DOI:
10.1039/C4RA06993E
(Paper)
RSC Adv., 2014,
4, 47402-47408
Simple solution-combusting synthesis of octahedral ZnFe2O4 nanocrystals and additive-promoted photocatalytic performance
Received
11th July 2014
, Accepted 22nd September 2014
First published on 22nd September 2014
Abstract
In this paper, we report a simple solution-combusting route for the one-step synthesis of octahedral ZnFe2O4 nanocrystals. Zn(CH3COO)2 and FeCl3 were employed as the starting Zn and Fe sources, oxygen in the atmosphere as the oxygen source, and a mixture of ethanol and ethylene glycol with a volume ratio of 60/40 as the solvent. The final product was characterized by powder X-ray diffraction (XRD), scanning electron microscopy (SEM), (high resolution) transmission electron microscopy (TEM/HRTEM), selected area electron diffraction (SAED) and energy dispersive X-ray spectrometry (EDS). Experiments showed that under the assistance of some additives such as H2O2, H2C2O4 and KOH, the as-obtained octahedral ZnFe2O4 nanocrystals could be used as monocomponent visible-light photocatalysts for the degradation of the organic dye methylene blue. The degradation mechanisms in various systems were discussed.
1. Introduction
Over the past decade, metal ferrite nanocrystals with normal and inverse spinel structures have attracted increased research interest owing to their remarkable magnetic, catalytic, electrical and optical properties and wide applications in magnetic and electronic devices, drug delivery and as photomagnetic materials.1–4 Among various metal ferrites, ZnFe2O4 with the normal spinel structure has been paid much attention due to its chemical stability and sensitivity to visible light, and potential applications in diverse areas such as permanent magnets, recording media, ferrofluids, gas sensors, and anode materials for Li-ion batteries.5–9 In particular, ZnFe2O4 is a narrow bandgap (1.86 eV) n-type semiconductor,5 and can effectively absorb visible light up to a wavelength of 653 nm. Thereby, ZnFe2O4 nanocrystals can be used as potential visible-light photocatalysts for the degradation of organic pollutants.10 However, photo-generated electron–hole pairs of ZnFe2O4 can rapidly recombine with each other, which is unfavorable for the application of ZnFe2O4 as the monocomponent photocatalyst.11 To conquer the above shortcoming, at present, small amounts of H2O2 are introduced to promote the photocatalytic property of ZnFe2O4;12 or polycomponent photocatalysts containing ZnFe2O4 are designed in practical applications.13–15
To obtain ZnFe2O4 nanocrystals, many methods have been developed, including hydrothermal synthesis,7,9 microemulsion,16 polyol route,17 micelle solution,18 sonochemical approach,19 sol–gel technique,20 coprecipitation,21 microwave irradiation,22 and so on. Among the above most methods, however, the alkali is often needed besides Zn2+ and Fe3+ ion sources. Can ZnFe2O4 nanocrystals be prepared without the presence of an alkali? In 2012, Sun et al. reported a solution combustion route to prepare sponge-like ZnFe2O4 under the ignition temperature of 573 K.23 Different from the above high-temperature combustion route, our group developed a mild solution-combusting method to successfully prepare some binary and ternary metal oxide nanocrystals in recent years, including ZnO, Fe2O3, TiO2 and NiTiO3.24–27 Metal sources were firstly dissolved into the mixed solvent of ethanol and ethylene glycol. Then, the goal products were obtained by combusting the above alcohol solution in air. Since combustion reactions are ignited at room temperature and only produce metal oxide, CO2 and water, the above solution-combusting method is mild and friendly-environment.
In the present paper, we employed the above solution-combusting method to realize the synthesis of octahedral ZnFe2O4 nanocrystals. Zn(CH3COO)2 and FeCl3 were used as the reactants, and O2 in air as the oxygen source. Compared with the reported methods to prepare ZnFe2O4, the present route is simple, safe, quick and environment-friend. It was found that the as-prepared octahedral ZnFe2O4 nanocrystals could be used as a monocomponent visible-light photocatalyst for the degradation of organic dye methylene blue under the assistances of different additives. Simultaneously, the photocatalytic mechanisms of the as-obtained octahedral ZnFe2O4 nanocrystals under the presences of various additives were discussed.
2. Experimental section
All reagents and chemicals are analytically pure, bought from Shanghai Chemical Company and used without further purification.
2.1 Synthesis of octahedral ZnFe2O4 nanocrystals
In a typical experimental procedure, 5 mmol Zn(CH3COO)2·2H2O and 10 mmol FeCl3 were dissolved in 100 mL mixed solvents of ethanol and ethylene glycol (EG) with the volume ratio of 60/40 under the magnetic stirring. After the solution was transferred into a spirit lamp with an absorbent cotton lampwick, the spirit lamp was fired. After a while, some reddish-brown products were produced on the top of the lampwick. The products were collected, washed several times with distilled water and dried at 60 °C in air.
2.2 Characterization
XRD patterns were obtained using a Shimadzu XRD-6000 X-ray diffractometer equipped with Cu Kα radiation (λ = 0.154060 nm). SEM images and energy dispersive spectrometry (EDS) were obtained on Hitachi S-4800 field emission scanning electron microscope, employing the accelerating voltage of 5 kV and 15 kV (for EDS). TEM images and selected area electron diffraction (SAED) of the product were carried out on JEOL-2010 high resolution transmission microscope employing an accelerating voltage of 200 kV.
2.3 Photocatalytic properties
To investigate the photocatalytic activity of the as-produced octahedral ZnFe2O4 nanocrystals under the visible light, organic dye, methylene blue (MB), was used as the pollute mode. In a typical experimental process, 15 mg of ZnFe2O4 nanocrystals were firstly dispersed in 40 mL MB solution with an initial concentration of 10 mg L−1 under the ultrasonic assistance. Then, the mixed system was stirred in the dark for another 30 min to ensure a sorption–desorption equilibrium. After different additives were separately introduced, the mixed system was irradiated under high-pressure Xenon lamp equipped with a UV filter for various durations. The concentration changes of MB solution were measured with Shimadzu UV-2450 UV-Vis-NIR spectrophotometer.
3. Results and discussion
3.1 Structure and morphology characterization
Fig. 1a shows a typical XRD pattern of the product prepared by the present solution-combusting method. All the diffraction peaks can be indexed as the cubic-phase ZnFe2O4 by comparison with JCPDS card files no. 22-1012 (see the bottom in Fig. 1a). The strong and narrow diffraction peaks indicate the high crystallinity of the product. No obvious impurity peak is found, implying that the product is pure. The formation of ZnFe2O4 was confirmed by the EDS analysis of the product. As shown in Fig. 1b, the strong Zn, Fe and O peaks can be readily found. According to the calculation of the peak areas, the Zn/Fe/O atomic ratio is 1/2.1/4.3, which is close to the stoichiometry of ZnFe2O4.
 |
| | Fig. 1 (a) The XRD pattern and (b) EDS analysis of the final product prepared by the present solution-combusting route. | |
The morphology of the product was characterized by SEM and TEM technologies. Fig. 2a shows a representative SEM image of the product, from which one can find that the product comprises of smooth octahedral nanocrystals with the sizes of 100–300 nm. Fig. 2b depicts a TEM image of the as-prepared ZnFe2O4 nanocrystals. A rhombus plate is shown, which is attributed to the vertical projection of an octahedron by the incident electron beams. A HRTEM image of the edge section of the octahedron is shown in Fig. 2c. The clear stripes indicate the good crystallinity of octahedral nanocrystals. The distance between the neighboring planes is measured to be 0.21 nm, corresponding to the (400) plane of cubic ZnFe2O4. The SAED pattern of the nanocrystals is shown in the inset of Fig. 2d. The clear diffraction dots further confirm the good crystallinity of the product.
 |
| | Fig. 2 (a) Typical SEM image, (b) TEM image, (c) HRTEM image and (d) SAED pattern of the octahedral ZnFe2O4 nanocrystals prepared by the present solution-combusting route. | |
According to previous reports24–27 and the present experimental results, a possible formation process of ZnFe2O4 nanocrystals can be suggested. In this simple combustion process, alcohols combusted to produce CO2 and H2O and to free abundant heat; then, Zn2+ and Fe3+ ions dissolved in alcohols reacted with O2 in air under the assistance of heat to form ZnFe2O4. The related chemical reactions can be described as follows:
| | |
C2H5OH + 3O2 → 2CO2 + 3H2O + Q
| (1) |
| | |
C2H6O2 + 2.5O2 → 2CO2 + 3H2O + Q
| (2) |
| | |
Zn2+ + 2Fe3+ + 2O2 + Q → ZnFe2O4
| (3) |
Generally, the matter with the fcc structure easily forms octahedral shape.8a,28 In the present work, it was difficult to monitor the growth process of octahedral nanocrystals. Nevertheless, it was possible that during the formation of ZnFe2O4 the 〈100〉 direction owned the faster growth rate than the 〈111〉 one due to the lowest energy of the {111} surfaces. Thus, octahedral ZnFe2O4 nanocrystals were finally obtained.8a
3.2 Visible light photocatalytic performances of the as-prepared ZnFe2O4 nanocrystals under the assistances of various additives
In the current work, the as-obtained octahedral ZnFe2O4 nanocrystals could be used as the monocomponent photocatalysts for the degradation of the organic pollutant in the presences of some additives including H2O2, H2C2O4 and KOH, respectively. Methylene blue dye was selected as the pollution mode due to its stability under the visible light. Fig. 3a depicts the absorption spectra of MB solution irradiated by the visible light for various durations in the presence of 15 mg ZnFe2O4. Although MB could be degraded with the prolonging of the irradiation time, the degradation efficiency was rather low. Only ∼16.5% and 56.4% of MB was degraded after irradiating for 60 min and 120 min, respectively. However, after 1 mL H2O2 was added into the above system, the degradation efficiency of MB reached nearly 100% within the irradiation time of 60 min (see Fig. 3b). The color changes of MB solutions are separately displayed in the insets of Fig. 3a and b. Fig. 3c compares the degradation efficiencies of MB solutions within 60 min in the presences of 15 mg ZnFe2O4, 1 mL H2O2, and 15 mg ZnFe2O4 + 1 mL H2O2, respectively. Obviously, the introduction of 1 mL H2O2 significantly promotes the photocatalytic activity of ZnFe2O4. According to previous reports,12,29 the photocatalytic mechanism of ZnFe2O4 in the system containing H2O2 can be described below: the electron–hole pairs are firstly generated on the surface of ZnFe2O4 nanocrystals under the irradiation of visible light, and the photogenerated electrons rapidly react with Fe3+ to form Fe2+. Then, Fe2+ and Fe3+ can react with H2O2 to form ˙OH and ˙OOH radicals.12,30 The above oxidative state change of Fe element was also proved by the cyclic voltammetry of ZnFe2O4 (see Fig. 4). Due to the strong oxidative ability of ˙OH radicals, organic dyes are rapidly degraded. Simultaneously, H2O2 is regenerated owing to the reaction between ˙OOH radicals.31 The related reactions are exhibited as follows:| | |
ZnFe2O4 + hν → ZnFe2O4 (hole) + e−
| (4) |
| | |
Fe2+ + H2O2 → Fe3+ + ˙OH + OH−
| (6) |
| | |
Fe3+ + H2O2 → Fe2+ + ˙OOH + H+
| (7) |
| | |
˙OOH + ˙OOH → H2O2 + O2
| (8) |
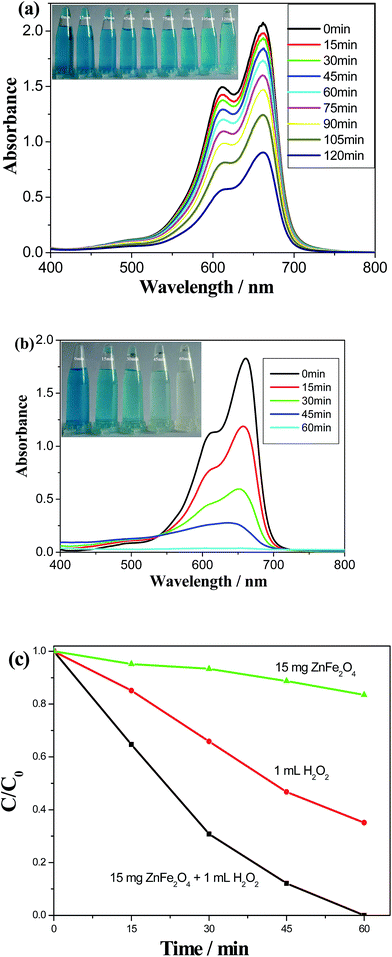 |
| | Fig. 3 The absorption spectra of MB solutions after irradiated by the visible light for various durations under the presence of different catalysts: (a) 15 mg ZnFe2O4 and (b) 15 mg ZnFe2O4 + 1 mL H2O2. (c) The correlation between the concentration of MB solution and the irradiation time under the presences of various catalysts. | |
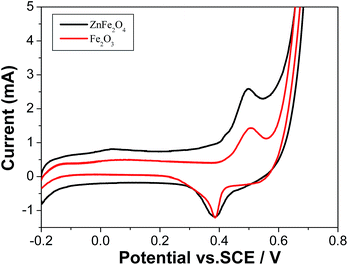 |
| | Fig. 4 CVs of ZnFe2O4 and Fe2O3 (as control) prepared under the same experimental conditions at the scan rate of 50 mV s−1. ZnFe2O4 exhibited the same redox potentials with Fe2O3. | |
It was found that proper amounts of H2C2O4 could also markedly promote the photocatalytic activity of octahedral ZnFe2O4 nanocrystals. The investigation discovered that the pH of the system containing MB and ZnFe2O4 nanocrystals was ∼7.0. After the pH of the system was adjusted to 3.0 by introducing 1.25 mol L−1 of H2C2O4, MB dye could be rapidly degraded under the same irradiation conditions (see Fig. 5a). Within 45 min, the color of MB solution changed from blue to colorless (see the inset in Fig. 5a). Compared with the system introducing 1 mL H2O2 (Fig. 3b), H2C2O4 owns the stronger ability to enhance the photocatalytic activity of octahedral ZnFe2O4 nanocrystals than H2O2. Further studies showed that the photocatalytic activity of ZnFe2O4 nanocrystals increased with the pH decrease of the system (see Fig. 5b). Namely, increasing the amount of H2C2O4 was favorable for the degradation of the pollutant in the present system.
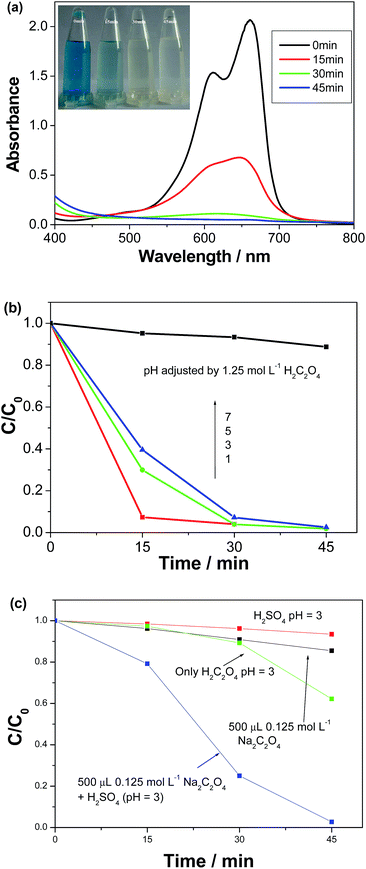 |
| | Fig. 5 (a) The absorption spectra of MB solutions irradiated by the visible light for various durations in the presences of 15 mg ZnFe2O4 and a proper amount of H2C2O4 (the system pH = 3.0). The inset is the according micrographs of MB solutions; (b) the photocatalytic efficiencies of the catalyst at different pH values adjusted by 1.25 mol L−1 H2C2O4; (c) the mechanism investigations of H2C2O4 promoting the photocatalytic efficiencies of the catalyst. | |
Usually, H2C2O4 is a weak acid and can partly decompose into H+, HC2O4−, and C2O42− ions in aqueous solution. To ascertain the role of H2C2O4, control experiments were designed. As seen from Fig. 5c, when the system pH was adjusted to 3.0 by adding diluted H2SO4 instead of H2C2O4, MB solution hardly faded under the same irradiation conditions. Similar experimental phenomena were also found when other inorganic and organic acids were employed instead of H2C2O4, including HCl, HCOOH and CH3COOH, implying that H+ ions could not promote the photocatalytic activity of octahedral ZnFe2O4 nanocrystals alone. When 500 μL 0.125 mol L−1 Na2C2O4 solution was added into the system, merely ∼14.6% of MB solution was degraded after irradiating for 45 min, which is far lower than equimolar H2C2O4 (see Fig. 5a). Distinctly, the photocatalytic activity of octahedral ZnFe2O4 nanocrystals cannot be promoted alone by H+ or C2O42− ions. The superior photocatalytic activity of ZnFe2O4 nanocrystals after introducing H2C2O4 should be attributed to the synergistic effect between H+ and C2O42− ions. To further confirm the above conclusion, H2SO4 was added into the system containing 500 μL 0.125 mol L−1 Na2C2O4 and adjusted the system pH 3.0. The photocatalytic efficiency of the catalyst was markedly enhanced. After irradiating for 45 min, the photocatalytic efficiency was very close to that of equimolar H2C2O4 (see Fig. 5c). Furthermore, experiments also uncovered that H2C2O4 could slightly promote the degradation of MB in the absence of the photocatalyst. As seen from Fig. 5c, ∼40% of MB was degraded when only H2C2O4 was used within 45 min.
Based on the above experimental facts, a possible photocatalytic mechanism was suggested. Under the irradiation of visible light, C2O42− ions could be oxidized by Fe3+ ions on the surface of ZnFe2O4 nanocrystals to form C2O4˙− radicals (see eqn (9)),32 which rapidly reacted with O2 dissolved in the system to convert into O2˙− radicals (eqn (10)). Due to the presence of a great amount of H+ ions in the system, O2˙− radicals could be reduced by Fe2+ ions to H2O2 (eqn (11)); Also, O2˙− radicals could be converted into ˙HO2 radicals (eqn (12)). Thereafter, reactions 6–8 occurred. Thus, the pollutant is successfully degraded.
| | |
Fe3+ + C2O42− + hν → Fe2+ + C2O4˙−
| (9) |
| | |
C2O4˙− + O2 → O2˙− + CO2
| (10) |
| | |
Fe2+ + O2˙− + 2H+ → Fe3+ + H2O2
| (11) |
Interestingly, the photocatalytic efficiency of octahedral ZnFe2O4 nanocrystals could also be promoted by an alkali. Fig. 6a depicts the absorption spectra of MB solution irradiated by the visible light for various durations in the presences of 15 mg ZnFe2O4 and 1.786 mol L−1 300 μL KOH. After irradiating for 60 min, ∼95% of MB was degraded. Distinctly, the introduction of KOH enhanced the photocatalytic activity of ZnFe2O4 nanocrystals compared with the absorption spectra of MB solution shown in Fig. 3a. According to eqn (4), the electron–hole pairs are firstly generated on the surface of ZnFe2O4 nanocrystals under the irradiation of the visible light. The produced holes could react with OH− ions to form ˙OH radicals with high oxidative capacity (see eqn (13)). Thus, organic dye was degraded under the illumination of the visible light.33
 |
| | Fig. 6 (a) Absorption spectra of MB solution irradiated by the visible light for various durations in the presences of 15 mg ZnFe2O4 and 1.786 mol L−1 300 μL KOH; (b) effect of the amount of KOH on photocatalytic degradation of MB. | |
Fig. 6b shows the degradation curves of MB solution under the presences of various KOH amounts. One could find that the photocatalytic efficiency of the catalyst was markedly enhanced with the increase of KOH amount from 0 to 300 μL; and thereafter, the photocatalytic efficiency hardly changed. The above facts indicated that 300 μL 1.786 mol L−1 KOH should be the optimum amount in the present experiment. This is understandable. Since the amount of the catalyst is kept unchanged, the number of the photogenerated holes should be constant under the same visible light conditions. According to eqn (13), the number of ˙OH radicals is determined by the number of the hole. Namely, excess OH− ions cannot raise the number of ˙OH radicals. As a result, the photocatalytic efficiency hardly changed when the volume of KOH increased from 300 to 500 μL. Moreover, experiments also found that MB was slightly degraded when only 300 μL 1.786 mol L−1 KOH was used in the system without ZnFe2O4. Namely, ZnFe2O4 was necessary in the KOH system for the degradation of MB dye.
The above experimental facts clearly showed that the as-prepared ZnFe2O4 could be used as a monocomponent visible-light photocatalyst for the degradation of organic pollutant MB in the presences of some additives, including H2O2, H2C2O4 and KOH. The corresponding catalytic efficiencies within 45 min were in turn ∼88% (for 1 mL H2O2), ∼98% (for pH 3.0 adjusted by 1.25 mol L−1 H2C2O4) and ∼85% (for 300 μL 1.786 mol L−1 KOH) under the same irradiation conditions. Among three additives, H2C2O4 showed the highest catalytic activity. This was possibly attributed to two reasons. Firstly, according to reactions 6–8, Fe2+ and Fe3+ ions could react with hydroxyl groups (H2O2 can be written as HO–OH) to form ˙OH and ˙OOH radicals. As an organic carboxylic acid, H2C2O4 contains two hydroxyl groups. Similarly to H2O2, thus, oxidative radicals can be formed, too. More importantly, C2O42− ion is a typical bidentate ligand, which can react with Fe3+ ion on the surface of ZnFe2O4 nanocrystals to form Fe(C2O4)n3−2n complex ion.34 Under the illumination of the light, ˙C2O4− radicals can be easily produced owing to the below reaction:34
| 2Fe(C2O4)n(3−2n) + hν → 2Fe2+ + (2n − 1) C2O42− + C2O4˙− |
Subsequently, the photodegradation reactions were initiated. Therefore, the photocatalytic performance was promoted.
4. Conclusions
In summary, octahedral ZnFe2O4 nanocrystals have been successfully prepared by a simple solution-combusting method, employing Zn(CH3COO)2, FeCl3 and O2 in air as the starting reactants without the assistance of any surfactant or template. Due to the combustion of mixed solvent of ethanol and ethylene glycol in air, abundant heat was freed, which promoted the reactions among Zn, Fe and O species. Thus, the final ZnFe2O4 nanocrystals were obtained. Experiments showed that the as-prepared product could be used as a monocomponent visible-light photocatalyst for the degradation of organic pollutant MB in the presences of some additives, such as H2O2, H2C2O4 and KOH. The corresponding catalytic efficiencies within 45 min were in turn ∼88% (for 1 mL H2O2), ∼98% (for pH 3.0 adjusted by 1.25 mol L−1 H2C2O4) and ∼85% (for 300 μL 1.786 mol L−1 KOH) under the same irradiation conditions. The present work is a powerful exploration for the application of ZnFe2O4 materials as a monocomponent visible-light photocatalyst in environmental treatment and protection. Also, it is of significant theoretically for other ternary semiconductor oxides in the application of environmental treatment as photocatalysts.
Acknowledgements
The authors thank the National Natural Science Foundation of China (21171005) and Key Foundation of Chinese Ministry of Education (210098) for the fund support.
References
- J. Haetge, C. Suchomski and T. Brezesinski, Inorg. Chem., 2010, 49, 11619 CrossRef CAS PubMed.
- T. J. Yoon, J. S. Kim, B. G. Kim, K. N. Yu, M. H. Cho and J. K. Lee, Angew. Chem., Int. Ed., 2005, 44, 1068 CrossRef CAS PubMed.
- K. Giri, K. Pellerin, W. Pongsaksawad, M. Sorescu and S. A. Majetich, IEEE Trans. Magn., 2000, 1, 3026 CrossRef.
- S. Bullita, A. Casu, M. F. Casula, G. Concas, F. Congiu, A. Corrias, A. Falqui, D. Loche and C. Marras, Phys. Chem. Chem. Phys., 2014, 16, 4843 RSC.
- K. J. McDonald and K. S. Choi, Chem. Mater., 2011, 23, 4863 CrossRef CAS.
- G. D. Prasanna, H. S. Jayanna and V. Prasad, J. Appl. Polym. Sci., 2011, 120, 2856 CrossRef CAS.
-
(a) M. Y. Wang, L. Sun, J. H. Cai, P. Huang, Y. F. Su and C. J. Lin, J. Mater. Chem., 2013, 1, 12082 RSC;
(b) Y. Y. Sun, W. W. Wang, L. Zhang, S. M. Sun and E. P. Gao, Mater. Lett., 2013, 98, 1 CrossRef PubMed.
-
(a) Z. Xing, Z. C. Ju, J. Yang, H. Y. Xu and Y. T. Qian, Nano Res., 2012, 5, 477 CrossRef CAS;
(b) J. Xie, W. T. Song, G. S. Cao, T. J. Zhu, X. B. Zhao and S. C. Zhang, RSC Adv., 2014, 4, 7703 RSC.
- R. Rameshbabu, R. Ramesh, S. Kanagesan, A. Karthigeyan and S. Ponnusamy, J. Mater. Sci.: Mater. Electron., 2013, 24, 4279 CrossRef CAS PubMed.
- H. A. Mohammed, A. Hamza, I. K. Adamu, A. Ejila, S. M. Wziri and S. I. Mustapha, J. Chem. Eng. Mater. Sci., 2013, 4, 80 CrossRef CAS.
- G. X. Tong, F. F. Du, W. H. Wu, R. N. Wu, F. T. Liu and Y. Liang, J. Mater. Chem. B, 2013, 1, 2647 RSC.
- M. H. Su, C. He, V. K. Sharma, M. A. Asi, D. H. Xia, X. Z. Li, H. Q. Deng and Y. Xiong, J. Hazard. Mater., 2012, 211, 95 CrossRef PubMed.
- L. Kong, Z. Jiang, T. C. Xiao, L. F. Lu, M. O. Jones and P. P. Edwards, Chem. Commun., 2011, 47, 5512 RSC.
-
(a) B. P. Zhang, J. L. Zhang and F. Chen, Res. Chem. Intermed., 2008, 34, 375 CrossRef CAS PubMed;
(b) S. H. Xu, D. L. Feng and W. F. Shangguan, J. Phys. Chem. C, 2009, 113, 2463 CrossRef CAS.
- Y. S. Fu and X. Wang, Ind. Eng. Chem. Res., 2011, 50, 7210 CrossRef CAS.
- H. L. Zhu, X. Y. Gu, D. T. Zuo, Z. K. Wang, N. Y. Wang and K. H. Yao, Nanotechnology, 2008, 19, 405 Search PubMed.
- S. Ammar, N. Jouini, F. Fiévet, Z. Beji, L. Smiri, P. Moliné, M. Danot and J. M. Grenèche, J. Phys.: Condens. Matter, 2006, 18, 9055 CrossRef CAS.
- J. F. Hochepied, P. Bonville and M. P. Pileni, J. Phys. Chem. B, 2000, 104, 905 CrossRef CAS.
- E. K. Nyutu, W. C. Conner, S. M. Auerbach, C. H. Chen and S. L. Suib, J. Phys. Chem. C, 2008, 112, 1407 CAS.
- M. H. Habibi and A. H. Habibi, J. Therm. Anal. Calorim., 2013, 113, 843 CrossRef CAS PubMed.
- E. C. Mendonça, C. B. R. Jesus, W. S. D Folly, C. T. Meneses and J. G. S. Duque, J. Supercond. Novel Magn., 2013, 26, 2329 CrossRef PubMed.
- R. Sai, S. D. Kulkarni, K. J. Vinoy, N. Bhat and S. A. Shivashankar, J. Mater. Chem., 2012, 22, 2149 RSC.
- S. Sun, X. Y. Yang, Y. Zhang, F. Zhang, J. J. Ding, J. Bao and C. Gao, Prog. Nat. Sci., 2012, 22, 639 CrossRef PubMed.
-
(a) Y. H. Ni, X. F. Cao, G. G. Wu, G. Z. Hu, Z. S. Yang and X. W. Wei, Nanotechnology, 2007, 18, 155603 CrossRef;
(b) T. T. Wu, Y. H. Ni, X. Ma and J. M. Hong, Mater. Res. Bull., 2013, 48, 4754 CrossRef CAS PubMed.
- X. Wang, L. Zhang, Y. H. Ni, J. M. Hong and X. F. Cao, J. Phys. Chem. C, 2009, 113, 7003 CAS.
- Y. H. Ni, Y. Zhu and X. Ma, Dalton Trans., 2011, 40, 3689 RSC.
- Y. H. Ni, X. H. Wang and J. M. Hong, Mater. Res. Bull., 2009, 44, 1797 CrossRef CAS PubMed.
-
(a) Y. Cheng, Y. Zheng, Y. Wang, F. Bao and Y. Qin, J. Solid State Chem., 2005, 178, 2394 CrossRef CAS PubMed;
(b) Y. Sun, W. Wang, L. Zhang, S. Sun and E. Gao, Mater. Lett., 2013, 98, 124 CrossRef CAS PubMed.
-
(a) H. J. Lv, L. Ma, P. Zeng, D. N. Ke and T. Y. Peng, J. Mater. Chem., 2010, 20, 3665 RSC;
(b) G. Fan, Z. Gu, L. Yang and F. Li, Chem. Eng. J., 2009, 155, 534 CrossRef CAS PubMed.
- W. Luo, L. Zhu, N. Wang, H. Q. Tang, M. J. Cao and Y. B. She, Environ. Sci. Technol., 2010, 44, 1786–1791 CrossRef CAS PubMed.
- Y. J. Cui, Z. X. Ding, P. Liu, M. Antonietti, X. Z. Fu and X. C. Wang, Phys. Chem. Chem. Phys., 2012, 14, 1455 RSC.
-
(a) Y. H. Huang, Y. J. Shih and C. H. Liu, J. Hazard. Mater., 2011, 188, 188 CrossRef CAS PubMed;
(b) L. R. Feng, F. T. Hu, C. B. Liu, F. Chen, N. Xu, S. Q. Liu and Z. G. Chen, Chin. J. Catal., 2012, 33, 1417 CAS.
- N. Daneshvar, D. Salari and A. R. Khataee, J. Photochem. Photobiol., A, 2004, 162, 317 CrossRef CAS.
- J. Jeong and J. Yoon, Water Res., 2005, 39, 2893 CrossRef CAS PubMed.
|
| This journal is © The Royal Society of Chemistry 2014 |
Click here to see how this site uses Cookies. View our privacy policy here. 





