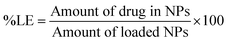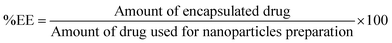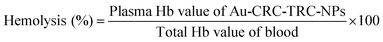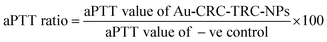Retracted Article: Radio frequency triggered curcumin delivery from thermo and pH responsive nanoparticles containing gold nanoparticles and its in vivo localization studies in an orthotopic breast tumor model
N. Sanoj Rejinolda,
Reju George Thomasb,
Muthunarayanan Muthiahc,
K. P. Chennazhia,
In-Kyu Park*c,
Yong Yeon Jeongb,
K. Manzoora and
R. Jayakumar*a
aAmrita Centre for Nanosciences and Molecular Medicine, Amrita Institute of Medical Sciences and Research centre, Amrita Vishwa Vidyapeetham University, Kochi-682041, India
bDepartment of Radiology, Chonnam National University Hwasun Hospital, Chonnam National University Medical School, Gwangju 501-746, South Korea
cDepartment of Biomedical Science and BK21 PLUS Center for Creative Biomedical Scientists, Chonnam National University Medical School, Gwangju 501-746, South Korea. E-mail: rjayakumar@aims.amrita.edu; jayakumar77@yahoo.com; pik96@chonnam.ac.kr; Fax: +91 484 2802020; Fax: +82-61-379-8455; Tel: +91 484 2801234 Tel: +82-61-379-8481
First published on 4th August 2014
Abstract
Non-invasive radiofrequency (RF) electric fields as an energy source for thermal activation of nanoparticles and thereby delivering drugs within cancer cells could be a valuable addition to nano-mediated RF based cancer therapies. Utilizing the high penetration of RF waves would be useful for the controlled release of encapsulated drug molecules from smart thermo and pH responsive nanoparticles. Herein, we demonstrate that breast cancer cells could selectively internalize hemocompatible, 170 ± 20 nm sized curcumin encapsulated chitosan-graft-poly(N-vinyl caprolactam) nanoparticles containing gold nanoparticles (Au-CRC-TRC-NPs). Au-CRC-TRC-NPs were predominantly accumulated within the cytoplasm. After “optimum RF exposure” at 40 watts for 5 minutes, Au-CRC-TRC-NPs absorbed and dissipated energy as heat in the range of 42 °C, which is the lower critical solution temperature (LCST) of chitosan-graft-poly(N-vinyl caprolactam), causing controlled curcumin release and inducing apoptosis to 4T1 breast cancer cells. Further, the tumor localization studies on orthotopic breast cancer models revealed that Au-CRC-TRC-NPs could selectively accumulate at primary and secondary tumors as confirmed by in vivo live imaging followed by ex vivo tissue imaging and HPLC studies. These preclinical results throw light on their feasibility as a better tumor targetable nanomedicine for RF-assisted breast treatment modalities.
Introduction
Breast cancer remains the second most leading cause of female mortality worldwide. In India, it is the same scenario, where 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000
000![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 new cases are being reported per year.1 Though there are number of conventional and advanced treatment regimens, the side effects associated with them are still lethal and unanswered.2
000 new cases are being reported per year.1 Though there are number of conventional and advanced treatment regimens, the side effects associated with them are still lethal and unanswered.2
Radio frequency (RF) assisted cancer therapy has tremendous advantages over conventional radiotherapy, as it is completely non-ionizing and non-hazardous.3 Since the RF waves have high penetration capability, deep rooted cancers can be well treated using RF therapy. There have been various attempts reported to improve RF therapy with special attention in replacing RF probe with metallic nanoparticles such as gold (Au-NPs) and iron oxide.4 In the current scenario, the major drawback in RF treatment is the invasive mode of the treatment. Most of the reported RF assisted cancer therapy has utilized Au-NPs as it is nontoxic, biocompatible and functional.5 Smart nanomaterials based on thermo and pH-responsive polymers, on the other hand, can take advantage of the innate tumor micro environment such as acidic pH,6 poor lymphatic drainage,7 and leaky vasculature, enhancing passive targeting of nanoparticles.8
Thermo-responsive polymers such as poly(N-vinyl caprolactam) [PNVCL] has been widely used for anti-cancer drug delivery.9–16 The function of these polymers are based on LCST (lower critical solution temperature) property, for delivering drug molecules. This LCST of polymer can be modified such that drug will be released when the polymer attains that specific temperature. Biopolymers such as chitosan can be conjugated to PNVCL, not only to tune its LCST, but also to impart a pH response.9
The naturally occurring polyphenolic phytoconstituent, curcumin, possesses anti-cancer, anti-oxidant, anti-inflammatory, anti-bacterial, wound healing and hepatoprotective activities.17 The therapeutic efficacy of curcumin is limited due to its poor oral bioavailability,18 which has been attributed to its poor aqueous solubility and extensive first pass metabolism. Various attempts have been made through encapsulation in polymeric nanoparticles;19–24 however, no work has been reported on RF-assisted curcumin delivery to cancer cells. Thus, combining thermo and pH responsive polymers with Au-NPs would be ideal for the effective RF-assisted curcumin delivery to cancer cells. The whole idea of incorporating Au-NPs is to induce RF assisted heating under optimum RF conditions, which in turn, sensitized by the thermo-responsive polymers allows the encapsulated curcumin molecules to be eluted out.
The research questions we focused on are as follows: (1) How would Au-CRC-TRC-NPs affect the thermo-responsive delivery to breast cancer cells at optimum RF conditions?; (2) How can these formulations be targeted toward the orthotopic breast tumor model?; and (3) What would be the tumor localization in a highly metastasized breast tumor model? We hypothesize that, since Au-NPs are RF heatable, the encapsulated curcumin can be released according to the thermo-sensitivity of TRC-NPs via the heat induced from Au-NPs at optimum RF conditions as shown in Fig. 1. The Au-CRC-TRC-NPs being smaller in size could easily be targeted to primary as well as metastasized tumors by the EPR (enhanced permeability and retention) effect. Thus, the major goal of our work is to understand the in vitro RF response of Au-CRC-TRC-NPs, its in vitro efficacy at optimum RF conditions, and finally in vivo breast tumor localization capability in detail.
Experimental
(a) Materials
Chitosan (viscosity average molecular weight, 20 kDa, degree of N-deacetylation, 75–80%) was purchased from Koyo chitosan Company, Japan, and used as received. N-Vinylcaprolactam (NVCL) was purchased from Sigma Aldrich and recrystallized from n-hexane before use. Azobis isobutyronitrile (AIBN), 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide (EDC) and N-hydroxy succinimide (NHS), penta sodium tripolyphosphate (TPP) were purchased from Sigma Aldrich, Bangalore, India, and used without further purification. Isopropyl alcohol and 3-mercaptopropionic acid (MPA) were supplied by Aldrich and used as supplied. Curcumin was purchased from Merck, Cochin, India. Acid dextrose citrate, sodium bicarbonate and sodium chloride were purchased from Merck, Cochin, for blood compatibility studies. The staining dyes were purchased from Sigma Aldrich, Bangalore. The cell lines were purchased from ATCC, USA.(b) Preparation of colloidal gold nanoparticles (Au-NPs)
The Au-NPs were prepared according to existing literature25 with further modifications for avoiding the ionic concentration in the Au-NPs suspension. 300 μL 0.l M HAuCl4·6H2O was treated with 10 mL 0.20% starch solution and then 200 μL 0.l M D-glucose was added and the reaction mixture was allowed to react at 50 °C. The pH was adjusted with 1% tris buffer solution. A deep wine red coloration after 5 min indicated the formation of 10 nm sized Au-NPs.(c) Determination of the lower critical solution temperature (LCST)
We analyzed the LCST of the systems by a UV spectrophotometer (Pharma spec, Japan) starting from a temperature range of 0–45 °C and an average of three values were taken as the LCST of the synthesized materials. This was further tested with a digital thermometer.(d) Preparation of curcumin encapsulated chitosan-graft-PNVCL nanoparticles (CRC-TRC-NPs)
CRC-TRC-NPs were prepared as per our pervious protocol with slight modifications to tune the LCST (42 °C).9 Chitosan-graft-PNVCL was dissolved in acetic acid (1% solution) and the resulting solution was treated with 5 mg of ethanolic curcumin (5 mg curcumin in 1 mL distilled ethanol), then cross-linked with (sodium tripoly-phosphate) TPP (Sigma-Aldrich) in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio. The resulting CRC-TRC-NPs were centrifuged at 20
1 ratio. The resulting CRC-TRC-NPs were centrifuged at 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 45 min, resuspended and pelletized several times in water until the pH became 7.4.
000 rpm for 45 min, resuspended and pelletized several times in water until the pH became 7.4.
(e) Preparation of CRC-TRC-NPs embedded Au-NPs (Au-CRC-TRC-NPs)
Based on a trial and error method, the Au concentration was optimized to be 0.54 mg for 1 mL of CRC-TRC-NPs. 0.54 mg of Au-NPs were suspended in 1 mL of CRC-TRC-NPs and incubated at room temperature for about 3 h to complete the reaction. The resulting solution was centrifuged at 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 5 min and redispersed in distilled water for further studies.
000 rpm for 5 min and redispersed in distilled water for further studies.
(f) Preparation of rhodamine-123 labelled-Au-CRC-TRC-NPs (rhod-123-Au-CRC-TRC-NPs)
Rhodamine-123 dye has active –NH2 sites, which could easily interact with –OH and or –COOH functionalities via hydrogen bonding. Rhodamine-123 encapsulation was performed to see the accumulation of nanoparticles in the cells by flow cytometry.The encapsulation of rhodamine-123 was performed by adding 40 μL dye in 1 mL precursor solution of chitosan-graft-PNVCL in acetic acid. The solution was then stirred for an hour and 1% TPP was added to enable the cross-linking reaction to form the final nanosuspension. The Au-NPs was directly added to this solution and centrifuged at 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 5 min to obtain the final pellets of rhodamine-123 encapsulated nanoparticles.
000 rpm for 5 min to obtain the final pellets of rhodamine-123 encapsulated nanoparticles.
(g) Synthesis of Indocyanin green (ICG) encapsulated Au-TRC-NPs (Au-ICG-TRC-NPs) and Au-CRC-TRC-NPs (Au-ICG/CRC-TRC-NPs)
ICG was encapsulated by dissolving 0.167 mg mL−1 ICG in 100% ethanol, which was then dissolved in the chitosan-graft-PNVCL/acetic acid solution. After a proper incubation time of 3 h, the whole solution was treated with 1% TPP solution, followed by 30 min stirring for an hour to obtain the nanosuspension. To this solution, Au-NPs was added and allowed to adsorb for 3 h. The resulting solution was centrifuged at 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 5 min, and redispersed in distilled water for further studies. The ICG was loaded similarly with Au-CRC-TRC-NPs as described above.
000 rpm for 5 min, and redispersed in distilled water for further studies. The ICG was loaded similarly with Au-CRC-TRC-NPs as described above.
(h) Leaching studies of ICG from Au-TRC-NPs and Au-CRC-TRC-NPs
The ICG was detected by IVIS Lumina (Xenogen, CA) with ICG excitation and Emission filters at an exposure value of one second after collecting the supernatants by centrifuging the ICG loaded nanoparticles at 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 5 min. The supernatants were collected at different time periods of day 1, 7, 14 and 30.
000 rpm for 5 min. The supernatants were collected at different time periods of day 1, 7, 14 and 30.
(i) The loading efficiency (LE) and encapsulation efficiency (EE)
The LE and EE were determined by centrifuging the drug-loaded nanoparticles at 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 30 min and separating the supernatant. The supernatant was assayed by a UV spectrophotometer (UV-1700 Pharma Spec, Japan) at 428 nm by dissolving it in ethanol. The calculated LE was 9.2% using the following formula:
000 rpm for 30 min and separating the supernatant. The supernatant was assayed by a UV spectrophotometer (UV-1700 Pharma Spec, Japan) at 428 nm by dissolving it in ethanol. The calculated LE was 9.2% using the following formula:The drug encapsulation efficiency (EE) was defined as the ratio of the mass of the encapsulated drug to the mass of the drug used for the nanoparticle preparation. The calculated EE was 92% using the following equation:
(j) In vitro quantification of curcumin
For in vitro quantification of curcumin, a standard solution of curcumin in ethanol was prepared by dissolving 5 mg of curcumin in 100 mL ethanol solution. A serial dilution from 0.2 to 2 mL was taken and diluted up to 25 mL and the system was assayed at 428 nm using a (UV-1700 Pharma Spec, Japan) UV spectrophotometer. The data was plotted to obtain a straight line for the quantification of the unknown drug in the nanoparticles.(k) Capacitively coupled RF heating of nanoparticle solutions
Fig. 2 shows the 13.56 MHz capacitive RF heating system (COMDEL, USA). A 35 × 10 mm Petri dish containing 4 mL of aqueous colloidal nanoparticles was positioned on a thin (∼0.5 cm) Teflon platform (not shown) located mid-plane between two insulated copper electrodes at a spacing of 7.5 cm. All suspensions were exposed to 40 W of RF generator power, resulting in a RF field of 15 kV m−1. Reflected RF power was monitored and minimized by impedance matching during RF exposure. The RF exposure system operated at a frequency of 13.56 MHz with an adjustable power output between ∼10 W and 1000 W. The RF generator/power amplifier (COMDEL, CX1250S/A, cooled RF generator) was connected through a type-N cable to a variable matching network (COMDEL/MATCH PRO CPMX 2500, CODEL INC), which matched the impedance of the power amplifier signal to the water cooled, solenoid antenna. Each sample was exposed to the RF signal at an amplifier setting at 40 W for 300 s. Temperature change of the solution was measured over time by an automatic digital thermometer. The background or unwanted heating was minimized by avoiding the use of a NaOH solution during the Au-NPs synthesis and replacing it with tris-buffer, which had no significant RF heating. The experiments were repeated several times to confirm the accuracy, as well the reproducibility under optimum RF conditions, i.e. RF exposure at 40 W for 300 s.(l) In vitro RF assisted curcumin release studies under optimum RF conditions
A known amount of lyophilized Au-CRC-TRC-NPs (50 mg) was dispersed in 10 mL deionized water (pH 7.4 adjusted by tris buffer solution) and the solution was added into 30 Petri dishes (500 μL each). These were then maintained under the RF chamber one by one and a RF pulse of 40 W for 5 min was given, followed by centrifugation of the released curcumin at 15000 rpm for 15 min, followed by dispersion in ethanol to quantify the released amount. The Au-CRC-TRC-NPs were exposed to RF pulses at predetermined time intervals of 5 min, 30 min, 1, 3, 6, 9, 12, 24, and 72 h to observe the RF-assisted curcumin release. The control CRC-TRC-NPs (LCST 42 °C) was also analyzed under RF conditions as above. The released curcumin was re-dissolved in 3 mL ethanol to assay it spectrophotometrically at 430 nm. The concentration of released curcumin was then calculated using the standard curve of curcumin in ethanol. The percentage of curcumin released was determined from the following equation:The same protocol was adapted for the RF-assisted drug release at 40 W for 5 min with pH 4.5. The samples were exposed under RF for 5 min at 40 W at predetermined time intervals. At each time point, an RF pulse of 40 W was given in all cases.
(m) Cell culture
4T1 (mouse mammary carcinoma cells) and IEC6 (mouse intestinal epithelial cells) were purchased from ATCC, USA. They were maintained in RPMI media supplemented with 10% fetal bovine serum (FBS). The cells were incubated with 5% CO2. After reaching confluency, the cells were detached from the flask with trypsin–EDTA. The cell suspension was centrifuged at 3000 rpm for 3 min, and then re-suspended in the growth medium for further studies.(n) Evaluation of cytocompatibility, cellular localization using confocal laser scanning microscopy (CLSM) and flow cytometry
For compatibility experiments, 4T1 and IEC6 cells were seeded on a 96 well plate with a density of 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cells per cm2. Different concentrations of the samples (0.1, to 1.0 mg mL−1) were prepared by diluting it with the media. For cellular localization studies, samples of concentration 1 mg mL−1 were used. Since Rhodamin-123 dye has innate green fluorescence, we stained the cells with anti-actin stain (TRITC-conjugated phalloidin dye), which is a red fluoresced dye, staining actin filaments of the cytoskeleton, so that the nanoparticles can be visualized well. Anti-actin dye was stained as per the manufacture's protocols. Confocal microscopy gives a better understanding of internalization of particles; thus, it was used to confirm the uptake of our samples by the cells. Since the laser for curcumin excitation (420 nm) was not available with our instrument, rhodamine-labelled samples were used (rhodamine-123 excitation at 511 nm and emission at 534 nm). Acid etched cover slips maintained in 24 well plates were seeded with 4T1 and IEC6 cells, respectively, with a seeding density of 2 × 104 cells per cover slip and incubated for 24 h to enable the cells to attach. After 24 h incubation, the media was removed and the wells were carefully washed with PBS buffer. Then, the particles at a concentration of 1 mg mL−1 were added along with the media in triplicate to the wells and incubated for a time period of 24 h. After the particular incubation time, anti-actin stain was added as mentioned above and the media with the sample was removed and the cover slips were processed for confocal microscopy. The processing involved washing the cover slips with PBS and fixing the cells in 3.7% paraformaldehyde (PFA), followed by a final PBS wash. The cover slips were air dried and mounted on to glass slides with DPX as the mountant. The slides were then viewed under a confocal microscope (Leica SP 5 II) to study the internalization of samples. IEC6 and 4T1 cells in its log phase were seeded at a density of 50
000 cells per cm2. Different concentrations of the samples (0.1, to 1.0 mg mL−1) were prepared by diluting it with the media. For cellular localization studies, samples of concentration 1 mg mL−1 were used. Since Rhodamin-123 dye has innate green fluorescence, we stained the cells with anti-actin stain (TRITC-conjugated phalloidin dye), which is a red fluoresced dye, staining actin filaments of the cytoskeleton, so that the nanoparticles can be visualized well. Anti-actin dye was stained as per the manufacture's protocols. Confocal microscopy gives a better understanding of internalization of particles; thus, it was used to confirm the uptake of our samples by the cells. Since the laser for curcumin excitation (420 nm) was not available with our instrument, rhodamine-labelled samples were used (rhodamine-123 excitation at 511 nm and emission at 534 nm). Acid etched cover slips maintained in 24 well plates were seeded with 4T1 and IEC6 cells, respectively, with a seeding density of 2 × 104 cells per cover slip and incubated for 24 h to enable the cells to attach. After 24 h incubation, the media was removed and the wells were carefully washed with PBS buffer. Then, the particles at a concentration of 1 mg mL−1 were added along with the media in triplicate to the wells and incubated for a time period of 24 h. After the particular incubation time, anti-actin stain was added as mentioned above and the media with the sample was removed and the cover slips were processed for confocal microscopy. The processing involved washing the cover slips with PBS and fixing the cells in 3.7% paraformaldehyde (PFA), followed by a final PBS wash. The cover slips were air dried and mounted on to glass slides with DPX as the mountant. The slides were then viewed under a confocal microscope (Leica SP 5 II) to study the internalization of samples. IEC6 and 4T1 cells in its log phase were seeded at a density of 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cells per cm2 into a 24-well plate. 1 mg mL−1 rhod-Au-CRC-TRC-NPs containing 25 μM were made by dilution with the media for this study. After attaining 90% confluency, the cells were washed with PBS buffer, and the abovementioned samples were added and incubated at 37 °C for 24 h. Intracellular rhod-123-Au-CRC-TRC-NPs fluorescence was analyzed by flow cytometry after excitation with a 488 nm argon laser using an FACS Aria II (Beckton and Dickinson, San Jose, CA). At 530/30 band pass and 502 long pass, fluorescence emission of 515–545 nm from 10
000 cells per cm2 into a 24-well plate. 1 mg mL−1 rhod-Au-CRC-TRC-NPs containing 25 μM were made by dilution with the media for this study. After attaining 90% confluency, the cells were washed with PBS buffer, and the abovementioned samples were added and incubated at 37 °C for 24 h. Intracellular rhod-123-Au-CRC-TRC-NPs fluorescence was analyzed by flow cytometry after excitation with a 488 nm argon laser using an FACS Aria II (Beckton and Dickinson, San Jose, CA). At 530/30 band pass and 502 long pass, fluorescence emission of 515–545 nm from 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cells was collected, amplified and scaled to generate a single parameter histogram.
000 cells was collected, amplified and scaled to generate a single parameter histogram.
(o) Anti-cancer efficacy of Au-CRC-TRC-NPs on 4T1 cells under optimum RF conditions
The MTT [3-(4,5-dimethylthiazole-2-yl)-2,5-diphenyl tetrazolium] assay for cytotoxic evaluation is a colorimetric test based on the selective ability of viable cells to reduce the tetrazolium component of MTT into purple-colored formazan crystals. After reaching 90% confluency, the cells were washed with PBS buffer and 1 mg mL−1 of sample was added and allowed for particle incubation of 24 h. The cells were then washed off with PBS for the RF experiments. RPMI10% contains lots of metallic ions, which could result in unwanted RF heating; therefore, to avoid this heating, we took a special media containing 40% FBS in deionized water for the RF exposure of cells and treated the cells at 40 W for 5 min. The special FBS media was further replaced with fresh RPMI and incubated further for 48 h. The treated and untreated cells were harvested and analyzed for the anti-cancer efficacy of Au-CRC-TRC-NPs. Cells in media alone, devoid of nanoparticles acted as a negative control and cells treated with Triton X-100 as a positive control for a period of 24 h. 5 mg of MTT (Sigma) was dissolved in 1 mL of PBS and the filter was sterilized. 10 μL of the MTT solution was further diluted to 100 μL with 90 μL of serum-free phenol red free medium. The cells were incubated with 100 μL of the above solution for 4 h to form formazan crystals by mitochondrial dehydrogenases. 100 μL of the solubilisation buffer (10% Triton X-100, 0.1 N HCl and Isopropyl alcohol) was added in each well and incubated at room temperature for 1 h to dissolve the formazan crystals. The optical density of the solution was measured at a wavelength of 570 nm using a Beckmann Coulter Elisa Plate Reader (BioTek Power Wave XS). Triplicate samples were analyzed for each experiment.(p) Apoptosis assay by flow cytometry
(q) Live/dead assay for RF treated and untreated samples
Live cells are distinguished by the presence of ubiquitous intracellular esterase activity, determined by the enzymatic conversion of the virtually nonfluorescent cell-permeant calcein AM to the intensely fluorescent calcein. The polyanionic dye calcein is well retained within live cells, producing an intense uniform green fluorescence (ex/em ∼495 nm/∼515 nm). EthD-1 enters cells with damaged membranes and undergoes a 40-fold enhancement of fluorescence upon binding to nucleic acids, thereby producing a bright red fluorescence in dead cells (ex/em ∼495 nm/∼635 nm). EthD-1 is excluded by the intact plasma membrane of live cells. The determination of cell viability depends on the physical and biochemical properties of the cells. Cytotoxic events that do not affect the cell properties may not be accurately assessed using this method. Background fluorescence levels are inherently low with this assay technique because the dyes are virtually non-fluorescent before interacting with cells. For live/dead assay tests, the cells were seeded on 6-well plates with a seeding density of 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cells. The samples were treated once they reached ∼80% confluency and assessed for RF experiments under optimum conditions. The RF exposed and unexposed samples were then stained with the live/dead assay kit and viewed under a fluorescent microscope for analysis.
000 cells. The samples were treated once they reached ∼80% confluency and assessed for RF experiments under optimum conditions. The RF exposed and unexposed samples were then stained with the live/dead assay kit and viewed under a fluorescent microscope for analysis.
(r) Blood compatibility studies
| Amount of plasma Hb (mg dL−1) = {(2A415) − [A380 + A450] × 76.25} |
The hemolytic property of nanoparticles was plotted as % hemolysis versus different concentrations of sample. Each concentration was evaluated in triplicate.
(s) HPLC procedure
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25 mixture of acetonitrile–5% acetic acid was used as the mobile phase and a C18 column (5 μm, 3 × 150 mm) was used. At a flow rate of 1 mL min−1, the retention time of curcumin was 1 ± 0.2 min. The detector was set at 429 nm. The peak area under the absorption time curve was noted for each sample and this was plotted against concentration to get the standard graph.
25 mixture of acetonitrile–5% acetic acid was used as the mobile phase and a C18 column (5 μm, 3 × 150 mm) was used. At a flow rate of 1 mL min−1, the retention time of curcumin was 1 ± 0.2 min. The detector was set at 429 nm. The peak area under the absorption time curve was noted for each sample and this was plotted against concentration to get the standard graph.The amount of curcumin detected in plasma and organ tissues were extracted as mentioned above and was quantified by a HPLC method. The sample collected was added to an equal volume of distilled ethanol and mixed well to dissolve the curcumin present in it. This was then centrifuged at 5000 rpm for 30 min, and the supernatant was filtered using a 22 μm syringe filter. 20 μL of this solution was injected into the HPLC system under the conditions mentioned earlier to determine the curcumin concentration.
(t) Analytical determinations
FT-IR spectroscopy of the materials were carried out using KBr tablets (1% w/w of product in KBr) with a resolution of 4 cm−1 and 100 scans per sample on a Perkin Elmer spectrum RX1 apparatus. The particle size was measured by dynamic light scattering (DLS-ZP /Particle Sizer Nicomp™ 380 ZLS) taking the average of 3 measurements. The surface morphology of nanoparticles was analyzed by SEM (JEOLJSM-6490LA) and TEM (Model: JEOL, JEM-2100F). In vivo live imaging followed by ex vivo tissue imaging were done using IVIS Lumina (Xenogen, CA) with ICG excitation and Emission filters at an exposure value of one second. ICP-AES was performed at CUSAT, STEC, Kerala, India.(u) Animal experiments
All animal experiments were performed according to the Ethics Committee of Chonnam National University Medical School and Chonnam National University Hospital, South Korea, Hwasun (CNU IACUC-H-2011-5). Athymic (nu/nu-ncr, Balb/c mice) 5–6 weeks old and 20–25 g weighed nude mice were used for the study. The mice were housed in sterile conditions in a laminar flow caging system (Thoren Caging Systems, Inc., Hazleton, PA), and all food, bedding, and water were autoclaved. The 4T1 cells were maintained in RPMI as described earlier. The 4T1 orthotopic tumor model was developed by orthotopic injection of 1 × 106 cells per 100 μL on the mammary fat pad.26 The animals were randomized in groups of 5 and the animals received i.v. injections of the control ICG, untreated animal, and Au-ICG-TRC-NPs and Au-ICG-CRC-TRC-NPs (low and high concentrations, i.e., 5 and 30 mg kg−1 of Au-CRC-TRC-NPs). The imaging was performed at different time periods from 5 min to one week. For analyzing the ex vivo tissue imaging, the animals were euthanized by cervical dislocation after anesthetizing in a gas anesthesia chamber containing isoflorane at a rate of 2.5% under oxygen supply. The organs, such as heart, liver, kidney, brain, stomach, intestine and spleen, were collected for imaging after wiping with saline (0.9%). Another set of samples of high concentrations via 30 mg kg−1 of Au-CRC-TRC-NPs and control curcumin were injected in tumor bearing mice (4 groups) to see the tumor localization after one and two weeks respectively.(v) Quantification of localized curcumin from Au-CRC-TRC-NPs
The tumor versus organ distribution of curcumin was evaluated by HPLC as described above.(w) Statistics analysis
The values were expressed in a format of mean ± standard deviation (SD). The results obtained were analyzed statistically. A Student's t-test was conducted to determine the significance. A probability level of p < 0.05 was considered to be statistically significant.Results
Different sized Au-NPs have been prepared using starch as the coating and D-glucose as the reducing agent in the presence of tris buffer to obtain different sized Au-NPs, as shown in the Fig. 4A–D. Depending on the size, the Au-NPs showed different colors starting from wine red to pale violet for 5 to 80 nm sized Au-NPs (Fig. 4A–H). This change in color is due to the difference in surface plasmon resonance (SPR) absorption of different sized Au-NPs. The Au-CRC-TR-NPs were prepared by incorporating 10 nm sized Au-NPs (Fig. 4I). The exact amount of Au-NPs in Au-CRC-TRC-NPs was found to be 17 ppm as per ICP-AES.The characteristic peak for Au-NPs at 520 nm (Fig. 4J) and curcumin's specific peak at 420 nm (Fig. 4L) were retained in the final nanosuspension of Au-CRC-TRC-NPs (Fig. 4K). The concentration of Au-NPs presented was validated by ICP and XPS studies. FTIR analysis was performed to see the potential interaction between the chemical moieties in the nanoformulation {Fig. 5A(a–d) and B(a–c)}.
 | ||
| Fig. 5 FTIR analysis for (A): (a) Au-NPs; (b) TRC-NPs; (c) Au-TRC-NPs; (d) curcumin; (e) Au-CRCC-TRC-NPs; (B): (a) ICG; (b) ICG/CRC-TRC-NPs and (c) Au-ICG/CRC-TRC-NPs, respectively. | ||
The control Au-NPs showed characteristic peaks of their starch coating at 3430 (–OH stretching vibrations), 2920 (–CH stretching vibration), 1740 (–CO stretching), 1640 (–OH bending vibration), 1462 (–CH deformation), 1375 (–C–CH3 stretching vibration), 1243 (–OH bending vibration), 1243 (–C–O (acetate)), 1100 cm−1 (–C–O stretching vibration), respectively (Fig. 5 A(a)). In Au-TRC-NPs (Fig. 5A(c)), there was a broadening at 3430 and sharpening at 1740 cm−1, which could be due to the combined stretching vibrations from the Au incorporated TRC-NPs. The peaks at 980 cm−1 also enhanced, which could be due to the presence of the aromatic ring structures of both TRC-NPs and starch coated Au-NPs. The increased –CH stretching vibrations could be attributed to the presence of starch coated Au-NPs on TRC-NPs.
In the control ICG (Fig. 5B(a)), the sharp and broad peaks at 3400 and 1740 cm−1 could be attributed to the –NH bending and –CO stretching vibrations, respectively. The 1410 cm−1 peaks representing the SO double bonds in ICG. In the Au-CRC-TRC-NPs, a sharp peak appeared at 1310 cm−1, which could be attributed to linking of the strong hydrogen bonds of CRC on Au-TRC-NPs via CRC's OH functionalities. This –OH perturbance in Au-TRC-NPs by the CRC was clear from –OH widening at 3430 cm−1 (Fig. 5A(c)). The Au-CRC/ICG-TRC-NPs showed an extra peak at 1629 cm−1, which could be due to the strong hydrogen bonding interaction of ICG with the Au-CRC-TRC-NPs Fig. 5B(c). Apart from this, peak broadening at 3430 cm−1 again confirmed hydrogen bonding within the nanosystems.
Particle size is the key, which can determine the circulation half-life and accumulation sites in the body.27 It can also influence the delivery volume and release characteristics for nanodrug carriers.28 TEM (Fig. 6A–C) images show the uniform spherical particles of Au-NPs. The size of Au-CRC-TRC-NPs was 160 ± 20 nm (Fig. 6D) and Au-ICG-CRC-TRC-NPs had 170 ± 15 nm (Fig. 6E). The plasma incubated Au-ICG/CRC-TRC-NPs showed almost same particle size of 170 ± 20 nm (Fig. 6F). The zeta potential for Au-CRC-TRC-NPs, Au-ICG/CRC-TRC-NPs and plasma incubated Au-ICG/CRC-TRC-NPs were found to be in the range of +18 ± 3, +19 ± 2 and +17 ± 5 mV respectively as shown in Table 1.
 | ||
| Fig. 6 A–C HR-TEM images for 10 nm sized Au-NPs at different magnifications; SEM analysis for (D) Control Au-CRC-TRC-NPs; (E) Au-ICG/CRC-TRC-NPs; (F) plasma incubated Au-ICG/CRC-TRC-NPs. | ||
| Samples | Particle size (nm) (DLS) | Particle size (nm) (SEM) | Zeta potential (mV) | Poly dispersity index (PDI) |
|---|---|---|---|---|
| Au-CRC-TRC-NPs | 170 ± 15 | 160 ± 20 | +18 ± 3 | 0.256 |
| Au-ICG/CRC-TRC-NPs | 185 ± 5 | 170 ± 15 | +19 ± 2 | 0.267 |
| Au-ICG/CRC-TRC-NPs (plasma incubated) | 180 ± 15 | 170 ± 20 | +17 ± 5 | 0.232 |
The cellular localization studies of Au-CRC-TRC-NPs were performed on 4T1 and IEC6 cell lines to see their internalization by CLSM Z-stacking, followed by flow cytometry. For this, we utilized CLSM z-stacking such that the exact internalization process can be validated properly. As visualized in the Fig. 7A and B, there was not much difference in internalization between the two cell lines, which are of epithelial origin. This was further validated by flow cytometry (Fig. 7C and D).
In general, NaOH is used to adjust pH in the preparation of Au-NPs; moreover, this could lead to extra heat generation due to the elemental contents of Na+ and Cl−. To avoid this situation, we used tris buffer to control the pH of the precursor solutions. The colloidal Au-NPs and Au-CRC-TRC-NPs were found to have RF power, as well as concentration dependent heating, as shown in Fig. 7E and F.
The cells exposed under RF along with RPMI media, can lead to extra heat due to the elemental contents in the media. Thus, we needed to replace the RPMI media with some other media while performing RF exposure for 5 min. After RF treatment, the cells can be maintained back in the same RPMI media for further analysis. With different trials, we found that only 40% FBS containing deionized water showed cell survival, which is significantly higher than that treated <40% FBS containing deionized water.
For conducting cellular experiments, we needed to replace the media (RPMI) with “specific media,” which does not contain many ions in it. Thus, 40% FBS containing deionized water was checked for its RF heating ability for 2 and 5 min, confirming its suitability to replace the cell media, which can avoid unwanted heating. Similarly, we checked the compatibility of this “special media” on IEC and 4T1 cells by the MTT assay after treating the cells with 40% FBS containing deionized water at 40 W for 5 min.
The RF-assisted drug release study was conducted in order to assess the effect of RF on the release properties. For this purpose, a higher LCST (42 °C) was used due to two important factors: (1) cells are more prone for cellular uptake and sensitive for treatment, and (2) most of the thermal treatments were performed at above 40 °C.29 As observed in Fig. 7G, the RF exposed samples were found to have curcumin release significantly higher than the un-exposed samples. The release rate was assessed with varying pH. The RF exposed samples with 4.5 pH had higher release compared to that with pH 7.4 (Fig. 7G and Fig. 8). However, even without RF, a significant amount of curcumin was released at an acidic pH of 4.5. The effect of Au-NPs on the RF assisted curcumin delivery was confirmed by exposing the control CRC-TRC-NPs alone under RF conditions as shown in Fig. 7G and H.
The Au-CRC-TRC-NPs were tested for its anti-cancer potential against 4T1 cells at optimum RF conditions. IEC6 cells were also tested as a control. There was no significant difference in cell death between the 4T1 and IEC6 cells. This nonspecificity could be explained due to the cell line origin, which is same for both 4T1 and IEC6 cells (epithelial cells). However, there was no significant toxicity shown by the treated cells without RF exposure (Fig. 9A and B).
The MTT results were further validated by a live/dead assay. As seen in Fig. 9C, the treated 4T1 cells on RF exposure showed a significant red fluorescence compared to the other samples, showing a bright green fluorescence, indicating that the cells are alive.
The apoptotic profile on 4T1 and IEC6 cells were studied after exposing Au-CRC-TRC-NPs treated cells at 40 W RF exposure for 5 min, followed by a pretreatment of 48 h prior to the apoptosis detection. There was not much difference in apoptosis between IEC6 and 4T1 cell lines (Fig. 10). To verify the effect of RF and LCST on apoptosis, we did the experiments without any RF exposure, which showed no significant apoptosis on both cell lines (Fig. 11).
The RF exposed 4T1 cells showed relatively higher apoptosis compared to that without RF exposure. There was ∼79 ± 3% apoptosis on RF exposed 4T1 cells compared to the unexposed samples. The unexposed samples had ∼10 ± 2% of apoptotic cells, which was significantly less compared to the RF exposed cells. The 1 mg mL−1 Au-CRC-TRC-NPs sample was found to be very effective as there was higher apoptosis on the 4T1 cells. The 10 ± 2% apoptosis in unexposed cells could be attributed to the pH-responsive delivery of curcumin within the cells.
It is mandatory to test the hemocompatibility of any drug delivery system prior to the in vivo assessments. Therefore, the Au-CRC-TRC-NPs with appropriate control samples were tested for blood compatibility by a series of experiments.
The interaction of Au-CRC-TRC-NPs with RBCs can be studied using the hemolysis test. In general, the interaction of RBCs with the nanoparticles depends on the size, surface charge and functionalities of the nanoparticles. In our study, the different concentrations of Au-CRC-TRC-NPs ranging from 0.01 to 10 mg mL−1 were exposed with the fresh human blood over a period of 6 h of incubation at room temperature. The % of hemolysis by the samples was tested by measuring the concentration of Hb in plasma obtained after centrifugation. As shown in Fig. 12A, compared to the positive control (Triton X-100 with ∼100% hemolysis), samples did not show any hemolysis up to 10 mg mL−1. These results were confirmed by taking the optical images shown in the Fig. 12B. The Triton-treated samples showed a red color due to Hb leakage in to the supernatant unlike the Au-CRC-TRC-NPs treated blood samples, which were shown to have a clear yellowish supernatant with unperturbed RBCs settling down at the bottom of the tubes. SEM analysis was performed to confirm that there was no damage to the RBCs as shown in the Fig. 12, D, and to show that the Au-CRC-TRC-NPs even with high concentrations were hemocompatible.
The basic function of hemostasis is to prevent loss of blood from an injured site and retain blood fluidity for smooth circulation. Circulating platelets and plasma proteins are key factors of the hemostatic system. Platelets have a size of ∼2 μm and are more fragile than RBCs. Platelets are responsible for the final step of hemostasis which is thrombus formation. The platelet-rich plasma (PRP) was incubated with the Au-NPs, CRC controls and Au-CRC-TRC-NPs to assess the platelet activation by monitoring the expression of granular membrane proteins (CD62P) and restricting the platelet surface membrane glycoprotein (CD42b) (Glycoprotein Ib). Phosphate-buffered saline (PBS) was used as a negative control.
The flow cytogram showed that PBS-treated platelets indicated a basal activation level of ∼3.4 ± 3% (Fig. 12E) and Au-CRC-TRC-NPs (with low and higher concentrations) treated platelets showed only ∼2.3% and ∼7.4 ± 1.1% activation (Fig. 12F and G), respectively, suggesting that the Au-CRC-TRC-NPs did not induce platelet activation. This was further confirmed through SEM imaging where the Au-CRC-TRC-NPs-treated platelets (Fig. 12H and I) remained discrete and maintained their discoidal shape.30
The above mentioned results were confirmed by conducting platelet aggregation potentials of the Au-CRC-TRC-NPs. To maintain hemostasis, the platelets with other blood components aggregate to form a clot to prevent excessive blood loss. However, a stroke can be expected if the platelet aggregation is excessive in nature.
The platelet count analysis was performed to confirm this platelet aggregation by the Au-CRC-TRC-NPs. The PRP treated Au-CRC-TRC-NPs were incubated for 30 min. Fig. 12K shows that while ADP-treated platelets yielded a significant reduction in the count, Au-CRC-TRC-NPs and all the controls, i.e. curcumin, and Au-NPs treated platelets maintained a normal count within the range of 50–250 K μL−1. This suggested that, as observed in the platelet activation study, neither CRC, Au-NPs nor Au-CRC-TRC-NPs caused platelet aggregation, confirming the effective evidence for their noninteraction with the function of platelets.
The mechanism of plasma coagulation consists of intrinsic and extrinsic pathways. These two pathways focused at a major point where factor X gets activated to Xa, activating prothrombin to thrombin, and subsequently converts fibrinogen to fibrin. It is well known that this kind of nanomaterial induced in vitro plasma coagulation studies can lay a better platform for the in vivo thrombogenic potential model. We assessed the possibility of Au-CRC-TRC-NPs interference with normal coagulation time using the prothrombin time (PT) and activated partial thromboplastin time (aPTT) tests. PPP isolated from freshly drawn human whole blood was exposed to different concentrations (0.2–0.4 mg/mL) of sample and analyzed. PT measurement detects abnormalities of the factors involved in the extrinsic pathway, including factors VII, X, V, II, and fibrinogen.
However, it was observed that PT values for all the tested concentrations fell well within the normal range of 12–15 s for all samples, as shown in Fig. 13A. Finally, we analyzed the effect of samples on intrinsic pathway of plasma coagulation, otherwise known as the contact activation pathway, which takes place during abnormal physiological conditions such as hyperlipidemic states or bacterial infiltration, which can lead to thrombosis. aPTT registers abnormalities in intrinsic clotting pathway factors, including I, II, V, VIII, IX, X, XI, XII, and proteins such as prekallikrein (PK), high-molecular-weight kininogen (HMWK), and fibrinogen. As shown in Fig.13B, the plasma treated samples were in the normal range of aPTT values, suggesting that it did not affect the intrinsic pathways.
Precisely, all the above results on hemostasis analysis show that Au-NPs, CRC and Au-CRC-TRC-NPs did not interfere with the normal functioning of platelets or influenced the coagulation pathways, and hence may remain non-thrombogenic. Thus, Au-CRC-TRC-NPs would be an ideal drug delivery agent for in vivo applications and can be directly injected into the blood stream via an intravenous mode of administration, which has more benefits than any other mode of administration, and thus there would be an enhanced therapeutic effect for bio applications.
Macrophages play an important role in the phagocytosis of foreign bodies, as well as in alerting the rest of the immune system against invaders, to elicit an innate or adaptive immune response. If the nanoparticles impart toxicity to macrophages, there will be significant impairment in the immune resistance capacity of the subjects. The Au-CRC-TRC-NPs treated macrophages did not show any uptake even after 6 h, as shown in Fig. 14.
 | ||
| Fig. 14 Macrophage response toward the Au-CRC-TRC-NPs on RAW 246.7 cells after 6 h of incubation period. | ||
4T1 cells are easily transplanted into the mammary gland such that the primary tumor grows in the anatomically correct site. As in human breast cancer, 4T1 metastatic disease develops spontaneously from the primary tumor. Moreover, the progressive spread of 4T1 metastases to the draining lymph nodes and other organs is very similar to that of human mammary cancer.31
As shown in the Fig. 15, the 4T1 tumor models started growing from day 3. There was sustained growth till day 21. The maximum tumor volume attained was ∼480 mm3 on 14th day.
 | ||
| Fig. 15 The growth progress of 4T1 orthotopic breast tumor model after (A) 3rd, (B) 5th, (C) 7th and (D) 14th days of tumor cells inoculations. | ||
However, the experiments were performed on a 4T1 tumor with a volume of 1500 mm3. From day 40, the mice showed an extended tumor on the back side of neck (nape), as shown in Fig. 16.
 | ||
| Fig. 16 Metastatic tumor derived from the primary 4T1 breast tumor. A perceptible secondary tumor is visible on the back side of the neck (nape). | ||
The leaching of ICG from Au-ICG-TRC-NPs and Au-ICG/CRC-TRC-NPs were studied by in vitro imaging using IVIS Lumina (Xenogen, CA) with ICG excitation and Emission filters at an exposure value of one second. It was necessary to confirm the leaching studies of ICG from the Au-CRC-TRC-NPs prior to its in vivo injection in the tumor mice models. Therefore, we tested the same with different time periods of one day to one month, suggesting that there was no significant amount of ICG leaching out from the Au-CRC-TRC-NPs. Fig. 17 shows the leaching studies from Au-ICG-TRC-NPs and Au-IGC/CRC-TRC-NPs.
The tumor localization of Au-TRC-NPs and the Au-CRC-TRC-NPs was studied by loading with the ICG dye as discussed previously. Au-ICG-TRC-NPs showed tumor localization within 24th h as shown in Fig. 18G. The samples were compared with the control ICG, which got eliminated after 6 h (Fig. 18D). The control animal (without any treatment) was also kept for avoiding food fluorescence. In the initial time of 5 min, all the Au-TRC-NPs were found to be in the liver. This was retained for 1 h, and then the Au-TRC-NPs moved toward the intestine with a slight intensity toward the tumor area. The same trends were sustained till 18 h, and then the intestine intensity was reduced and maximum tumor accumulation was observed (Fig. 18F). On the second day, the tumor localization was more clear as most of the Au-TRC-NPs clearly localized on the 4T1 breast tumor, as shown in the Fig. 18H. The tumor localization of Au-TRC-NPs was sustained even up to 7 days (Fig. 18I–J) with maximum retention as shown in the higher magnified images (Fig. 18K–L).
The Au-CRC-TRC-NPs were also analyzed for their tumor accumulation in a 4T1 orthotopic breast tumor model. The tumor localization was different from the control Au-TRC-NPs. However, the accumulation started from 6 to 18 h, as shown in Fig. 19 M–X. On day 1, there was prominent accumulation with retention of Au-CRC-TRC-NPs at the intestinal region. The accumulation retained was same as in 24th h till 72 h.
However, there was prominent distribution, both toward the tumor and intestinal regions from day 4. The 5th day also had the same trend; however, on day 7, maximum accumulation was observed in the tumor. The experiments were extended up to 2 weeks, confirming the accumulation of Au-CRC-TRC-NPs in the tumor for 2 weeks. This high retention of Au-CRC-TRC-NPs could be beneficial for a better regimen for cancer management. There was no reduction in the tumor as the TRC-NPs had the LCST in the range of 42 °C. This was expected as encapsulated curcumin could be released only when it attains the LCST at 42 °C. Thus, it was proven that the TRC-NPs could deliver curcumin only when it attained the maximum LCST range as described.
These results were further validated with ex vivo imaging by sacrificing the animals on day 7. As shown in Fig. 20A–C, the tumor had maximum accumulation, followed by the intestines and liver. Note that the other organs had no significant accumulation.
Further, these results were validated with HPLC quantification as given in Fig. 20D. The tumor accumulation was significantly higher at 3.6 μg g−1, whereas other organs did not contain a much amount of curcumin.
As shown in Fig. 21, the localization pattern of Au-CRC-TRC-NPs was different in the highly metastasized tumor model, compared to the normal orthotopic 4T1 tumor model. During the initial 30 min, the Au-CRC-TRC-NPs were found to be in liver followed by a drop in liver accumulation over time. Then, the intensity was higher in intestinal regions. By the 6 and 18 h, this intensity was found to be enhanced on the intestines with negligible intensity on other major organs.
However, on day 1, the primary tumor showed localization of the Au-CRC-TRC-NPs with a comparatively higher intensity and maximum localization in the intestinal region. The trend had been found to be same even in day 2. From day 3, there was a clear localization specifically on the tumor (Fig. 21(J)). The localization of the Au-CRC-TRC-NPs on the 3rd, 5th and 7th day is shown in the magnified images (Fig. 21 (M, N and O)). The top view images shown in the Fig. 22 clearly demonstrates the accumulation of Au-CRC-TRC-NPs on the secondary tumor, which developed on the back side of neck (nape). It was very clear that during the initial stages of 5 and 10 min, there was a slight intensity on the nape with maximum accumulation in the liver. However, this trend had been reduced after 30 min to 1 h. From the third hour onwards, the nape intensity was prominent, which further intensified at the sixth hour, as shown in Fig. 22(A–O). Thus, it was clear that the nape accumulation of Au-CRC-TRC-NPs was sustained for seven days (Fig. 22P).
Thus, it is evident that EPR is the key factor for nanoparticle accumulation in the breast tumor. Since the 4T1 orthotopic breast tumor model was highly metastasizing, the internal organs could also be accumulated with the Au-CRC-TRC-NPs. Ex vivo imaging was performed to prove this concept. Contrary to the in vivo imaging results on the seventh day, ex vivo imaging showed accumulation in the major organs, which could be due to the less penetrating capability of NIR into the internal organs (Fig. 23), whereas, the tumors were protruded, enabling an easy detection for NIR imaging. Ex vivo studies clearly showed that the 4T1 tumor must have metastasized into the liver, spleen, intestine and kidneys. Note that there have been reports for highly metastatic trends for 4T1 breast tumor models.32
Discussion
Chitosan-graft-PNVCL has sulphur functionalities in their PNVCL backbone. The incorporated Au-NPs would have strong binding with them.33 In addition, the unreacted amines and –OH functionalities could also have strong interactions with the incorporated Au-NPs, as shown in Fig. 1. The Au-NPs has SPR at 520 nm (Fig. 4J), which was evident even after incorporation with CRC-TRC-NPs (Fig. 4L), suggesting that there was no perturbance on its SPR. This could be due to the retention of Au-NPs without any changes in its structure within the CRC-TRC-NPs. The possible interaction of Au-NPs thus could be with the sulphur atom present in the chitosan-graft-PNVCL back bone. In the UV spectrum for Au-CRC-TRC-NPs, the peaks at 430 nm showed the presence of curcumin, which again confirmed the balance between two moieties such as Au-NPs and the CRC-TRC-NPs within a single nano-carrier. The optimized concentration of Au-NPs and curcumin for this balanced formulation was 17 ppm and 25 μM CRC. There have been reports for the strong interaction of Au-NPs with sulphur links, i.e. thiol groups.34 Thus, there would not be further leaching of Au-NPs from the TRC-NPs. On the other hand, chitosan-graft-PNVCL has strong unreacted amine functionalities, which could increase the hydrogen bonding with Au-NPs and would be more stable. Fig. 1 shows the expected interaction of Au-NPs within the nanosystem.It has already been reported that the permeability of tumor vasculature to nanoparticles is dependent on the tumor type.35 Some tumors have pore cut off sizes between 400 and 600 nm.36 Thus, ideally, the particle size must be below 600 nm for an effective passive delivery towards the tumor sites through the EPR effect.
The decrease in zeta potential for CRC-TRC-NPs from control TRC-NPs could be due to the higher loading efficiency of CRC. Similarly, after the Au-NPs incorporation, the zeta potential shifted to a lower value, due to the presence of –OH functionalities from the starch coated Au-NPs. The ICG-loaded Au-TRC-NPs had almost the same surface charges as the Au-CRC-TRC-NPs. Since both curcumin and ICG have similar functional groups, i.e. –OH functionalities, there would not be any changes in their surface charge. The mode of encapsulation of both curcumin and ICG were similar. To mimic the exact drug loaded TRC-NPs’ nature we used the hydrophobic ICG for imaging. Thus, the synthesized nanosystems have shown a particle size within the optimum range for an efficient drug delivery in vivo. Since our nanosystems were in the range of <250 nm, a better efficacy would be achieved in vivo.
Au-CRC-TRC-NPs did not undergo macrophage uptake, which could be explained on the basis of the surface charge as well as the chemical compositions of the chitosan-graft-PNVCL backbone. It has been reported that the particle size and surface hydrophilicity play an important role to determine the long-term circulation of nanoparticles in the blood stream.37 The larger particles can be rapidly taken up by the MPS (mono nuclear phagocyte system) cells present in the liver, spleen, and to a lesser extent, in the bone marrow.38 Hydrophobic surfaces are more prone to opsonization compared to hydrophilic nanoparticles.39 The surface charge of nanoparticles may also be responsible for suppressing plasma proteins approach. The opsonization existing between the complement proteins involved on the opsonization and the nanoparticles are more favorable to occur on the hydrophobic surfaces than on the hydrophilic ones.40 Au-CRC-TRC-NPs is expected to possess a hydrophilic surface with a high affinity for water due to the hydrophilic amines and carboxyl molecules, helping to introduce hydrophilic moieties that camouflage Au-CRC-TRC-NPs and make them invisible to phagocytes.
There have been many reports suggesting that the RF heating of Au-NPs could be due to the “Joule heating”. Precisely, the increased resistivity of small sized Au-NPs could impart a higher RF heating due to the increased surface to electron scattering, as Au-NPs' size is smaller than the mean free path of electrons in it. A similar trend in RF heating of Au-NPs and Au-CRC-TRC-NPs throws light on the fact that the Au-NPs did not lose its integrity even after incorporation with the CRC-TRC-NPs as proven by UV analysis, where we could see the significant SPR peak in the Au-CRC-TRC-NPs.
Since the Au-TRC-NPs have lot of unreacted amines and acetamide, it could easily swell in acidic environment, enhancing the pH responsive drug release as well. Thus, the TRC-NPs could release curcumin not only with the thermal stimulus, but also with the pH. However, there was a significant difference with RF and without RF in acidic pH environment. It was very clear that even with pH 7.4 and RF, the drug release was very high owing to the thermal stimuli responsive delivery of curcumin from the Au-TRC-NPs. The high release profile of curcumin above LCST could be basically explained by the increased entropic changes along with temperature. Since temperature is directly proportional to entropy (according to Gibb's free energy equation, ΔG = ΔH − TΔS), with increased temperature above LCST, the hydrogen bonding interaction of curcumin with TRC-NPs would be weak and the polymer-polymer interaction would be higher. Since there is a hydrophobic hydration, the released curcumin would have more affinity toward water than the Au-TRC-NPs.
Tager et al. have already reported certain thermo-dynamic conditions for a PNVCL solution in the vicinity of the LCST. This could be fulfilled when favorable conditions such as negative enthalpy ΔH < 0 and a negative entropy ΔS < O are attained.41 The partial Gibbs energy (ΔG), partial enthalpy (ΔH), and partial entropy (ΔS) of mixing PNVCL solutions were negative over the entire range of compositions. Increasing temperature resulted in a decrease in the exothermal character of mixing. Excessive heat capacity values, calculated from the dependencies of enthalpy of mixing on temperature, were positive over the entire composition range. The heat capacity of the dilute solutions was measured at 298 K and partial heat capacity of PNVCL at infinite dilution was shown to be positive. The data obtained pointed out the hydrophilic and hydrophobic hydration of PNVCL in aqueous solutions. Hydrophobic hydration dominates at temperatures close to the binodal curve. As a result, the mutual mixing of the polymer with water is decreased and phase separation takes place. This could be the reason why a prominent release was above the LCST with respect to the applied RF power. There are so many controversial results for the hemocompatibility associated with chitosan nanoparticles based on the mode of preparation or the solvents that were used. A basic understanding should be given to the sample preparation for hemocompatibility tests. In most of the cases, hemolysis would have happened due to the remnant solvents present in the nanoparticle system, which has to be drained out properly by high centrifugation at 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 10 min. The resulting pellet should always be redispersed in either saline or PBS to know the actual blood compatibility rather than mixing along with solvents for blood compatibility assessments. However, these issues could be avoided through proper modification via sulfonation, carboxymethylation or grafting. The resulting modified chitosan, thus, would be ideal for in vivo applications, including drug delivery. Thus, the synthesized Au-CRC-TRC-NPs could be an ideal drug delivery system for in vivo applications, in which hemocompatibility is an essential factor. Furthermore, maximum dosage of Au-CRC-TRC-NPs could be given intravenously since they are hemocompatible even up to 10 mg mL−1 concentrations.
000 rpm for 10 min. The resulting pellet should always be redispersed in either saline or PBS to know the actual blood compatibility rather than mixing along with solvents for blood compatibility assessments. However, these issues could be avoided through proper modification via sulfonation, carboxymethylation or grafting. The resulting modified chitosan, thus, would be ideal for in vivo applications, including drug delivery. Thus, the synthesized Au-CRC-TRC-NPs could be an ideal drug delivery system for in vivo applications, in which hemocompatibility is an essential factor. Furthermore, maximum dosage of Au-CRC-TRC-NPs could be given intravenously since they are hemocompatible even up to 10 mg mL−1 concentrations.
The tumor localization capability of passively targeted nanoparticles mainly depends on its size, shape and surface charge with chemical compositions.42 However, there are many parameters which plays a crucial role in tumor localization, including the EPR effect. The EPR effect can facilitate easy accumulation of the injected nanoparticles to a great extent. EPR is the key for many macromolecular and lipid based anti-tumor delivery agents. It has been reported that macromolecule based anticancer agents can substantially accumulate in a tumor,43 depending on the molecular weight of the anticancer agents. The macromolecules, in particular, those with slight higher molecular weights are well known for their tumor accumulation via the EPR effect.
The EPR effect for macromolecules has already been observed in many experimental and human solid tumors like Ehrlich carcinoma, mice colon 38 adenocarcinoma, Yoshida AH136B, Walker 2565 carcinoma and rat tumors, VX-2 carcinoma in rabbits and many human tumors (hepatoma, renal, lung and brain tumors).44
In metastasis, the cancer can spread from one organ or part to another non-adjacent organ or part. The new occurrences of the disease thus generated are referred as metastases.45 Breast cancer is characterized by a distinct metastatic pattern, involving the regional lymph nodes, bone marrow, lungs and liver. Tumour cell migration and metastasis share many similarities with leukocyte trafficking, which is critically regulated by chemokines and their receptors. 4T1 cancers are well known for its metastatic tumorigenicity similar to human breast cancers. As shown in Fig. 16, the primary tumor metastasized perceptibly toward the back side of neck (nape) and developed a tumor with 8.5 mm3 height.
It is crucial step to see the tumor localization in a metastatic model. As in many human breast cancers, metastasis is a major phenomenon to spread the tumor cells to other unaffected organs and body parts. Since the targeting of our Au-CRC-TRC-NPs was only via a passive mode, it has to be accumulated in the secondary tumors too. In addition, the 4T1 breast tumor can spread toward the lungs, spleen, kidneys, or intestine.46 Since EPR is the major key for tumor accumulation of nanoparticles, the Au-CRC-TRC-NPs accumulated well in organs, where angiogenesis was prominent through metastasis.
Conclusions
We demonstrated the feasibility of hemocompatible Au-CRC-TRC-NPs as an ideal thermo and pH-responsive RF-assisted curcumin delivery agent against breast tumor cells. They also showed potential anti-cancer efficacy on 4T1 tumor cells at optimum RF conditions. In addition, the developed Au-CRC-TRC-NPs were well accumulated in the primary and secondary tumors with maximum retention for seven days. This long term tumor retention of Au-CRC-TRC-NPs is ideal and could replace daily injections, enabling an enhanced efficacy toward the breast tumors.Acknowledgements
Dr R Jayakumar acknowledges the Department of Biotechnology (DBT), Government of India, for supporting this work under the Nanosciences & Nanotechnology program (Ref. no BT/PR10850/NNT/28/127/2008). Sanoj Rejinold acknowledges the Department of Science and Technology (DST, India) and Ministry of Science, Education and Technology (MEST), South Korea, for awarding “Indo Korea-Internship Programme-2013 (Award no: INT/ROK/IKRI-1/2013/6/2/2013)” and Council of Scientific and Industrial Research (CSIR) for the financial support through Senior Research Fellowship (SRF Award no: 9/963 (0017)2K11-EMR-I). The authors are thankful to Mr Sajin. P. Ravi (SEM studies), Sarath S. (confocal Imaging), Sreerekha P. R. (Flow cytometry), Dr A. K. K. Unni, Dr P. Reshmi, Sunil, Sajith, Shantini Narayanan, (Amrita Animal Lab), Dr T. V. Anil Kumar (SCTIMST Trivandrum), Lee Hwa Jeong, and Myeong Ju Moon (Department of Biomedical Science, Research Institute of Medical Sciences, Chonnam National University Medical School, Gwangju 501-746, South Korea) for their technical help.Notes and references
- K. M. Kelly, M. K. Shetty and J. H. T. G. Fregnani, Breast cancer screening and cervical cancer prevention in developing countries: strategies for the future, ed. M. K. Shetty, Breast and Gynecological Cancers, 2013, pp. 301–329 Search PubMed.
- S. Borges, H. R. Döppler and P. Storz, Breast Cancer Res. Treat., 2014, 144, 79–91 CrossRef CAS.
- N. S. Rejinold, R. Ranjusha, A. Balakrishnan, N. Mohammed and R. Jayakumar, RSC Adv., 2014, 4, 5819–5825 RSC.
- G. Ferro-Flores, B. E. Ocampo-García, C. L. Santos-Cuevas, E. Morales-Avila and E. Azorín-Vega, Curr. Med. Chem., 2014, 21, 124–138 CrossRef CAS.
- S. Halani, J. Coccagna, E. Chow, K. Hynynen and G. J. Czarnota, Bone Metastases: Cancer Metastasis-Biology and Treatment, ed. V. Vassiliou, E. Chow and D. Kardamakis, Springer, Netherlands, 2014, vol. 21, pp. 275–300, Series ISSN 1568-2102 Search PubMed.
- P. Pellegrini, A. Strambi, C. Zipoli, M. Hägg-Olofsson, M. Buoncervello, S. Linder and A. D. Milito, Autophagy, 2014, 10, 6–15 CrossRef.
- H. K. Patra and A. P. F. Turner, Trends Biotechnol., 2014, 32, 21–31 CrossRef CAS.
- D. Peer, J. M. Karp, S. Hong, O. C. Farokhzad, R. Margalit and R. Langer, Nat. Nanotechnol., 2007, 2, 751–760 CrossRef CAS.
- N. S. Rejinold, M. Muthunarayanan, V. V. Divyarani, P. R. Sreerekha, K. P. Chennazhi, S. V. Nair, H. Tamura and R. Jayakumar, J. Colloid Interface Sci., 2011, 360, 39–51 CrossRef.
- N. S. Rejinold, K. P. Chennazhi, S. V. Nair, H. Tamura and R. Jayakumar, Carbohydr. Polym., 2011, 83, 776–786 CrossRef CAS.
- D. Schmaljohann, Thermo- and pH-responsive polymers in drug delivery, Adv. Drug Delivery Rev., 2006, 58, 1655–1670 CrossRef CAS.
- N. S. Rejinold, T. Baby, K. P. Chennazhi and R. Jayakumar, Colloids Surf., B, 2014, 114, 209–217 CrossRef CAS.
- T. T. N. Vo, R. Yang, F. Aldabbagh, W. Carroll, M. Meere and Y. Rochev, J. Therm. Sci. Eng. Appl., 2014, 021012, 1–9 Search PubMed.
- B. Y. Swamy, J. H. Chang, H. Ahn, W.-K. Lee and I. Chung, Cellulose, 2013, 20, 1261–1273 CrossRef CAS.
- S. Shah, A. Pa and S. Devi, J. Appl. Polym. Sci., 2013, 127, 4991–4999 CrossRef CAS.
- K. Madhusudana Rao, K. S. V. Krishna Rao, P. Sudhakar, K. Chowdoji Rao and M. C. Subha, J. Appl. Pharm. Sci., 2013, 3, 60–69 Search PubMed.
- M. M. Yallapu, B. K. Gupta, M. Jaggii and S. C. Chauhan, J. Colloid Interface Sci., 2010, 351, 19–29 CrossRef CAS.
- P. Anand, A. B. Kunnumakkara, R. A. Newman and B. B. Aggarwal, Mol. Pharm., 2007, 4, 807–818 CrossRef CAS.
- R. V. Tikekar, Y. Pan and N. Nitin, Food Res. Int., 2013, 51, 370–377 CrossRef CAS.
- D. Patra, R. Aridi and K. Bouhadir, Microchim. Acta, 2013, 180, 59–64 CrossRef CAS.
- A. L. Palange, D. Di Mascolo, C. Carallo, A. Gnasso and P. Decuzzi, Nanomedicine (N. Y., NY, U. S.), 2014, 10, 991–1002 CAS.
- N. M. Khalil, T. C. Frabel do Nascimento, D. M. Casa, L. F. Dalmolin, A. C. de Mattos, I. Hoss, M. A. Romano and R. M. Mainardes, Colloids Surf., B, 2013, 101, 353–360 CrossRef CAS.
- P. Verderio, P. Bonetti, M. Colombo, L. Pandolfi and D. Prosperi, Biomacromolecules, 2013, 14, 672–682 CrossRef CAS.
- R. K. Gangwar, G. B. Tomar, V. A. Dhumale, S. Zinjarde, R. B. Sharma and S. Datar, J. Agric. Food Chem., 2013, 61, 9632–9637 CAS.
- E. Christian, H. S. Karsten, Z. Jingdong, C. W. Anna, S. J. Palle and U. Jens, J. Mater. Chem., 2009, 19, 7839–7847 RSC.
- S. N. Jiang, T. X. Phan, T. K. Nam, V. H. Nguyen, H. S. Kim, H. S. Bom, H. E. Choy, Y. Hong and J. J. Min, Mol. Ther., 2010, 18, 635–642 CrossRef CAS.
- A. Nagayasu, K. Uchiyama, T. Nishida, Y. Yamagiwa, Y. Kawai and H. Kiwada, Biochim. Biophys. Acta, 1996, 1278, 29–34 CrossRef.
- A. Ferdous, T. Ishida, M. Shinohara, H. Harashima and H. Kiwada, Biopharm. Drug Dispos., 1996, 17, 145–154 CrossRef CAS.
- J. A. Dickson and B. E. Oswald, Br. J. Cancer, 1976, 34, 262–271 CrossRef CAS.
- A. Anitha, N. S. Rejinold, J. D. Bumgardner, S. V. Nair and R. Jayakumar, Approaches for functional modification or cross-linking of chitosan, Chitosan-based systems for biopharmaceuticals: delivery, targeting and polymer therapeutics, John Wiley & Sons, Ltd, 2012, pp. 107–124 Search PubMed.
- B. A. Pulaski and S. Ostrand-Rosenberg, Mouse 4T1 breast tumor model, Curr. Protoc. Immunol., 2001 May, ch. 20, unit 20.2, DOI:10.1002/0471142735.im2002s39.
- L. Chen, T. G. Huang, M. Meseck, J. Mandeli, J. Fallon and S. LC Woo, Mol. Ther., 2007, 1512, 2194–2202 CrossRef.
- W. Wang, Q. Q. Wei, J. Wang, B. C. Wang, S. H. Zhang and Z. Yuan, J. Colloid Interface Sci., 2013, 15, 223–239 CrossRef.
- H. Hannu, Nat. Chem., 2012, 4, 443–455 CrossRef PubMed.
- F. Yuan, H. A. Salehi, Y. Boucher, U. S. Vasthare, R. F. Tuma and R. K. Jain, Cancer Res., 1994, 54, 4564–4568 CAS.
- F. Yuan, M. Dellian, D. Fukumura, M. Leunig, D. A. Berk, V. Torchilin and R. K. Jain, Cancer Res., 1995, 55, 3752–3756 CAS.
- G. Altankov, K. Richau and T. Groth, Materialwiss. Werkstofftech, 2003, 34, 1120–1128 CrossRef CAS.
- M. Gaumet, A. Vargas, R. Gurny and F. Delie, Eur. J. Pharm. Biopharm., 2008, 69, 1–9 CrossRef CAS.
- Y. Sheng, Y. Yuan, C. Liu, X. Tao, X. Shan and F. Xu, J. Mater. Sci.: Mater. Med., 2009, 20, 1881–1891 CrossRef CAS.
- S. Bruno, M. Donatella, C. B. Maria, P. N. Ana, C. M. Maria do and S. Vitor, Carbohydr. Polym., 2011, 84, 919–925 CrossRef.
- Q. Kun, T. Ge, L. Peng, C. Qiang, Z. Run, K. Yi-quan, X. Zhi-cheng and J. Xiao-dan, J. Neuroimmunol., 2012, 242, 9–15 CrossRef.
- A. Frank, P. Eric, K. M. Linda and C. F. Omid, Mol. Pharm., 2008, 5, 505–515 CrossRef.
- J. G. Adam, L. Nate and G. Hamidreza, Enhanced delivery of polymer therapeutics to solid tumors, tailored polymer architectures for pharmaceutical and biomedical applications, 2013, ch. 10, vol. 1135, pp. 151–185, ACS Symposium Series, ISBN13: 9780841227989e, ISBN: 9780841227996, DOI:10.1021/bk-2013-1135.ch010.
- H. Maeda, J. Wu, T. Sawa, Y. Matsumura and K. Hori, Tumor vascular permeability and the EPR effect in macromolecular therapeutics: a review, J. Controlled Release, 2000, 65, 271–284 CrossRef CAS.
- M. Anja, H. Bernhard, S. Hortensia, G. Nianfeng, C. Daniel, E. B. Matthew, T. McClanahan, M. Erin, W. Yuan, N. W. Stephan, L. B. Jose, M. Alejandro, V. Emma and Z. Albert, Nature, 2001, 410, 50–56 CrossRef.
- T. Kai, F. Min, A. Joseph and G. Gary Sahagian, BMC Cancer, 2008, 8, 228–233 CrossRef.
| This journal is © The Royal Society of Chemistry 2014 |