Highly permeable poly(4-methyl-1-pentyne)/NH2-MIL 53 (Al) mixed matrix membrane for CO2/CH4 separation
Reza Abedini,
Mohammadreza Omidkhah* and
Fatereh Dorosti
Faculty of Chemical Engineering, Tarbiat Modares University, P.O. Box 14115-143, Tehran, Iran. E-mail: r.abedini@modares.ac.ir; omidkhah@modares.ac.ir; Fax: +98 21 82883334; Tel: +98 21 82883334
First published on 4th August 2014
Abstract
Poly(4-methyl-1-pentyne) (PMP) as a polymer matrix together with synthesized NH2-MIL 53 metal organic framework (MOF) as a filler were used to fabricate a mixed matrix membrane (MMM). Various characterization methods as well as a series of CO2/CH4 gas separation tests (i.e. pure and mixed gas tests) were conducted in order to determine the effect of NH2-MIL 53 on the properties of the prepared MMMs and their gas transport characteristics. The results of TGA and DMA showed that both degradation temperature (Td) and glass transition temperature (Tg) increased by increasing the NH2-MIL 53 loading. SEM images also demonstrated that uniform dispersion of NH2-MIL 53 particles in the PMP matrix was achieved with no noticeable voids in the polymer-filler interfaces. It was also found that incorporation of NH2-MIL 53 in PMP results in an increase of gas permeability (especially for CO2) and higher CO2/CH4 selectivity. In contrast with the increment of CO2 solubility due to the presence of MOF in the polymer matrix, the solubility of CH4 decreases. Although the CO2 solubility was improved with the addition of NH2-MIL 53, its diffusivity remained almost constant with no significant changes. Lastly, it was observed that increasing the MOF loading along with higher feed pressure provide a condition to overcome the Robeson upper bound.
1. Introduction
Gas separation by means of membrane technology has increasingly gained interest, mainly because of the lower required energy, higher flexibility, working at low temperature and compact system structure.1–3 Among the various types of polymers, polymeric membranes have been widely used in membrane fabrication for gas separation processes mainly due to ease of fabrication and lower operating cost. Because of low chemical and thermal stability, and no acceptable selectivity of some polymeric membranes, fabrication of mixed matrix membranes (MMMs) was developed. Fabricated MMMs are based on the polymers and incorporation of different fillers such as metal oxides and carbon nano-tubes (CNTs), and also some porous materials such as zeolites, so that there is a potential to overcome the Robeson trade-off.4 However, the impact of novel materials such as metal organic frameworks (MOFs) on the performance of MMMs has not comprehensively studied.5 MOFs are specific categories of materials which are composed of metal ions as connectors and organic bridging ligands as linkers.5 Creating the non-selective voids at the filler-polymer interface caused that recent studies have turned to the usage of fillers with higher affinity to polymer matrix. MOFs with broad functionalized ligands interact with polymer chain properly leading to prevention of non-selective voids in MOF and polymer interface.6 Moreover, MOFs with high pore volume and specific cavities are able to absorb and separate particular gases which results in higher selectivity.7 Impact of different MOFs on performance of MMMs has been investigated in various studies.8–12The inclusion of amines in MOFs for the superior separation of CO2 from different gases has been examined. The effects of these functionalized MOFs on the separation performance and polymer-filler compatibility of mixed matrix membranes have been investigated.
Seoane et al. studied the effects of NH2-MIL 53 (Al) and NH2-MIL 101 (Al) in sulfur-containing copolyimide mixed matrix membranes for gas separation process. 6FDA![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DSDA/4MPD
DSDA/4MPD![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4,40-SDA 1
4,40-SDA 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (polymer P1) and 6FDA/4MPD
1 (polymer P1) and 6FDA/4MPD![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4,40-SDA 1
4,40-SDA 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (polymer P2) were used for based polymer matrix. The gas separation properties of MMMs evaluated through permeation of H2, CH4 and CO2. The loading of NH2-MIL 53 in MMMs were 10 and 15 wt% whereas 5 and 10 wt% for NH2-MIL 101. Using 10 wt% of NH2-MIL 101 in P1 caused that the permeability of H2, CH4 and CO2 improved to 114, 1.7 and 71 Barrer respectively and the performance of MMMs was closeness to the Robeson upper bound for H2/CH4 and CO2/CH4 separation.13
1 (polymer P2) were used for based polymer matrix. The gas separation properties of MMMs evaluated through permeation of H2, CH4 and CO2. The loading of NH2-MIL 53 in MMMs were 10 and 15 wt% whereas 5 and 10 wt% for NH2-MIL 101. Using 10 wt% of NH2-MIL 101 in P1 caused that the permeability of H2, CH4 and CO2 improved to 114, 1.7 and 71 Barrer respectively and the performance of MMMs was closeness to the Robeson upper bound for H2/CH4 and CO2/CH4 separation.13
Impacts of NH2-MIL 53 incorporation with polyimide were reported by Chen et al. NH2-MIL 53 was added up to 36 wt% to the polyimide. The permeability of CO2 increased significantly by increasing MOF loading but remained unchanged for CH4 in part. Like permeability, increasing of MOF loading led to the ideal selectivity (PCO2/PCH4) and consequently the separation factor (α CO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CH4 = 50
CH4 = 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50) was improved. The separation performance of MMM was decreased by 36 wt% loading of MOF which could be due to the lower compatibility of NH2-MIL 53 at higher loading and formation of non-selective voids in polymer-filler interfaces.14
50) was improved. The separation performance of MMM was decreased by 36 wt% loading of MOF which could be due to the lower compatibility of NH2-MIL 53 at higher loading and formation of non-selective voids in polymer-filler interfaces.14
Ghaffari Nik et al. investigated the effects of NH2-UiO-66 (Zr-ABDC) and NH2-MOF-199 as filler in polyimide MMMs. Both MOFs added to the polymer matrix with 25 wt% loading. For the case of NH2-UiO-66, presence of MOF in MMM resulted in permeability decline for both CO2 and CH4, while the ideal selectivity of CO2/CH4 increased. The results for NH2-UIO-66 showed that the presence of “–NH2” functional groups lead to creating rigidified chain at the polymer/filler interface, so that permeability reduced while selectivity improved. Unlike NH2-UiO-66, incorporation of NH2-MOF-199 with polyimide improved permeability of both gases (especially for CO2) together with CO2/CH4 ideal selectivity.15
Chen et al. reported the effects of NH2-MIL 53 with the loading of 8, 15 and 25 wt% on gas separation characteristics of co-polyimide. Through addition of MOF, the permeability of MMMs enhanced compared to neat co-polyimide. They also showed that increasing the drying temperature of membrane after casting process resulted in better gas permeability together with higher selectivity.16
Impact of NH2-MIL 53 particles on performance of PSF membrane was investigated by Valero et al. Results from Young's modulus and contact angle measurement suggested well dispersion of filler in polymer matrix. The optimum H2/CH4 separation factor of 67.34 with hydrogen permeability of 19.5 Barrer was achieved at 8 wt% loading of NH2-MIL 53.17
Incorporation of NH2-MIL 53 in Matrimid®5218 membrane improved the CO2/CH4 separation performance of MMM. At 15 wt% loading of NH2-MIL 53, CO2/CH4 selectivity increased up to 36.4. Although, the best selectivity obtained at 15 wt% incorporated of NH2-MIL 53, the permeability of CO2 decreased from 10.4 Barrer at 8 wt% loading to 9.2 Barrer at 15 wt% loading of NH2-MIL 53.18
Rodenas et al. investigated the performance of Matrimid®5218/NH2-MIL 53 based MMM. NH2-MIL 53 incorporated to Matrimid with 8, 15 and 25 wt%. Increasing of NH2-MIL 53 loading resulted in CO2 permeability improvement from 7.3 Barrer to 14.6 Barrer at 8 and 25 wt% loading of MOF respectively. Unlike permeability improvement, CO2/CH4 selectivity decreased from 38.3 to 34.8 at temperature of 308 K and pressure of 3 bar.19
Among different types of MOFs, MIL 53 has great tendency for selective CO2 adsorption compared to other gases. It was found that isotherm of CH4 and CO2 differ significantly due to isotypical porous trephthaliates MIL 53. The molecular structure of MIL 53 with chemical formula of [M(OH)(O2C–C6H4–CO2), (M = Al3+, Cr+3, Fe3+)] comprise from infinite chains which share M4(OH)2 octahedra, interconnected with the dicarboxylate groups.20
Applications of glassy polymers which have high free volume (i.e., resulted from unoccupied space between molecules) have acquired considerable interests. These polymers such as PTMSP,21,22 PTMGP23,24 and PMP25,26 are relatively more permeable to condensable large gases.
Poly(4-methyl-1-pentyne) (PMP) which is known as TPX, is a member of polyolefin group. PMP with low density, good chemical resistance, high thermal stability and especially high gases permeability is a promising nominee as a high permeable membrane.27
Therefore PMP is one of the alternative materials for fabrication of gas permeable membrane, in which the gas transport properties could be improved by embedding different kinds of filler in the polymer matrix that can surpass the Robeson upper bound.
The aim of this study is fabrication of MMM with appropriate filler and high free volume polymer. Thus, NH2-MIL 53 (Al) with high affinity to CO2 adsorption and high permeable PMP were selected as filler and based polymer respectively. The CO2 and CH4 adsorption–desorption test was conducted to evaluate adsorption properties of NH2-MIL 53 (Al). Different MMMs were fabricated by adding MOF to the polymer from 0 to 30 wt%. MOF and the prepared membranes were characterized by X-ray diffraction (XRD), FT-IR spectrum, thermal gravimetric analysis (TGA), dynamic mechanical analysis (DMA) scanning electron microscopy (SEM) and N2 adsorption. Lastly, performance of pure PMP and MMMs were analyzed by constant volume gas permeation test and the results were compared with Robeson upper bound.
2. Materials and methods
2.1. Materials
A PMP (Sigma Aldrich) with medium molecular weight was used for polymeric matrix of membrane. CCl4 supplied by Merck was also used to prepare the casting solution. N,N-Dimethylformamide or DMF (Merck), 2-amino terephthalic acid (NH2–H2BDC, Sigma-Aldrich with purity of 99%), aluminium nitrate nonahydrate (Al(NO3)3·9H2O, Sigma Aldrich with purity of 99%) and deionized water were employed for synthesis of amine functionalized MIL 53. MIL 53 (Al) from Sigma Aldrich without any preparing process was used to determine the exact effect of –NH2 functional group on synthesized NH2-MIL 53 (Al).2.2. Synthesis of amine functionalized MIL 53
The synthesis of NH2-MIL 53 (Al) was carried out according to the method proposed by Ahnfeldt et al.28 First, 3.1 g of aluminium nitrate nonahydrate was dissolved in 1.5 g of 2-aminotereohthalic acid through Teflon-lined stainless steel autoclave. Once the solution was homogenised, 22.8 mL of deionized water was added to the solution. Thereafter, the prepared mixture was heated at the temperature of 150 °C and for a period of 5 h. Then the product was washed with acetone and thereafter collected by a centrifuge set at the speed of 6000 rpm for 10 min. To remove the incorporated NH2–H2BDC, the synthesized NH2-MIL 53 washed with DMF twice, under 150 °C for 48 h. Finally, the produced powder was dried in a vacuum oven at temperature of 150 °C for 48 h.2.3. Membrane preparation
PMP/NH2-MIL 53 (Al) flat sheet membranes were prepared via solution casting method by applying priming technique. First, NH2-MIL 53 (Al) with pre-specified weight percent (wt%) were mixed with CCl4 under stirring for 6 hours. To ensure homogenous dispersion of MOFs in solvent, the mixture was sonicated for 10 min. Then PMP (5 wt% by weight of solution) was added to the mixture (NH2-MIL 53 + CCl4) and dissolved in the solvent by 24 hours stirring. The solution was remained in the vacuumed oven with ambient temperature for six hours in order to remove any bubble which created during stirring process. Final mixture with admissible viscosity was casted on glass plate. For slow evaporation of solvent and to avoid of any defect in membrane formation, a small thin glassy dish was put over the casted film to allow 24 hours for solvent evaporation. To ensure that solvent was evaporated completely, formatted membrane was placed in a vacuumed oven with temperature of 50 °C for 12 hours. At the end, the membrane was simply separated from glass plate. The concentration of NH2-MIL 53 (Al) in the casting solution varied from 0 to 30 wt%. Table 1 presents the composition of each casting solution.| Membrane code | Polymer (5 wt%) | Solvent (wt%) CCl4 | |
|---|---|---|---|
| PMP | NH2-MIL 53 (Al) | ||
| M1 | 100 | 0 | 95 |
| M2 | 95 | 5 | 95 |
| M3 | 90 | 10 | 95 |
| M4 | 85 | 15 | 95 |
| M5 | 80 | 20 | 95 |
| M6 | 75 | 25 | 95 |
| M7 | 70 | 30 | 95 |
2.4. Characterization
X-ray diffraction (XRD) was conducted at room temperature using X'Pert MPD (Philips) diffractometer with the cupper as an anode and graphite as a cathode. The wave length of X-ray, its voltage and current were 1.54056 Å, 40 kV and 30 mA, respectively. The data were collected from 2θ = 2–40° with the scanning rate of 0.02° s−1.Fourier transform infrared spectroscopy (FTIR) was performed using a PerkinElmer-0.03.06 FTIR spectrometer. Spectra were recorded with an average of 50 scans in the wavenumber range of 4000–450 cm−1, and with a resolution of 5 cm−1.
The thermal degradation of polymer and the actual amount of NH2-MIL 53 in polymer matrix were analysed by means of thermal gravimetric analyzer (TGA-50, Shimadzu). 20 mg of sample was loaded in a pretarred platinum pan which was pre-heated to 120 °C in order to remove moisture. When the sample was cooled down, it was reheated from temperature of 20 to 800 °C with a rate of 10 °C min−1.
Dynamic mechanical analysis (DMA) was carried out to examine the glass transition of MMMs and their mechanical properties. The temperature was increased from ambient to 90 °C with a rate of 5 °C min−1. Young's modulus of the membranes was also determined at various temperatures in the range of 25–200 °C with 5 °C min−1 heating rate and an amplitude of 75 μm. Tensile strength and elongation at break were also calculated at temperature of 25 °C and action force up to 20 N.
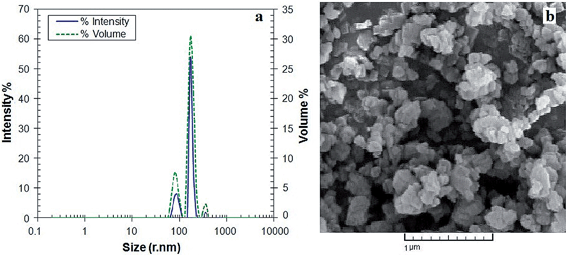
Diffraction light scattering (DLS) analysis was utilized to determine the size of NH2-MIL 53. In order to investigate the membrane structure and the quality of dispersion of NH2-MIL 53 in polymer matrix, scanning electron microscopy (SEM) was performed using CamScan SEM model MV2300 microscope. The membranes were snapped under liquid nitrogen to give a generally unfailing and clean cut. The membranes were then sputter-coated with thin film of gold and mounted on brass plates with double-sided adhesive tape in a lateral position.
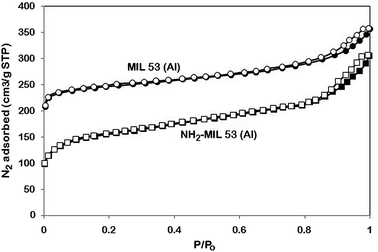
Nitrogen adsorption/desorption analysis (Belsorp mini II, BelJapan) was carried out to determine the textural characteristic of MOFs. The data were collected at temperature of 77 K and in the range of relative pressure (P/Po) between 0.02 and 1.0. In order to calculate the transparent properties of MMMs, CO2 and CH4 adsorption isotherms were conducted on MIL 53 and NH2-MIL 53. For each test, 0.5 g of particles was placed in a container at 303 K. The adsorption data was collected at pressures in the range of 0–10 bar and analysed by Langmuir eqn as follow:
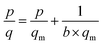 | (1) |
2.5. Pure gas permeation
Pure CH4 and CO2 permeabilities were determined by means of constant volume system and calculated using the following equation:
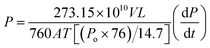 | (2) |
The multiplication of solubility and diffusivity of each gas, gives the related permeability of the gas as presented as follows:
| P = S × D | (3) |
The ideal selectivity is defined as the permeability ratio of two types of gas as it is given:
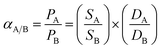 | (4) |
To calculate the diffusivity coefficient of each gas in membrane, the modified time lag method for mixed matrix membranes was used. Paul and Kemp proposed this method to represent the relationship between diffusivity (D) and time-lag (θ) in MMMs as:28
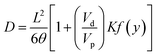 | (5) |
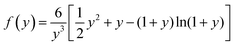 | (6) |
| K = qmb/KH | (7) |
The main advantage of modified time lag method compared to time lag one (were D = L2/6θ) is that this method considers the effect of filler phase on gas diffusion in MMMs. It is worthwhile to mention that L2/6θ can be derivate from eqn (5) for the case of neat membrane. In neat membrane, Vd (the volume fraction of filler) equals to zero and eqn (5) gives time lag method equation.
2.6. Mixed gas permeation
A binary mixture of CO2 and CH4 which containing 10% of CO2 was used as a feed gas in mixed gas test and permeability measurement of each gas conducted at 30 °C and 2 bar using the following equation:
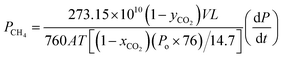 | (8) |
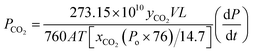 | (9) |
The gas mixture selectivity was calculated by the following equation:
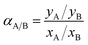 | (10) |
3. Results and discussions
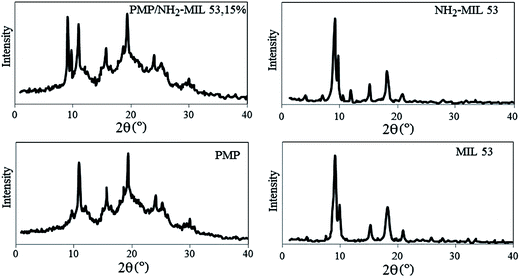
3.1. Characterization of NH2-MIL 53 and prepared MMMs
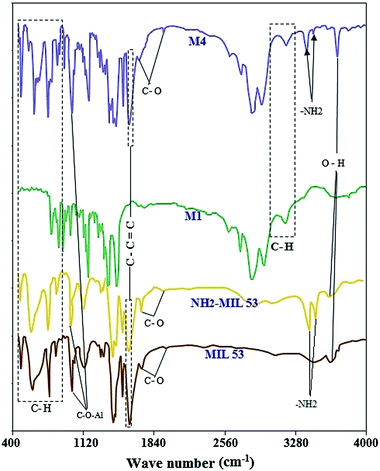
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond (1687 cm−1) in the pores of MOF is replaced by the C
O bond (1687 cm−1) in the pores of MOF is replaced by the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond of the DMF molecules (1670 cm−1). There are only two peaks at 3450 and 3617 cm−1 in the MIL 53 curve at wavenumbers above 2000 cm−1. These peaks correspond to the O–H bond and can be seen in other MMMs FTIR results too (at 3622 cm−1 for 15 wt% MOF loading).
O bond of the DMF molecules (1670 cm−1). There are only two peaks at 3450 and 3617 cm−1 in the MIL 53 curve at wavenumbers above 2000 cm−1. These peaks correspond to the O–H bond and can be seen in other MMMs FTIR results too (at 3622 cm−1 for 15 wt% MOF loading).
Two monomeric residue units in poly(4-methyl-1-pentyne) are repeated in every structural unit. Since there are methyl group in the side chain which the asymmetric stretching modes of –CH3 are assigned to 2973 and 2954 cm−1. CH3 symmetric stretching modes corresponded to very strong bands at 2888 and 2869 cm−1.31
The polymethylene chain with –CH2–CH–(CH3)2 as the side group is attached to every second carbon atom. The bands at 2971, 2949, 2928 and 2887 cm−1 all corresponded to the CH asymmetric stretching in CH3 and the bands of 2864 cm−1 shows the CH symmetric stretching. Moreover the C–C stretching is specified via bands of 1102, 1063, 1054 and 996 cm−1. The C–C bonds in plane bending were shown by the bands with frequency of 618, 540, 528 and 449 cm−1.31
The MMM with 15 wt% loading of NH2-MIL 53 has the similar peaks compare to the PMP neat membrane. There exists some shifting which are mainly due to the interaction between NH2-MIL 53 and polymer chains. The peaks at 3421 and 3488 cm−1 indicated the presence of –NH2 group and the amino terephthalic acid in the pores. The bands at 1021 cm−1 in MIL 53, NH2-MIL 53 and M4 corresponded to C–O–Al bond.
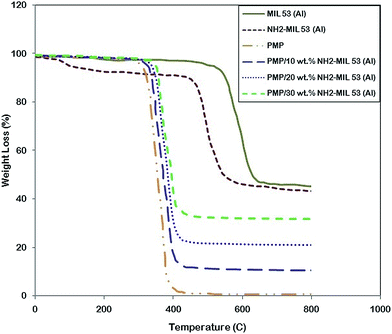 | ||
| Fig. 3 Thermal gravimetric analysis (TGA) curve of MIL 53, NH2-MIL 53 and MMMs with loading of 10, 20 and 30 wt% of MOF. | ||
The amount of NH2-MIL 53 as a filler in polymer matrix can be accurately measured via the remained weight loss in TGA curves. Except for NH2-MIL 53 and because of its –NH2 group, the first and third section of TGA curves for each sample, shows no significant weight loss which is mainly due to the weight have been occupied with remained solvent.
Fig. 4 shows the tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ curve and modulus (E′) for pure PMP and selected MMMs as a change in temperature. The peaks of tan
δ curve and modulus (E′) for pure PMP and selected MMMs as a change in temperature. The peaks of tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ curve (i.e., d
δ curve (i.e., d![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) tan
tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ/dT equal to zero), show the glass transition temperature of prepared membranes. For instance, it is seen that a peak at 33 °C represents the Tg of pure PMP which is the same as the previously reported Tg for PMP.35,36 The Tg of MMMs increased with increase of NH2-MIL 53 loading. The chain movement restriction as a result of the interaction between polymer chains and MOFs leads to the Tg increment.37,38 As shown, the storage modulus of pure PMP was 1370 MPa at 0 °C. Storage modulus of PMP deceases by increasing temperature and reaches 301 MPa at 88 °C. The MMMs show higher storage modulus over the entire temperature. The presence of NH2-MIL 53 strengthens and reinforces the PMP chains which subsequently results in increase of the modulus storage for MMMs.
δ/dT equal to zero), show the glass transition temperature of prepared membranes. For instance, it is seen that a peak at 33 °C represents the Tg of pure PMP which is the same as the previously reported Tg for PMP.35,36 The Tg of MMMs increased with increase of NH2-MIL 53 loading. The chain movement restriction as a result of the interaction between polymer chains and MOFs leads to the Tg increment.37,38 As shown, the storage modulus of pure PMP was 1370 MPa at 0 °C. Storage modulus of PMP deceases by increasing temperature and reaches 301 MPa at 88 °C. The MMMs show higher storage modulus over the entire temperature. The presence of NH2-MIL 53 strengthens and reinforces the PMP chains which subsequently results in increase of the modulus storage for MMMs.
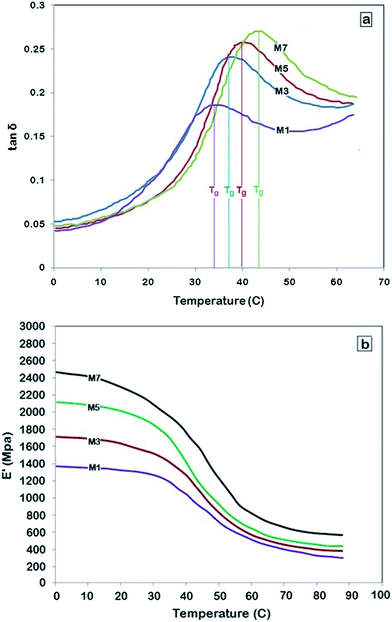 | ||
Fig. 4 (a) tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ curve, (b) storage modulus versus temperature for PMP (M1), PMP/10 wt% MOF (M3), PMP/20 wt% MOF (M5) and PMP/30 wt% MOF (M7). δ curve, (b) storage modulus versus temperature for PMP (M1), PMP/10 wt% MOF (M3), PMP/20 wt% MOF (M5) and PMP/30 wt% MOF (M7). | ||
Table 2 presents the mechanical properties of filled PMP membranes with 10, 20 and 30 wt% loading of NH2-MIL 53 loading. Tensile strength and elongation at break decreased by increasing the NH2-MIL 53 loading. During the membrane formation, NH2-MIL 53 may agglomerate. These agglomerated decrease tensile strength and elongation at break with two ways. First, chain disruptions can occur and the second, the concentration of stress is higher around of agglomerated MOFs.9 A significant enhancement of Young's modulus might be due to well dispersion of fillers in polymer matrix and acceptable interaction between polymer chains and NH2-MIL 53.10
| Membrane | Tensile strength (MPa) | Elongation at break (%) | Young modulus (GPa) |
|---|---|---|---|
| PMP | 132 ± 2 | 116 ± 5 | 3.65 ± 0.2 |
| PMP/10 wt% MOF | 120 ± 3 | 109 ± 2 | 3.89 ± 0.1 |
| PMP/20 wt% MOF | 112 ± 2 | 101 ± 3 | 4.18 ± 0.1 |
| PMP/30 wt% MOF | 107 ± 4 | 92 ± 3 | 4.34 ± 0.3 |
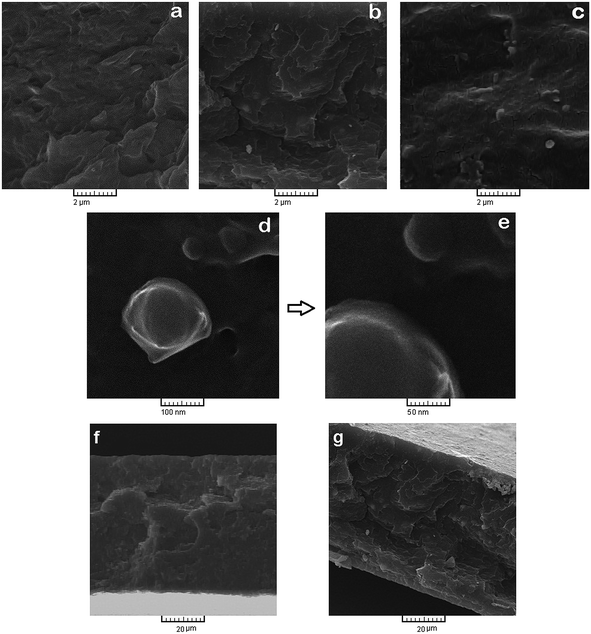
Quality of NH2-MIL 53 dispersion within the polymer matrix was investigated by means of scanning electron microscopy analysis. Fig. 6 shows the SEM photograph of top layer of prepared membranes. Some particles agglomeration can be seen on the top layer of MMMs with 10 and 30 wt% of NH2-MIL 53, however the dispersion of particles was appropriately uniform. Fig. 6d shows the interface of polymer and particle with higher magnification.
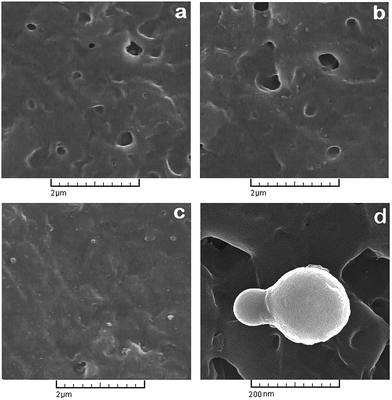 | ||
| Fig. 6 Top surface SEM image of (a) neat PMP, (b) PMP/10 wt% MOF and (c) PMP/30 wt% MOF, (d) NH2-MIL 53 particle on the surface of membrane. | ||
Fig. 7 also depicts the cross sectional photograph of the prepared membranes. For the MMM with 30 wt% loading of particles, NH2-MIL 53 dispersed uniformly in the membrane. Fig. 7d and e illustrate the NH2-MIL 53 and its interface with polymer matrix with higher magnification. As it is shown, there are almost no noticeable voids at the polymer/filler interface. Furthermore, cross sectional images confirm that at higher loading of NH2-MIL 53 (i.e., 30 wt%), some particle agglomeration formed in the polymer matrix. Fig. 7f and g reveals the cross sectional image of M1 and M4 with lower magnification. As Fig. 7f and g shown, prepared membranes have a thickness of 60 to 70 nm.
| Sample | SBET (m2 g−1) | Smeso (m2 g−1) | Vmicro (cm3 g−1) | Vmeso (cm3 g−1) | Vtotal (cm3 g−1) | DP (nm) |
|---|---|---|---|---|---|---|
| MIL 53 (al) | 1408 | 254 | 0.462 | 0.283 | 0.547 | 0.91 |
| NH2-MIL 53 (al) | 795 | 143 | 0.258 | 0.137 | 0.338 | 0.83 |
| Reduction percentage | 43.5 | 43.7 | 45.1 | 51.5 | 38.2 | 8.7 |
Since the –NH2 group was connected to –H2BDC ligands, the pore diameter of functionalized MIL 53 reduced to 0.83 nm compared to 0.91 nm of MIL 53. The percentage reduction of all textural characteristics for MIL 53 and NH2-MIL 53 is given in Table 3.
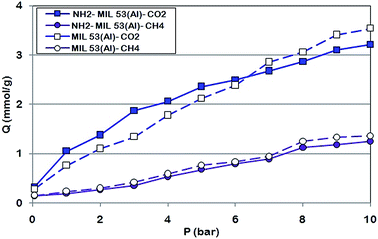
To investigate the adsorption properties of NH2-MIL 53 compared to those of MIL 53, the CO2 and CH4 adsorption isotherms were conducted at different pressures up to 10 bar and temperature of 303 K and the results for each MOF are shown in Fig. 9. CO2 uptake of MIL 53 and NH2-MIL 53 increased with increased adsorption pressure. The strong interaction between positive charges on the unsaturated open Aluminium and quadro-polarity of CO2 results in significant amount of CO2 adsorption.29 CO2 adsorption isotherm indicated that MIL 53 has greater potential to adsorb CO2 more than NH2-MIL 53. This behaviour confirms the BET results and reported previously for other similar amine-functionalized MOF.14,15 At pressures lower than 6.3 bar, more amount of CO2 adsorbed by NH2-MIL 53 compared to MIL 53 mainly due to presence of –NH2 group. This behaviour of amine-functionalized MOF was reported for other similar adsorbent.40–42 For CH4, the amount of CH4 uptake increased because of increase in adsorption pressure.
Unlike CO2, the amount of adsorbed CH4 was imperceptible at pressure lower than 2 bar which is in agreement with the results reported by Couck et al.43 This difference between adsorbed CO2 and CH4 is as a result of the presence of –NH2 groups located on the aromatic linker of terephthalic acid which reduces the polar adsorption sites.44
The overall CO2/CH4 sorption selectivities for MIL 53 and NH2-MIL 53 were 2.60 and 2.56, respectively, where similar to the reported by Stavitiski et al.45 At pressures lower than 6.3 bar, the selectivity of NH2-MIL 53 was more than that of MIL 53.
The adsorption characteristics of MIL 53 and NH2-MIL 53 were analysed based on the Langmuir equation. The values of qm and b of Langmuir determined via regression analysis of the data shown in Fig. 9 and are listed in Table 4. It was found that the qm for MIL 53 which has higher surface area and pore volume is higher than that for NH2-MIL 53 with lower surface area and pore volume. Henry's constant, which reflect the adsorption strength of gases was presented in Table 4 for both CO2 and CH4. KH of gases for MIL 53 adsorbent was higher than NH2-MIL 53 which confirms the superior adsorption capability of MIL 53 compared to amine functionalized one. Although Henry's constant used for adsorption analysis at lower pressure, but according to Langmuir model which emphasis on one layer adsorption of gases, KH can be calculated through multiplication of qm and b.
| Sample | Gas | qm (mmol g−1) | b × 103 (kPa−1) | KH × 102 (mmol g−1 kPa−1) |
|---|---|---|---|---|
| MIL 53 (al) | CH4 | 1.42 | 3.76 | 2.79 |
| CO2 | 3.84 | 4.27 | 2.35 | |
| NH2-MIL 53 (al) | CH4 | 1.36 | 3.81 | 2.57 |
| CO2 | 3.75 | 4.49 | 2.16 |
3.2. Pure gas permeation properties
| Membrane code | Permeability (barrer) | PCO2/PCH4 | |
|---|---|---|---|
| CO2 | CH4 | ||
| M1 | 98.74 | 11.32 | 8.72 |
| M2 | 107.32 | 9.05 | 11.85 |
| M3 | 118.74 | 9.43 | 12.59 |
| M4 | 139.56 | 8.87 | 15.72 |
| M5 | 164.78 | 8.92 | 18.46 |
| M6 | 203.44 | 10.08 | 20.18 |
| M7 | 226.37 | 10.12 | 22.36 |
Effect of NH2-MIL 53 loading on free volume of polymer was determined through calculation of fractional free volume (FFV) as follows:
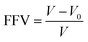 | (11) |
| Membrane code | M1 | M3 | M5 | M7 |
|---|---|---|---|---|
| FFV | 0.283 | 0.307 | 0.322 | 0.359 |
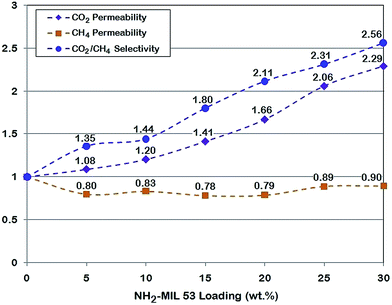
The selectivity of CO2/CH4 increased from 11.85 with 5 wt% of MOF loading to 22.36 when the MOF loading increased to 30 wt%. The increase in the selectivity of CO2/CH4 depends on three main mechanisms:
(i) The high free volume of PMP caused that gases pass through the membrane with dominant solubility. In glassy polymer also, presence of different kinds of particles (in this case MOF) can disrupt chains and increase the free volume of polymer. By this means, between CO2 and CH4, CO2 with higher condensability permeates more than CH4.46
(ii) The R–NH2 group in NH2-MIL 53 reacts reversibly with CO2 molecules and forms the carbamate.47 According to Fig. 10, this reaction enhances the permeability of CO2 compared to CH4 and results in the increment of CO2/CH4 selectivity.
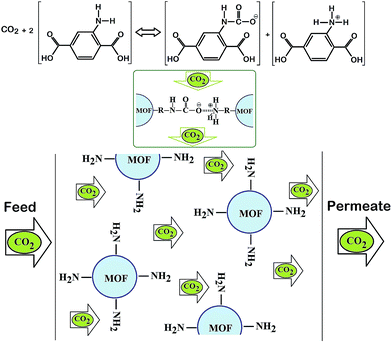 | ||
| Fig. 10 Schematic view of reaction between CO2 and R–NH2 group to form carbomate formation where facilitated the CO2 permeation. | ||
(iii) Functional –NH2 group significantly increases the affinity between polymer chains and MOF because of formatted hydrogen bond between –NH2 of MIL 53 and polymer. Although this bond rigidifies the polymer chain and consequently reduces the probability of non-selective voids formation, thereby it noticeably improves the selectivity of CO2/CH4.
Normalized permeability of CO2 and CH4 together with normalized CO2/CH4 ideal selectivity are shown in Fig. 11. This figure depicts the variation of both gas permeability and the CO2/CH4 selectivity more obvious. In contrast with non-modified MOF, the modified one has a smaller pore size which is mainly attributed to additional –NH2 group. This can be considered as the main reason for CH4 permeability reduction. It is noteworthy to mention that the similar trend was seen for CO2 permeability and CO2/CH4 selectivity.
| Membrane code | Diffusivity (10−7 cm2 s−1) | Solubility (10−3 cm3 (STP) cm−3 cmHg−1) | DCO2/DCH4 | SCO2/SCH4 | ||
|---|---|---|---|---|---|---|
| CO2 | CH4 | CO2 | CH4 | |||
| M1 | 61.37 | 16.67 | 16.10 | 6.82 | 3.82 | 2.47 |
| M2 | 62.14 | 16.61 | 17.24 | 5.45 | 3.74 | 3.16 |
| M3 | 63.29 | 17.15 | 18.76 | 5.58 | 3.69 | 3.42 |
| M4 | 62.75 | 17.87 | 22.24 | 4.97 | 3.51 | 4.47 |
| M5 | 61.53 | 16.76 | 26.78 | 5.17 | 3.57 | 5.17 |
| M6 | 61.20 | 17.63 | 33.24 | 5.72 | 3.47 | 5.81 |
| M7 | 60.47 | 15.84 | 37.43 | 6.38 | 3.81 | 5.86 |
The diffusivity coefficient was calculated using the modified time-lag method. The diffusivity of CO2 and CH4 almost remained constant by increasing NH2-MIL 53 loading. The diffusivity is influenced by free volume of MMMs, diffusion through MOF pore, and quality of surface compatibility between polymer chains and functional MOF.48 Each of aforementioned parameters has a different impact on diffusivity. Increasing the free volume of membrane due to the addition of NH2-MIL 53 facilitates and enhances the diffusion of gases. Despite free volume, the pore size of NH2-MIL 53 is smaller than that of MIL 53 which inhibits the passage of gases. A proper compatibility between polymer chains and MOF decreases the probability of non-selective void formation. The effect of this compatibility is obvious at higher NH2-MIL 53 loading which resulted the diffusivity of both CO2 and CH4 to be decreased.
Solubility coefficient of CO2 increased from 17.24 to 37.43 barrer by increasing the NH2-MIL 53 loading from 5 to 30 wt% in MMM, respectively. High affinity of NH2-MIL 53 to adsorb CO2 and higher condensability led to such significant increase in solubility coefficient of CO2. In contrast with CO2, lack of noticeable interaction between –NH2 group and CH4 along with lower condensability of CH4 caused that the solubility of CH4 did not effectively improved.
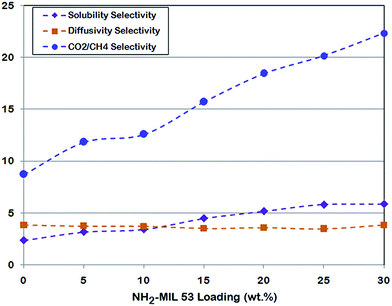
As it is presented in Table 7, all diffusivity selectivity (DCO2/DCH4) values for MMMs are lower than those for pure PMP. On the other hand, solubility selectivity (SCO2/SCH4) increased by increasing NH2-MIL 53 loading. Compared to neat PMP, all MMMs have higher solubility selectivity because of impressive CO2 sorption by amine functionalized MIL 53. It is important to notify that the NH2-MIL 53 fillers in MMM separate gases based on preferential adsorption of CO2, not size sieving. Consequently this phenomenon may leads to the more solubility selectivity of MMM compared with pure PMP.
Fig. 12 illustrates the variation of solubility, diffusivity and ideal selectivity as a change of NH2-MIL 53 loading in membranes. It is seen that solubility selectivity has a significant responsibility in ideal selectivity increment.
| Feed pressure (bar) | Membrane code | MIL 53 loading (wt%) | CO2 permeability (barrer) | CH4 permeability (barrer) |
|---|---|---|---|---|
| 2 | M1 | 0 | 98.74 | 11.32 |
| M4 | 15 | 139.56 | 8.87 | |
| M7 | 30 | 226.37 | 10.12 | |
| 4 | M1 | 0 | 97.82 | 11.20 |
| M4 | 15 | 208.68 | 11.92 | |
| M7 | 30 | 290.55 | 12.57 | |
| 6 | M1 | 0 | 97.05 | 11.13 |
| M4 | 15 | 231.63 | 12.11 | |
| M7 | 30 | 328.48 | 13.75 | |
| 8 | M1 | 0 | 96.54 | 10.95 |
| M4 | 15 | 264.26 | 13.53 | |
| M7 | 30 | 358.18 | 14.70 |
As Table 8 shows, the permeability of CO2 and CH4 increased from 98.74 and 11.32 barrer to 358.18 and 14.70 barrer (for M7 at 8 bar) respectively.
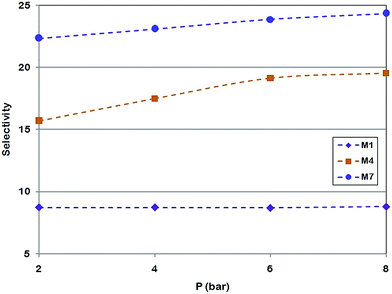
Fig. 13 depicts the effect of feed pressure on ideal selectivity of MMMs. The selectivity of neat PMP (M1) increased slightly from 8.72 to 8.81. In addition, it was found that the ideal selectivity of MMMs (M4 and M7) is increased. The CO2/CH4 selectivity of MMM with 15 wt% loading of NH2-MIL 53 increased from 15.72 at pressure of 2 bar to 19.53 at 8 bar. For 30 wt% loading of NH2-MIL 53 (M7), the CO2/CH4 selectivity increased from 22.36 at 2 bar to 24.35 at the pressure of 8 bar. This effect can be attributed to a more favourable condensability as well as more sorption of CO2 by NH2-MIL 53. Since there is a lower condensability along with less sorption for CH4, CO2 permeability increased significantly at higher pressures compared to the permeability of CH4 and higher selectivity was achieved.50 Cohen and Turnball proposed a correlation to determine the diffusion coefficient using free volume of polymer as it is given:51
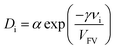 | (12) |
According to eqn (12), increasing the free volume of polymer results in reduction of the impact of diffusivity selectivity (DA/DB) in ideal selectivity. Therefore, solubility selectivity is a key factor and playing an important role in overall selectivity. It means that solubility is a dominant factor due to the increasing in free volume of PMP. In this case, presence of NH2-MIL 53 leads to the higher free volume of membrane. Moreover, sorption of CO2 by MOF increased as pressure increased, and this higher amount of sorption along with higher free volume leads to the enhancement of CO2/CH4 selectivity.
3.3. Mixed gas permeation
The mixed gas permeation property of MMMs was investigated through the gas mixtures of CO2/CH4 ratios of 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 90 v/v. After passing of gas mixtures through the membrane, the impermeable flow was removed and thereafter, the gas composition of permeable gas flow was determined by utilizing the GC. The results of the permeability and selectivity of gas mixture at pressure of 2 bar and temperature of 30 °C are presented in Table 9.
90 v/v. After passing of gas mixtures through the membrane, the impermeable flow was removed and thereafter, the gas composition of permeable gas flow was determined by utilizing the GC. The results of the permeability and selectivity of gas mixture at pressure of 2 bar and temperature of 30 °C are presented in Table 9.
| Membrane code | Permeability (barrer) | αCO2/CH4 | |
|---|---|---|---|
| CO2 | CH4 | ||
| M1 | 83.35 | 10.95 | 8.05 |
| M2 | 97.65 | 9.68 | 9.87 |
| M3 | 112.39 | 10.02 | 10.04 |
| M4 | 126.72 | 9.49 | 10.71 |
| M5 | 150.69 | 9.57 | 12.10 |
| M6 | 191.82 | 10.64 | 14.37 |
| M7 | 210.21 | 10.58 | 17.00 |
Comparing the results presented in Table 9 with those of the pure gas permeability data (i.e., Table 6) reveals that in all of the MMMs, the permeability values of CO2 as well as the selectivity of CO2/CH4 are lower than those of pure gases. The reduction of CO2 permeability and the subsequent decrease in the selectivity values are due to the presence of CH4 in the mixture. In fact, the co-presence of CH4 with the CO2 prevents the pass of CO2 through the membrane. The presence of the CH4 in mixture reduces the CO2 permeability mainly due to the two following reasons:
(i) The presence of CH4 in the mixture prevents further absorption of CO2 by MOF particles.
(ii) The presence of CH4 in the mixture inside the membrane, more specifically inside the free volume of the membrane, prevents the more condensation of CO2 which can reduce the CO2 solubility.
The results also showed that the presence of CH4 reduces the selectivity of the mixture so that, the maximum selectivity of PMP/NH2-MIL 53 MMM decreased from 22.36 (in pure permeation) to 17.00 in the CO2/CH4 mixture.
Table 10 presents the impact of feed pressure on the performance of MMMs containing MOF particles in the separation of gas mixture. The trend of changes in permeability and selectivity were almost the same as pure gas permeability and ideal selectivity of CO2/CH4 selectivity, respectively. It was also found that the permeability of CO2 and the real selectivity were lower than corresponding values of pure permeability and ideal selectivity. For CO2 case, the permeability of 339.48 Barrer and selectivity of 22.87 were the most efficient performance of PMP/NH2-MIL 53 which was observed at pressure of 8 bar.
| Feed pressure (bar) | Membrane code | MIL 53 loading (wt%) | CO2 permeability (barrer) | αCO2/CH4 |
|---|---|---|---|---|
| 2 | M1 | 0 | 83.35 | 7.61 |
| M4 | 15 | 126.72 | 13.34 | |
| M7 | 30 | 204.44 | 19.18 | |
| 4 | M1 | 0 | 81.18 | 7.74 |
| M4 | 15 | 189.75 | 14.88 | |
| M7 | 30 | 272.18 | 19.88 | |
| 6 | M1 | 0 | 80.21 | 7.94 |
| M4 | 15 | 222.51 | 17.15 | |
| M7 | 30 | 311.22 | 20.12 | |
| 8 | M1 | 0 | 80.10 | 8.12 |
| M4 | 15 | 244.61 | 18.24 | |
| M7 | 30 | 339.48 | 22.87 |
The results presented in Table 10 indicate that although the interaction between the gases in the gas mixture prevents the pass of CO2 through membrane, the MMMs show an appropriate performance to separate the CO2 mixtures.
3.4. Permeability and selectivity at higher loading of NH2-MIL 53
According to Fig. 14 adding NH2-MIL 53 at higher loading values results in the reduction of the permeability of CO2 and CH4. It was observed that the permeability of CO2 and CH4 was decreased at 40 wt% loading of NH2-MIL 53 to 216.71 and 9.87 Barrer compared to MMM with containing 35 wt% of NH2-MIL 53 (i.e. CO2 permeability and CO2/CH4 selectivity of 224.47 barrer and 9.87 respectively). In addition, the selectivity values at 30, 35, and 40 wt% loadings of NH2-MIL 53 were found to be almost unchanged. The main reason for permeability reduction of CO2 at higher loading of NH2-MIL 53 is attributed to the considerable decrease in its diffusion coefficient as a result of the presence of high amount of particles in the polymer matrix. High amount of NH2-MIL 53 particles in the polymer matrix of membrane rigidifies polymer chain extremely and elongates the diffusion path of CO2, thereby reduces the diffusion coefficient of the CO2. In addition, the high percentage of NH2-MIL 53 particles improves CO2 solubility coefficient together with solubility selectivity. Therefore, it was concluded that the 30 wt% NH2-MIL 53 is the optimum loading in the membrane matrix and provides the most appropriate performance compared to other MMMs.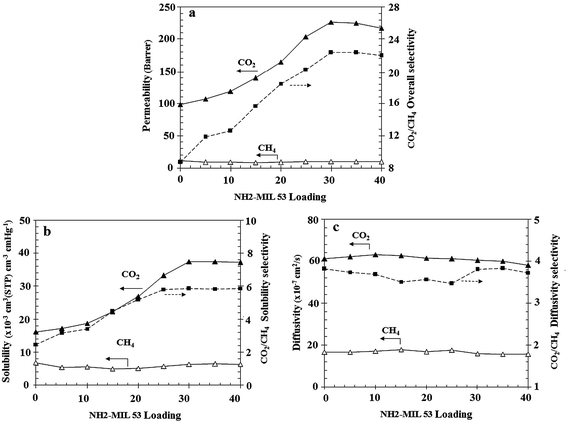 | ||
| Fig. 14 The effect of NH2-MIL 53 loading on (a) permeability and CO2/CH4 selectivity, (b) solubility and CO2/CH4 solubility selectivity (c) diffusivity and CO2/CH4 diffusivity selectivity. | ||
3.5. Separation performance of MMMs
Table 11 summarizes the results of CO2/CO4 separation using MMM in which MOF was used as filler. As it is shown, the permeability and selectivity of each MMM are strong functions of MOF structure, polymer properties, process parameters, and its affinity to CO2. Comparison of the results of this study with those of previously published work showed that PMP/NH2-MIL 53 MMMs have significantly higher permeability along with satisfactory range of selectivity.| Polymer | MOF | Loading (wt%) | Operating conditions | PCO2 | PCH4 | PCO2/PCH4 | Ref. | |
|---|---|---|---|---|---|---|---|---|
| P | T | |||||||
| a Permeability calculated in terms of GPU.b Mixed gas permeability. | ||||||||
| Matrimid® | CU-BPY-HFS | 10 | 2.7 bar | 35 °C | 7.8 | 0.24 | 31.9 | 8 |
| Matrimid® | CU-BPY-HFS | 30 | 2.7 bar | 35 °C | 10.4 | 0.38 | 27.5 | 8 |
| Matrimid® | ZIF-8 | 30 | 5 bar | 35 °C | 0.68a | 0.021a | 31.5 | 10 |
| Matrimid® | MIL 53 IAl) | 30 | 5 bar | 35 °C | 0.71a | 0.022a | 32.0 | 10 |
| Matrimid® | Cu3(BTC)2 | 30 | 5 bar | 35 °C | 0.65a | 0.019a | 33.0 | 10 |
| Copolyimide (P1) | NH2-MIL 101 | 10 | 3 bar | 35 °C | 70.9 | 1.7 | 41.6 | 13 |
| Copolyimide (P2) | NH2-MIL 101 | 10 | 3 bar | 35 °C | 151.0 | 5.1 | 29.6 | 13 |
| Copolyimide (P1) | NH2-MIL 53 | 10 | 3 bar | 35 °C | 56.9 | 1.6 | 35.8 | 13 |
| Copolyimide (P2) | NH2-MIL 53 | 10 | 3 bar | 35 °C | 137.0 | 5.0 | 27.2 | 13 |
| 6FDA-ODA | NH2-MIL 53 | 15 | 150 psi | 30 °C | 14.3 | 0.27 | 52. 6 | 14 |
| 6FDA-ODA | NH2-MIL 53 | 30 | 150 psi | 30 °C | 16.2 | 0.23 | 70.43 | 14 |
| 6FDA-ODA | UiO-66 | 25 | 150 psi | 35 °C | 40.4 | 1.10 | 44.1 | 15 |
| 6FDA-ODA | NH2-UiO-66 | 25 | 150 psi | 35 °C | 13.7 | 0.27 | 46.1 | 15 |
| 6FDA-ODA | MOF-199 | 25 | 150 psi | 35 °C | 21.8 | 0.43 | 51.2 | 15 |
| 6FDA-ODA | NH2-MOF-199 | 25 | 150 psi | 35 °C | 26.6 | 0.45 | 59.6 | 15 |
| PSF | ZIF-8 | 16 | 2 bar | 30 °C | 12.1 | 0.61 | 19.8 | 52 |
| PEES | ZIF-8 | 30 | 5 bar | 35 °C | 50 | 2.40 | 20.8 | 53 |
| PMP | NH2-MIL 53 | 30 | 8 bar | 30 °C | 358.2 | 14.7 | 24.4 | This work |
| PMP | NH2-MIL 53 | 30 | 8 bar | 30 °C | 339.5b | 14.9b | 22.8 | This work |
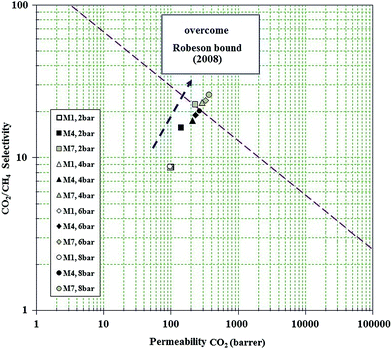
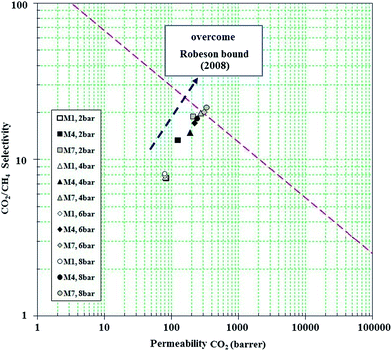
The performance of all synthesized membranes for CO2/CH4 separation is presented in Fig. 15 and 16. It is shown that higher pressures provide the condition to approach the Robeson bound. Fig. 15 and 16 confirm that incorporation of NH2-MIL 53 in PMP along with higher upstream pressure enhance the selectivity of CO2/CH4. Furthermore, the trend of selectivity versus CO2 permeability shows the behaviour against the Robeson curve trade off. The high free volume of polymer, more condensability of CO2, high affinity of NH2-MIL 53 to adsorb CO2, and higher upstream pressure are the main parameters providing the condition to overcome the Robeson upper bound. Generally, membranes with high permeability and acceptable selectivity are definitely more attractive to industry. Significant increase of CO2 permeability (i.e., from 98.74 barrer at 2 bar and no loading of MOF to 358.18 barrer at 8 bar and 30 wt% loading of MOF in pure permeation) and noticeable improvement of selectivity (i.e., from 8.72 to 24.4) were obtained via using NH2-MIL 53 for the prepared MMMs. Thus, it is believed that amine-functional groups (e.g., NH2-MIL 53) are appropriate fillers for MMMs. Fig. 16 also confirms that against the reduction in CO2 permeability and CO2/CH4 selectivity in mixed gas test (i.e. 339.5 Barrer and 22.8), the performance of MMM at 8 bar and 30 wt% loading of MOF surpasses Robeson upper bound.
4. Conclusions
In this study, synthesized NH2-MIL 53 was incorporated in PMP to prepare MMMs with extensive gas permeation properties. TGA and DMA analyses revealed that the thermal and mechanical properties such as degradation temperature, tensile strength and Young's modulus of MMMs improved by increasing the MOF loading. It was found that presence of NH2-MIL 53 in PMP matrix results in noticeable improvement of CO2 permeability. In addition, increasing the free volume of MMMs together with high affinity of NH2-MIL 53 to adsorb CO2 led to the higher CO2 permeability and solubility coefficient. Unlike CO2, enhancing the diffusivity coefficient of CH4 increased CH4 permeability slightly. CO2/CH4 selectivity in MMMs increased from 8.72 to 22.36 at pressure of 2 bar by increasing the MOF loading with dominant solubility selectivity. Moreover, it was obtained that increasing the feed pressure improves the permeability and selectivity of MMMs. A similar trend in permeability and CO2/CH4 selectivity was a remarkable property of PMP/NH2-MIL 53 MMMs. This character together with higher feed pressure ranging from 2 to 8 bar provided a condition for MMMs to overcome the Robeson upper bound. Finally the permeability and selectivity of MMMs (i.e., M7) obtained in this study were significantly improved compared with those of pure PMP. Generally, it seems that –NH2 functionalized MIL 53 is an appropriate filler to incorporate with PMP based on MMM for CO2/CH4 separation.Symbols
| p | Adsorption pressure (kPa) |
| q | Adsorbed gas (mmol g−1) |
| qm | Maximum adsorbed gas (mmol g−1) |
| b | Langmuir eqn parameter |
| KH | Henry's law constant |
| P | Permeability (Barrer) |
| V | Constant volume vessel (cm3) |
| L | Membrane thickness (cm) |
| A | Membrane surface area (cm2) |
| Po | Feed pressure (Psia) |
| T | Temperature (K) |
| S | Gas solubility (cm3 (STP) cm−3 cmHg−1) |
| D | Gas diffusivity (cm2 s−1) |
| αA/B | Selectivity |
| θ | Time-lag (s) |
| Vd | Volume fraction of filler |
| Vp | Volume fraction of polymeric phase |
| X | Mole fractions in the feed |
| y | Mole fractions in the permeate |
| FFV | Fractional free volume |
| V0 | Volume occupied by PMP chains |
| VW | van der Waals volume |
| VFV | Average free volume of polymer |
| γνA | Penetrant size |
References
- M. Zamani Pedram, M. R. Omidkhah and A. Ebadi Amooghin, Synthesis and characterization of diethanolamine-impregnated cross-linked polyvinylalcohol/glutaraldehyde membranes for CO2/CH4 separation, J. Ind. Eng. Chem., 2014, 20, 74–82 CrossRef CAS.
- L. Shao and T. S. Chung, In situ fabrication of cross-linked PEO/silica reverse-selective membranes for hydrogen purification, Int. J. Hydrogen Energy, 2009, 34, 6492–6504 CrossRef CAS.
- R. Abedini, S. M. Mousavi and R. Aminzadeh, A novel cellulose acetate (CA) membrane using TiO2 nanoparticles: Preparation, characterization and permeation study, Desalination, 2011, 277, 40–45 CrossRef CAS.
- I. Erucar, G. Yilmaz and S. Keskin, Recent advances in metal–organic framework-based mixed matrix membranes, Chem.–Asian J., 2012, 41, 14003–14027 Search PubMed.
- L. Lin, L. Zhang, C. Zhang, M. Dong, C. Liu, A. Wang, Y. Chu, Y. Zhang and Z. Cao, Membrane adsorber with metal organic frameworks for sulphur removal, RSC Adv., 2013, 3, 9889–9896 RSC.
- J. Caro, Are MOF membranes better in gas separation than those made of zeolites?, Curr. Opin. Chem. Eng., 2011, 1, 77–83 CrossRef CAS.
- X. Duan, Y. He, Y. Cui, Y. Yang, R. Krishna, B. Chen and G. Qian, Highly selective separation of small hydrocarbons and carbon dioxide in a metal–organic framework with open copper (II) coordination sites, RSC Adv., 2014, 4, 23058–23063 RSC.
- Y. Zhang, I. H. Musselman, J. P. Ferraris and K. J. Balkus, Gas permeability properties of Matrimid® membranes containing the metal-organic framework Cu–BPY–HFS, J. Membr. Sci., 2008, 313, 170–181 CrossRef CAS.
- F. Dorosti, M. Omidkhah and R. Abedini, Fabrication and characterization of Matrimid/MIL-53 mixed-matrix membranes for CO2/CH4 separations, Chem. Eng. Res. Des., 2014 DOI:10.1016/j.cherd.2014.02.018.
- S. Basu, A. Cano-Odena and I. F. J. Vankelecom, MOF-containing mixed-matrix membranes for CO2/CH4 and CO2/N2 binary gas mixture separations, Sep. Purif. Technol., 2011, 81, 31–40 CrossRef CAS.
- R. Abedini, M. Omidkhah and F. Dorosti, CO2/CH4 separation by using poly methyl pentene/MIL 53 particles mixed matrix membrane, Iran. J. Polym. Sci. Technol., 2014, 27, 18–27 Search PubMed.
- E. V. Perez, K. J. Balkus, J. P. Ferraris and I. H. Musselman, Mixed-matrix membranes containing MOF-5 for gas separations, J. Membr. Sci., 2009, 328, 165–173 CrossRef CAS.
- B. Seoane, C. Téllez, J. Coronas and C. Staudt, NH2-MIL-53(Al) and NH2-MIL-101(Al) in sulfur-containing copolyimide mixed matrix membranes for gas separation, Sep. Purif. Technol., 2013, 111, 72–81 CrossRef CAS.
- X. Y. Chen, H. Vinh-Thang, D. Rodrigue and S. Kaliaguine, Amine-functionalized MIL-53 metal−organic framework in polyimide mixed matrix membranes for CO2/CH4 Separation, Ind. Eng. Chem. Res., 2012, 51, 6895–6906 CrossRef CAS.
- O. Ghaffari Nik, X. Y. Chen and S. Kaliaguine, Functionalized metal organic framework-polyimide mixed matrix membranes for CO2/CH4 separation, J. Membr. Sci., 2012, 413–414, 48–61 Search PubMed.
- X. Y. Chen, H. Vinh-Thang, D. Rodrigue and S. Kaliaguine, Optimization of continuous phase in amino-functionalized metal–organic framework (MIL-53) based co-polyimide mixed matrix membranes for CO2/CH4 separation, RSC Adv., 2013, 3, 24266–24279 RSC.
- M. Valero, B. Zornoza, C. Téllez and J. Coronas, Mixed matrix membranes for gas separation by combination of silica MCM-41 and MOF NH 2-MIL-53(Al) in glassy polymers, Microporous Mesoporous Mater., 2014, 192, 23–24 CrossRef CAS.
- T. Rodenas, M. Dalen, P. Serra-Crespo, F. Kapteijn and J. Gascon, Mixed matrix membranes based on NH 2-functionalized MIL-type MOFs: Influence of structural and operational parameters on the CO 2/CH 4 separation performance, Microporous Mesoporous Mater., 2014, 192, 35–42 CrossRef CAS.
- T. Rodenas, M. Dalen, E. García-Pérez, P. Serra-Crespo, B. Zornoza, F. Kapteijn and J. Gascon, Visualizing MOF Mixed Matrix Membranes at the Nanoscale: Towards Structure-Performance Relationships in CO2/CH4 Separation Over NH2-MIL-53(Al)@PI, Adv. Funct. Mater., 2014, 24, 249–256 CrossRef CAS.
- A. Boutin, F. X. Coudert, M. A. Springuel-Huet, A. V. Neimark, G. Férey and A. H. Fuchs, The Behavior of Flexible MIL-53(Al) upon CH4 and CO2 Adsorption, J. Phys. Chem. C, 2010, 114, 22237–22244 CAS.
- I. Pinnau, C. G. Casillas, A. Morisato and B. D. Freeman, Hydrocarbon/hydrogen mixed gas permeation in poly(1-trimethylsilyl-1-propyne) (PTMSP), poly(1-phenyl-1-propyne) (PPP), and PTMSP/PPP blends, J. Polym. Sci., Part B: Polym. Phys., 1996, 34, 2613–2621 CrossRef CAS.
- S. Matteucci, A. Kusuma-Victor, D. Sanders, S. Swinea and B. D. Freeman, Gas transport in TiO2 nanoparticle-filled poly(1-trimethylsilyl-1-propyne), J. Membr. Sci., 2008, 307, 196–217 CrossRef CAS.
- V. Khotimskiy, M. Chirkova, E. Litvinova, M. Konrad, N. Lencerf, W. Yave, S. Shishatskiy and K. V. Peinemann, Poly(1-trimethylgermyl-1-propyne): synthesis, characterisation and transport properties of pure polymers and nanocomposites, Desalination, 2006, 199, 198–209 CrossRef CAS.
- A. Trusov, S. Legkov, L. J. P. Broeke, E. Goetheer, V. Khotimsky and A. Volkov, Gas/liquid membrane contactors based on disubstituted polyacetylene for CO2 absorption liquid regeneration at high pressure and temperature, J. Membr. Sci., 2011, 383, 241–249 CrossRef CAS.
- W. Yave, S. Shishatskiy, V. Abetz, S. Matson, E. Litvinova and V. Khotimskiy, A novel poly(4-methyl-2-pentyne)/TiO2 hybrid nanocomposite membrane for natural gas conditioning: butane/methane separation, Macromol. Chem. Phys., 2007, 208, 2412–2418 CrossRef CAS.
- T. C. Merkel, B. D. Freeman, R. J. Spontak, Z. He, I. Pinnau, P. Meakin and A. J. Hill, Sorption, transport, and structural evidence for enhanced free volume in poly(4-methyl-2-pentyne)/fumed silica nanocomposite membranes, Chem. Mater., 2003, 15, 109–118 CrossRef CAS.
- S. D. Wanjale and J. P. Jog, Poly(4-methyl-1-pentyne)/clay nanocomposites: effect of organically modified layered silicates, Polym. Int., 2004, 53, 101–105 CrossRef CAS.
- T. Ahnfeldt, D. Gunzelmann, T. Loiseau, D. Hirsemann, J. Senker, G. Férey and N. Stock, Synthesis and Modification of a Functionalized 3D Open-Framework Structure with MIL-53 Topology, Inorg. Chem., 2009, 48, 3057–3064 CrossRef CAS PubMed.
- D. R. Paul and D. R. Kemp, The diffusion time lag in polymer membranes containing adsorptive fillers, J. Polym. Sci., Polym. Symp., 1973, 41, 79–93 CrossRef.
- H. Fan, H. Xia, C. Kong and L. Chen, Synthesis of thin amine-functionalized MIL-53 membrane with high hydrogen permeability, Int. J. Hydrogen Energy, 2013, 38, 10796–10802 Search PubMed.
- E. J. Samuel and S. Mohanb, FTIR and FT Raman spectra and analysis of poly(4-methyl-1-pentyne), Spectrochim. Acta., 2004, 60, 19–24 CrossRef.
- R. Abedini, M. Omidkhah and F. Dorosti, Hydrogen separation and purification with poly(4-methyl-1-pentyne)/MIL 53 mixed matrix membrane based on reverse selectivity, Int. J. Hydrogen Energy, 2014, 56, 7897–7909 CrossRef.
- Y. Hu, L. Wu, J. Shen and M. Ye, Amino-functionalized multiple-walled carbon nanotubes-polyimide nanocomposite films fabricated by in situ polymerization, J. Appl. Polym. Sci., 2008, 110, 701–705 CrossRef CAS.
- R. Abedini, S. M. Mousavi and R. Aminzadeh, Effect of sonochemical synthesized TiO2 nanoparticles and coagulation bath temperature on morphology, thermal stability and pure water flux of asymmetric cellulose acetate nanocomposite membranes prepared via phase inversion method, Chem. Ind. Chem. Eng. Q., 2012, 18, 385–398 CrossRef CAS.
- S. Bourrelly, P. L. Llewellyn, C. Serre, F. Millange, T. Loiseau and G. Férey, Different adsorption behaviors of methane and carbon dioxide in the isotypic nanoporous metal terephthalates MIL-53 and MIL-47, J. Am. Chem. Soc., 2005, 127, 13519–13521 CrossRef CAS PubMed.
- T. Tanigami, K. Yamaura, S. Matsuzawa and K. Miyasaka, Thermal expansion of crystal lattice of isotactic poly(4-methyl-1-pentyne), Polym. J., 1986, 18, 35–40 CrossRef CAS.
- B. G. Ranby, K. S. Chan and H. Brumberger, Higher-order transitions in poly(4-methyl-1-pentyne), J. Polym. Sci., 1962, 58, 545–552 CrossRef CAS.
- X. Y. Chen, H. Vinh-Thang, D. Rodrigue and S. Kaliaguine, Effect of macrovoids in nano-silica/polyimide mixed matrix membranes for high flux CO2/CH4 gas separation, RSC Adv., 2014, 4, 12235–12244 RSC.
- E. P. Barrett, L. G. Joyner and P. P. Halenda, The determination of pore volume and area distributions in porous substances: Computations form nitrogen isotherms, J. Am. Chem. Soc., 1951, 73, 373–380 CrossRef CAS.
- R. V. Siriwardane, M. S. Shen, E. P. Fisher and J. A. Poston, Adsorption of CO2 on molecular sieves and activated carbon, Energy Fuels, 2001, 15, 279–284 CrossRef CAS.
- T. H. Bae, J. S. Lee, W. Qiu, W. J. Koros, C. W. Jones and S. Nair, A high-performance gas-separation membrane containing submicrometer-sized metal-organic framework crystals, Angew. Chem., Int. Ed., 2010, 49, 9863–9866 CrossRef CAS PubMed.
- Q. Yang and C. Zhong, Molecular simulation of carbon dioxide/methane/hydrogen mixture adsorption in metal-organic frameworks, J. Phys. Chem. B, 2006, 110, 17776–17783 CrossRef CAS PubMed.
- S. Couck, J. F. M. Denayer, G. V. Baron, T. Rémy, J. Gascon and F. Kapteijn, An amine-functionalized MIL-53 metal-organic framework with large separation power for CO2 and CH4, J. Am. Chem. Soc., 2009, 131, 6326–6327 CrossRef CAS PubMed.
- F. Zhang, X. Zou, X. Gao, S. Fan, F. Sun, H. Ren and G. Zhu, Hydrogen selective NH2-MIL-53(Al) MOF membranes with high permeability, Adv. Funct. Mater., 2012, 22, 3583–3590 CrossRef CAS.
- E. Stavitski, E. A. Pidko, S. Couck, T. Remy, E. J. M. Hensen, B. M. Weckhuysen, J. Denayer, J. Gascon and F. Kapteijn, Complexity behind CO2 capture on NH2-MIL-53(Al), Langmuir, 2011, 27, 3970–3976 CrossRef CAS PubMed.
- F. Dorosti, M. R. Omidkhah, M. Z. Pedram and F. Moghadam, Fabrication and characterization of polysulfone/polyimide–zeolite mixed matrix membrane for gas separation, Chem. Eng. J., 2011, 171, 1469–1476 CrossRef CAS.
- M. Caplow, Kinetics of carbamate formation and breakdown, J. Am. Chem. Soc., 1968, 90, 6795–6803 CrossRef CAS.
- F. Moghadam, M. R. Omidkhah, E. Vasheghani-Farahani, M. Z. Pedram and F. Dorosti, The effect of TiO2 nanoparticles on gas transport properties of Matrimid5218-based mixed matrix membranes, Sep. Purif. Technol., 2011, 77, 128–136 CrossRef CAS.
- T. C. Merkel, B. D. Freeman, R. J. Spontak, Z. He, I. Pinnau, P. Meakin and A. J. Hill, Ultrapermeable, reverse-selective nanocomposite membranes, Science, 2002, 296, 519–522 CrossRef CAS PubMed.
- J. Li, Y. Ma, M. C. McCarthy, J. Sculley, J. Yub, H. K. Jeong, P. B. Balbuenab and H. C. Zhoua, Carbon dioxide capture-related gas adsorption and separation in metal-organic frameworks, Coord. Chem. Rev., 2011, 255, 1791–1823 CrossRef CAS.
- M. H. Cohen and D. Turnbull, Molecular transport in liquids and glasses, J. Chem. Phys., 1959, 31, 1164–1169 CrossRef CAS.
- B. Zornoza, B. Seoane, J. M. Zamaro, C. Téllez and J. Coronas, Combination of MOFs and zeolites for mixed-matrix membranes, Chem. Phys. Chem., 2011, 12, 2781–2785 CrossRef CAS PubMed.
- K. Diaz, L. Garrido, M. Lopez-Gonzalez, L. F. del Castillo and E. Riande, CO2 transport in polysulfone membranes containing zeolitic imidazolate frameworks as determined by permeation and PFG NMR techniques, Macromolecules, 2010, 43, 316–325 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2014 |