Determination of critical micelle concentration of cationic surfactants by surface-enhanced Raman scattering
Abstract
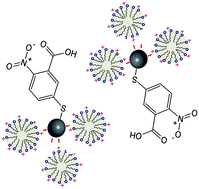
A novel approach, based on surface-enhanced Raman scattering (SERS) and 5,5′-dithiobis-(2-nitrobenzoic acid) (DTNB) functionalized silver nanoparticles, was developed to determine the critical micelle concentration (CMC) of cationic surfactants. A graph was generated by plotting the Raman intensity ratio between the aromatic ring vibration of DTNB at 1558 cm−1 and its symmetric nitro stretching at 1333 cm−1 as a function of surfactant concentration. An abrupt change of slope at a particular concentration was shown to reliably predict the CMC of cetyltrimethylammonium bromide (CTAB).

 Please wait while we load your content...
Please wait while we load your content...