Silica/CdTe/silica fluorescent composite nanoparticles via electrostatic assembly as a pH ratiometer†
Zhipeng Ran and
Wuli Yang *
*
State Key Laboratory of Molecular Engineering of Polymers and Department of Macromolecular Science, Fudan University, Shanghai 200433, China. E-mail: wlyang@fudan.edu.cn; Fax: +86 21-6564 0293; Tel: +86 21-6564 2385
First published on 13th August 2014
Abstract
In this report, we present a ratiometric pH nanoprobe based on stable fluorescent colloidal silica composite nanoparticles encapsulating hydrophilic CdTe quantum dots. Quantum dots, owing to their fascinating optical properties, have been widely used for sensors and bioimaging. To avoid the inherent chemical instability and serious photoluminescence quenching of quantum dots, a facile electrostatic assembly method is developed to prepare sandwich-like silica/CdTe quantum dots/silica composite nanoparticles stabilized by mercaptopropyl trimethoxysilane (SQMS). This approach is of high efficiency, e.g., 98.9% of quantum dots are instantly adsorbed on the surface of silica nanospheres, and nearly 80% of the original fluorescence of the quantum dots is retained for SQMS while traditional silica coating processes caused dramatic quenching. Finally, the bright SQMS with remarkable stability is modified with pH-sensitive fluorescein isothiocyanate to fabricate a high-resolution pH ratiometric nanoprobe. Above all, SQMS shows a uniform sandwich-like structure, narrow size distribution, and stable fluorescence in strongly acidic and highly salted solutions, and the remarkable stability favours quantitative analyses and nanoprobes.
1. Introduction
II–VI Semiconductor nanocrystals (or quantum dots, QDs), on account of quantum confinement effects, exhibit unique optical properties such as particle size-dependent fluorescence, broad excitation range, high quantum efficiency, and simultaneous excitation by a single light source for all colours. QDs have attracted great interest in various fields, such as nanosensors1–6 and theranostics.7,8 However, the II–VI semiconductor nanocrystals suffer from inherent poor photoluminescence (PL) stability in harsh chemical environments, such as solutions containing plenty of metal ions or hydrions.9 The intrinsically unstable PL of QDs is a deadly shortcoming for their application of sensors, even though lots of ratiometric sensors have been fabricated by combining QDs with another fluorophore.4,5 Since the fluorescence intensity is not specifically sensitive to a selected analyte, fluorescence fluctuation induced by multiple factors always leads to confused results in quantitative analyses. In addition, cytotoxicity caused by the release of heavy metal ions precludes the practical applications of QDs.9 Thus, many efforts have been dedicated to exploring suitable coating techniques to resolve the fluorescence instability and toxicity, e.g., incorporating the nanocrystals into polymer spheres10,11 and silica nanoparticles.12,13Silica nanoparticles are one of the biocompatible, inert and water dispersible materials, and a number of silica-coating procedures of QDs have undergone intensive investigations.14–25 In addition to being expected to dispel the cytotoxicity as well as chemical and colloidal instability, the silica coatings ulteriorly enable surface functionalization with diverse groups. Nevertheless, the classical approaches to coat QDs with silica shell, i.e., Stöber method and reverse microemulsion approach, not only induce a dramatic decrease in QD fluorescence, but also cannot retain QD PL from harsh chemical environments.26,27 To the best of our knowledge, only 57% of the initial fluorescence of CdTe QDs is retained for core/shell type of CdTe/SiO2 nanoparticles that are prepared by reverse microemulsion method in a successful case.28 Afterwards, a series of researches have focused on the silica coating methodology, such as electrostatic assembly between silica microspheres and CdSe/CdS QDs,19 and in situ growth of CdTe QDs on mercapto silica nanoparticles followed by further coating of another silica layer.29,30 Although silica-coated QDs with higher relative quantum yield (QY) are acquired from these recent researches, few reports are simultaneously devoted to overcoming the fluorescence instability in harsh environments. For the significance of applications on nanosensors, it is thus highly desirable to give an in-depth and detailed illumination to preparation of silica coated QDs composites with uniform morphology, bright fluorescence, remarkable chemical stability and low cytotoxicity.
Here, we present an efficient preparation of sandwich-like silica/CdTe quantum dots/silica composite nanoparticles (SQMS) by a handy electrostatic assembly between 3-mercaptopropionic acid (MPA)-capped CdTe QDs and amino-functionalized SiO2 nanoparticles. The colloidal composite nearly retains 80% of initial fluorescence of CdTe QDs, and its fluorescence possesses remarkable stability in saline solution with high concentration or in buffer solution with pH values from 2 to 8. We find that the mercapto ligand exchange on the surface of QDs plays a key role in the chemical stability of fluorescence. Finally, the SQMS with remarkable stability is employed as an internal reference fluorophore and further modified with pH sensitive fluorescein isothiocyanate (FITC) to construct a pH nanoprobe (SQMSF). The QDs and FITC in SQMSF are able to be excited synchronously without dependence on fluorescence resonance energy transfer (FRET), resulting in high-resolution for the pH ratiometric sensor.
2. Experimental section
2.1 Chemicals
Tellurium powder (98%) was acquired from Aldrich. CdCl2 (99%), NaBH4 (99%), and 3-mercaptopropionic acid (MPA) (99%+) were purchased from ACROS. Mercaptopropyl trimethoxysilane (MPTS), tetraethyl orthosilicate (TEOS), and 3-aminopropyltriethoxysilane (APS) were obtained from Shanghai Chemical Reagents Company. Fluorescein isothiocyanate (FITC) was purchased from Beijing Huafeng United Technology Company. Ammonium hydroxide (25–28%) was received from Taican Zhitang Chemical Co., Ltd.2.2 Synthesis of CdTe QDs stabilized by MPA
The MPA-capped CdTe QDs were prepared according to a modified hydrothermal approach reported previously.31 Briefly, 100 mg of sodium borohydride (NaBH4) was added in a flask containing 20 mL of distilled water. The flask was degassed under continuous nitrogen flow at 4 °C. Half an hour later, 127.6 mg of tellurium powder was added quickly with vigorous stirring at nitrogen atmosphere. After tellurium powder completely dissolved, purple fresh solution of sodium hydrogen telluride (NaHTe) was produced and set aside under nitrogen gas. Meanwhile, 20 mL of the solution with 20 mmol L−1 of CdCl2 and 36 mmol L−1 of MPA was prepared. 2 mol L−1 of NaOH was continuously added to the solution until the pH hit 9.0. And the solution was transferred to another flask and degassed under nitrogen for 0.5 hours. 1.0 mL of as-synthesized NaHTe solution was rapidly injected into the flask with protection of nitrogen. Then, the precursor mixture was sealed into a Teflon-lined stainless steel autoclave and heated to 185 °C. After a certain time, autoclave was cooled to room temperature in cold water bath. The as-prepared CdTe QDs were precipitated by alcohol and separated by centrifugation. The time of thermal treatment would lead to different size of QDs. QD670 (top right number is the maximum emission wavelength, nm) was acquired by adjusting the reaction time to 60 min.2.3 Synthesis of amino-functionalized silica nanoparticles (SiO2–NH2)
The monodispersed SiO2–NH2 was fabricated by a modified Stöber method.32 In detail, 15 mL ammonium hydroxide and 250 mL ethanol were mixed into a flask at 40 °C with mechanical stirring (200 rpm). 0.5 hours later, 2.5 mL of TEOS was added. And the mixture was stirred overnight continuously at 40 °C. Another 1.25 mL TEOS and 0.1 mL APS were added into flask successively with an interval of one hour. After 12 hours, the product mixture was centrifuged to collect the silica nanoparticles. The SiO2–NH2 was further washed with water and ethanol several times to remove the unreacted chemicals.2.4 Synthesis of SQ, SQS and SQMS
Electrostatic self-assembly approach was explored to prepare SQMS with strong fluorescence.19 In detail, 0.5 mg of QDs was dispersed in 10 mL of deionized water (DI water). 30 mg of SiO2–NH2 and 0.5 mL of phosphate buffer solution (PBS, pH = 5.5) were added to the QDs suspension successively. Several minutes later, the QDs would be electrostatically adsorbed on the surface of SiO2–NH2 to form silica/CdTe quantum dots (SQ) and the SQ was separated by centrifugation and washed with water and ethanol for further use. And the supernate was collected to determine amount of unassembled QDs. The Stöber method was utilized again to coat the SQ with another outer layer of silica. In a typical procedure, the SQ was dispersed in 10 mL of ethanol with 0.5 mL of ammonium hydroxide. After 2 μL MPTS and 50 μL TEOS were added in succession, the mixture was vigorously stirred at room temperature for 6 hours. The silica/CdTe quantum dots/silica composite nanoparticles (SQMS) were centrifuged from suspension and purified by washing with water and ethanol. Compared with SQMS, SQS were also prepared at the absence of MPTS.2.5 Synthesis of SQMSF
The mixture of 6 μL APS and 2 mg FITC was dissolved in 2 mL ethanol, and stirred for 12 hours to form FITC–APS conjugation. Here, the excess APS was used and the FITC–APS conjugation was not isolated and directly used in the next step. The as-prepared SQMS was redispersed in a flask containing 10 mL ethanol and 0.5 mL ammonium hydroxide. For modification of FITC, 40 μL FITC–APS was added in the flask simultaneously and stirred for 4 hours continuously. The products were separated and purified by centrifugation and marked as SQMSF.2.6 Characterization
Hydrodynamic size distribution and zeta potential of particles were measured by Malvern Zetasizer Nano. Fluorescent spectra were collected by FLS920 at 25 °C. The UV-vis absorption was measured on Lambda 35. JEM-2100F field emission transmission electron microscope (FETEM) was used to analyse the size and morphology.3. Result and discussion
3.1 Synthesis of SQMS
Indeed, silica coatings of QDs have been studied extensively.33–35 Thereinto, a large proportion of the researches are based on Stöber method and reverse microemulsion approach. Both methods, however, suffer from a dramatic decrease in the fluorescence. Regardless of the mechanism of PL quenching, the compromise on fluorescence hampers further applications of these colloidal nanoparticles. Moreover, the silica shell seems deficient to overcome the fluorescent instability of QDs.16,20,36Here, to avoid the huge drop of QD PL during encapsulating QDs within silica shells, a facile electrostatic assembly approach is developed to prepare sandwich-like silica nanoparticles with a middle layer of QDs.19 As briefly outlined in Scheme 1, first we prepare monodispersed silica nanoparticles with amino groups on the surface. The existence of amino group is verified by zeta-potential. The zeta potential of SiO2–NH2 is higher than SiO2, implying that SiO2–NH2 is successfully amino-functionalized (Table 1). The electrostatic interactions between particles greatly depend on their zeta potentials. As presented in Table 1, we monitor zeta-potential of samples in following steps. QDs are dispersed in DI water in a beaker, and a suspension of SiO2–NH2 nanoparticles is dropped into the QD dispersion. The pH value of the mixture is adjusted to 5.5 with PBS, inducing zeta potential of SiO2–NH2 dramatically changing to +15 mV with a reversal from negative to positive charge, while zeta potential of QDs drops to −35 mv on the sharp contrary. The QDs are assembled on the surface of SiO2–NH2 completely and immediately to form SQ by strong binding interaction between the surface amino groups of SiO2–NH2 and QDs.37,38 Owing to the anchor of QDs on SiO2–NH2, the zeta potential of SQ drops to −17 mv at pH 5.5. To increase their fluorescence and colloidal stability, another silica layer is then coated onto the surface of SQ via Stöber method. The further decrease of zeta potential of SQMS indicates that the SQ is completely coated by the outer silica layer.
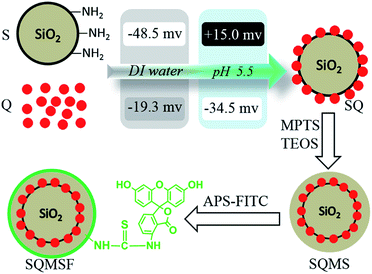 | ||
| Scheme 1 Electrostatic assembly of amino-functionalized silica nanoparticles (SiO2–NH2) and MPA-capped CdTe quantum dots, and preparation of pH ratiometer. | ||
| Samples | Diameter/nm | PDI | Zeta potential/mV (pH = 5.5) |
|---|---|---|---|
| SiO2 | 81 | 0.13 | −21 |
| SiO2–NH2 | 91 | 0.11 | 15 |
| QD | — | — | −35 |
| SQ | 101 | 0.08 | −17 |
| SQMS | 144 | 0.07 | −30 |
Fig. 1 shows the morphology and structure for QDs, SiO2–NH2, SQ and SQMS observed in detail by high-resolution TEM. Lower magnification images with more particles and size information are presented in ESI (Fig. S1†). The QD crystals with the maximum emission peak of 670 nm are about 5 nm in diameter. SiO2–NH2 shows uniform spheres with flat surface and average size of 65 ± 5 nm in diameter. Fig. 1c shows clearly that some black dots on the surface of SiO2–NH2 for SQ and no obvious aggregation of QDs is observed. The average size of SQMS becomes slightly larger and increases to 71 ± 7 nm, another silica layer about 3 nm is coated on SQ. The hydrodynamic diameter of samples is also investigated by dynamic light scattering (DLS) to estimate size distribution as shown in Fig. 2. The hydrodynamic size is 101 nm for SQ, slightly larger than 91 nm of SiO2–NH2, and increases to 144 nm for SQMS after coated with the outer silica layer. All of SiO2–NH2, SQ and SQMS nanospheres present narrow size distribution with a polydispersity index of 0.11, 0.08 and 0.07 respectively.
Then fluorescent methodology is employed to calculate the loading efficiency of QDs. After addition of PBS (pH 5.5) to aqueous dispersion of QDs and SiO2–NH2, QDs are immediately adsorbed on the surface of SiO2–NH2. Considering QDs are very stable in aqueous dispersion and not able to be precipitated by centrifugation as results of smaller size and stabilization of MPA, we centrifuge the mixture to separate SQ and collect the supernate that contains unloaded QDs (Fig. 3a). Under irradiation of an ultraviolet lamp, the QDs dispersion and SQ precipitate show bright red fluorescence while nearly no emission from supernate is observed by naked eye. The result manifests almost all of QDs are loaded onto SQ. And a dramatic increase of absorption for SQ also indicates the incorporation of QDs (Fig. S2†). Further quantitative analysis by fluorescent spectra indicates that about 98.9% of QDs are loaded to SQ and the assembly process has little effect on QY of QDs (Fig. 3b). These findings strongly demonstrate that the current electrostatic assembly is more rapid to fabricate composite silica nanoparticles encapsulating QDs with uniform sandwich-like structure and high loading efficiency.
 | ||
| Fig. 3 (a) Water dispersion of QD and SQ excited under a 365 nm ultraviolet (UV) lamp. (b) Quantitative analysis by fluorescent spectra. | ||
3.2 Fluorescence and stability of SQMS
Stability of QDs fluorescence is of particular importance, and the goal of the silica coating is to improve the chemical stability of QDs and retain fairly considerable fluorescence at the same time. QDs should be at least stable between pH 4 and pH 8 when used for biology, since pH values in human body fall in this range.27 For broader applications, especially as fluorescent probes, the chemical instability of QDs remains a problem. The PL stability of QDs is surveyed in buffer solution and NaCl solution (Fig. S3–S5†).20 As Fig. 4b and c show, PL of pure QDs is completely quenched at pH 4.0 or 200 mM NaCl solution. Even in NaCl solution at concentration of 150 mM corresponding with ionic strength in human body, the fluorescence of QDs is seriously quenched and only about 10% of initial fluorescence is retained. These conspicuous fluorescence instabilities under different environments seriously hamper QD applications in quantitative imaging. In the most successful solution, stable QDs have been acquired by combining the silica and amphiphilic polymer encapsulation techniques, but huge drop of fluorescence still exists.27In this work, we present a simple silica coating approach with ligand exchange. After encapsulated in silica, nearly 80% of the original fluorescence is preserved for SQMS, and aqueous dispersion of SQMS shows bright red fluorescence, almost commensurate with pure QDs (Fig 4a). The considerable fluorescence retention can be attributed to pre-anchor of QDs on the surface of SiO2–NH2 before the following silica coating via Stöber method. And the ligand exchange from MPA to MPTS induces blue shift of about 5 nm for maximum emission as described elsewhere (Fig. 4a).29
The ligand exchange is in favor of the stability of SQMS. After coated with silica shell, the PL of SQS and SQMS becomes inert for NaCl at concentration below 200 mM, indicating the silica shell plays a key role in PL stability. However, H+ still slightly quenches the PL of SQS. On one hand the carboxyl group of MPA ligand on QDs has significant effect on pH sensitivity of PL.9 On the other hand due to pore-structure and hydrophilic essence of silica nanoparticles prepared by Stöber method or reverse microemulsion method, silica coating is not enough to protect QDs from permeation of hydrogen ions.20,26 So MPTS is employed to stabilize the SQ in coating process. SQMS shows remarkable stability to H+ at pH values between 2 to 8, even no fluorescence quenching appears in buffer solution at pH 4 within 16 hours (Fig. 4d), and indicating the ligand exchange between MPTS and MPA facilitates the improvement of PL stability. The bright fluorescence and remarkable stability of SQMS are of great benefit to biology use, such as imaging and biosensors.
3.3 pH ratiometric sensor
Finally, we show that the SQMS with stable fluorescence can be used to configurate a pH probe.39 Since the fluorescence of QDs are often sensitive to H+,9 diverse metal ions,1 proteins,33 etc., they have been widely used to probe pH values in biomedical researches through the change of their fluorescence emission intensity.40 However, the intensity of excitation light, absolute concentration of QDs, and loss due to photobleaching also have significant effect on fluorescence intensity, resulting in that the single QDs sensors are of little use. Therefore numerous recent works have built QD-based ratiometric sensors by combining QDs with another fluorophore.5,41 These internal reference/sensitivity dual emissive pairs rather than single QDs generate fluorescence intensity ratios irrespective of factors aforementioned. And it will be better if the reference fluorophore and sensor dye are able to be excited at the same time by a monochromatic light rather than excited respectively. To this end, pH ratiometric sensors based on FRET have been intriguing considerable interests.4,5 However, owing to the essence of FRET, the PL peaks of reference and sensor dye are too close to be distinguished for the sensor. Besides, fluorescence of QDs is not specifically sensitive to pH values. Credible result is elicited from neither single QDs probes nor QDs based ratiometric sensors because the presence of other ions could also change the fluorescence intensity of QDs. Thus, instability of QDs should be eliminated before using QDs as pH sensors as well as other probes.Here, to construct a high resolution pH ratiometer, stable SQMS with the maximum emission of 670 nm is used as an internal reference and further modified with pH-sensitive FITC with maximum emission of 516 nm. Due to the effective overlap of absorption spectra for QDs and FITC, they are able to be synchronously excited by a monochromatic light of 488 nm without the assistance of FRET (Fig. 5a), which is also proved by the fluorescence lifetime measurements (Fig. S6†). And the size-dependent tuneable emission for QDs endows QD/FITC pairs with high distinguishability for their PL peaks (Fig. 5b). As pH increases from 4.1 to 8.1, the PL intensity ratio of FITC to QDs (I516/I670) rises from 0.2 to 2.1 (Fig. 5d). The dependence of I516/I670 versus pH values is similar to that of FITC with pKa of about 6.4 (ref. 5) (Fig. S7†), while PL of QDs keeps constant at pH values from 4.1 to 8.1 (Fig 4c). There is almost no overlap for PL peaks of FITC (516 nm) and QDs (670 nm) excited by a monochromatic light of 488 nm simultaneously (Fig. 5c), indicating high resolution of SQMSF for pH. In the stability test, I516/I670 has little change at pH 8.1 or 4.1 for SQMSF kept in water, indicating SQMSF has remarkable stability and reversibility in water (Fig. 6, red circle and green triangle). However, one must notice that slight leakage of FITC in alkaline aqueous solution cannot be avoided due to cleavage of Si–O–Si induced by self-catalysis of amine groups of APS (Scheme S2†).42,43
4. Conclusions
In conclusion, highly fluorescent and stable sandwich-like SQMS is prepared by a simple and efficient electrostatic self-assembly approach. Nearly 98.9% of QDs are loaded onto composite nanoparticles. SQMS is 71 ± 7 nm in diameter, retaining 80% of initial fluorescence, with uniform morphology and robust stability in NaCl solution and at pH values from 4 to 8 due to the ligand exchange from MPA to MPTS. Then SQMS is modified with FITC for the application as a pH ratiometric sensor. Owing to tuneable PL and large Stokes shift for QDs, the SQMSF is able to be simultaneously excited by a monochromatic light and shows high resolution in PL peeks. I516/I670 changes from 0.2 to 2.1 as pH values increasing from 4 to 8, indicating SQMSF can be used to determine H+ accurately.Acknowledgements
We acknowledge the support from the National Science Foundation of China (Grant no. 51273047) and the “Shu Guang” Project (12SG07) supported by Shanghai Municipal Education Commission and Shanghai Education Development Foundation.Notes and references
- P. Wu, T. Zhao, S. Wang and X. Hou, Nanoscale, 2014, 6, 43 RSC.
- B. J. Kim, K. Paek, H. Yang, J. Lee and J. W. Park, ACS Nano, 2014, 8, 2848 CrossRef PubMed.
- Q. Ma, Y. Li, Z. H. Lin, G. C. Tang and X. G. Su, Nanoscale, 2013, 5, 9726 RSC.
- P. T. Snee, R. C. Somers, G. Nair, J. P. Zimmer, M. G. Bawendi and D. G. Nocera, J. Am. Chem. Soc., 2006, 128, 13320 CrossRef CAS PubMed.
- T. Jin, A. Sasaki, M. Kinjo and J. Miyazaki, Chem. Commun., 2010, 46, 2408 RSC.
- S. Huang, F. Zhu, H. Qiu, Q. Xiao, Q. Zhou, W. Su and B. Hu, Colloids Surf., B, 2014, 117, 240 CrossRef CAS PubMed.
- Y. P. Ho and K. W. Leong, Nanoscale, 2010, 2, 60 RSC.
- X. Michalet, F. F. Pinaud, L. A. Bentolila, J. M. Tsay, S. Doose, J. J. Li, G. Sundaresan, A. M. Wu, S. S. Gambhir and S. Weiss, Science, 2005, 307, 538 CrossRef CAS PubMed.
- Y. Li, L. Jing, R. Qiao and M. Gao, Chem. Commun., 2011, 47, 9293 RSC.
- Y. Yang, Z. Wen, Y. Dong and M. Gao, Small, 2006, 2, 898 CrossRef CAS PubMed.
- N. Tomczak, R. R. Liu and J. G. Vancso, Nanoscale, 2013, 5, 12018 RSC.
- T. Nann and P. Mulvaney, Angew. Chem., Int. Ed., 2004, 43, 5393 CrossRef CAS PubMed.
- L. Jing, C. Yang, R. Qiao, M. Niu, M. Du, D. Wang and M. Gao, Chem. Mater., 2009, 22, 420 CrossRef.
- Z. Lin, X. Fei, Q. Ma, X. Gao and X. Su, New J. Chem., 2014, 38, 90 RSC.
- G. Stoica, I. C. Serrano, A. Figuerola, I. Ugarte, R. Pacios and E. Palomares, Nanoscale, 2012, 4, 5409 RSC.
- Y. Zhang, M. Wang, Y. G. Zheng, H. Tan, B. Y. W. Hsu, Z. C. Yang, S. Y. Wong, A. Y. C. Chang, M. Choolani, X. Li and J. Wang, Chem. Mater., 2013, 25, 2976 CrossRef CAS.
- C. L. Li and N. Murase, J. Colloid Interface Sci., 2013, 411, 82 CrossRef CAS PubMed.
- W. S. Song, J. H. Kim and H. Yang, Mater. Lett., 2013, 111, 104 CrossRef CAS PubMed.
- M. Cho, K. Lim and K. Woo, Chem. Commun., 2010, 46, 5584 RSC.
- C. Wang, Q. Ma, W. Dou, S. Kanwal, G. Wang, P. Yuan and X. Su, Talanta, 2009, 77, 1358 CrossRef CAS PubMed.
- S. Wang, C. Li, P. Yang, M. Ando and N. Murase, Colloids Surf., A, 2012, 395, 24 CrossRef CAS PubMed.
- K. Han, Z. Xiang, Z. Zhao, C. Wang, M. Li, H. Zhang, X. Yan, J. Zhang and B. Yang, Colloids Surf., A, 2006, 280, 169 CrossRef CAS PubMed.
- B. H. Jun, D. W. Hwang, H. S. Jung, J. Jang, H. Kim, H. Kang, T. Kang, S. Kyeong, H. Lee, D. H. Jeong, K. W. Kang, H. Youn, D. S. Lee and Y.-S. Lee, Adv. Funct. Mater., 2012, 22, 1843 CrossRef CAS PubMed.
- C. Wu, J. Zheng, C. Huang, J. Lai, S. Li, C. Chen and Y. Zhao, Angew. Chem., Int. Ed., 2007, 46, 5393 CrossRef CAS PubMed.
- M. F. Foda, L. Huang, F. Shao and H. Y. Han, ACS Appl. Mater. Interfaces, 2014, 6, 2011 CAS.
- M. Szekeres, J. Toth and I. Dekany, Langmuir, 2002, 18, 2678 CrossRef CAS.
- X. G. Hu and X. H. Gao, ACS Nano, 2010, 4, 6080 CrossRef CAS PubMed.
- L. Jing, C. Yang, R. Qiao, M. Niu, M. Du, D. Wang and M. Gao, Chem. Mater., 2010, 22, 420 CrossRef CAS.
- Y. Zhu, Z. Li, M. Chen, H. M. Cooper, G. Q. Lu and Z. P. Xu, Chem. Mater., 2012, 24, 421 CrossRef CAS.
- Y. Zhu, Z. Li, M. Chen, H. M. Cooper and Z. P. Xu, J. Mater. Chem. B, 2013, 1, 2315 RSC.
- J. Guo, W. Yang and C. Wang, J. Phys. Chem. B, 2005, 109, 17467 CrossRef CAS PubMed.
- Z. Ran, Y. Sun, B. Chang, Q. Ren and W. Yang, J. Colloid Interface Sci., 2013, 410, 94 CrossRef CAS PubMed.
- R. Koole, M. M. van Schooneveld, J. Hilhorst, C. de Mello Donegá, D. C. 't Hart, A. van Blaaderen, D. Vanmaekelbergh and A. Meijerink, Chem. Mater., 2008, 20, 2503 CrossRef CAS.
- M. Darbandi, R. Thomann and T. Nann, Chem. Mater., 2005, 17, 5720 CrossRef CAS.
- Q. Ma, I. C. Serrano and E. Palomares, Chem. Commun., 2011, 47, 7071 RSC.
- P. Yang, N. Murase and J. Yu, Colloids Surf., A, 2011, 385, 159 CrossRef CAS PubMed.
- C. Graf, S. Dembski, A. Hofmann and E. Ruhl, Langmuir, 2006, 22, 5604 CrossRef CAS PubMed.
- N. Liu, B. S. Prall and V. I. Klimov, J. Am. Chem. Soc., 2006, 128, 15362 CrossRef CAS PubMed.
- M. F. Frasco and N. Chaniotakis, Sensors, 2009, 9, 7266 CrossRef CAS PubMed.
- I. L. Medintz, M. H. Stewart, S. A. Trammell, K. Susumu, J. B. Delehanty, B. C. Mei, J. S. Melinger, J. B. Blanco-Canosa, P. E. Dawson and H. Mattoussi, Nat. Mater., 2010, 9, 676 CrossRef CAS PubMed.
- M. Amelia, A. Lavie-Cambot, N. D. McClenaghan and A. Credi, Chem. Commun., 2011, 47, 325 RSC.
- M. Etienne, Talanta, 2003, 59, 1173 CrossRef CAS.
- O. Seitz, P. G. Fernandes, R. Tian, N. Karnik, H.-C. Wen, H. Stiegler, R. A. Chapman, E. M. Vogel and Y. J. Chabal, J. Mater. Chem., 2011, 21, 4384 RSC.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra05897f |
| This journal is © The Royal Society of Chemistry 2014 |





