Synthesis of fully alternating polycarbonate with low Tg from carbon dioxide and bio-based fatty acid†
Ying-Ying Zhang,
Xing-Hong Zhang*,
Ren-Jian Wei,
Bin-Yang Du,
Zhi-Qiang Fan and
Guo-Rong Qi
MOE Key Laboratory of Macromolecular Synthesis and Functionalization, Department of Polymer Science and Engineering, Zhejiang University, Hangzhou, 310027, China. E-mail: xhzhang@zju.edu.cn; Fax: +86-571 87953732; Tel: +86-571 87953732
First published on 31st July 2014
Abstract
The selective copolymerization of CO2 and an epoxide to form fully alternating polycarbonates is a great challenge via catalysis with the zinc–cobalt(III) double metal cyanide complex [Zn–Co(III) DMCC]. We describe the first perfectly alternating copolymerization of CO2 with a bio-based epoxide. The resultant polycarbonate had a low Tgs of −38 to −44 °C and two end hydroxyl groups, which were then used to initiate ring-opening polymerization of L-lactide via metal-free catalysis, affording a biodegradable triblock copolymer. This study provides a new platform copolymer for making various advanced polymers with biodegradable properties.
Introduction
In recent years, there has been an increasing demand for the sustainable use of renewable natural resources as raw chemical materials due to the depletion of the oil reserves and environmental problems.1 Bio-based plastics, produced from renewable feedstocks, such as biomass, could decrease our dependency on petroleum and help efforts to curb global warming. Furthermore, using renewable raw materials and taking full advantage of the synthetic potential of nature can meet the requirements of green and sustainable chemistry well.2 Plant oils are considered as the most important renewable raw materials for the production of bio-based polymers because of their renewability, worldwide availability, relatively low price, and wide possible applications. Castor oil, a non-edible oil extracted from the seeds of the castor bean plant is, as with many other plant oils, abundantly available and potentially capable of being transformed into various value-added monomers.3 For instance, 10-undecenoic acid can be obtained by heating ricinoleic acid under vacuum,4 or efficiently produced by hydrolysis of methyl undecenoate5 at 558–638 °C with yields of 44.6–45.7%. Therefore, 10-undecenoic acid could potentially be used as a valuable precursor for making chemicals and materials, such as antitumor compounds, antibiotics,6 Nylon 11,7 and polyurethane (PU).8 A complete review of the possible applications of undecylenic acid has been published by Van der Steen and Stevens.4On the other hand, as a renewable natural C1 feedstock, carbon dioxide (CO2) is a promising low-cost nontoxic resource for chemical synthesis, especially for polymer synthesis.9 Recently, much research has focused on the use of CO2 as a C1 building block for polymers. In particular, polycarbonates represent a promising class of materials. Since the landmark discovery of the ZnEt2/H2O system for CO2/propylene oxide (PO) copolymerization by Inoue et al. in 1969,10 much progress has been made in developing various epoxides and catalyst systems for CO2 copolymerization with epoxides.11 However, nearly all studies have focused on the copolymerization of petrochemically derived epoxides with CO2. Only a few papers have reported the copolymerization of bio-renewable epoxides. For example, Coates and co-workers reported the alternating copolymerization of limonene oxide and CO2 using β-diiminate zinc complexes, affording a polycarbonate with a high glass transition temperature (Tg) of 113.9 °C.12 To date, many kinds of fully alternating CO2–epoxide copolymers were reported with relatively high values of Tg, such as the poly(indene carbonate)s with a Tg of 138 °C, formed by the coupling of indene oxides with CO2 using bifunctional cobalt(III) catalysts reported by Darensbourg et al.13 Lu and coworkers recently reported the selective synthesis of perfectly alternating CO2 copolymers with a high Tg (140 °C) from 4,4-dimethyl-3,5,8-trioxabicyclo[5.1.0]octane through desymmetrization copolymerization mediated by the enantiopure dinuclear Co(III)-complex.14 The other direction for CO2-based copolymers is the synthesis of more “soft” or functional copolymers, such as polycarbonate diols with low molecular weights and low Tg.15 Yamada and co-workers described the alternating polymerization of propylene oxide, terminated epoxides with long side chains, and CO2 using cobalt(III) complex catalysts.15f The Tg of the obtained polymers could be tuned by varying the feeding ratios of the epoxides as well as the length of the long side group.15f These CO2-based copolymers could potentially be used as the precursors for making rubber or elastomers. In this case, it is necessary to develop a low Tg polycarbonate with at least two end –OH groups, which are helpful for subsequent reactions. To the best of our knowledge, the lowest Tg for a polycarbonate is −22 °C, reported by Coates and co-workers, namely poly(l,2-hex-5-ene carbonate),16 which was obtained from the copolymerization of CO2 with a petrochemically-derived epoxide catalyzed by β-diiminate zinc complexes.
In this work, we describe the synthesis of a new polycarbonate derived from C1 feedstock and a bio-renewable monomer, epoxy methyl 10-undecenoate (EMU), by using a zinc–cobalt(III) double metal cyanide complex [Zn–Co(III) DMCC] catalyst (Scheme 1), and its application for synthesizing C1- and bio-based triblock copolymers. The resultant polycarbonate was fully alternating (carbonate linkage content was estimated to be >99%) with two end –OH groups and a low Tg of −44 °C.
 | ||
| Scheme 1 Synthesis of bio-based epoxide from 10-undecenoic acid and alternating copolymerization of epoxy methyl 10-undecenoate and CO2. | ||
Experimental section
Materials
10-undecenoic acid (95%), m-chloroperbenzoic acid (mCPBA), sulphuric acid (98%), anhydrous sodium sulfate, sodium chloride, methanol, and dichloromethane (DCM) were purchased from Aladdin and used without further purification. L-Lactide was recrystallized from ethanol and dried under vacuum at 40 °C overnight. DBU (1,8-diazabicyclo[5.4.0]undec-7-ene) was purchased from Aladdin and distilled under reduced pressure over CaH2. THF was distilled from sodium and stored under nitrogen. CO2 (99.995%) was used as received.Characterization
1H and 13C NMR spectra of the products were performed on a Bruker Advance DMX 500 MHz spectrometer. Chemical shift values were referenced to TMS as internal standard at 0.0 ppm for 1H NMR (500 MHz) and against CDCl3 at 77.0 ppm for 13C NMR (100 MHz). The molecular weights and polydispersity index (PDI) of the polycarbonates were determined by using a PL-GPC220 chromatograph (Polymer Laboratories) equipped with an HP 1100 pump (Agilent Technologies). The GPC columns were eluted with THF at 1.0 mL min−1 at 40 °C. The sample concentration was approximately 0.3 wt% and the injection volume was 50 μL. Calibration was performed using monodisperse polystyrene standards covering the molecular-weight range from 500 to 500![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 Da. Infrared spectra were obtained by using a Bruker Vector 22 FT-IR spectrophotometer. Differential scanning calorimetric (DSC) tests were conducted on a TAQ200 instrument (New Castle, DE) with a heating rate of 10 °C min−1 from −80 to 220 °C under an N2 atmosphere. Tg and Tm were determined from the second run. Thermogravimetric analysis was carried out on a Perkin-Elmer Pyris 1 instrument at a heating rate of 10 °C min−1 from room temperature to 500 °C under an N2 atmosphere. Samples for thermal analyses were all purified. ESI-MS analyses were performed by using an Esquire3000 plus mass spectrometer with methylene chloride–methanol as a solvent for dissolving the copolymer. The water content in EMU was determined using a MaxTitra20Q moisture meter (Shanghai Tianmei Scientific Instruments Co., Ltd. China) based on a colorimetric method.
000 Da. Infrared spectra were obtained by using a Bruker Vector 22 FT-IR spectrophotometer. Differential scanning calorimetric (DSC) tests were conducted on a TAQ200 instrument (New Castle, DE) with a heating rate of 10 °C min−1 from −80 to 220 °C under an N2 atmosphere. Tg and Tm were determined from the second run. Thermogravimetric analysis was carried out on a Perkin-Elmer Pyris 1 instrument at a heating rate of 10 °C min−1 from room temperature to 500 °C under an N2 atmosphere. Samples for thermal analyses were all purified. ESI-MS analyses were performed by using an Esquire3000 plus mass spectrometer with methylene chloride–methanol as a solvent for dissolving the copolymer. The water content in EMU was determined using a MaxTitra20Q moisture meter (Shanghai Tianmei Scientific Instruments Co., Ltd. China) based on a colorimetric method.
Synthesis of methyl 10-undecenoate
A 250 mL three-necked round-bottomed flask equipped with a water cooled condenser was charged with 10-undecenoic acid (60 mL), an excess of methanol (110 mL). Concentrated sulphuric acid (15 mL) was added dropwise with a dropping funnel over 30 min. The reaction mixture was heated at 65 °C for 16 h under magnetic stirring. The mixture was then washed with 10% sodium bicarbonate solution followed by brine until the solution was neutral. The upper layer liquid was collected and dried over anhydrous magnesium sulfate, and filtered to obtain methyl 10-undecenoate. Yield: 95%; FT-IR: 1640 cm−1 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1742 cm−1 (COOCH3), 3077 cm−1 (C
C), 1742 cm−1 (COOCH3), 3077 cm−1 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–H); 1H NMR (CDCl3, 500 MHz, δ ppm): 1.40–1.17 (m, 10H), 1.67–1.52 (m, 2H), 2.05–1.93 (q, 2H), 2.33–2.21 (t, 2H), 3.63 (s, 3H), 5.01–4.84 (m, 2H), 5.85–5.68 (m, 1H); 13C NMR (CDCl3, 100 MHz, δ ppm): 174.2 (1C), 139.1 (1C), 114.2 (1C), 51.4 (1C), 34.1 (1C), 33.9 (1C), 29.4–29.0 (5C), 25.0 ppm (1C).
C–H); 1H NMR (CDCl3, 500 MHz, δ ppm): 1.40–1.17 (m, 10H), 1.67–1.52 (m, 2H), 2.05–1.93 (q, 2H), 2.33–2.21 (t, 2H), 3.63 (s, 3H), 5.01–4.84 (m, 2H), 5.85–5.68 (m, 1H); 13C NMR (CDCl3, 100 MHz, δ ppm): 174.2 (1C), 139.1 (1C), 114.2 (1C), 51.4 (1C), 34.1 (1C), 33.9 (1C), 29.4–29.0 (5C), 25.0 ppm (1C).
Epoxidation of methyl 10-undecenoate
A 500 mL bottomed flask was charged with methyl 10-undecenoate (30 mL, 0.13 mol). A DCM solution of mCPBA (250 mL, 0.16 mol mCPBA) was added in small portions at 0 °C. The reaction mixture was then stirred at room temperature for 12 h. After the reaction, 10% (w/v) of saturated aqueous sodium sulfite was added followed by saturated aqueous sodium hydrogen carbonate and brine. The organic phase was dried over anhydrous magnesium sulfate and evaporated under reduced pressure to obtain EMU. Yield: 93%; FT-IR: 1741 cm−1 (COOCH3), 837 cm−1 (epoxy); 1H NMR (CDCl3, 500 MHz, δ ppm): 1.34–1.22 (m, 10H), 1.52–1.46 (m, 2H), 1.66–1.53 (m, 2H), 2.31–2.23 (t, 2H), 2.91–2.83 (m, 1H), 2.76–2.69 (m, 1H), 2.90–2.83 (m, 1H), 3.64 (s, 3H); 13C NMR (CDCl3, 100 MHz, δ ppm): 174.1 (1C), 52.3 (1C), 51.4 (1C), 47.0 (1C), 34.0 (1C), 32.5 (1C), 29.4–29.1 (4C), 26.0 (1C), 25.0 ppm (1C).Preparation of the nanolamellar Zn–Co(III) DMCC catalyst
The catalyst was synthesized according to our reported method.20 Elemental analysis of the catalyst: Co: 12.48; Zn: 27.29; N: 16.57; C: 23.34; H: 2.27; Cl: 9.50.Representative copolymerization of CO2 and epoxy methyl 10-undecenoate
A 10 mL autoclave with a small magnetic stirrer was dried at 120 °C for 3 h, and cooled to room temperature in a closed desiccator. The Zn–Co(III) DMCC catalyst (16.0 mg) and EMU (2.0 mL) were transferred into the autoclave. The autoclave was sealed and filled with CO2, then heated to the target temperature in a pre-heated oil bath. The system was adjusted to the set pressure and stirred for the set time. After the copolymerization, the autoclave was cooled with an ice-water bath and the pressure was slowly vented. A small amount of crude product was collected for 1H NMR spectroscopy. The remaining products were dissolved with small amounts of DCM and precipitated by excess methanol, dried at 60 °C under vacuum to a constant weight. The obtained copolymer was colorless and viscous.FT-IR: 3450 cm−1 (OH), 1741 cm−1 (COOCH3); 1H NMR (CDCl3, 500 MHz, δ ppm): 1.43–1.16 (m, 10H), 1.70–1.49 (m, 4H), 2.33–2.21 (t, 2H), 3.64 (s, 3H), 4.42–3.95 (m, 2H), 4.86 (s, 1H); 13C NMR (CDCl3, 100 MHz, δ ppm): 174.3 (1C), 154.7 (1C), 75.8 (1C), 68.2 (1C), 51.5 (1C), 34.1 (1C), 30.5 (1C), 29.2 (4C), 25.0 ppm (2C).
Representative procedure for the synthesis of triblock copolymers from L-lactide
Polycarbonate (Mn = 4400 g mol−1, 0.1645 g) and L-lactide (5.586 mmol, 0.805 g) were placed in a 25 mL Schlenk flask under a nitrogen atmosphere and dissolved in THF (3 mL). DBU (45 μL) in DCM (2 mL) was added to prepare the triblock polymer, and the mixture was stirred for 3 h. Benzoic acid was added to quench the reaction. The crude polymer was precipitated from methanol three times, and the white polymer was dried under vacuum. 1H NMR (CDCl3, 500 MHz, δ ppm): 1.26 (s), 1.55 (m), 2.27 (t), 3.64 (s, 3H), 4.42–3.95 (m), 4.86 (s), 5.13 (q); 13C NMR (CDCl3, 100 MHz, δ ppm): 174.3 (1C), 169.5 (1C), 154.7 (1C), 75.8 (1C), 68.2 (1C), 68.9 (1C), 51.5 (1C), 34.1 (1C), 30.5 (1C), 29.2 (4C), 25.0 (2C), 16.6 (3C).Results and discussion
Zn–Co(III) DMCC is a typical catalyst that has been developed as a highly efficient catalyst for CO2–epoxide copolymerization.17 In Zn–Co(III) DMCC, the zinc and cobalt atoms are associated with cyanide bridges, affording a three-dimensional backbone. The empirical formula of the catalyst prepared at below 40 °C in the presence of tert-butanol (t-BuOH) is Zn3[Co(CN)6]2·xZnCl2·yt-BuOH·zH2O (x, y, and z vary based on the preparation conditions).17f,18 We have previously reported a nanolamellar Zn–Co(III) DMCC catalyst synthesized at 75 °C. The Zn/Co molar ratio of this catalyst was 2.0 based on the elemental analysis.17b The catalytic center of the catalyst was confirmed to be a Zn–OH bond on the surface of the catalyst, as shown in Fig. S1.†19 For Zn–Co(III) DMCC-catalyzed CO2–epoxide copolymerization, it is still a big challenge to obtain a fully alternating copolymer. The best reported alternation of the styrene oxide–CO2 copolymer is 99.4% from Zn–Co(III) DMCC, in our recent study.20 This kind of Zn–Co(III) DMCC catalyst exhibited nanolamellar structures with thicknesses of 20–80 nm and high surface areas of ca. 600 m2 g−1, which could provide more active sites than the conventional catalyst.20 Herein, we used this nanolamellar Zn–Co(III) DMCC catalyst directly for the copolymerization of CO2 with EMU. A polycarbonate with a perfectly alternating structure was expected because of the long bulky side groups in EMU (Scheme 1).The bio-based epoxide, EMU, was synthesized from 10-undecenoic acid by two methods21 that are often used in the laboratory, as shown in Scheme 1. Firstly, the esterification of 10-undecenoic acid with methanol was carried out using sulphuric acid as the catalyst.21a Then, the methyl 10-undecenoate was oxidized by mCPBA at 25 °C for 12 h using DCM as the solvent. The FT-IR (Fig. S2†) and 1H NMR spectra (Fig. S3†) proved the successful synthesis of EMU, with a yield of 93%.21b Note that this epoxy monomer could be synthesized by sustainable methods, for example, direct pyrolysis of methyl ricinoleate could produce methyl 10-undecenoate4,5 and an environmentally friendly oxidant, such as hydrogen peroxide, could be used for the epoxidation.22
A series of EMU–CO2 copolymerizations were successfully catalyzed by nanolamellar Zn–Co(III) DMCC. The experimental conditions and the results are summarized in Table 1. No products were collected when the reaction temperature was 30 °C (entry 1, Table 1) for 24 h. Increasing the temperature from 40 to 100 °C (entries 2–8) resulted in an increase in the production of cyclic carbonate from 4.8 to 20.0 wt% and a decrease in the Mn of the resultant copolymer from 18.6 to 7.1 kg mol−1. For obtaining copolymers with relative low Mn, water was used as the chain transfer agent in the copolymerization system.15b As shown in entries 9–11, when the water content in the monomer was 400 ppm, the Mns of the copolymer was decreased to 3.7–4.4 kg mol−1 without reducing the monomer's conversion at 50 °C. In this case, increasing the CO2 pressure from 3.0 to 5.0 MPa (entries 9–11) resulted in a clear increase in the catalyst productivity and a slight increase in the weight percentage of the cyclic carbonate in the total product (Wcc) from 1.9 to 5.7 wt%. Except from entries 1 and 2 in Table 1, EMU in entries 3–11 was completely converted to the product within 12 h, according to the 1H NMR spectra of the crude copolymers (Fig. S5–S13†). The optimized reaction temperature and CO2 pressure were 50 °C and 3.0 MPa, respectively, because a small amount of the cyclic product was produced, as shown in entries 3, 9–11.
| Entry | Temp. (°C) | Press (MPa) | EMUb (conv.%) | FCO2c (%) | Wccc (wt%) | Mnd (kg mol−1) | PDI | Tge (°C) | |
|---|---|---|---|---|---|---|---|---|---|
| a Reactions were performed in a 10 mL dry autoclave for 12 h, with EMU (2.0 mL), and the Zn–Co(III) DMCC catalyst (16.0 mg). Water contents of entries 1–8 and entries 9–11 were 120 and 400 ppm, respectively.b EMU was converted to both poly(carbonate-co-ether) and cyclic carbonate, and EMU was not observed in the 1H NMR spectra of entries 3–11 (Fig. S5–S13†).c FCO2 (%) indicates the molar fraction of carbonate linkages in the polymer product. FCO2 (%) = A4.9/[A4.9 + (A3.4–3.8 − A3.6)/3]; Wcc (wt%) indicates the weight percentage of cyclic carbonate in the total crude product, determined by using 1H NMR spectra. Wcc (wt%) = 258A4.5/[258A4.5 + 258A4.9 + 214 (A3.4–3.8 − A3.6)/3] (Fig. S4–S13†).d Determined by gel permeation chromatography in THF at 40 °C, calibrated with standard monodispersed polystyrene.e Data obtained from the second DSC scan (10 °C min−1, N2). | |||||||||
| 1 | 30 | 5 | ND | ND | ND | ND | ND | ND | |
| 2 | 40 | 5 | 78 | >99 | 4.8 | 18.6 | 2.1 | −40 | |
| 3 | 50 | 5 | 100 | >99 | 7.4 | 17.7 | 2.4 | −40 | |
| 4 | 60 | 5 | 100 | >99 | 14.5 | 16.6 | 2.6 | −38 | |
| 5 | 70 | 5 | 100 | >99 | 15.3 | 11.8 | 2.1 | −39 | |
| 6 | 80 | 5 | 100 | >99 | 16.7 | 9.9 | 2.8 | −40 | |
| 7 | 90 | 5 | 100 | 92 | 18.0 | 8.5 | 2.8 | −42 | |
| 8 | 100 | 5 | 100 | 90 | 20.0 | 7.1 | 2.9 | −44 | |
| 9 | 50 | 3 | 100 | >99 | 1.9 | 3.7 | 2.2 | −41 | |
| 10 | 50 | 4 | 100 | >99 | 3.9 | 4.2 | 2.3 | −41 | |
| 11 | 50 | 5 | 100 | >99 | 5.7 | 4.4 | 2.3 | −41 | |
The formation of the ether units is usually thermodynamically favourable during Zn–Co(III) DMCC catalysis;15b however, in this case, the alternating degree (FCO2) of all the resultant copolymers obtained at 40–80 °C were estimated as >99% because no ether unit signals were observed in their 1H NMR spectra (e.g., Fig. 1 and S14–S18†). To our knowledge, this is the first example of an alternating CO2–epoxide copolymer with an FCO2 of >99% obtained via Zn–Co(III) DMCC catalysis. The bulky side group led to full alternating copolymerization of EMU with CO2 via heterogeneous Zn–Co(III) DMCC catalysis. However, the steric effect of the bulky groups in EMU on the FCO2 of the resultant copolymer was weakened when the reaction temperature was elevated to 90 and 100 °C (entries 7 and 8, Table 1). This is in close agreement with the typical catalytic behaviour of Zn–Co(III) DMCC. Moreover, the resultant copolymer had a regio-irregular structure, as the 13C NMR spectra of entry 5 in Table 1 shows (Fig. S24†), which means that the attack of the propagating species at the CH and CH2 sites had nearly equal probability.
 | ||
| Fig. 1 Selected 1H NMR spectra of (a) the crude product and (b) the purified copolymer shown in entry 3 in Table 1 (CDCl3, 500 MHz). | ||
The EMU–CO2 copolymer obtained with Zn–Co(III) DMCC has two end hydroxyl (–OH) groups, which was confirmed by the electrospray ionization-tandem mass spectrometry results in positive mode for the low Mn copolymer [Mn = 2000 and PDI = 2.4 (GPC result), FCO2 = 95.2%, Fig. S25†], which was obtained at 90 °C for 5 h (Fig. S26†). All the captured species in the range of m/z 600–2000 contained two –OH groups. One end –OH group came from the initiation Zn–OH bond, and the other was produced by the chain transfer reaction of the propagating chain with H2O (or other proton-bearing species).19 This was consistent with the results of the copolymerization of other aliphatic epoxides with CO2 catalyzed by the same Zn–Co(III) DMCC catalyst.15b,19
These EMU–CO2 copolymers exhibited low values of Tg of −38 to −44 °C (Fig. 2). This could be attributed to the internal plasticization effect or the micro-Brownian motion15f of the long alkyl side chain with the ester groups. To the best of our knowledge, this is the first example of a CO2-based polycarbonate with such a low Tg. Moreover, the Tg of this copolymer is close to that of typical commercial bio-compatible polyols, such as PEO (Tg of about −54 °C)23 that are widely used as precursors for functional polymers. At the same time, the EMU–CO2 copolymers also exhibited a high initial thermal decomposition temperature (Td, 5%) of 240 °C (Fig. S27†), which is clearly higher than that of PEO. Therefore, the EMU–CO2 copolymer could be potentially used as a platform for making various advanced polymers.
 | ||
| Fig. 2 The DSC curves of the CO2–EMU resultants at various temperatures (curves 1–7 represent entries 2–8 in Table 1). | ||
It was demonstrated that the EMU–CO2 copolymer could be used as a macroinitiator to initiate ring-opening polymerization (ROP) of L-lactide via a metal-free catalytic route, as shown in Scheme 2. Note that L-lactide is a common bio-based monomer. DBU (1, 8-diazabicyclo [5.4.0] undec-7-ene) was used as the catalyst for L-lactide ROP at a mild temperature of 25 °C. The weight percentage of the EMU–CO2 copolymer in this block polymer was 20.1% based on the L-lactide conversion. GPC traces of the resultant triblock copolymers and EMU–CO2 copolymer (Fig. 3) showed a clear increase in the Mn, whereas the control experiment showed that L-lactide ROP did not occur without using the EMU–CO2 copolymer initiator under the same reaction conditions. The structure of the triblock copolymer was well characterized by the 1H and 13C NMR spectra (Fig. S28 and S29†). The thermal properties of the resulting triblock copolymers were determined by differential scanning calorimetry (DSC) at a heating rate of 20 °C min−1 (Fig. S30†). Two values of Tg at −35 and 45 °C were observed, which could be attributed to the polycarbonate block and the poly-L-lactide (PLLA) block, respectively. A cold crystallizing peak at 108 °C and two melting peaks at 131 and 142 °C were also observed. The cold crystallizing peak indicated the confined or incomplete crystallization of the sample, whereas the double melting peaks showed the existence of dual (or multiple) crystal structures24 in this sample. This was because the polycarbonate macroinitiator presented a relative large polydispersity (PDI) of 2.30, which meant that the triblock copolymers had different length ratios for the poly(EMU–CO2) block and PLLA block. Still, the CO2 and bio-based triblock copolymer, PLLA–PC–PLLA, has many thermal transitions and potential applications in functional materials.
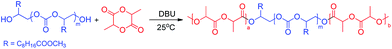 | ||
| Scheme 2 Synthesis of CO2- and bio-based triblock copolymers from ROP of L-lactide initiated by the EMU–CO2 copolymer. | ||
 | ||
| Fig. 3 GPC curves of the EMU–CO2 copolymer macroinitiator (Table 1, entry 11), and the resultant triblock copolymer, PLLA-PC-PLLA. | ||
Conclusions
In summary, a biodegradable full alternating polycarbonate and its triblock copolymer were synthesized from CO2 and renewable platform chemicals. The fully CO2-based polycarbonates with a low Tg and two end hydroxyl groups are good candidates for making thermoplastic elastomers that are not from petroleum resources.Acknowledgements
The authors are grateful for financial support by the National Science Foundation of the People's Republic of China (no. 21274123 and 21074106), the Fundamental Research Funds for the Central Universities, and Zhejiang University K. P. Chao's High Technology Development Foundation.Notes and references
- C. K. Williams and M. A. Hillmyer, Polym. Rev., 2008, 48, 1–10 CrossRef CAS.
- M. Eissen, J. O. Metzger, E. Schmidt and U. Schneidewind, Angew. Chem., Int. Ed., 2002, 41, 414–436 CrossRef CAS.
- H. Mutlu and M. A. Meier, Eur. J. Lipid Sci. Technol., 2010, 112, 10–30 CrossRef CAS PubMed.
- M. Van der Steen and C. V. Stevens, ChemSusChem, 2009, 2, 692–713 CrossRef CAS PubMed.
- H. Guobin, L. Zuyu, Y. Suling and Y. Rufeng, J. Am. Oil Chem. Soc., 1996, 73, 1109–1112 CrossRef CAS.
- V. Rahman, S. Mukhtar, W. H. Ansari and G. Lemiere, Eur. J. Med. Chem., 2005, 40, 173–184 CrossRef CAS PubMed.
- R. Aelion, Ind. Eng. Chem. Res., 1961, 53, 826–828 CAS.
- A. S. More, T. Lebarbé, L. Maisonneuve, B. Gadenne, C. Alfos and H. Cramail, Eur. Polym. J., 2013, 49, 823–833 CrossRef CAS PubMed.
- K. Yao and C. Tang, Macromolecules, 2013, 46, 1689–1712 CrossRef CAS.
- S. Inoue, H. Koinuma and T. Tsuruta, J. Polym. Sci., Part B: Polym. Phys., 1969, 7, 287–292 CAS.
- (a) G. W. Coates and D. R. Moore, Angew. Chem., Int. Ed., 2004, 43, 6618–6639 CrossRef CAS PubMed; (b) X.-B. Lu, W.-M. Ren and G.-P. Wu, Acc. Chem. Res., 2012, 45, 1721–1735 CrossRef CAS PubMed; (c) D. J. Darensbourg and S. J. Wilson, Green Chem., 2012, 14, 2665–2671 RSC; (d) M. R. Kember, F. Jutz, A. Buchard, A. J. White and C. K. Williams, Chem. Sci., 2012, 3, 1245–1255 RSC; (e) M. W. Lehenmeier, S. Kissling, P. T. Altenbuchner, C. Bruckmeier, P. Deglmann, A. K. Brym and B. Rieger, Angew. Chem., Int. Ed., 2013, 52, 9821–9826 CrossRef CAS PubMed.
- C. M. Byrne, S. D. Allen, E. B. Lobkovsky and G. W. Coates, J. Am. Chem. Soc., 2004, 126, 11404–11405 CrossRef CAS PubMed.
- D. J. Darensbourg and S. J. Wilson, Macromolecules, 2013, 46, 5929–5934 CrossRef CAS.
- Y. Liu, M. Wang, W.-M. Ren, K.-K. He, Y.-C. Xu, J. Liu and X.-B. Lu, Macromolecules, 2014, 47, 1269–1276 CrossRef CAS.
- (a) Y. Gao, L. Gu, Y. Qin, X. Wang and F. Wang, J. Polym. Sci., Part A: Polym. Chem., 2012, 50, 5177–5184 CrossRef CAS PubMed; (b) X.-H. Zhang, R.-J. Wei, X.-K. Sun, J.-F. Zhang, B.-Y. Du, Z.-Q. Fan and G.-R. Qi, Polymer, 2011, 52, 5494–5502 CrossRef CAS PubMed; (c) J. Geschwind and H. Frey, Macromolecules, 2013, 46, 3280–3287 CrossRef CAS; (d) X. Wu, H. Zhao, B. Nörnberg, P. Theato and G. A. Luinstra, Macromolecules, 2014, 47, 492–497 CrossRef CAS; (e) L. Gu, Y. Gao, Y. Qin, X. Chen, X. Wang and F. Wang, J. Polym. Sci., Part A: Polym. Chem., 2013, 51, 282–289 CrossRef CAS PubMed; (f) A. Okada, S. Kikuchi and T. Yamada, Chem. Lett., 2011, 40, 209–211 CrossRef CAS; (g) S. D. Thorat, P. J. Phillips, V. Semenov and A. Gakh, J. Appl. Polym. Sci., 2003, 89, 1163–1176 CrossRef CAS PubMed.
- S. D. Allen, C. M. Byrne and G. W. Coates, Feedstocks for the Future, American Chemical Society, Washington, DC, 2006, vol. 921, pp. 116–128 Search PubMed.
- (a) I. Kim, M. J. Yi, K. J. Lee, D.-W. Park, B. U. Kim and C.-S. Ha, Catal. Today, 2006, 111, 292–296 CrossRef CAS PubMed; (b) X. K. Sun, X. H. Zhang, F. Liu, S. Chen, B. Y. Du, Q. Wang, Z. Q. Fan and G. R. Qi, J. Polym. Sci., Part A: Polym. Chem., 2008, 46, 3128–3139 CrossRef CAS PubMed; (c) R.-J. Wei, X.-H. Zhang, Y.-Y. Zhang, B.-Y. Du, Z.-Q. Fan and G.-R. Qi, RSC Adv., 2014, 4, 3188–3194 RSC; (d) R.-J. Wei, X.-H. Zhang, B.-Y. Du, Z.-Q. Fan and G.-R. Qi, Polymer, 2013, 54, 6357–6362 CrossRef CAS PubMed; (e) X. K. Sun, S. Chen, X. H. Zhang and G.-R. Qi, Prog. Chem., 2012, 9, 1776–1784 Search PubMed; (f) X.-H. Zhang, Z.-J. Hua, S. Chen, F. Liu, X.-K. Sun and G.-R. Qi, Appl. Catal., 2007, 325, 91–98 CrossRef CAS PubMed.
- S. Chen, Z. Hua, Z. Fang and G. Qi, Polymer, 2004, 45, 6519–6524 CrossRef CAS PubMed.
- X. K. Sun, X. H. Zhang, R. J. Wei, B. Y. Du, Q. Wang, Z. Q. Fan and G. R. Qi, J. Polym. Sci., Part A: Polym. Chem., 2012, 50, 2924–2934 CrossRef CAS PubMed.
- R.-J. Wei, X.-H. Zhang, B.-Y. Du, X.-K. Sun, Z.-Q. Fan and G.-R. Qi, Macromolecules, 2013, 46, 3693–3697 CrossRef CAS.
- (a) M. J. Haas, S. Bloomer and K. Scott, J. Am. Oil Chem. Soc., 2000, 77, 373–379 CrossRef CAS PubMed; (b) T. Vijai Kumar Reddy, B. Prabhavathi Devi, R. Prasad, P. Sujitha and C. Ganesh Kumar, Eur. J. Med. Chem., 2013, 67, 384–389 CrossRef CAS PubMed.
- G. Yadav and D. Satoskar, J. Am. Oil Chem. Soc., 1997, 74, 397–407 CrossRef CAS.
- A. Nijenhuis, E. Colstee, D. Grijpma and A. Pennings, Polymer, 1996, 37, 5849–5857 CrossRef CAS.
- (a) P. Pan, W. Kai, B. Zhu, T. Dong and Y. Inoue, Macromolecules, 2007, 40, 6898–6905 CrossRef CAS; (b) X. Ling and J. E. Spruiell, J. Polym. Sci., Part B: Polym. Phys., 2006, 44, 3200–3214 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra06157h |
| This journal is © The Royal Society of Chemistry 2014 |
