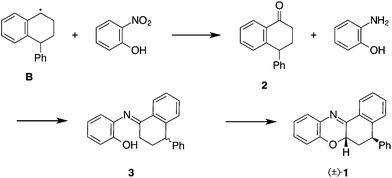
A new intermediate in spontaneous styrene polymerisation inhibited by 2-nitrophenol†
Robert J. Thatcher‡
a,
Thomas E. Newbya,
Peter Priceb,
Colin Loynsb and
Victor Chechik*a
aUniversity of York, Heslington, York, YO10 5DD, UK. E-mail: victor.chechik@york.ac.uk; Fax: +44 (0)1904-322516; Fax: (0); Tel: +44 Tel: 1904-324185
bNufarm UK Limited, Wyke Lane, Wyke, Bradford, BD12 9EJ, UK
First published on 29th July 2014
Abstract
The polymerisation inhibitor, 2-nitrophenol reacts with radicals generated in self-initiated thermal styrene polymerisation to form the unexpected benzoxazine 1. We propose the mechanism for the formation of 1 and show that it is produced by a radical mediated C–H activation.
1 Introduction
Styrene is arguably the archetypal feedstock for polymer synthesis, most notably for the production of polystyrene as well as a number of other commercially important co-polymers.1 In order to efficiently purify the monomer by distillation, it is necessary to treat styrene with additives to prevent spontaneous radical-initiated polymerisation. The common compounds used for this purpose are o-nitrophenols, such as 2,4-dinitrophenol (DNP), and 2,4-dinitro-6-sec-butyl phenol (DNBP), which, unlike other inhibitors, are effective even in the absence of oxygen. While there is a plethora of data available on the relative inhibition efficiencies of dinitrophenols, the underlying mechanisms of action for these compounds are largely unknown, save that they act as radical scavengers.2 The proximity of the nitro and phenol groups is key to the inhibition efficiency. For example, 2-nitrophenol acts as a reasonable inhibitor for styrene polymerisation, however 4-nitrophenol does not show any significant inhibition properties.In order to better understand the mechanisms behind the inhibition of styrene polymerisation with nitrophenols, we were motivated to isolate reaction products resulting from the inhibitor within the reaction mixture using the simple model compound, 2-nitrophenol. Herein we report the isolation of an unexpected byproduct of 2-nitrophenol inhibited styrene polymerisation, propose the mechanism of formation and discuss its relevance in the inhibition process. We also present an independent synthesis of the title compound from an intermediate imine via a radical initiated C–H activation step.
2 Results and discussion
The spontaneous polymerisation of styrene is initiated by radical formation under thermal conditions. Formation of diradical species A and radicals of the type B and C has been proposed by Flory and Mayo, respectively (Scheme 1), although the latter mechanism involving hydrogen abstraction from a Diels–Alder adduct by a styrene molecule is widely accepted as being the most important for the self-initiation of polymerisation.3–7Direct observation of species A–C has not been possible, with their existence inferred from isolation of their respective H˙ adducts.8,9 It is reasonable to suggest that in the absence of oxygen, inhibitors could intercept any of these radical species to suppress chain polymerisation, however, to date there are few fully characterised adducts of A, B or C with inhibitor molecules.10–12
Notably, Bushby and coworkers reported the detection of benzoxazole D (Fig. 1) from residues obtained at an industrial styrene fractionation still, and demonstrated its formation from reaction of styrene and the inhibitor 2,4-dinitro-6-sec-butylphenol (DNBP).12 The reaction was proposed to proceed via a condensation of intermediate benzaldehyde and DNBP-derived aniline.
 | ||
| Fig. 1 Isolated adduct from DNBP inhibited styrene polymerisation.12 | ||
Model reactions carried out by heating 2-nitrophenol in styrene at 110 °C were monitored by mass spectroscopy and revealed the development of peaks at m/z 110 and 312. The former can be attributed to the reduction of the nitro-group by benzyl radicals to give 2-aminophenol.13 NMR spectroscopy of the crude material showed a complex mixture of products mainly consisting of unreacted styrene and short chain styrene oligomers, as well as other unidentified species.
To isolate the major products of the inhibited polymerisation, the crude reaction mixtures were dissolved in diethyl ether and the organic phase washed with 5 N hydrochloric acid. The 1H-NMR spectrum of the crude residue obtained from evaporation of the acidic phase was identical to 2-aminophenol hydrochloride, confirming the formation of 2-aminophenol in the reaction mixture.
The organic phase was then washed with 5 N NaOH(aq) and brine. The filtrate was evaporated to dryness and the resulting oil purified by column chromatography using 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 DCM/hexane with the major product 1 eluting with an Rf of 0.33. Product 1 was obtained as a yellow oil and was characterised by a combination of mass spectrometry, IR and NMR spectroscopies. The high resolution ESI-MS of 1 exhibits a signal at 312.1370, compatible with the formula [C22H17NO+H]+ (expected m/z 312.1383) and corroborated by the 1H-NMR spectrum which shows 17 protons (Fig. 2). The IR spectrum of 1 shows a relatively weak absorbance at 1612 cm−1 which can be attributed to the C
1 DCM/hexane with the major product 1 eluting with an Rf of 0.33. Product 1 was obtained as a yellow oil and was characterised by a combination of mass spectrometry, IR and NMR spectroscopies. The high resolution ESI-MS of 1 exhibits a signal at 312.1370, compatible with the formula [C22H17NO+H]+ (expected m/z 312.1383) and corroborated by the 1H-NMR spectrum which shows 17 protons (Fig. 2). The IR spectrum of 1 shows a relatively weak absorbance at 1612 cm−1 which can be attributed to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N stretch of a conjugated imine, an assignment consistent with the signal observed in 13C-NMR spectrum at 158.5 ppm. The 1H-NMR spectrum recorded at 700 MHz indicates the presence of 13 protons in the aromatic region, which can be identified as two 1,2-disubstituted phenyl rings and one pendant phenyl ring from the COSY, HSQC and HMBC spectra.
N stretch of a conjugated imine, an assignment consistent with the signal observed in 13C-NMR spectrum at 158.5 ppm. The 1H-NMR spectrum recorded at 700 MHz indicates the presence of 13 protons in the aromatic region, which can be identified as two 1,2-disubstituted phenyl rings and one pendant phenyl ring from the COSY, HSQC and HMBC spectra.
The remaining four protons are identified as aliphatic from HSQC correlations to three carbon atoms, which is indicative of a diastereotopic –C(H)–C(H2)–C(H)– moiety. Collectively the data for 1 are consistent with the structure shown in Fig. 3 (full assignments and correlations are presented in ESI†). Interestingly, the assigned signal for H(10) at 8.65 ppm exhibits a line shape significantly distorted from the expected doublet of doublets pattern. This is attributable to a ‘virtual coupling’ effect due to disparate interaction with H(8) and H(9) which coincidently resonate at the same chemical shift. A similar distortion is observed for the H(7) signal. This proposed structure of 1 has two stereocentres, thus the NMR data may correspond to one of two pairs of diastereoisomers, with the relative configurations of the racemic mixture confirmed by single crystal X-ray diffraction.
 | ||
| Fig. 3 Molecular structure of 1 with numbering scheme. Ellipsoids drawn at 50% probability level. H atoms corresponding to aromatic rings omitted for clarity. | ||
Crystals suitable for X-ray diffraction were grown by slow evaporation of a diethyl ether–pentane solution of 1. Compound 1 crystallised in the P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) space group with the two stereogenic centres exhibiting opposite configurations. The pendant phenyl group adopts an axial position with respect to the cyclohexylene ring and a cis configuration with respect to the H(2) atom. The relative stereochemistry determined by diffraction is consistent with the 1H-NMR data for 1 as H(4) does not exhibit a strong trans coupling (JH–H > 7 Hz) with either H(3a) or H(3b) as would be expected if H(4) occupied an apical position.
space group with the two stereogenic centres exhibiting opposite configurations. The pendant phenyl group adopts an axial position with respect to the cyclohexylene ring and a cis configuration with respect to the H(2) atom. The relative stereochemistry determined by diffraction is consistent with the 1H-NMR data for 1 as H(4) does not exhibit a strong trans coupling (JH–H > 7 Hz) with either H(3a) or H(3b) as would be expected if H(4) occupied an apical position.
Compound 1 is clearly derived from a combination of the 2-nitrophenol inhibitor and the radical B. A plausible mechanism for the formation of 1 from these starting materials is given in Scheme 2. As reported by Mayo, styrene undergoes Diels–Alder dimerisation and a styrene molecule can abstract a hydrogen from the resulting adduct to give radical B.8 It has been reported that such radicals can be oxidised via the concomitant reduction of 2-nitrophenol to give the substituted cyclohexanone, 2.12,13
We propose that 2 reacts with 2-aminophenol to give the intermediate imine 3, which must then cyclise to give the final product, 1. The production of 1 under these conditions is particularly interesting as it requires the activation of a C–H bond on the incoming cyclohexyl ring. To justify the proposed mechanism, we sought to prepare compounds 2, 3 and 1 independently and study their reactivity. Compound 2 was conveniently prepared from E-cinnamic acid and benzene according to the procedure of Rendy and coworkers.14 The reaction of 2 with 3 equivalents of 2-aminophenol in the presence of 3 Å molecular sieves proceeded in toluene at 110 °C over 3 days to give the desired imine, 3. Aliquots of the reaction mixture were analysed by 1H-NMR indicating the emergence of a single new species and consumption of the starting material, however, the isolated yield of the imine product after work up was low. Compound 3 is air sensitive and is easily hydrolysed to give the starting ketone and 2-aminophenol and, as such, must be handled under an inert atmosphere.
Crystals suitable for X-ray diffraction study were grown by slow evaporation of a saturated pentane solution of 3. The obtained structure of 3 was solved in the P21/c space group and modelled as disordered about the cyclohexylene and phenyl moieties due to the presence of the diastereoisomer with inverted configuration at C(4) (Fig. 4 and ESI†).
 | ||
| Fig. 4 Molecular structure of 3. Major component of disordered structure shown. Ellipsoids drawn at 50% probability level. All except aliphatic H atoms omitted for clarity. | ||
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N distance of 1.286(2) Å typical for unprotonated imines.16 The imine group substituents adopt Z-geometry with the phenol ring lying at an angle of 71.36(12)° to the plane of the C–N
N distance of 1.286(2) Å typical for unprotonated imines.16 The imine group substituents adopt Z-geometry with the phenol ring lying at an angle of 71.36(12)° to the plane of the C–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C fragment. The imine N atom acts as a intermolecular hydrogen bond acceptor for the phenol –OH group of a neighbouring molecule (D–H⋯A d = 2.7303(17) Å, ∡ DHA 169(2)°), leading to a one-dimensional herring-bone motif parallel to the c-axis.
C fragment. The imine N atom acts as a intermolecular hydrogen bond acceptor for the phenol –OH group of a neighbouring molecule (D–H⋯A d = 2.7303(17) Å, ∡ DHA 169(2)°), leading to a one-dimensional herring-bone motif parallel to the c-axis.
The solid state IR spectrum of 3 exhibits two intense absorbances at 1617 and 1590 cm−1, attributable to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N and C
N and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching modes respectively, as well as a broad feature characteristic of a hydrogen bonded phenol O–H stretch at 2975 cm−1.17 The NMR data for compound 3 is consistent with a single configuration about the imine. With the exception of the phenolic proton there is a negligible change in the proton chemical shifts of 3 between 260 and 320 K as might be expected if there was isomerisation about the imine bond in this temperature range.
C stretching modes respectively, as well as a broad feature characteristic of a hydrogen bonded phenol O–H stretch at 2975 cm−1.17 The NMR data for compound 3 is consistent with a single configuration about the imine. With the exception of the phenolic proton there is a negligible change in the proton chemical shifts of 3 between 260 and 320 K as might be expected if there was isomerisation about the imine bond in this temperature range.
The reaction of imine 3 to give compound 1 is not observed under thermal conditions even after extended heating at 130 °C in toluene, and the cyclisation is most likely to be radically initiated by the thermally generated styrene radicals A, B and/or C. To support this view, a portion of imine 3 was added to a polymerising sample of styrene at 130 °C and the temperature was maintained for 24 hours. After heating, an aliquot of the reaction mixture was analysed by tandem mass spectrometry revealing the formation of a signal with identical MS/MS data to that of compound 1. Similarly, the reaction of 3 with dicumyl peroxide (DCP) in toluene at 120 °C leads to conversion of the starting material into a product for which a signal attributable to compound 1 is observed in the mass spectrum. The reaction of 3 with DCP proceeds with the formation of numerous impurities as gauged by TLC, however, compound 1 can be clearly observed in the 1H-NMR of the crude products (ESI†) confirming its formation under these conditions.
A similar cyclisation has been reported by Tauer and Grellman, who demonstrated that N-(1-o-tolyl-propylidene)-o-aminophenol is oxidized by air in the absence of light to give 2-methyl-3-o-tolyl-2H-1,4-benzoxazin-2-ol (Scheme 3) and proposed the formation of a hydroperoxide intermediate.18
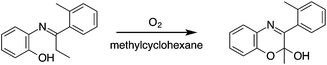 | ||
| Scheme 3 Oxidation of N-(1-o-tolyl-propylidene)-o-aminophenol to 2-methyl-3-o-tolyl-2H-1,4-benzoxazin-2-ol (modified from ref. 18). | ||
Formation of compound 1 from 3 cannot proceed by this process as it would lead to overoxidation at the C(2) centre, however, a plausible mechanism initiated by phenol H-abstraction is given in Scheme 4. Abstraction of the phenolic hydrogen is facilitated by the continuous generation of radicals A–C to give the intermediate 4.19 It follows that 4 can act as a H˙ donor for an incoming radical acceptor, such as styrene, to give the azadienone 5. Hydrogen abstraction is facilitated by the resonance stabilisation of azadienone 5 and this intermediate may then undergo an 1,6-electrocyclisation to give compound 1. Thermally-initiated electrocyclisations of similar azadienones have been reported in the literature.20
In contrast to the work of Bushby et al., we did not observe the formation of benzoxazoles similar to D. We note that the prior work used t-Bu peroxide initiated polymerisation that produced benzaldehyde as a by-product, and that the imine derivative of the benzaldehyde cannot form benzoxazine as it lacks the β-hydrogen. Presumably, in the self-initiated styrene polymerisation, oxidation of styrene to benzaldehyde is limited, which may explain formation of a different product (i.e., benzoxazine 1) in our case. We would like to note that our results were obtained under laboratory conditions and the isolation of the identified compounds from production streams would be required to test if these reactions occur at industrial plants.
3 Conclusions
We have isolated the benzoxazine 1 from 2-nitrophenol inhibited, thermally-initiated styrene polymerisation. The benzoxazine is proposed to result from the reaction of 2-aminophenol and a ketone derivative of a styrene dimer. This mechanism is supported by the independent synthesis of 1 from 2-aminophenol and the ketone via the proposed imine intermediate.Bushby's earlier report on dinitrophenol-inhibited styrene polymerisation described formation of a related benzoxazole which is also derived from the condensation of 2-aminophenol and a carbonyl compound (e.g., benzaldehyde). We conclude therefore that 2-nitrophenols intercept alkyl radicals formed during inhibited styrene polymerisation, and are reduced to 2-aminophenols while the incoming alkyl radicals are oxidised to the corresponding carbonyl derivatives. A condensation of 2-aminophenol with these carbonyl compounds leads to heterocyclic products that can be isolated from the polymerisation mixtures.
4 Experimental
4.1 General experimental considerations
With the exception of experiments involving styrene, which were carried out under aerobic conditions, all synthetic procedures were carried out under an Argon atmosphere using standard Schlenk procedures and glovebox technique. All solvents were dried using Innovative Technologies anhydrous solvent engineering system and degassed with argon prior to use. Styrene was purchased from Aldrich and was passed through an Alumina column to remove the commercial inhibitor additive before use. Aldrich brand 2-nitrophenol was dried in vacuo at 50 °C before use. Molecular sieves were heated at 230 °C for 24 h and stored under nitrogen prior to use. CDCl3 was dried over CaH2 and vacuum transferred prior to use. NMR spectra were recorded at the probe temperature on JEOL 400, Bruker AV-500 and Bruker AMX-700 instruments. Chemical shifts are described in parts per million, downfield shifted from SiMe4, and are consecutively reported as position (δ H or δ C), multiplicity (s = singlet, d = doublet, t = triplet, q = quartet, m = multiplet, br = broad, sep = septet ps. = pseudo), coupling constant (J in Hz), relative integral, and assignment. Assignments of the resonances are supported by 2D experiments. Proton NMR spectra were referenced to the chemical shift of residual proton signals. Carbon NMR spectra were referenced to a 13C resonance of the solvent. 1H–1H COSY, 13C HSQC and Gradient HMBC experiments were performed by using standard Bruker or JEOL pulse sequences. Infrared spectra were recorded on an Avatar 370 FT-IR Thermo Nicolet instrument. Electrospray ionization mass spectra and tandem MS/MS data were recorded on a Bruker Daltronic microTOF instrument. Major fragments are given as percentages of the base-peak intensity (100%). Diffraction data for 1 and 2 were collected at 110 K on an Agilent SuperNova diffractometer with Mo Kα radiation (λ = 0.71073 Å). Data collection, unit cell determination, and frame integration were carried out with “CrysalisPro” software. Absorption corrections were applied using crystal face-indexing and the ABSPACK absorption correction software within CrysalisPro. Structures were solved and refined using Olex2 implementing SHELX algorithms.21,22 Structures were solved by using the superflip algorithm23 and refined by full-matrix least-squares using SHELXL-97. All non-hydrogen atoms were refined anisotropically. Carbon-bound hydrogen atoms were placed at calculated positions and refined using a riding model. The O-bound H atom of 3 was located by difference map and allowed to refine without restraint.4.2 Compound 1, isolation from 2-nitrophenol inhibited styrene polymerisation
In a typical experiment, 3.5 g of 2-nitrophenol was heated in 50 mL of styrene at 110 °C for 72 h. After cooling, the reaction mixture was diluted with 250 mL diethyl ether and washed consecutively with 5 N HCl (3 × 300 mL), 5 N NaOH (3 × 300 mL) and brine (300 mL). Volatiles were removed under reduced pressure and the solid material obtained was then purified on a silica plug by elution with diethyl ether. Volatiles were removed under reduced pressure and the crude product was purified by column chromatography using 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 DCM/hexane to give 1 as a yellow oil. Rf 0.33. Typical yields: 200 mg.
1 DCM/hexane to give 1 as a yellow oil. Rf 0.33. Typical yields: 200 mg.
1H NMR (400 MHz, CDCl3): δ 8.59–8.62(m, 1H, H10), 7.46–7.50(m, 3H, H8, H9 & H15), 7.26(t, JH–H = 8 Hz, 2H, H19), 7.21–7.23(m, 1H, H7), 7.19(t, JH–H = 7, 1H, H20), 7.12(td, JH–H = 8, 1.5 Hz, 1H, H13), 7.04(td, JH–H = 8, 1.5 Hz, 1H, H14), 7.00(d, JH–H = 8, 2H, H18), 6.87(dd, JH–H = 8, 1 Hz, H12), 4.59(dd, JH–H = 4.3 Hz, 1H, H4), 4.54(dd, JH–H = 12, 5 Hz, 1H, H2), 2.79(ddd, JH–H = 12, 5, 3 Hz, H3i), 2.69(td, JH–H = 12, 5 Hz, 1H, H3ii); 13C NMR (100 MHz, CDCl3): δ 36.1(C3), 43.5(C4), 68.3(C2), 115.7(C12), 122.5(C14), 126.4(C10), 126.7(C20), 127.5(C15), 127.8(C8 or C9), 128.1(C18), 128.3(C13), 128.6(C19), 130.0(C7), 131.8(C8 or C9), 131.9(C6), 135.1(C16), 141.4(C5), 143.4(C17), 148.0(C11), 158.5(C1); ESI-MS: calcd for C22H17NO [M + H]+ m/z 312.1383, observed m/z 312.1370 (100%); IR (KBr): 1612 cm−1 (conjugated C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N).
N).
4.3 Compound 2
Ketone 2 was prepared according to the published procedure using 1 g of E-cinnamic acid, 20 mL toluene and 20 mL trifluoromethanesulfonic acid.14 Yield: 1.23 g (90%).1H NMR (400 MHz, CDCl3): δ 2.31(dddd, JH–H = 13.5, 9, 8, 4.5 Hz, 1H, CH), 2.48 (ddt, JH–H = 9, 8, 4.5 Hz, 1H, CH), 2.63 (ddd, JH–H = 17, 9, 4.5 Hz, 1H, CH), 2.74(ddd, JH–H = 17, 8, 4.5 Hz, 1H, CH), 4.31(dd, JH–H = 8, 4.5 Hz, 1H, CH), 6.99(d, JH–H = 7 Hz, 1H, p-Ph-H), 7.10–7.13(m, 2H, o-Ph-H), 7.27(tt, JH–H = 7, 1.5 Hz, HAr), 7.30–7.38(m, 3H, m-Ph-H + HAr), 7.44(td, JH–H = 7.5, 1.5 Hz, 1H, HAr), 8.12(dd, JH–H = 8, 1.5 Hz, 1H, HAr); 13C NMR (100 MHz, CDCl3): δ 31.9(CH2), 36.8(CH2), 45.3(CH), 126.8(CH), 127.1(CH), 127.1(CH), 128.7(CH), 128.7(CH), 129.6(CH), 132.8(Cq), 133.7(Cq), 143.7(Cq), 146.3(Cq), 198.1(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O); ESI-MS: calcd for C16H15O [M + H]+m/z 223.1117, observed m/z = 223.1112 (15%), calcd for C16H14ONa [M + Na]+ = 245.0937 (20%), observed m/z = 245.0936 (100%); IR (KBr): 1683 cm−1 (C
O); ESI-MS: calcd for C16H15O [M + H]+m/z 223.1117, observed m/z = 223.1112 (15%), calcd for C16H14ONa [M + Na]+ = 245.0937 (20%), observed m/z = 245.0936 (100%); IR (KBr): 1683 cm−1 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O).
O).
4.4 Compound 3
An ampoule was charged with 2 (217 mg, 0.98 mmoles), 2-nitrophenol (323 mg, 2.94 mmoles), 3 Å molecular sieves and toluene (4 mL), sealed and the reaction mixture heated at 110 °C for 3 days without stirring. After cooling, the supernatant was retained and the molecular sieves and remaining solids washed with DCM. The combined solvent layers were evaporated to dryness and the residue extracted with 10 mL diethyl ether. Solvent was removed in vacuo to give the product as a beige solid. Yield: 140 mg (46%).1H NMR (500 MHz, CDCl3): δ 2.12(dddd, JH–H = 13, 9, 7.5, 4 Hz, 1H, H3), 2.31(JH–H = 13, 9, 4.5 Hz, 1H, H3), 2.76(ddd, JH–H = 16.5, 9, 4 Hz, 1H, H2), 2.90(ddd, JH–H = 16, 9, 4 Hz, 1H, H2), 4.31(dd, JH–H = 7.5, 5 Hz, 1H, H4), 6.20(s, 1H, H(O)), 6.80(dd, JH–H = 8, 1.5 Hz, H12), 6.85(td, JH–H = 7.5, 1.5 Hz, H12), 6.99–7.02(m, 2H, H7 + H15), 7.09(ddd, JH–H = 8, 7, 1.5 Hz, H14), 7.13(ps. d, JH–H = 7 Hz, 2H, H18), 7.27(tt, JH–H = 7, 1 Hz, 1H, H20), 7.33(ps. t, JH–H = 7 Hz, 2H, H19), 7.35–7.39(m, 2H, H8 + H9), 8.44–8.47(m, 1H, H10); 13C NMR (126 MHz; CDCl3): δ 27.8(C2), 32.0(C3), 45.4(C4), 114.8(C15), 119.7(C13), 120.4(C12), 126.5(C14), 126.6(C10), 126.8(C20), 127.1(C9), 128.7(C19), 128.8(C18), 129.9(C7), 131.5(C8), 134.3(C6), 135.9(C11), 143.5(C5), 144.5(C17), 150.2(C16), 168.0(C1); IR (KBr): 2900–3600 (broad, OH & NH2), 1617 (s, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N), 1590 cm−1 (s, C
N), 1590 cm−1 (s, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N); ESI-MS: calcd for C22H20NO [M + H]+ m/z 314.1539, observed m/z = 314.1527 (100%).
N); ESI-MS: calcd for C22H20NO [M + H]+ m/z 314.1539, observed m/z = 314.1527 (100%).
4.5 Synthesis of compound 1 from 3
An ampoule was charged with 3 (70 mg, 0.22 mmoles), dicumylperoxide (135 mg, 0.50 mmoles) and toluene (3 mL), sealed in a pressure vessel and heated at 120 °C for 24 h. The reaction mixture was cooled and volatiles were removed under reduced pressure. ESI-MS of the reaction mixture showed the formation a signal attributable to 1. The crude solid was purified by preparative TLC using 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 DCM/Hexane. The desired product co-eluted with several other impurities as determined by 1H-NMR, however, compound 1 could be identified in the NMR spectra by comparison of the purified material from the styrene inhibition experiment (ESI†).
1 DCM/Hexane. The desired product co-eluted with several other impurities as determined by 1H-NMR, however, compound 1 could be identified in the NMR spectra by comparison of the purified material from the styrene inhibition experiment (ESI†).
4.6 Crystal data for compounds 1 and 3
![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) , a = 8.6089(7), b = 9.9620(9), c = 10.0051(8), α = 77.586(7), β = 79.970(7), γ = 71.479(8), V = 789.38(12), Z = 2, Dc = 1.310 g cm−1, F(000) = 328, μ = 0.080 cm−1, radiation Mo Kα, T = 110 K, 2θ limit = 60.046°, 15
, a = 8.6089(7), b = 9.9620(9), c = 10.0051(8), α = 77.586(7), β = 79.970(7), γ = 71.479(8), V = 789.38(12), Z = 2, Dc = 1.310 g cm−1, F(000) = 328, μ = 0.080 cm−1, radiation Mo Kα, T = 110 K, 2θ limit = 60.046°, 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 984 reflections observed, 3500 reflections used (I > 2.00σ(I)), number of variables = 217, R = 0.0395, Rw = 0.0969.†
984 reflections observed, 3500 reflections used (I > 2.00σ(I)), number of variables = 217, R = 0.0395, Rw = 0.0969.†Acknowledgements
We gratefully acknowledge the assistance of Dr Pedro M. Aguiar in the measurement of 700 MHz NMR spectra and Mr Karl Heaton for obtaining MS/MS data. TEN acknowledges Chemistry Innovation KTN and Nufarm for a CASE award to support this work.Notes and references
- J. Maul, B. G. Frushour, J. R. Kontoff, H. Eichenauer, K.-H. Ott and C. Schade, Polystyrene and Styrene Copolymers, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, Germany, 2000 Search PubMed.
- J. Brandrup, E. H. Immergut and E. A. Grulke, Polymer Handbook, Wiley-Interscience, 1999 Search PubMed.
- P. J. Flory, J. Am. Chem. Soc., 1937, 59, 241–253 Search PubMed.
- R. A. Gregg and F. R. Mayo, J. Am. Chem. Soc., 1953, 75, 3530–3533 CrossRef CAS.
- F. R. Mayo, J. Am. Chem. Soc., 1953, 75, 6133–6141 CrossRef CAS.
- Y. K. Chong, E. Rizzardo and D. H. Solomon, J. Am. Chem. Soc., 1983, 105, 7761–7762 CrossRef CAS.
- K. S. Khuong, W. H. Jones, W. A. Pryor and K. N. Houk, J. Am. Chem. Soc., 2005, 127, 1265–1277 CrossRef CAS PubMed.
- F. R. Mayo, J. Am. Chem. Soc., 1968, 90, 1289–1295 CAS.
- W. G. Brown, Die Makromolekulare Chemie, 1969, 128, 130–136 CrossRef CAS PubMed.
- K. R. Kopecky and M. C. Hall, Can. J. Chem., 1981, 59, 3095–3104 CrossRef CAS.
- G. Moad, E. Rizzardo and D. Solomon, Polym. Bull., 1982, 6, 11–12 CrossRef.
- R. J. Bushby, N. Lord, I. McRobbie and A. Pryce, Polymer, 1998, 39, 5567–5571 CrossRef CAS.
- R. A. Jackson and W. A. Waters, J. Chem. Soc., 1960, 1653–1657 Search PubMed.
- Y. Rendy, A. Zhang, A. McElrea, Gomez and D. A. Klumpp, J. Org. Chem., 2004, 69, 2340–2347 CrossRef PubMed.
- A. G. J. Ligtenbarg, R. Hage, A. Meetsma and B. L Feringa, J. Chem. Soc., Perkin Trans. 2, 1999, 807–812 Search PubMed.
- F. H. Allen, O. Kennard, D. G. Watson, L. Brammer, A. G. Orpen and R. Taylor, J. Chem. Soc., Perkin Trans. 2, 1987, S1–S19 RSC.
- D. Lin-Vien, N. B. Colthup and J. G. Grasselli, The Handbook of Infrared and Raman Characteristic Frequencies of Organic Molecules, Elsevier Inc., 1991 Search PubMed.
- E. Tauer and K. H. Grellmann, J. Org. Chem., 1981, 46, 4252–4258 Search PubMed.
- The abstraction of phenolic hydrogen by such carbon-centred radicals is possible but not facile. However, in the presence of oxygen these radicals will rapidly form peroxyl radicals which in turn will more efficiently abstract phenolic hydrogen.
- A. Khlebnikov, M. S. Novikov, A. F. Khlebnikov and N. V. Rostovskii, Tetrahedron Lett., 2009, 6509–6511 CrossRef PubMed.
- O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, J. A. K. Howard and H. Puschmann, J. Appl. Crystallogr., 2009, 42, 339–341 CrossRef CAS.
- G. Sheldrick, Acta Crystallogr., Sect. A: Found. Crystallogr., 2008, 64, 112–122 Search PubMed.
- L. Palatinus and G. Chapuis, J. Appl. Crystallogr., 2007, 40, 789 Search PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available: NMR data for compounds 1, 2 & 3 and CIFs for 1 and 3. CCDC 987438–987439. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c4ra05443a |
| ‡ Current address: University of Bristol, Dept. Physiology & Pharmacology, Medical Sciences Building, University Walk, Bristol, BS8 1TD. |
| This journal is © The Royal Society of Chemistry 2014 |