Manganese ferrite nanoparticle catalyzed tandem and green synthesis of spirooxindoles†
Ramin Ghahremanzadeha,
Zahra Rashidb,
Amir-Hassan Zarnanic and
Hossein Naeimi*b
aNanobiotechnology Research Center, Avicenna Research Institute, ACECR, Tehran, Iran. E-mail: r.ghahremanzadeh@avicenna.ac.ir; Fax: +98-02122432021; Tel: +98-02122432020
bDepartment of Organic Chemistry, Faculty of Chemistry, University of Kashan, Kashan, 87317, Iran. E-mail: Naeimi@kashanu.ac.ir; Fax: +98-03615511121; Tel: +98-03615912388
cReproductive Immunology Research Center, Avicenna Research Institute, ACECR, Tehran, Iran. E-mail: zarnania@gmail.com; Fax: +98-02122432021; Tel: +98-02122432020
First published on 5th September 2014
Abstract
An environmentally benign and efficient method for the synthesis of spirooxindoles has been developed via a one-pot and three-component reaction of isatins, malononitrile, and anilinolactones in the presence of a catalytic amount of manganese ferrite nanoparticles in PEG-400, as a nontoxic, green, and reusable solvent. The significant advantages of this protocol are; the use of a magnetically recoverable and reusable catalyst, high to excellent product yields, operational simplicity and the use of PEG-400 as an environmentally-friendly solvent.
Introduction
Multicomponent reactions (MCRs) are special types of synthetically important organic reactions in which three or more different starting materials come together in a single reaction vessel to produce a final product containing diverse substituents from all the reactants.1 Such strategies are excellent tools in modern organic synthesis and medicinal chemistry due to the product diversity, operational simplicity, reduction in reaction steps and work-up, less time and energy consumption and a high degree of atom economy.2,3 In the last decade, with increasing environmental concerns, the design of new MCRs with eco-friendly, green procedures has drawn significant attention, especially in the fields of drug discovery, organic synthesis, and materials science.4,5 The use of polyethylene glycol (PEG), as a green solvent for organic synthesis has attracted extensive attention recently, due to its many advantages such as; water solubility, thermal stability over a wide range of temperatures, recoverability, non-volatility, non-explosiveness, commercial availability, and low toxicity.6 A number of organic reactions have been reported using PEG as a solvent medium or support for various organic transformations.7–9 Therefore, additional MCRs have become a critical and demanding research area in organic chemistry for the synthesis of molecular complexity and heterocyclic compounds such as spirooxindoles.10,11Spirooxindole cores are an important constituent in many natural and synthetic biologically active compounds, as well as in many drug molecules,12 in which an indole system is joined to varied heterocyclic motifs at the C-3 position through a spiro carbon atom. Molecules containing the spirooxindole moiety are widely found in a number of natural products such as spirotryprostatin A, horsfiline, and elacomine (Fig. 1).13 These natural products have been shown to possess a variety of important biological activities such as; anti-tumor,14 anti-tuberculosis,15 anti-microbial,16 anti-mycobacterium,17 anti-fungus,18 anti-malaria,19 and anti-oxidation.20 Consequently, looking for efficient, new and concise synthetic methods to prepare spirooxindole fused heterocycles is a major challenge and a popular field in chemistry.21–23 In recent years several methods using a variety of reagents and catalysts have been reported for the promoting preparation of spirooxindoles. One of an interesting catalysts for the synthesis of spirooxindole derivatives is magnetic nanoparticles.24,25
Magnetic nanoparticles are a group of nanostructured materials of considerable interest, largely due to their advanced technological and medical applications, envisioned or realized.26,27 In recent years they have emerged as a suitable group of heterogeneous catalysts because of their extremely small size, large surface to volume ratio, and because they can achieve many of the goals of green chemistry. Magnetic nanoparticles open up new opportunities to come up with an amazing and efficient system to facilitate catalyst recovery in organic reactions, because the magnetic nature of these particles allows for simple recovery and recycling of the catalysts by an external magnet, and magnetic separation is an attractive alternative to filtration or centrifugation as it prevents the loss of catalyst and increases reusability.28,29
In this research we report an environmentally benign synthetic method to uncover a green protocol for one-pot three-component synthesis of 2-amino-2′,5-dioxo-1-phenyl-5,7-dihydro-1H-spiro[furo[3,4-b]pyridine-4,3′-indoline]-3-carbonitrile derivatives. This reaction was carried out by using manganese ferrite nanoparticles as an efficient, reusable, and recoverable catalyst in PEG-400, as a safe, inexpensive, reusable, and biodegradable polymeric solvent.
Experimental
The chemicals used in this work were obtained from Fluka and Merck and were used without purification. Melting points were measured on an Electrothermal 9200 apparatus. IR spectra were recorded as KBr pellets on a Perkin-Elmer 781 spectrophotometer and an Impact 400 Nicolet FT-IR spectrophotometer. 1H NMR and 13C NMR spectra were recorded in DMSO-d6 solvents on a Bruker DRX-400 spectrometer with tetramethylsilane as internal reference. The elemental analyses (C, H, N) were obtained from a Carlo ERBA Model EA 1108 analyzer. Nanostructures were characterized using a Holland Philips Xpert X-ray powder diffraction (XRD) diffractometer (Cu K, radiation, k = 0.154056 nm), at a scanning speed of 2° min−1 from 10° to 100°/(2θ). Scanning electron microscope (SEM) was performed on a FEI Quanta 200 SEM operated at a 20 kV accelerating voltage. The purity determination of the substrates and reaction monitoring were accomplished by TLC on silica-gel polygram SILG/UV 254 plates (from Merck Company).Typical experimental procedure for the preparation of catalyst
MnFe2O4 nanoparticles have been prepared following the reported standard protocol by co-precipitation of MnCl2 and FeCl3 in water in the presence of sodium hydroxide. Briefly, MnCl2·4H2O and FeCl3·6H2O were taken in molar ratio of Mn2+![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fe3+ = 1
Fe3+ = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 to prepare 0.3 mol L−1 metal ion solution of 100 mL containing 0.1 mol L−1 Mn2+ and 0.2 mol L−1 Fe3+. Then, it was slowly dropped into 100 mL NaOH solution of 3 mol L−1 at the preheated temperature of 95 °C. After aging for 2 h with continuous stirring, the mixture was filtered, washed and dried at 60 °C for 12 h.30
2 to prepare 0.3 mol L−1 metal ion solution of 100 mL containing 0.1 mol L−1 Mn2+ and 0.2 mol L−1 Fe3+. Then, it was slowly dropped into 100 mL NaOH solution of 3 mol L−1 at the preheated temperature of 95 °C. After aging for 2 h with continuous stirring, the mixture was filtered, washed and dried at 60 °C for 12 h.30
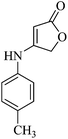
Typical procedure for the preparation of 2-amino-2′,5-dioxo-1-p-tolyl-5,7-dihydro-1H-spiro[furo[3,4-b]pyridine-4,3′-indoline]-3-carbonitrile (5b)
A mixture of isatin 1a (1 mmol), malononitrile 2 (1 mmol), 4-(4-methylphenylamino)furan-2(3H)-one 3b (1 mmol), and MnFe2O4 (5 mol%) were taken in PEG-400 (1 mL). The resulting mixture was stirred at 90 °C for an appropriate time. After completion of the reaction as indicated by TLC, the mixture was magnetically concentrated with the aid of an external magnet to separate the catalyst. The separated catalyst washed with acetone several times followed by EtOH, then dried under vacuum and reutilized four times for the same reaction. After separation of the catalyst, H2O (10 mL) was added to the reaction mixture and was shaken for a few minutes to dissolve PEG and precipitate the product. The crude product (insoluble in water) was filtered and re-crystallized by ethanol for more purification. The desired pure product was identified as white powder (yield: 83%). mp > 300 °C. IR (KBr) (νmax/cm−1): 3454, 2185, 1721, 1682. 1H NMR (DMSO-d6, 400 MHz): δppm: 2.37 (3H, s, CH3), 4.47–4.64 (2H, m, OCH2), 5.99 (2H, s, NH2), 6.82–7.44 (8H, m, ArH), 10.48 (1H, s, NH). 13C NMR (DMSO-d6, 100 MHz): δppm: 21.2, 48.2, 60.0, 66.1, 99.0, 109.8, 119.5, 122.5, 125.3, 128.9, 129.3, 131.2, 131.7, 134.3, 140.3, 141.8, 152.8, 159.6, 170.1, 178.0. Anal. calcd for C22H16N4O3: C, 68.74; H, 4.20; N, 14.58%. Found C, 68.69; H, 4.24; N, 14.53%. MS: m/z 384.Results and discussion
Due to the unique properties of spirooxindole compounds and in continuation of our research for the efficient preparation of spirooxindole heterocycles31–33 via a simple and environmentally benign synthetic method, a three component synthesis was planned based on foreseeing a one-pot reaction among isatins 1, malononitrile 2, and anilinolactones 3 for the synthesis of spirooxindoles (Scheme 1).Anilinolactones are versatile synthetic intermediates in organic synthesis that combine the nucleophilicity of an enamine and the electrophilicity of an enone. They are commonly applied in the preparation of heterocyclic compounds.34,35 As shown in Scheme 2, when tetronic acid was reacted with an equimolar amount of various anilines in 1,4-dioxane at room temperature, the corresponding products were obtained in excellent yields and purity.36
Recent studies on the preparation of spirooxindoles revealed that one of the usual conditions for their synthesis uses water as a solvent in the presence of p-toluenesulfonic acid (p-TSA) as an economical, non-toxic catalyst under reflux conditions.37–40 Hence, the reaction of isatin 1a, malononitrile 2, and 4-(4-methylphenylamino)furan-2(3H)-one 3b in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 : 1 molar ratio as a model substrate was refluxed for 8 h in water with p-TSA (20%). After completion and work-up of the reaction a powdery product was obtained and purified (Scheme 3).
1 : 1 molar ratio as a model substrate was refluxed for 8 h in water with p-TSA (20%). After completion and work-up of the reaction a powdery product was obtained and purified (Scheme 3).
Product structure was characterized based on mass, 1H NMR, and 13C NMR spectra. In the 1H NMR spectrum of product (as shown in Fig. 2), the signal at δ = 10.70 ppm indicates the presence of –NH proton of oxindole ring (D2O exchangeable), the NH2 protons resonated at δ = 7.70 ppm with two integral values (exchangeable with D2O), the aromatic protons exhibited multiplets in the region δ = 6.85–7.26 ppm with four integral values, the signals around δ = 5.06–5.21 ppm with two integral values are assigned to the protons of OCH2 of tetronic acid. The 13C NMR spectrum showed 15 distinct signals; also the mass spectrum of product displayed the molecular ion peak at m/z: 295. Surprisingly, the reaction did not proceed according to expectation and spectral data were inconsistent with the expected structure 5b. Indeed, the data were in good agreement with the structure of an unprecedented product, and showed the structure of the unexpected product 4. Also, the structure of the identity of the obtained product was confirmed by comparing its melting point with that of this previously.41
 | ||
| Fig. 2 The 1H NMR spectrum of 2-amino-2′,5-dioxo-5,7-dihydrospiro[furo[3,4-b]pyran-4,3′-indoline]-3-carbonitrile 4. | ||
Although the detailed mechanism of the above reaction has not yet been clarified, we proposed the possible pathway to form the spiro product 4 via domino reactions. As shown in Scheme 4, compound 4 could be synthesized via sequential condensation, addition, hydrolysis, cyclization and tautomerization. The reaction may proceed in a stepwise manner, in which the isatin 1a can be firstly condensed with malononitrile 3 to afford isatylidene malononitrile 6 in the presence of p-TSA in water. This step was regarded as a fast Knoevenagel condensation reaction. Then, compound 6 is attacked by a Michael type addition with 4-(4-methylphenylamino)furan-2(3H)-one 3b to produce the intermediate 7. We suspect in the presence of p-TSA as a Bronsted acid and water, the iminium group in the intermediate 7, was hydrolyzed followed by an intramolecular cyclization and tautomerization to afford product 4 (Scheme 4). In the proposed mechanism, p-TSA may be able to catalyze the reaction steps due to its acidic nature.
In order to produce the expected product 5, we continued to explore different catalysts and media on the model reaction (Scheme 5). The results are summarized in Table 1. As shown in this table, when we tested on the model reaction in PEG 400 or in ionic liquids (IL) with magnetic nanoparticles as the catalyst, the product 5b was obtained (Table 1, Entries 10–14). PEG-400 in the presence of MnFe2O4 as the catalyst proved to be the best system tested based on its reaction rate as well as yield while under other conditions as seen in Table 1, compound 4 was produced.
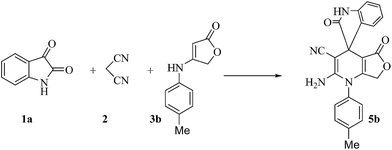 | ||
| Scheme 5 Model reaction for the synthesis of 2-amino-2′,5-dioxo-1-p-tolyl-5,7-dihydro-1H-spiro[furo[3,4-b]pyridine-4,3′-indoline]-3-carbonitrile 5b. | ||
| Entry | Medium | Catalyst | Product | Time (h) | Yieldb (%) |
|---|---|---|---|---|---|
| a Reaction conditions isatin 1a (1 mmol), malononitrile 2 (1 mmol), 4-(4 methylphenylamino)furan-2(3H)-one 3b (1 mmol).b Isolated yields. | |||||
| 1 | H2O (90 °C) | p-TSA (20 mol%) | 4 | 8 | 75 |
| 2 | H2O (90 °C) | Alum (20 mol%) | 4 | 12 | 68 |
| 3 | EtOH (70 °C) | p-TSA (20 mol%) | 4 | 12 | 69 |
| 4 | CH3CN (70 °C) | p-TSA (20 mol%) | 4 | 12 | 48 |
| 5 | [Bmim]Br (90 °C) | p-TSA (20 mol%) | 4 | 4 | 70 |
| 6 | [Bmim]PF6 (90 °C) | p-TSA (20 mol%) | 4 | 4 | 73 |
| 7 | PEG-400 (100 °C) | p-TSA (20 mol%) | 4 | 6 | 62 |
| 8 | PEG-400 (100 °C) | CH3COOH (50 mol%) | 4 | 6 | 51 |
| 9 | PEG-400 (100 °C) | Nano MnFe2O4 (10 mol%) | 5b | 6 | 83 |
| 10 | PEG-400 (100 °C) | Nano CuFe2O4 (10 mol%) | 5b | 6 | 70 |
| 11 | PEG-400 (100 °C) | Nano Fe3O4 (10 mol%) | 5b | 8 | 46 |
| 12 | [Bmim]PF6 (90 °C) | Nano MnFe2O4 (10 mol%) | 5b | 8 | 54 |
| 13 | [Bmim]PF6 (90 °C) | Nano CuFe2O4 (10 mol%) | 5b | 8 | 51 |
| 14 | PEG-400 (100 °C) | — | — | 6 | — |
| 15 | H2O (90 °C) | — | — | 12 | — |
| 16 | [Bmim]PF6 (90 °C) | — | — | — | |
Characterization of the catalyst
The manganese ferrite nanoparticles were prepared by co-precipitation of MnCl2 and FeCl3 in basic solution at 95 °C using the previous reported method. The synthesized MnFe2O4 was characterized by X-ray diffraction (XRD), scanning electron microscope (SEM), and vibrating sample magnetometer (VSM). The position and relative intensities of all peaks were confirmed as well as with standard XRD pattern of MnFe2O4 (JCPDS card no. 73-1964). The calcined manganese ferrite at 800 °C present a particle size of 33 nm, calculated from the broadening of the peak at 2θ = 35.31 using the Scherrer equation (Fig. 3). The SEM image revealed that manganese ferrite nanoparticles have a mean diameter of about 30–35 nm (Fig. 4). The magnetization curve for MnFe2O4 nanoparticles is shown in Fig. 5. It is of great importance that a catalyst should possess sufficient magnetic and super paramagnetic properties for its practical application. Magnetic hysteresis measurements on MnFe2O4 were conducted in an applied magnetic field at room temperature, with the field sweeping from −10 000 to +10 000 oersted. As shown in Fig. 5, the hysteresis loop for the sample was completely reversible confirming its super paramagnetic nature. The catalyst showed high permeability in magnetization and high reversibility in the hysteresis loop.In the next step, in order to optimize the more suitable reaction conditions, we evaluated the amount of catalyst required, and the effect of temperature for this transformation. Our optimization studies revealed that when the model reaction was carried out in the presence of 2 mol% of catalyst, 62% yield is obtained. It was found that catalyst loadings above 5 mol% did not improve the reaction rate of yield. Thus 5 mol% of catalyst was chosen as the maximum quantity of the catalyst for the reaction (Table 2, Entry 2). Also, the effect of temperature was studied by carrying out the model reaction in PEG-400 at different temperatures in the presence of 5 mol% of catalyst. As shown in Table 2, when the reaction temperature was 25 °C or 40 °C (Table 2, Entries 5 and 6), the reaction was proceeded, but the obtained yield remained low even after longer reaction time until 24 h. However, at elevated temperature (40–90 °C) using PEG-400 gave better results in terms of yield and reaction time. It was realized that when temperature increased up further to 110 °C (Table 2, Entry 8), there was no significant improvement of the rate as well as yield of the reaction. Thus, the temperature of 90 °C was found to be the most suitable reaction temperature for an optimum yield of desired product (Table 2, Entry 2).
| Entry | Catalyst (mol%) | Temperature (°C) | Time (h) | Yieldb (%) |
|---|---|---|---|---|
| a Reaction conditions: isatin 1a (1 mmol), malononitrile 2 (1 mmol), 4-(4 methylphenylamino)furan-2(3H)-one 3b (1 mmol), PEG-400 (1 mL).b Isolated yields. | ||||
| 1 | 2 | 90 | 8 | 62 |
| 2 | 5 | 90 | 6 | 83 |
| 3 | 10 | 90 | 6 | 83 |
| 4 | 20 | 90 | 6 | 84 |
| 5 | 5 | 25 | 24 | <50 |
| 6 | 5 | 40 | 24 | <50 |
| 7 | 5 | 70 | 10 | 72 |
| 8 | 5 | 110 | 6 | 83 |
Most remarkably, we were also able to recycle the catalyst for five times with almost the same catalytic activity as illustrated in Fig. 5. The catalyst was recovered in excellent yield (96–98%) after each of the new set of reaction. Isatin, malononitrile, and 4-(4-methylphenylamino)furan-2(3H)-one were also employed as the reactants of the model reaction for the reusability study of the catalyst at 90 °C in PEG-400. In this procedure, after completion of the reaction, the catalyst could be magnetically recovered by an external magnetic field and the retained catalyst was washed with acetone to remove the residual product. After being dried, catalyst was subjected to other reaction runs. After separation of the catalyst, water (10 mL) was added to the reaction mixture and was shaken for a few minutes to dissolve PEG and precipitated the product. The crude product (insoluble in water) was filtered and washed with ethanol for further purification. The procedure was repeated and the results indicated that in five consecutive runs. The isolated yields were remained similar with no detectable loss (Fig. 6).
 | ||
| Fig. 6 Catalyst recyclability study on the synthesis of 2-amino-2′,5-dioxo-1-p-tolyl-5,7-dihydro-1H-spiro[furo[3,4-b]pyridine-4,3′-indoline]-3-carbonitrile 5b. | ||
Finally, we examined the recyclability of the PEG after the extraction of the product. In order to prove that the use of polyethylene glycol as environmentally benign solvent is also practical; it must be conveniently recycled with minimum loss and decomposition. In this procedure, after completion of the reaction, the crude product (insoluble in water) was filtered and recrystallized from ethanol for further purification. In order to recover the PEG, H2O was evaporated under reduced pressure, and the result was washed with diethyl ether, and dried under reduced pressure. The recycled PEG does not change in its reactivity but approximately 5% weight loss of PEG was observed from cycle to cycle (Table 3).
We have not established an exact mechanism for the formation of 5b, however, a reasonable possibility based on literatures42,43 is shown in Scheme 6. Compound 5b could be synthesized via sequential condensation, addition, cyclization and tautomerization. The process represents a typical domino reaction in which the activated isatin 1, may be firstly condensed with malononitrile 2 to afford isatylidene malononitrile 6 in the presence of manganese ferrite nanoparticles as a catalyst in PEG-400. This step was regarded as a fast Knoevenagel condensation. Then, compound 6 is attacked by Michael addition of 4-(4-methylphenylamino)furan-2(3H)-one 3b to give the intermediate 7, followed by intra-molecular cyclization and tautomerization to afford the target product 5b. The manganese ferrite nanoparticles as a Lewis acid probably can catalyze the reaction steps.
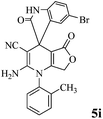
In order to generalize the optimum conditions and check the feasibility of this protocol, different derivatives of 2-amino-2′,5 dioxo-1-phenyl-5,7-dihydro-1H-spiro[furo[3,4-b]pyridine-4,3′ indoline]-3-carbonitrile 5a–l were prepared from the one-pot reaction mixture of isatins 1a–e, malononitrile 2, and anilinolactones 3a–g in the presence of a catalytic amount of MnFe2O4 (5 mol%) in PEG-400 at 90 °C. The results are summarized in Table 4. Compounds 5a–l are stable solids and the structures of which were determined by IR, Mass, 1H and 13C NMR spectroscopy, and elemental analysis.
| Entry | Isatin 1 | Anilinolactone 3 | Product 5 | Time (h) | Yieldb (%) |
|---|---|---|---|---|---|
| a Reaction conditions: isatin 1a–e (1 mmol), malononitrile 2 (1 mmol), anilinolactones 3a–g (1 mmol), MnFe2O4 (5 mol%), PEG-400 (1 mL), 90 °C.b Isolated yields. | |||||
| 1 | 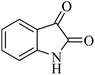 |
 |
 |
5 | 79 |
| 2 |  |
 |
 |
6 | 83 |
| 3 |  |
 |
 |
5 | 80 |
| 4 |  |
 |
 |
5 | 77 |
| 5 |  |
 |
 |
5 | 80 |
| 6 |  |
 |
 |
5 | 79 |
| 7 |  |
 |
 |
5 | 75 |
| 8 |  |
 |
 |
4 | 73 |
| 9 |  |
 |
 |
4 | 76 |
| 10 |  |
 |
 |
3 | 81 |
| 11 |  |
 |
 |
3 | 84 |
| 12 |  |
 |
 |
3 | 75 |
To determine the percent leaching of the manganese ferrite nanoparticles, the model reaction was carried out in the presence of catalyst for 1 h, and at that point the catalyst was separated by external magnet. The residue was then allowed to react, but no significant progress was observed after 24 h. Also, it was determined the amount of Fe and Mn metals in product 4a as a model reaction by atomic absorption in that the quantity of the residue of Fe and Mn metals was not detectable.
Conclusions
In conclusion, we have developed an efficient and more environmentally friendly protocol for the one-pot synthesis of spirooxindoles via a three-component condensation reaction of isatins, malononitrile, and anilinolactones by using manganese ferrite nanoparticles as a powerful catalyst in PEG-400. The notable features of this procedure are mild and green reaction conditions, convenient workup, recyclability and reusability of the magnetic catalyst, high to excellent product yields, and recyclability of PEG.Acknowledgements
This study is part of Zahra Rashid Ph. D thesis entitled: “Synthesis, modification and functionalization of magnetic nanoparticles for catalytic application in the synthesis of heterocyclic compounds and biomedical applications” which has been conducted in the Nanobiotechnology Research Center, Avicenna Research Institute. And also, we are thankful from University of Kashan for supporting this work by grant number 159148/15.References
- A. Dömling, Chem. Rev., 2006, 106, 17–89 CrossRef PubMed.
- H. Wang and D. Shi, ACS Comb. Sci., 2013, 15, 261–266 CrossRef CAS PubMed.
- F. Shi, Z. Xiao-Ning, G. Zhang, N. Ma, B. Jiang and S. Tu, Bioorg. Med. Chem. Lett., 2011, 21, 7119–7123 CrossRef CAS PubMed.
- F. Alonso, Y. Moglie, G. Radivoy and M. Yus, Org. Biomol. Chem., 2011, 9, 6385–6395 CAS.
- S. Rajesh, B. Bala, S. Perumal and J. C. Menendez, Green Chem., 2011, 13, 3248–3254 RSC.
- (a) V. V. Kouznetsov, D. R. Merchan Arenas and A. R. Romero Bohorquez, Tetrahedron Lett., 2008, 49, 3097–3100 CrossRef CAS PubMed; (b) B. Das, M. Krishnaiah, P. Thirupathi and K. Laxminarayana, Tetrahedron Lett., 2007, 48, 4263–4265 CrossRef CAS PubMed; (c) S. Chandrasekhar, N. R. Reddy, S. S. Sultana, C. Narsihmulu and K. V. Reddy, Tetrahedron, 2006, 62, 338–345 CrossRef CAS PubMed.
- A. Hasaninejed, M. Rasekhi Kazerooni and A. Zare, Catal. Today, 2012, 196, 148–153 CrossRef CAS PubMed.
- K. Pradhan, P. Bhattachary, S. Paul and A. Das, Tetrahedron Lett., 2012, 53, 5840–5844 CrossRef CAS PubMed.
- A. Kumar, A. Saxena, M. Dewan, A. De and S. Mozumdar, Tetrahedron Lett., 2011, 52, 4835–4839 CrossRef CAS PubMed.
- S. Pal, V. Singh, P. Das and L. H. Choudhury, Bioorg. Chem., 2013, 48, 8–15 CrossRef CAS PubMed.
- S. Rostamnia, A. Nuri, H. Xin, A. Pourjavadi and H. Hosseini, Tetrahedron Lett., 2013, 54, 3344–3347 CrossRef CAS PubMed.
- (a) M. M. Khafagy, A. H. F. A. El-Wahas, F. A. Eid and A. M. El-Agrody, Farmaco, 2002, 57, 715–722 CrossRef CAS; (b) P. R. Sebahar and R. M. Williams, J. Am. Chem. Soc., 2000, 122, 5666–5667 CrossRef CAS.
- (a) A. Rahman, W. S. J. Silva, K. A. Alvi and K. T. D. De Silva, Phytochemistry, 1987, 26, 865–868 CrossRef; (b) C. B. Cui, H. Kakeya and H. Osada, Tetrahedron, 1996, 52, 12651–12666 CrossRef CAS; (c) C. B. Cui, H. Kakeya and H. Osada, J. Antibiot., 1996, 49, 832–835 CrossRef CAS; (d) C. B. Cui, H. Kakeya, G. Okada, R. Onose and H. Osada, J. Antibiot., 1996, 49, 527–533 CrossRef CAS; (e) C. B. Cui, H. Kakeya and H. Osada, J. Antibiot., 1996, 49, 534–540 CrossRef CAS; (f) C. B. Cui, H. Kakeya and H. Osada, Tetrahedron, 1997, 53, 59–72 CrossRef CAS; (g) T. H. Kang, K. Matsumoto, Y. Murakami, H. Takayama, M. Kitajima, N. Aimi and H. Watanabe, Eur. J. Pharmacol., 2002, 444, 39–45 CrossRef CAS; (h) R. M. Williams and R. J. Cox, Acc. Chem. Res., 2003, 36, 127–139 CrossRef CAS PubMed; (i) C. V. Galliford and K. A. Scheidt, Angew. Chem., Int. Ed., 2007, 46, 8748–8758 CrossRef CAS PubMed.
- K. Ding, Y. P. Lu, Z. Nikolovska-Coleska, G. P. Wang, S. Qiu, S. Shangary, W. Gao, D. G. Qin, J. Stuckey, K. Krajewski, P. P. Roller and S. M. Wang, J. Med. Chem., 2006, 49, 3432–3435 CrossRef CAS PubMed.
- P. Prasanna, K. Balamurugan, S. Perumal, P. Yogeeswari and D. Sriram, Eur. J. Med. Chem., 2010, 45, 5653–5661 CrossRef CAS PubMed.
- A. Nandakumar, P. Thirumurugan, P. T. Perumal, P. Vembu, M. N. Ponnuswamy and P. Ramesh, Bioorg. Med. Chem. Lett., 2010, 20, 4252–4248 CrossRef CAS PubMed.
- S. U. Maheswari, K. Balamurugan, S. Perumal, P. Yogeeswari and D. Sriram, Bioorg. Med. Chem. Lett., 2010, 20, 7278–7282 CrossRef CAS PubMed.
- A. Thangamani, Eur. J. Med. Chem., 2010, 45, 6120–6126 CrossRef CAS PubMed.
- B. K. S. Yeung, B. Zou, M. Rottmann, S. B. Lakshminarayana, S. H. Ang, S. Y. Leong, J. Tan, J. Wong, S. Keller-Maerki, C. Fischli, A. Goh, E. K. Schmitt, P. Krastel, F. Erancotte, K. Kuhen, D. Plouffe, K. Henson, T. Wagner, E. A. Winzeler, F. Petersen, B. Reto, V. Dartois, T. T. Diagana and T. H. Keller, J. Med. Chem., 2010, 53, 5155–5164 CrossRef CAS PubMed.
- N. Karali, O. Guzel, N. Ozsoy, S. Ozbey and A. Salman, Eur. J. Med. Chem., 2010, 45, 1068–1077 CrossRef CAS PubMed.
- A. Alizadeh, A. Rezvanian and L. G. Zhu, J. Org. Chem., 2012, 77, 4385–4390 CrossRef CAS PubMed.
- B. Maheshwar Rao, G. Niranjan Reddy, T. Vijaikumar Reddy, B. L. A. Prabhavathi Devi, R. B. N. Prasad, J. S. Yadav and B. V. Subba Reddy, Tetrahedron Lett., 2013, 54, 2466–2471 CrossRef CAS PubMed.
- S. Paul and A. Das, Tetrahedron Lett., 2013, 54, 1149–1154 CrossRef CAS PubMed.
- A. Bazgir, G. Hosseini and R. Ghahremanzadeh, ACS Comb. Sci., 2013, 15, 530–534 CrossRef CAS PubMed.
- A. Khalafi-Nezhad and S. Mohammadi, ACS Comb. Sci., 2013, 15, 512–518 CrossRef CAS PubMed.
- (a) M. Casula, A. Corrias, P. Arosio, A. Lascialfari, T. Sen, P. Floris and I. J. Bruce, J. Colloid Interface Sci., 2011, 357, 50–55 CrossRef CAS PubMed; (b) M. R. Nejadmoghaddam, M. Chamankhah, S. Zarei and A. H. Zarnani, J. Nanobiotechnol., 2011, 9, 1–11 CrossRef PubMed.
- S. Thayyil, S. Schievano, N. J. Robertson, R. Jones, L. S. Chitty, N. J. Sebire and A. Taylor, Eur. J. Radiol., 2009, 72, 321–326 CrossRef PubMed.
- (a) B. Li, L. Gao, F. Bian and W. Yu, Tetrahedron Lett., 2013, 54, 1063–1066 CrossRef CAS PubMed; (b) A. Maleki, Tetrahedron, 2012, 68, 7827–7833 CrossRef CAS PubMed.
- (a) J. Deng, L. Mo, F. Zhao, Z. Zhang and S. Liu, ACS Comb. Sci., 2012, 14, 335–341 CrossRef CAS PubMed; (b) M. Sheykhan, H. Mohammadnejad, J. Akbari and A. Heydari, Tetrahedron Lett., 2012, 53, 2959–2964 CrossRef CAS PubMed.
- H. Aijun, L. Juanjuan, Y. Mingquan, L. Yan and P. Xinhua, Chin. J. Chem. Eng., 2011, 19, 1047–1051 CrossRef.
- S. Ahadi, R. Ghahremanzadeh, P. Mirzaei and A. Bazgir, Tetrahedron, 2009, 65, 9316–9321 CrossRef CAS PubMed.
- R. Ghahremanzadeh, S. Ahadi, G. Imani Shakibaei and A. Bazgir, Tetrahedron Lett., 2010, 51, 499–502 CrossRef CAS PubMed.
- A. Bazgir, S. Ahadi, R. Ghahremanzadeh, H. Khavasi and P. Mirzaei, Ultrason. Sonochem., 2010, 17, 447–452 CrossRef CAS PubMed.
- C. Cheng, B. Jiang, S. J. Tu and G. Li, Green Chem., 2011, 13, 2107–2115 RSC.
- S. L. Wang, C. Cheng, F. Y. Wu, B. Jiang, F. Shi, S. J. Tu, T. Rajale and G. Li, Tetrahedron, 2011, 67, 4485–4493 CrossRef CAS PubMed.
- Y. Hitotsuyanagi, M. Kobayashi, M. Fukuyo, K. Takeya and H. Itokawa, Tetrahedron Lett., 1997, 38, 8295–8296 CrossRef CAS.
- Y. Zou, H. Wu, Y. Hu, H. Liu, X. Zhao, H. Ji and D. Shi, Ultrason. Sonochem., 2011, 18, 708–712 CrossRef CAS PubMed.
- R. Ghahremanzadeh, F. Fereshtehnejad, Z. Yasaei, T. Amanpour and A. Bazgir, J. Heterocycl. Chem., 2010, 47, 967–972 CrossRef CAS.
- J. Quiroga, S. Portillo, A. Pérez, J. Gálvez, R. Abonia and B. Insuasty, Tetrahedron Lett., 2011, 52, 2664–2666 CrossRef CAS PubMed.
- K. Rad-Moghadam and L. Youseftabar-Miri, Synlett, 2010, 1969–1973 CrossRef CAS PubMed.
- Y. Li, H. Chen, C. Shi, D. Shi and S. Ji, J. Comb. Chem., 2010, 12, 231–237 CrossRef CAS PubMed.
- A. Dandia, A. k. Laxkar and R. Singh, Tetrahedron Lett., 2012, 53, 3012–3017 CrossRef CAS PubMed.
- A. R. Karimi and F. Sedaghatpour, Synthesis, 2010, 1731–1735 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra05756b |
| This journal is © The Royal Society of Chemistry 2014 |










