Three-dimensional flower-like hybrid BiOI–zeolite composites with highly efficient adsorption and visible light photocatalytic activity
Abstract
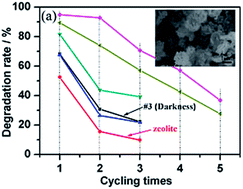
Three-dimensional flower-like hybrid BiOI–zeolite composites were synthesized via a simple, facile and environmentally-benign hydrothermal method. And the synthetic mechanism of the flower-like BiOI–zeolite hierarchical structures was discussed. The as-obtained BiOI–zeolite samples exhibited excellent performance in the degradation of methylene blue solution under visible light irradiation. The influence of different Bi/I molar ratios and surfactants was studied. The degradation rate of MB by BiOI–zeolite composite photocatalysts can reach 94.8% under visible light irradiation for 10 min, when the Bi/I molar ratio is 1 : 1 and is surfactant free. This implies its huge application potential as photocatalysts in degradation of pollutants under visible light.

 Please wait while we load your content...
Please wait while we load your content...