) using the tapping mode with scan rate 0.75 Hz. The cantilever was a SiO2 probe (model: TGS1, NT-MDT, Moscow, Russia) with 2 nm probe tip size and 144 kHz resonant frequency.
The transmission electron microscopy (TEM) images were taken with a Hitachi-700 transmission electron microscope operated at 100 kV accelerating voltage. Positive stained specimens were prepared as follows: 10 μL of particle solution was dispensed on a carbon-coated copper grid and the excess solution was instantly removed by a filter paper, then the particle solution (10 μL) and 0.5 wt% uranyl peroxide in DI water (10 μL) were sequentially dropped on the copper grid and the extra liquid was also removed by filter paper. The grid was dried in air for about 1 h before TEM characterization.
Extraction of albumin and alpha-fetoprotein to ALB MMIPs, AFP MMIPs and MNIPs
Freshly trypsinized HepG2 cells were seeded in T-25 flasks (500![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cells in 4 mL of medium per flask). The cells were placed in the incubator (37 °C and 5% CO2) for a week or three days. The culture medium was then collected for MMIPs adsorption. The collected culture medium was diluted 50 or 10 times and centrifuged at 3000 rpm for 10 minutes to remove wastes and dead cells. Magnetic molecularly imprinted polymers of diluted culture medium (to reduce the usage amount of MMIP particles, 1 mg and 1 mL respectively) were added to an eppendorf tube for stationary adsorption for up to 5 hours. After the adsorption at various time points, the tubes were placed on a magnetic plate to separate particles from solution. The magnetic particles were then washed with 1 mL PBS for 30 minutes. One milliliter of the diluted culture medium, post-adsorption extracted culture medium and washing solutions were stored in an eppendorf microcentrifuge tube at 4 °C and analyzed for microalbumin and alpha-fetoprotein with the ARCHITECT ci 8200 system using the manufacturer's protocols (Abbott Laboratories. Abbott Park, Illinois, U.S.A.). The details of the protocol could be found in the web page: https://www.abbottdiagnostics.com/en-us/products/ARCHITECT-ci8200.html. All binding experiments in this work were carried out with at least three replicates; data are expressed as means and standard deviations.
000 cells in 4 mL of medium per flask). The cells were placed in the incubator (37 °C and 5% CO2) for a week or three days. The culture medium was then collected for MMIPs adsorption. The collected culture medium was diluted 50 or 10 times and centrifuged at 3000 rpm for 10 minutes to remove wastes and dead cells. Magnetic molecularly imprinted polymers of diluted culture medium (to reduce the usage amount of MMIP particles, 1 mg and 1 mL respectively) were added to an eppendorf tube for stationary adsorption for up to 5 hours. After the adsorption at various time points, the tubes were placed on a magnetic plate to separate particles from solution. The magnetic particles were then washed with 1 mL PBS for 30 minutes. One milliliter of the diluted culture medium, post-adsorption extracted culture medium and washing solutions were stored in an eppendorf microcentrifuge tube at 4 °C and analyzed for microalbumin and alpha-fetoprotein with the ARCHITECT ci 8200 system using the manufacturer's protocols (Abbott Laboratories. Abbott Park, Illinois, U.S.A.). The details of the protocol could be found in the web page: https://www.abbottdiagnostics.com/en-us/products/ARCHITECT-ci8200.html. All binding experiments in this work were carried out with at least three replicates; data are expressed as means and standard deviations.
Results and discussion
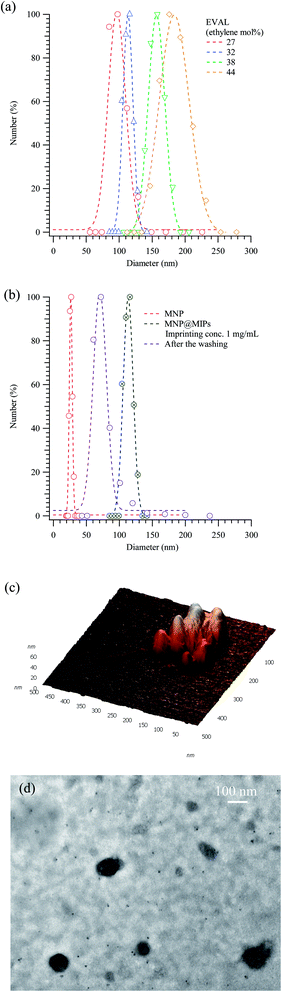
Although natural antibodies have high specificity toward their corresponding antigen, ligand–receptor binding usually depends on epitope recognition. Accordingly, peptides or structural protein fragments are sometimes used as recognition targets in a biological assay. Owing to the similarity between the amino acidic sequences of albumin and alpha-fetoprotein, the recognition of albumin may encounter difficulty when samples contain alpha-fetoprotein. Compared with other MMIPs preparation methods, the instant phase inversion technique22 is simpler than the conventional polymerization of functional and cross-linking monomers.24 Moreover, the smaller particle sizes obtained even without a silica layer coating24 could be used for successive delivery of bioactive molecules.32To study the binding efficiency of MMIPs, particles thereof and their surface morphology were examined. Fig. 1(a) compares the distribution of sizes of magnetic particles with that of molecularly imprinted polymers containing various mole percentages of ethylene. Interestingly, the average size of the albumin-imprinted EVAL composite particles before the removal of albumin increased from 55.8 to 181 nm as the ethylene content of EVAL increased from 27 to 44 mol%. Fig. 1(b) displays the size distributions of the magnetic particles and of the composite particles before and after the removal of the template and re-adsorption of template molecules. The mean sizes are 27 ± 2, 116 ± 8 and 72 ± 8 nm for non-imprinted magnetic particles, and molecularly imprinted polymers before and after removal of template molecules, respectively, suggesting that the presence of albumin molecules significantly increases the diameter of the composite particles (p = 0.0025). The magnetic molecularly imprinted polymeric nanoparticles are observed in the atomic force and transmission electron images in Fig. 1(c) and (d). Those figures are consistent with the size distribution in the graphs.
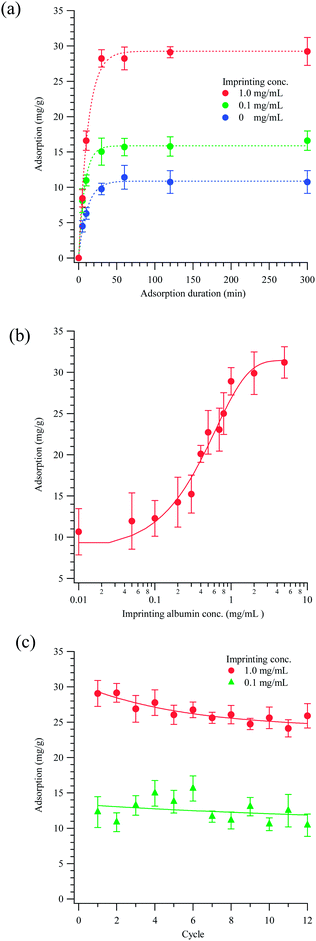
Fig. 2 presents the binding kinetics, effect of imprinted concentration and reusability of the magnetic molecularly imprinted polymeric nanoparticles. For a given rebinding concentration of albumin (0.05 mg mL−1), as shown in Fig. 2(a), magnetic molecularly imprinted polymers saturate in around 30 min, independent of the imprinting concentrations. In Fig. 2(b), the amount adsorbed at saturation is increases three fold with the imprinted concentration from 0.1 mg mL−1 to 1.0 mg mL−1, indicating that the imprinted albumin may change its orientation on EVALs from a loose arrangement to a more compact one, and a higher imprinting concentration may induce molecular aggregation of albumin. Fig. 2(c) plots the reusability of the magnetic albumin-imprinted polymeric nanoparticles at imprinted concentrations of 0.1 and 1 mg mL−1. MMIPs with a 1 mg mL−1 imprinted albumin appear to undergo a greater reduction in rebinding capacity after 12 cycles of washing compared with the lower concentration (15.17% for 1 mg mL−1 vs. 10.93% for 0.1 mg mL−1). However, their binding capacity still remains significantly higher. The loss of particles during the washing process may contribute to the reduction in rebinding capacities of MMIPs.
The extraction of alpha-fetoprotein was performed using composite nanoparticles of magnetic albumin-imprinted EVAL. In Table 1 we report the data on protein extraction from human hepatocellular carcinoma (HepG2) cellular culture medium, but not from pure protein solutions. The medium was incubated for three days and then diluted by a factor of 50, before being treated with the albumin-imprinted MMIPs. Initial albumin and alpha-fetoprotein concentrations in the stock culture medium were 2.8 and 0.43 μg mL−1 respectively. The binding amounts of MMIPs were in the range 0.3–0.6 and 0.015–0.046 mg of protein per g of nanoparticles for albumin and alpha-fetoprotein, respectively. A significantly (p < 0.001) higher adsorption for alpha-fetoprotein was observed when the EVAL contained ethylene 32 mol% than other ratios.
| EVAL | Albumin (μg g−1) | AFP (μg g−1) | ||||
|---|---|---|---|---|---|---|
| MIPs | NIPs | IF | MIPs | NIPs | IF | |
| a The albumin and α-fetoprotein concentrations in three days culture medium is diluted 50×. The albumin and alpha-fetoprotein concentrations in the media were initially 2.8 and 0.43 μg mL−1 as measured by the ARCHITECT ci 8200 system. | ||||||
| 27 | 500 ± 10 | 350 ± 50 | 1.43 | 40 ± 8 | 37 ± 12 | 1.08 |
| 32 | 550 ± 50 | 300 ± 0 | 1.83 | 46 ± 9 | 14 ± 8 | 3.13 |
| 38 | 450 ± 50 | 350 ± 50 | 1.29 | 44 ± 4 | 36 ± 1 | 1.21 |
| 44 | 450 ± 50 | 350 ± 50 | 1.29 | 42 ± 18 | 17 ± 5 | 2.46 |
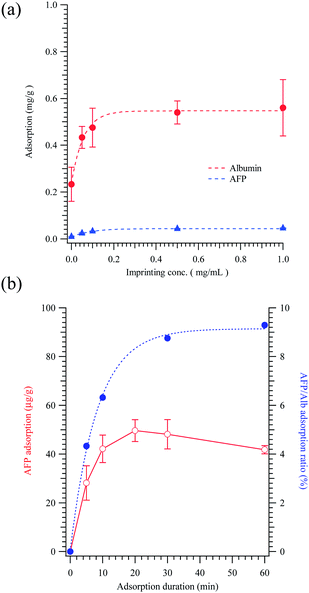
The imprinting concentration of albumin also influences the adsorption and desorption of alpha-fetoprotein. Fig. 3(a) plots the competitive adsorption of alpha-fetoprotein and human albumin. The MMIPs are saturated with proteins from the culture medium when the imprinting concentration exceeds 0.2 mg mL−1, and this imprinting concentration is also associated with the highest adsorption ratio between alpha-fetoprotein and albumin (Fig. 3(b)). The adsorption ratio is increased from 0.04 for non-imprinted polymers to 0.08 for imprinted polymers. Fig. 3(b) reveals that increasing the adsorption duration increased capacity of MMIPs to adsorb albumin. Although alpha-fetoprotein saturated in 10 minutes, the adsorption ratio of alpha-fetoprotein to albumin saturated after 20 minutes. This result demonstrates that increasing the adsorption duration may reduce the adsorption of albumin due to a Vroman-like effect.
The alpha-fetoprotein adsorbed on the ALB MMIPs was recovered by washing with PBS, as reported in Fig. 4. The recovered amounts were 3.22 ± 0.35 μg g−1 for NIP and 5.13 ± 0.75 μg g−1 for MMIPs (p < 0.03), which were imprinted with 1.0 mg mL−1 of albumin. To compare the imprinting of albuminoids, AFP MMIPs were also prepared, but at a lower imprinting concentration (e.g. 0.1 and 0.5 mg mL−1), for culture medium extraction.
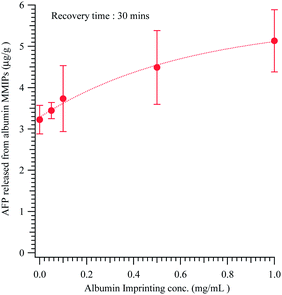 | ||
| Fig. 4 The extraction and recovery of AFP from the HepG2 cellular culturing medium by the ALB MMIPs with different imprinting albumin concentrations (30 min adsorption). | ||
As showed in Table 2, the higher imprinting concentration of MMIPs offers more cavities for the rebinding of AFP. However, the binding capacity of albumin imprinted MMIPs for albumin is about ten-fold higher than for AFP-imprinted MMIPs. Thus, despite the 59.1% structural similarity between alpha-fetoprotein and albumin, the particles can distinguish between the two proteins, suggesting that the imprinted cavities have high specificity.4 The alpha-fetoprotein adsorbed on the AFP MMIPs was recovered by washing with de-ionised water. Using the magnetic AFP-imprinted polymeric composite with an imprinting concentration of 0.1 mg mL−1 allowed cyclic extraction and release of 41.42 ± 2.56 μg g−1 for ten cycles.
| HepG2 culture medium target conc. (μg mL−1) | Adsorption (μg g−1) | ||||
|---|---|---|---|---|---|
| Imprinting albumin conc. (mg mL−1) | Imprinting alpha-fetoprotein conc. (mg mL−1) | ||||
| 0.1 | 0.5 | 0.1 | 0.5 | ||
| ALB | 0.90 | 460 ± 49 | 500 ± 63 | 60 ± 50 | 50 ± 50 |
| AFP | 0.77 | 34 ± 5 | 60 ± 6 | 170 ± 19 | 185 ± 17 |
Conclusions
Using cancer cells as bioreactors for the production of biomolecules provides the opportunity to reduce the cost of biomarkers in the synthesis of molecularly imprinted polymers. Using molecularly imprinted polymers as a sensing element may provide the advantages of low cost, and ease of preparation and storage. Accordingly, magnetic albuminoid-imprinted poly(ethylene-co-vinyl alcohol) composites may be adopted to preconcentrate alpha-fetoprotein from the human hepatocellular carcinoma HepG2 cellular culturing medium.Acknowledgements
We appreciate financial supports from National Science Council of ROC under Contract no. NSC 100-2314-B-390-001-MY3 and NSC 103-2923-E-390-001-MY2. Support from the CNR-Taiwan bilateral agreement is also acknowledged.Notes and references
- B. Knowles, C. Howe and D. Aden, Science, 1980, 209, 497–499 CAS.
- F. X. Bosch, J. Ribes, M. Díaz and R. Cléries, Gastroenterology, 2004, 127, S5–S16 CrossRef PubMed.
- A. El-Aneed and J. Banoub, Anticancer Res., 2006, 26, 3293–3300 CAS.
- T. Morinaga, M. Sakai, T. G. Wegmann and T. Tamaoki, Proc. Natl. Acad. Sci. U. S. A., 1983, 80, 4604–4608 CrossRef CAS.
- G. J. Mizejewski, Exp. Biol. Med., 2001, 226, 377–408 CAS.
- M. J. Whitcombe, I. Chianella, L. Larcombe, S. A. Piletsky, J. Noble, R. Porter and A. Horgan, Chem. Soc. Rev., 2011, 40, 1547–1571 RSC.
- N. Ghasemzadeh, F. Nyberg and S. Hjertén, J. Sep. Sci., 2008, 31, 3954–3958 CrossRef CAS PubMed.
- W. Zhang, L. Qin, X.-W. He, W.-Y. Li and Y.-K. Zhang, J. Chromatogr. A, 2009, 1216, 4560–4567 CrossRef CAS PubMed.
- Y. Lin, S. Tang, X. Mao and L. Bao, J. Biomed. Mater. Res., Part A, 2008, 85A, 573–581 CrossRef CAS PubMed.
- S. Takka and A. Gürel, AAPS PharmSciTech, 2010, 11, 460–466 CrossRef CAS PubMed.
- K. Zhao, U. JWei, G. Cheng, C. Yang and L. Chen, J. Appl. Polym. Sci., 2009, 113, 1133–1140 CrossRef CAS PubMed.
- F. Bonini, S. Piletsky, A. P. F. Turner, A. Speghini and A. Bossi, Biosens. Bioelectron., 2007, 22, 2322–2328 CrossRef CAS PubMed.
- H. Wang, W. Li, X. He, L. Chen and Y. Zhang, React. Funct. Polym., 2008, 68, 1291–1296 CrossRef CAS PubMed.
- Y. Lu, C.-L. Yan and S.-Y. Gao, Appl. Surf. Sci., 2009, 255, 6061–6066 CrossRef CAS PubMed.
- C.-H. Hu and T.-C. Chou, Microchim. Acta, 2009, 165, 399–405 CrossRef CAS.
- C.-H. Hu and T.-C. Chou, Microchem. J., 2009, 91, 53–58 CrossRef CAS PubMed.
- M. Soares da Silva, E. R. Vão, M. Temtem, L. Mafra, J. Caldeira, A. Aguiar-Ricardo and T. Casimiro, Biosens. Bioelectron., 2010, 25, 1742–1747 CrossRef CAS PubMed.
- Q.-Q. Gai, F. Qu, T. Zhang and Y.-K. Zhang, J. Chromatogr. A, 2011, 1218, 3489–3495 CrossRef CAS PubMed.
- X. Pang, G. Cheng, S. Lu and E. Tang, Anal. Bioanal. Chem., 2006, 384, 225–230 CrossRef CAS PubMed.
- C. J. Tan, H. G. Chua, K. H. Ker and Y. W. Tong, Anal. Chem., 2008, 80, 683–692 CrossRef CAS PubMed.
- C.-Y. Huang, T.-C. Tsai, J. L. Thomas, M.-H. Lee, B.-D. Liu and H.-Y. Lin, Biosens. Bioelectron., 2009, 24, 2611–2617 CrossRef CAS PubMed.
- H.-Y. Lin, M.-S. Ho and M.-H. Lee, Biosens. Bioelectron., 2009, 25, 579–586 CrossRef CAS PubMed.
- M.-H. Lee, J. L. Thomas, M.-H. Ho, C. Yuan and H.-Y. Lin, ACS Appl. Mater. Interfaces, 2010, 2, 1729–1736 CAS.
- L. Xie, R. Jiang, F. Zhu, H. Liu and G. Ouyang, Anal. Bioanal. Chem., 2014, 406, 377–399 CrossRef CAS PubMed.
- L. Li, X. He, L. Chen and Y. Zhang, Chem.–Asian J., 2009, 4, 286–293 CrossRef CAS PubMed.
- M. Zhang, Y. Wang, X. Jia, M. He, M. Xu, S. Yang and C. Zhang, Talanta, 2014, 120, 376–385 CrossRef CAS PubMed.
- Q.-Q. Gai, F. Qu, Z.-J. Liu, R.-J. Dai and Y.-K. Zhang, J. Chromatogr. A, 2010, 1217, 5035–5042 CrossRef CAS PubMed.
- T. Jing, H. Xia, Q. Guan, W. Lu, Q. Dai, J. Niu, J.-M. Lim, Q. Hao, Y.-I. Lee, Y. Zhou and S. Mei, Anal. Chim. Acta, 2011, 692, 73–79 CrossRef CAS PubMed.
- Z. Zhang, X. Yang, X. Chen, M. Zhang, L. Luo, M. Peng and S. Yao, Anal. Bioanal. Chem., 2011, 401, 2855–2863 CrossRef CAS PubMed.
- X. Li, B. Zhang, W. Li, X. Lei, X. Fan, L. Tian, H. Zhang and Q. Zhang, Biosens. Bioelectron., 2014, 51, 261–267 CrossRef CAS PubMed.
- T.-H. Young, L.-P. Cheng, C.-C. Hsieh and L.-W. Chen, Macromolecules, 1998, 31, 1229–1235 CrossRef CAS.
- M.-H. Lee, J. L. Thomas, H.-Y. Wang, C.-C. Chang, C.-C. Lin and H.-Y. Lin, J. Mater. Chem., 2012, 22, 24644–24651 RSC.
| This journal is © The Royal Society of Chemistry 2014 |