Enhanced performances of nonenzymatic glucose sensors by attaching Au nanoparticles onto the surfaces of Cu2O@Cu nanocable arrays†
Abstract
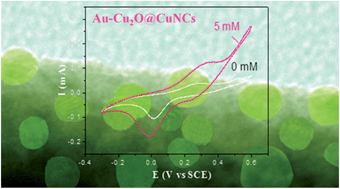
Nanoarchitectures (Au–Cu2O@CuNCs) have been fabricated successfully for the first time by attaching Au nanoparticles onto the surfaces of Cu2O@Cu nanocables. The hybrid material nanoelectrodes exhibit highly enhanced performances in detecting glucose nonenzymatically, compared to those made with Cu2O@Cu nanocable arrays or Au nanoparticles.

 Please wait while we load your content...
Please wait while we load your content...