Cellular uptake, imaging and pathotoxicological studies of a novel Gd[III]–DO3A-butrol nano-formulation†
Elham Mohammadia,
Massoud Amanloub,
Seyed Esmaeil Sadat Ebrahimib,
Morteza Pirali Hamedanib,
Abdolkarim Mahroozc,
Bita Mehravid,
Baharak Abd Emamie,
Mohammad Reza Aghasadeghif,
Ahmad Bitarafan-Rajabig,
Hamid Reza Pour Ali Akbarh and
Mehdi Shafiee Ardestani*i
aDepartment of Biochemistry, Faculty of Biochemistry, Mazandaran University of Medical Sciences, Mazandaran, Iran
bDepartment of Medicinal Chemistry, Faculty of Pharmacy, Tehran University of Medical Sciences, Tehran, Iran
cMolecular and Cell Biology Research Center, Mazandaran University of Medical Sciences, Sari, Iran
dFaculty of Advanced Technologies in Medicine, Iran University of Medical Sciences, Tehran, Iran
eNational Cell Bank, Pasteur Institute of Iran, Tehran, Iran
fDepartment of Hepatitis and AIDS, Pasteur Institute of Iran, Tehran, Iran
gCardiovascular Interventional Research Centre, Department of Nuclear Medicine, Rajaei Cardiovasular, Medical & Research Center, Iran University of Medical Sciences, Tehran, Iran
hCardiovasular, Medical & Research Center, Radiology Department, Iran University of Medical Sciences, Tehran, Iran
iDepartment of Radiopharmacy, Faculty of Pharmacy, Tehran University of Medical Sciences, Tehran, Iran. E-mail: shafieeardestani@gmail.com; shafieeardestani@sina.tums.ac.ir; Fax: +98-021-66953311; Tel: +98-021-66953311
First published on 26th August 2014
Abstract
The high adaptability of dendrimer-based contrast agents (CAs) is ideal for the reliable molecular imaging of cancerous tissues. Gadobutrol (Gd3+[III]–DO3A-butrol or Gadovist) is a commercially produced contrast agent used in clinical settings. In this study, gadobutrol was nano-formulated by adding different ratios (X, 2X) of the anionic linear globular dendrimer G2 (gadobutrol–ALGD-G2 nano-formulation) to evaluate its intracellular uptake, pathotoxicity and use for in vitro and in vivo molecular imaging in cancer cells. Drug stability studies were carried out to ensure the correct formulation, after which the percentage of drug entry and levels of cytotoxicity (HEK cell line, KB cell line) were evaluated in cancer cells using the KB cell line. The intracellular uptake of gadobutrol–ALGD-G2 was measured quantitatively using inductively coupled plasma atomic emission spectrometry. The relaxometry of this CA and tumor imaging were determined using 1.5 T magnetic resonance imaging. The results indicated that the cellular uptake of gadobutrol–ALGD-G2 was about 71%. The r1 relaxation of this CA was measured as 4.75 mM−1 s−1 and on a per gadolinium [Gd3+] basis. In addition, the nano-formulated Gd[III]–DO3A-butrol was able to enter the KB cancer cells. The apoptosis assay and pathological evidence verified that the cells did not show any significant toxicity on exposure to the drug. The use of magnetic resonance imaging is feasible for the quantitative evaluation of the delivery of the drug and the detection of cancer cells. These results suggest that the new nano-formulation of gadobutrol–ALGD-G2 provides guidance for the selection of appropriate CAs for medical nanotechnology applications.
1 Introduction
Cancer is defined as the uncontrolled growth of unusual cells in humans or animals. Despite the large amount of effort devoted to its prevention, diagnosis and treatment, cancer is still one of the most significant causes of morbidity worldwide.1 Metastases are the most common reason for the unsuccessful treatment of cancer. In addition, many different types of cancer do not show any clinical symptoms in the early stages.2The early diagnosis or detection of tumors is one of the key factors in the treatment of malignancies. Many of the traditional methods of tumor treatment are unsuccessful and have a poor prognosis due to high costs, the side-effects of drugs and late diagnosis. Many cancer treatments are expensive and unpleasant for patients, so the best methods of cancer treatment rely on early detection by non-invasive techniques, including molecular imaging.3,4
Many pharmaceutical drugs have been developed for use in molecular imaging and as a safe, effective approach for the imaging of early stage cancers.3 Magnetic resonance imaging (MRI) is one of the most useful molecular imaging tools. MRI is a non-invasive imaging method for the diagnosis of pathologies (e.g. tumors) at an early stage. The basis of the MRI method is imaging of the hydrogen nucleus in water contained in tissues. The water content and relaxation ratios of protons are different in tumor tissues and in the surrounding normal tissues.1,5–7
Contrast agents (CAs) have been used in MRI for the detection and diagnosis of tumors. Gd3+[III]-based CAs are routinely used to improve diagnostic imaging. Gd3+–diethylene triamine-penta-acetic acid (DTPA)–dimeglumine [Magnevist] was the first CA approved for clinical use in MRI in 1988.8–10 There are many barriers to obtaining suitable specific molecular CAs, including: their low specificity, poor cellular uptake and the difficulty of providing sufficient relaxation of the cancerous tissues due to the extracellular distribution of the CA. There is therefore a need to design tumor-targeting CAs for use in MRI. There have been many published reports on the design and evaluation of novel molecules with specific membrane permeability.2,11–18
Many new developments in molecular imaging have been reported. One of these approaches includes the synthesis of covalently or non-covalently bound macromolecular nanoparticle-based Gd[III] ion chelates, such as dendrimers.3,7,19 Previous studies on the impact of Gd in MRI have shown that Gd results in the largest decrease in relaxation time (T1) and in the T2 value between compounds among the other available paramagnetic lanthanide metal ions, such as Fe, Cu and Mn.8 Compounds containing Gd are considered to be among the best CAs in MRI. Gd3+-DOTA (Gadovist; Gd[III]–DO3A-butrol or gadobutrol), a macrocyclic chelate, has a higher thermodynamic and kinetic stability than linear chelates such as Gd3+–DTPA (Magnevist), which is rapidly eliminated from the body after administration.20,21
A major limitation in the clinical application of small molecular Gd[III] chelates is the relatively low relaxation, but macromolecular Gd[III] complexes improve the pharmacokinetics and relaxation of Gd[III]-based agents. In addition, macromolecular Gd[III] complexes circulate in the blood for a long time and accumulate in tumor tissues by the EPR effect, allowing a long time span for imaging, and show none of the significant pathotoxicological effects seen with Magnevist or Gadovist.22,23
Enhanced relaxation of the Gd[III]-based CAs was observed with Gd[III] chelates attached to macromolecules such as nano-polymeric structures. Diverse chemically designed dendrimers have been increasingly used as the best candidates for the delivery of CAs in molecular imaging due to their biocompatibility and biodegradability. In addition, as a result of their controllable properties, such as water solubility, drug loading ability, monodispersity and large numbers of functional groups available for bioconjugation, dendrimers are of interest in drug delivery systems.3,7,24–27
In order to improve the molecular paramagnetic magnetic resonance CAs, many workers have demonstrated that chemically designed macromolecular dendrimer structures are able to load or even conjugate with multiple Gd3+-based chelates.11–16 Such highly branched chemically designed nanomaterials have a partial molecular weight (due to polymeric structure and polydispersity) and clearly identified numbers of terminal groups.17,18 Gd3+–DTPA molecules have been conjugated with poly(amidoamine) (PAMAM) and poly(propylene imine) (PPI) dendrimers by the incorporation of isothiocyanate-activated p-nitrophenylalanine-based DTPA chelates. Such dendritic magnetic resonance molecular imaging agents were found to be useful for in vivo molecular angiography, molecular lymphography, the assay of the distribution or clearance of molecular imaging contrast radiopharmaceuticals, and for target-specific imaging.16,19–22 In addition, the r1 relaxivity value significantly increases almost linearly with the molecular weight of the chemically designed dendrimer-based molecular CA developed as a conjugate or loaded by DTPA as a Gd3+-chelating agent.16
High toxicity, difficulties in the synthetic pathway and the high production costs of first-generation dendrimers such as PAMAM and PPI have limited their use. To improve these problems, two important strategies were employed: (1) the use of a biodegradable core of polyethylene glycol (PEG; low cost and readily available); and (2) surface engineering, e.g. a facile and one-pot synthetic pathway for acetic acid modification. In this work, PEG was used as a biodegradable, low-cost, readily available core. Citric acid was used as the surrounding group to produce an anionic linear globular dendrimer (ALGD); both of these molecules are completely and safely metabolized in the human body. PEG has been reported to be significantly accumulated in cancerous tissues without any targeting agent, whereas the citric acid surface makes the negatively charged dendrimer soluble. The negative charge of the citric acid groups in the surrounding dendrimer prevents interaction with the cell surface, avoiding damage to the cell membrane and subsequent pathotoxicological effects. Citric acid is quickly metabolized in the body, so it is biodegradable and non-toxic. Such dendrimers are therefore recognized as biodegradable and biocompatible nano-polymers with no significant toxicity.3,28,29
In this study, Gd[III]–DO3A-butrol, formally known as gadobutrol, was nano-formulated based on an ALGD G2 (GALGD). Its physicochemical properties and cellular uptake were studied; imaging and pathotoxicological studies were carried out in vitro and in vivo to investigate its use as a novel molecular tumor CA in MRI. In vivo, DTPA is a more unstable metal chelator than the DOTA incorporated in the gadobutrol chemical structure. As a result, such nano-formulations are more desirable than those reported previously.16–20
2 Results and discussion
2.1 Size and zeta potential distribution
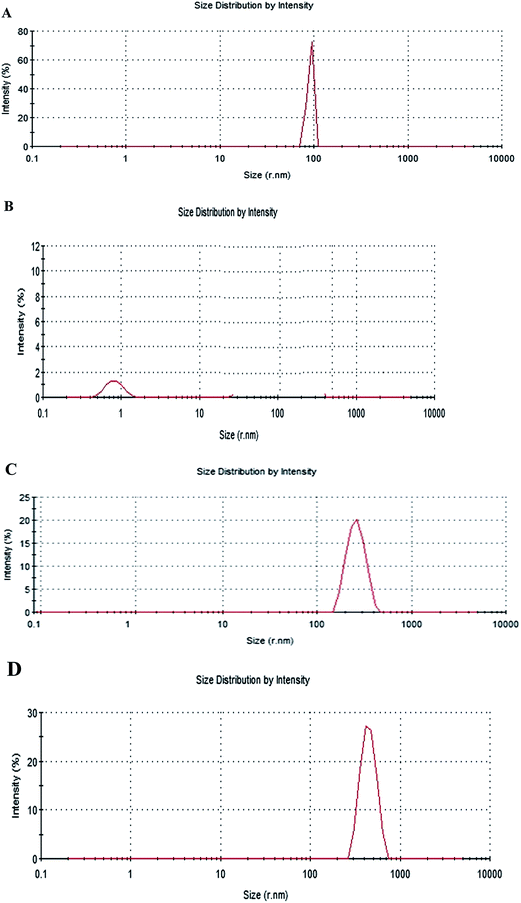
Table 1 gives the reduction zeta potential for gadobutrol after binding to dendrimers as a result of the existence of negative groups on the dendrimer surface. The zeta size was 90 nm for the G2 size dendrimer alone and 169.4 and 525.5 nm for the nano-formulation sizes G + XD and G + 2XD, respectively (Fig. 1). The same confirming evidence was obtained by AFM and SEM imaging (see ESI†), which showed roughly spherical particles (SEM after sonification) and larger particles on AFM (without sonification). This indicated the inherent ability of the nanoparticles to aggregate.| Zeta potential of gadobutrol (mV) | Zeta potential of GALGD2 [G + 2XD] (mV) | Zeta potential of GALGD2 [G + XD] (mV) |
|---|---|---|
| −5.50 | −18.5 | −12.7 |
2.2 Apoptosis assay
Apoptosis is a mode of cell death that occurs under normal physiological conditions and in which the cell is an active participant in its own death. No statistically significant response was seen in effect of GALGD-G2s (G + XD, G + 2XD) on apoptotic cells compared with gadobutrol (Fig. 2). Apoptotic and/or necrotic cells were determined using the Annexin V-propidium iodide staining kit using the manufacturer's protocols. Fig. 3 shows the percentage of apoptotic cells determined by flow cytometry. The same confirming evidence was observed using an MTT cellular assay (see ESI†).2.3 Intracellular uptake of GALGD into tumor cells
The affinity of tumor cells to the GALGD-G2 is shown in Fig. 4. The ICP-AES results show the cell entering level for GALGD-G2 (Gad + XD, Gad + 2XD) at two different concentrations is significantly increased in comparison to the unformulated gadobutrol.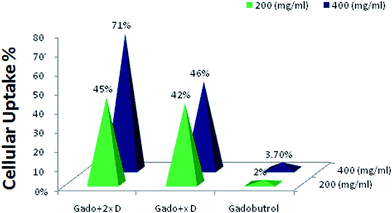 | ||
| Fig. 4 Cell uptake of GALGD-G2. The results show the effect of the dendrimer on intracellular uptake (p < 0.05). | ||
2.4 Relaxation
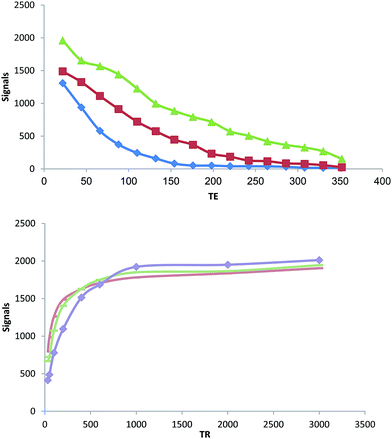
The MRI relaxation times for GALGD-G2 were measured using a 1.5 T MRI scanner. The GALGD-G2 showed large longitudinal (r1) and transverse (r2) relaxations. The r1 and r2 values were 4.57 mM−1 s−1 and 10.18 mM−1 s−1, respectively, while the r2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) r1 ratio was 2.3 (Fig. 5).
r1 ratio was 2.3 (Fig. 5).
2.5 Tumor imaging
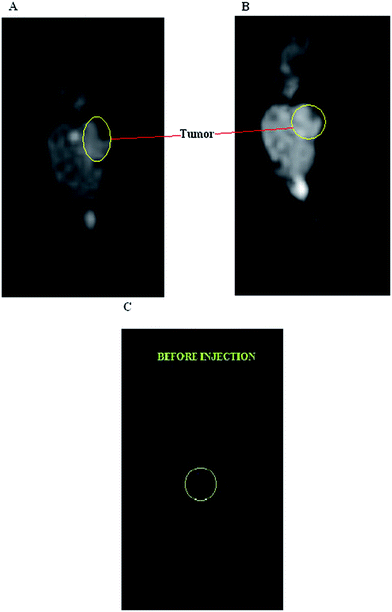
An MRI study was carried out in an animal model to evaluate the in vivo capability of the gadobutrol–GALGD-G2 nano-formulation and to discriminate between the tumor and normal tissues. The results indicated that images of the tumor were obtained a few minutes after the injection and the amount of GALGD-G2 internalized was large enough to have a significant effect on the MRI signal intensity (Fig. 6). Fig. 7 shows that the tumor is clearly visible after injecting the gadobutrol–GALGD-G2 nano-formulation.2.6 Pathological findings
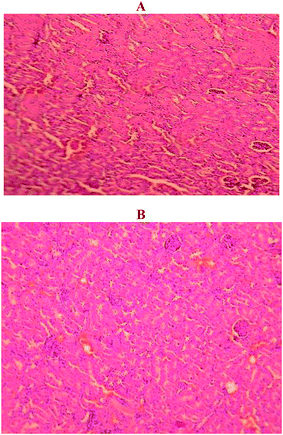
Fig. 8 shows that no significant pathological defect was observed in either the control or treated animals.2.7 Protein corona
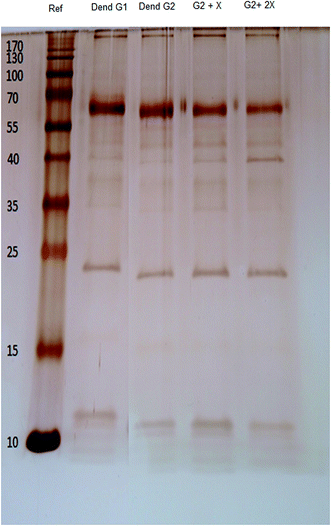
It has been shown that the interaction between nanoparticles and physiological fluids leads to the formation of a protein corona on the surface of dendrimer-based gadobutrol.32–34 The type and amount of associated proteins at the surface of nanoparticles may determine their biological fate. However, based on our results from SDS-PAGE, the protein profile of the nano-objects did not change the protein profile (Fig. 9).In this study, an ALGD was used as the nano-carrier and relaxation-enhancing agent for the Gd[III]–DO3A-butrol CA as it is a low-cost, readily available and safe nanoparticle with a facile synthetic pathway. The development of novel CAs that relatively or specifically accumulate at cancerous targets is important in cancer cell imaging, specifically in MRI. The nano-formulation of Gd[III]–DO3A-butrol dendrimer can be used to increase the specific accumulation of these nano-carriers within the target cancerous tissue by increasing the intracellular uptake and kinetic attraction of the PEG moiety to the tumor.
Recent studies have attempted to design novel synthetic strategies to reduce the toxicity of the dendrimers.35 In our experiment, PEG was used as a biodegradable core and citric acid was applied as the surrounding groups. PEG has been shown to accumulate in cancerous tissues without any manipulation of the targeting agent and it improves the solubility of the dendrimer.35
The release of the Gd3+ ion from the chelator may cause severe kidney damage.30–35 The previously reported nano-formulations often used DTPA as the chelator; the in vivo stability of DTPA is significantly lower than that of the class of macrocyclic chelators, such as the DOTA used in the current experiments. Our proposed nano-formulation will result in a more stable complex than the previously reported DTPA chelates. The negative charges surrounding the citric acid groups prevent any interaction with the cell membrane and therefore any subsequent cellular damage. Citric acid is quickly metabolized by the body and is therefore biodegradable and non-toxic.3,7,19,29
We assessed the intracellular uptake of the Gd[III]–DO3A-butrol–GALGD-G2 nano-formulation was using ICP-AES. The intracellular uptake of the gadobutrol–GALGD-G2 nano-formulation was also studied using transverse in vitro relaxations (r1) with increasing concentrations of labeled cells dispersed in homogenous phantoms. The in vitro studies on human cancer cell lines showed that the gadobutrol–GALGD-G2 nano-formulation is taken up by cells two to seven times more efficiently than that of Gd[III]–DO3A-butrol. However, this nano-formulation also leads to a significant cellular uptake of Gd3+ ions into cancerous cells.
The ALGD nano-polymer has numerous pores for loading drugs such as Gd[III]–DO3A-butrol. The loading ability could potentially be increased by adding more citric acid groups and increasing the generation of the dendrimer;36 therefore a second generation ALGD G2 was chosen as the nano-formulation base.
The standard gadobutrol drug is an extracellular contrast medium and cannot enter cells. However, based on the results of Gd[III]–DO3A-butrol loading into the dendrimer, significant amounts of the drug entered the cancerous cells. Receptor-mediated endocytosis is a proposed mechanism for the entry of dendrimers into cells; after entering the cell, the dendrimers are passed to the lysosomes for cellular digestion.25,30–37
The ICP-AES results showed that the intracellular uptake of the gadobutrol–GALGD-G2 nano-formulation by cancerous cells was on average 4.4 × 10−3 pg Gd3+ per cell, which correlates with other studies using serine-derived carbon nanotubes to label the KB cell line, Gd3+-based single-wall carbon nanotubes in a mouse macrophage cell line 37 and glucosylated Gd3+-based meso-porous silica nanospheres.15
Biocompatibility25–30 is a major concern when the gadobutrol–GALGD-G2 nano-formulation is introduced into cells. After the cells had been treated with the gadobutrol–GALGD-G2 nano-formulation, the results showed no significant change in normal cell viability compared with the control group. The results, including an apoptosis assay and MTT assay (see ESI†), agree with the results of previous studies.15,18
The relaxation data for the gadobutrol–GALGD-G2 nano-formulation designed as a novel molecular CA for MRI are important. The r1 and r2 values were 4.57 and 10.18 mM−1 s−1 in a phantom, respectively, and the r2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) r1 ratio was 2.3. This shows that the gadobutrol–GALGD-G2 nano-formulation is a good T1-weighted CA. We attributed the increased MR relaxation to the accessibility of water molecules at the Gd3+ moiety. The T1-weighted image enhancements were also related to the reduction in T1 relaxation times. Recent reports have used GD–ALGD2-C595 as a CA in breast cancer.15 These results showed the potential of GD–ALGD2-C595 in the selective and molecular targeting of breast tumor cells.7,19
r1 ratio was 2.3. This shows that the gadobutrol–GALGD-G2 nano-formulation is a good T1-weighted CA. We attributed the increased MR relaxation to the accessibility of water molecules at the Gd3+ moiety. The T1-weighted image enhancements were also related to the reduction in T1 relaxation times. Recent reports have used GD–ALGD2-C595 as a CA in breast cancer.15 These results showed the potential of GD–ALGD2-C595 in the selective and molecular targeting of breast tumor cells.7,19
In conclusion, the Gd[III]–DO3A-butrol nano-formulation has shown a more significant relaxation and cellular uptake than Gd [III]–DO3A-butrol alone and also similar previously reported Gd-based CAs, such as gadopentatedimeglumine, gadodiamide or their nano-conjugates.3,38 Some studies have reported higher relaxation values for more recent dendrimer-based nanoconjugate imaging agents than the Gd[III]–DO3A-butrol nano-formulation, but there is no significant difference between the Gd[III]–DO3A-butrol nano-formulation and other proposed CAs such as Gd3+–DTPA–PAMAM or PPI nano-conjugates.16 This is because the apparatus used (H-NMR or relaxometer) in those experiments is different from the current experiment (MRI); also, Gd3+–DTPA was conjugated to the chemical structure of the dendrimers, whereas we loaded gadobutrol onto the dendrimer. In addition, MRI relaxation data (which is similar to the clinical use of MRI radiopharmaceuticals) is usually obtained at higher levels than with H-NMR or relaxometers and, as a result, these data are not comparable. Previous studies used DTPA as a chelator for radioactive metals, for which the in vivo stability is significantly lower than for the DOTA used in the current experiments. The lower cost, facile synthetic pathway, biocompatibility and biodegradability of our ALGD-G2 makes it a promising chemically designated dendrimer for future use in clinical practice and in the manufacture of CAs for MRI. The effectiveness of the gadobutrol–GALGD-G2 nano-formulation was evaluated in vivo in a similar manner to other CAs for MRI. The results show the ability of the gadobutrol–GALGD-G2 nano-formulation to enhance the T1-weighted images. It demonstrated the efficacy of the gadobutrol–GALGD-G2 nano-formulation, as shown by the T1 CAs in vivo. The tumor images were taken a few minutes after the injection of the gadobutrol–GALGD-G2 nano-formulation and Gd[III]–DO3A-butrol alone into the tail vein of mice (0.2 mL per body weight), indicating the use of Gd[III]–DO3A-butrol–GALGD-G2 nano-formulation as a potential tumor and gastrointestinal CA for MRI.
3 Experimental methods
Fetal bovine serum (FBS), penicillin, streptomycin, phosphate-buffered saline (powder, pH 7.4) and adipic acid dihydrazide (≥98%) were purchased from Sigma-Aldrich (USA). Anhydrous ethanol (EtOH, 99.5%), anhydrous N and N-dimethyl formamide (99.8%) were provided by Acros Co. (Belgium). A dialysis bag with 500 Da cut-off was obtained from Spectrum Laboratory (USA). Gadovist (gadobutrol) was obtained from Bayer HealthCare Pharmaceuticals Inc. (Montville, NJ, USA). Dicyclohexylcarbodiimide (DCC), polyethylene glycol 600 (PEG), citric acid anhydride and Sephadex G-15 fine were purchased from Merck (Darmstadt, Germany).Cells from the Mouth Epidermal Carcinoma cell line (KB), Human Embryonic Kidney 293 cell line (HEK 293) and Murine Mammary Adenocarcinoma cells (MMAC) derived from the M05 cell line were provided by the National Cell Bank of the Pasteur Institute of Iran.
3.1 Instrumentation
The Gd3+ ions were determined using ICP-AES (JY138 Spectroanalyzer, Horiba JobinYvon, Inc., Edison, NJ, USA). The zeta potential of GALGD was determined using apparatus from Malvern Instruments (Malvern, Worcestershire, UK). Flow cytometry analyses were carried out using an Epics Altra Hyper sort flow cytometer (Beckman Coulter, Brea, CA, USA) with an air-cooled argon ion laser (488 nm, 15 mW). This standard instrument is equipped with two light-scattering detectors that measure the forward scatter as well as the side scatter. The data were analyzed using Beckman Coulter software.3.2 Animals
The animal experiments were conducted according to the relevant guidelines of Tehran University of Medical Sciences in agreement with the Helsinki Declaration. All inbred female BALB/c mice (6–8 weeks old, purchased from the Iran Pasteur Institute) were kept in large group-houses under 12 h dark and light cycles and were given access to food and water ad libitum.3.3 GALGD-G2 synthesis and gadobutrol–GALGD-G2 nano-formulation
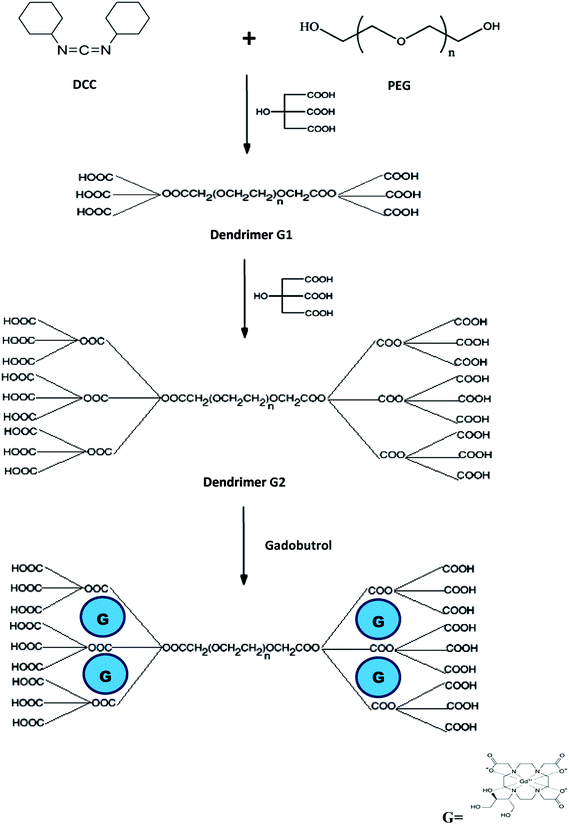
ALGD-G2 was synthesized according to previously reported methods with slight modifications.24,30 Compared with the previously reported methods, the present procedure has a better performance without the risk of using thionyl chloride (better biosafety and green chemistry) and the synthesis time was decreased.PEG-600 was selected as the core and was reacted with citric acid in the presence of excess amounts of dicyclohexylcarbodiimide in anhydrous N-dimethyl formamide. A dialysis bag was used for purification. Gel-filtration chromatography with Sephadex G-15 fine was used to purify the ALGD-G2. Each external 1 mL of eluent buffer for the reaction mixture was collected separately in a different tube and its UV–OD/iodine TLC spots were monitored to detect ALGD-G2.
To formulate the gadobutrol–GALGD-G2, 2 mL of standard gadobutrol (from commercially available Gadovist) were added to two different concentrations (7.26 mL, 100 mg dendrimer to provide gadobutrol + x dendrimer concentration; and 14.5 mL, 200 mg dendrimer to provide gadobutrol + 2x dendrimer concentration) of dendrimer and sonicated for at least 15 min. Excess gadobutrol was removed by dialyzing against PBS or, alternatively, by gel-filtration chromatography. The iodine TLC spots showed only one spot, which indicated that there was no starting material present. The schematic synthesis of GALGD-G2 and the gadobutrol–GALGD-G2 nano-formulation is shown in Fig. 10.
3.4 Cell culture
The Mouth Epidermal Carcinoma cell line (KB) and Human Embryonic Kidney cell line (HEK 293) were cultured at 37 °C in 5% CO2 using standard cell culture media containing Dulbecco's Modified Eagle Medium. The cell culture medium was supplemented with 10% FBS and 1% penicillin/streptomycin.3.5 Apoptosis/necrosis assay
Apoptosis is considered to be a specific mechanism for cell death. Apoptosis was determined using an Annexin V–propidium iodide staining kit following the manufacturer's recommendations. For the cell viability assay, the KB cell line (1 × 105 cells per well) and the HEK cell line (1 × 105 cells per well) were incubated with GALGD-G2 (gadobutrol + x dendrimer, 5 μL; gadobutrol + 2x dendrimer, 5 μL) and gadobutrol alone (5 μL) in a 24-well micro-plate for 24 h, with untreated cells as a positive control. Each concentration was tested in duplicate (Fig. 2 and 3).3.6 Nano-formulation intracellular uptake
To detect the intracellular uptake of the gadobutrol–GALGD-G2 nano-formulation at two concentrations (gadobutrol + x dendrimer and gadobutrol + 2x dendrimer), the KB cell line was re-plated into six-well plates at a concentration of 1 × 105 cells per well and incubated at 37 °C and 5% CO2 for 24 h. GALGD-G2 (0.2 and 0.4) and gadobutrol (0.2 and 0.4) were added to each well (1 mL media). Cells were incubated at 37 °C in 5% CO2 for 1.5 h, washed twice with PBS (500 μL) and then centrifuged at 2500 rev min−1 for 5 min and reconstituted in 100 μL of PBS. The intracellular uptake of Gd3+ ions was quantitatively determined by ICP-AES. The measurements were performed in duplicate and the mean ± SD of the results was calculated (Fig. 4). The same procedure was used for Gadovist.3.7 Evaluation of protein corona
One-dimensional SDS-PAGE (18%) was used to investigate the protein profiles of the corona-coated particles. A 20 μL aliquot of protein-loading buffer was added to 80 μL of the hard corona-coated nanoparticles. After boiling for 4 min, 15 μL of each sample were loaded into each well. Silver staining was performed to detect the protein bands. As temperature has a crucial effect on the composition of the protein corona, the incubation temperature was fixed at 37 °C.30,314 Magnetic resonance measurements and stability studies
The relaxation times of the gadobutrol–GALGD-G2 nano-formulation and Gadovist were measured at different concentrations using different spin echo and gradient echo protocols in a 1.5 T MRI scanner with a head coil. Multiple spin echo protocols were used for T2 measurements. In total, 32 echoes were obtained with an echo spacing of 2 ms. The first echo time (TE) was 22 ms with a TR of 3000 ms (matrix = 256 × 256; slice thickness = 1.5 mm, non-averaged). A FLASH protocol was used to calculate the T1 maps (TR = 32, 50, 100, 200, 400, 600, 1000, 2000 and 3000 ms; TE = 12 ms; matrix = 256 × 256; slice thickness = 1.5 mm, non-averaged). For quantitative analysis, the MRI images were analyzed by DICOM software, version 1.3.0.5. (MEDAV GmbH Co.). A similar procedure was performed 3 and 6 months after the formulation to investigate any unwanted instabilities that may have occurred.4.1 In vivo MRI preparation
Adult mice (provided by the Pasteur Institute of Iran and treated according to the Helsinki Declaration), injected with cancer cells and weighing about 20 g, were used for the in vivo study. Mouse 4T1 breast cancer cells were trypsinized and re-suspended in a 10-fold excess of culture medium. After centrifugation, the cells were re-suspended in serum-free medium and 1 × 106 cells were injected subcutaneously into the left flank of BALB/c mice under ketamine and xylazine (10 mg kg−1, i.p) anesthesia in a final volume of 0.1 mL using a 21-gauge needle. Tumor growth was visible 4–6 weeks after injection. Tumor images were obtained 5 min after injection of 0.2 mL per kg body weight of gadobutrol–GALGD-G2 nano-formulation into the tail vein (each 1 mL contained 12 mg of gadobutrol and Gadovist and each 1 mL contained 120 mg of gadobutrol) using the MRI scanner.185 Pathological findings
One week after imaging, all the injected animals were killed ethically and kidney tissues of both healthy and treated animals were collected in formalin and stained with H&E. All tissues were evaluated for any pathological defects.6 Statistical analysis
Multi-group comparisons were carried out by one-away ANOVA and the post hoc Tukey test. Statistical significance for cell toxicity was set at p < 0.05. The results are presented as mean ± SD values.7 Conclusion
The large and linear transverse relaxation of the Gd3+–DO3A-butrol–GALGD-G2 nano-formulation provides a CA with good sensitivity and low pathotoxicological effects for cellular and molecular imaging in 1.5 T MRI. Future experiments are needed to study the cellular uptake mechanisms of the Gd(III)–DO3A-butrol–GALGD-G2 nano-formulation and its subcellular localization. These findings provide helpful insights into the design of efficient CAs for nano-carrier delivery of drugs to cancerous tissues.Disclosure
This research forms the major part of an MS thesis in Biochemistry by Elham Mohammadi at the Mazandaran University of Medical Science. All authors declare that they have no conflicts of interest associated with this work.Acknowledgements
We deeply appreciate all the academic staff and technicians for their kind support during the course of these experiments.References
- J. Moscow and K. Cowan, Cecil Medicine, Saunders Elsevier Publications, Philadelphia, 23rd edn, 2007 Search PubMed.
- K. N. Raymond and V. C. Pierre, Bioconjugate Chem., 2005, 16(1), 3–8 CrossRef CAS PubMed.
- D. T. Mohamadi, M. Amanlou, N. Ghalandarlaki, B. Mehravi, M. Shafiee Ardestani and P. Yaghmaei, ISRN Pharm., 2013, 1–14, DOI:10.1155/2013/378452.
- P. Anand, A. B. Kunnumakara, C. Sundaram, K. B. Harikumar, S. T. Tharakan, O. S. Lai, B. Sung and B. B. Aggarwal, Pharm. Res., 2008, 25(9), 2097–2116 CrossRef CAS PubMed.
- D. Weishaupt, V. D. Köchli and B. Marincek, Springer, 2003.
- S. Koenig, R. Brown and R. Gupta, CRC Press, Boca Raton, FL, 1987.
- M. Mirzaei, M. Mohagheghi, D. Shahbazi-Gahrouei and A. Khatami, J. Biomater. Nanobiotechnol., 2013, 4(1), 22–29 CrossRef.
- H. J. Weinmann, R. C. Brasch, W. R. Press and G. E. Wesbey, AJR, Am. J. Roentgenol., 1984, 142(3), 619–624 CrossRef CAS PubMed.
- Z. Zhou and Z. R. Lu, Wiley Interdiscip. Rev.: Nanomed. Nanobiotechnol., 2013, 5(1), 1–18 CrossRef CAS PubMed.
- C. Westbrook, John Wiley & Sons, 2013.
- M. J. Allen and T. J. Meade, JBIC, J. Biol. Inorg. Chem., 2003, 8(7), 746–750 CrossRef CAS PubMed.
- R. Bhorade, R. Weissleder, T. Nakakoshi, A. Moore and C. H. Tung, Bioconjugate Chem., 2000, 11(3), 301–305 CrossRef CAS PubMed.
- P. Wunderbaldinger, L. Josephson and R. Weissleder, Bioconjugate Chem., 2002, 13(2), 264–268 CrossRef CAS PubMed.
- M. Lewin, N. Carlesso, C. H. Tung, X. W. Tang, D. Cory, D. T. Scadden and R. Weissleder, Nat. Biotechnol., 2000, 18(4), 410–414 CrossRef CAS PubMed.
- B. Mehravi, M. Ahmadi, M. Amanlou, A. Mostaar, M. S. Ardestani and N. Ghalandarlaki, Int. J. Nanomed., 2013, 8, 3209 CrossRef PubMed.
- B. Mehravi, M. Ahmadi, M. Amanlou, A. Mostaar, M. S. Ardestani and N. Ghalandarlaki, Int. J. Nanomed., 2013, 8, 3383 CrossRef CAS PubMed; S. Langereis, Q. Lussanet, G. E. W. Meijer, R. G. H. Beets-Tan, A. W. Griffioen, J. M. A. V. Engelshoven and W. H. Backes, NMR Biomed., 2006, 19, 133–141 CrossRef PubMed.
- C. H. Dodd, H. C. Hsu, W. J. Chu, P. Yang, H. G. Zhang, J. D. Mountz, K. Zinn, J. Forder, L. Josephson and R. Weissleder, J. Immunol. Methods, 2001, 256(1), 89–105 CrossRef CAS.
- B. Mehravi, M. S. Ardestani, M. Damercheli, H. Soltanghoraee, N. Ghanaldarlaki, A. M. Alizadeh, M. A. Oghabian, M. S. Shirazi, S. Mahernia and M. Amanlou, Mol. Imaging Biol., 2014, 1–10 Search PubMed.
- M. Mirzaei, M. Mohagheghi, D. Shahbazi-Gahrouei and A. Khatami, J. Nanomed. Nanotechnol., 2012, 3(7), 1000147–1000154 Search PubMed.
- E. Wiener, M. Brechbiel, H. Brothers, R. Magin, O. Gansow, D. Tomalia and P. Lauterbur, Magn. Reson. Med., 1994, 31(1), 1–8 CrossRef CAS.
- M. A. Perazella, Clin. J. Am. Soc. Nephrol., 2009, 4(2), 461–469 CrossRef CAS PubMed.
- M. Botta and L. Tei, Eur. J. Inorg. Chem., 2012, 12, 1945–1960 CrossRef.
- H. Daldrup, D. M. Shames, M. Wendland, Y. Okuhata, T. M. Link, W. Rosenau, Y. Lu and R. C. Brasch, AJR, Am. J. Roentgenol., 1998, 171(4), 941–949 CrossRef CAS PubMed.
- I. Haririan, M. S. Alavidjeh, M. R. Khorramizadeh, M. S. Ardestani, Z. Z. Ghane and H. Namazi, Int. J. Nanomed., 2010, 5, 63 CrossRef CAS.
- M. S. Alavidjeh, I. Haririan, M. R. Khorramizadeh, Z. Z. Ghane, M. S. Ardestani and H. Namazi, J. Mater. Sci.: Mater. Med., 2010, 21(4), 1121–1133 CrossRef CAS PubMed.
- B. Klajnert and M. Bryszewska, Acta Biochim. Pol., 2000, 48(1), 199–208 Search PubMed.
- P. Antoni, Y. Hed, A. Nordberg, D. Nyström, H. Von Holst, A. Hult and M. Malkoch, Angew. Chem., Int. Ed. Engl., 2009, 121(12), 2160–2164 CrossRef.
- K. Jain, P. Kesharwani, U. Gupta and N. Jain, Int. J. Pharm., 2010, 394(1), 122–142 CrossRef CAS PubMed.
- A. D'Emanuele and D. Attwood, Adv. Drug Delivery Rev., 2005, 57(15), 2147–2162 CrossRef PubMed.
- M. Mahmoudi, S. E. Lohse, C. J. Murphy, A. Fathizadeh, A. Montazeri and K. S. Suslick, Nano Lett., 2014, 14(1), 6–12 CrossRef CAS PubMed.
- M. Mahmoudi, A. M. Abdelmonem, S. Behzadi, J. H. Clement, S. Dutz, M. R. Ejtehadi, R. Hartmann and K. Kantner, ACS Nano, 2013, 7(8), 6555 CrossRef CAS PubMed.
- M. Mahmoudi, I. Lynch, M. R. Ejtehadi, M. P. Monopoli, F. B. Bombelli and S. Laurent, Chem. Rev., 2011, 111, 5610–5637 CrossRef CAS PubMed.
- V. Mirshafiee, M. Mahmoudi, K. Lou, J. Cheng and M. L. Kraf, Chem. Commun., 2013, 49(25), 2557–2559 RSC.
- M. Ghavami, S. Saffar, B. Abd Emamy, A. Peirovi, M. A. Shokrgozar, V. Serpooshan and M. Mahmoudi, RSC Adv., 2013, 3, 1119–1126 RSC.
- M. Mirzaei, M. Mohagheghi and D. Shahbazi-Gahrouei, J. Biomater. Nanobiotechnol., 2013, 4, 22–29 CrossRef.
- R. Abu-Rmaileh, D. Attwood and A. Emanuele, Dendrimers in cancer therapy, Drug Delivery Syst. Sci., 2003, 3, 65–70 CAS.
- A. M. Tang, J. S. Ananta, H. Zhao, B. T. Cisneros, E. Y. Lam, S. T. Wong, L. J. Wilson and K. K. Wong, Contrast Media Mol. Imaging, 2011, 6(2), 93–99 CrossRef CAS PubMed.
- N. Ghalandarlaki, T. D. Mohammadi, R. Agha Babaei, M. A. Tabasi, P. Keyhanvar, B. Mehravi, P. Yaghmaei, R. A. Cohan and M. S. Ardestani, Drug Res., 2014, 64(2), 57–65 CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra05596a |
| This journal is © The Royal Society of Chemistry 2014 |