An efficient method for the synthesis of heteroaryl C–O bonds in the absence of added transition metal catalysts†
Katie Walsha,
Helen F. Sneddonb and
Christopher J. Moody*a
aSchool of Chemistry, University of Nottingham, University Park, Nottingham NG7 2RD, U.K. E-mail: c.j.moody@nottingham.ac.uk; Fax: +44 (0)115 951 3564
bGreen Chemistry Performance Unit, GlaxoSmithKline R&D Ltd, Medicines Research Centre, Gunnels Wood Road, Stevenage, Hertfordshire SG1 2NY, U.K
First published on 18th June 2014
Abstract
Reaction of 2-chloropyrazine and 2-chloropyrimidine with phenols and alcohols in the presence of K2CO3 in DMSO results in high yielding SNAr coupling. The reaction works particularly well with phenols and yields are comparable or superior to the metal-catalysed couplings described in the literature.
Introduction
The formation of aryl and heteroaryl C–O bonds in organic chemistry is an important synthetic tool, with the aryl ether products present in a variety of both natural and synthetic bioactive molecules. Examples include purmorphamine, which activates the Hedgehog-signalling pathway, an important regulator of stem cell renewal and cancer growth,1 and bispyribac-sodium, which is used as a herbicide (Fig. 1).2Traditionally the formation of these aryl C–O bonds was performed using nucleophilic aromatic substitution (SNAr), however poor substrate scope and reactivity has led to more elaborate synthetic strategies. For instance, the palladium-catalysed Buchwald–Hartwig reaction,3–5 or modern copper-catalysed variants on the classical Ullmann reaction.6–9
However, although the use of palladium and copper has substantially promoted these aryl C–O bond forming processes, these precious metals are still being employed for couplings involving more activated substrates where catalysis may be unnecessary; for instance, 2-chloropyrimidine is 1014 times more reactive than chlorobenzene towards SNAr reactions.10 Some recent examples of metal-catalysed couplings with these more activated heteroaryl halide substrates are shown in Scheme 1,11–14 with some examples demonstrating low loadings of the transition metals.15,16 Whilst these catalysed reactions can be carried out in good yield, the conditions remain quite forcing, and often require expensive ligands. In fact the use of transition metals may not be necessary to carry out these processes on such activated halides. As a consequence, we sought to develop an optimised approach to these C–O bond forming reactions that avoids the use of precious metals, which others have noted.17,18 In continuation of our interest in alternatives to transition-metal catalysed C–O and C–N coupling reactions, for example in amination chemistry,19 we now report the results of this study.
 | ||
| Scheme 1 Examples of palladium- and copper-catalysed formation of heteroaryl ethers [NMP = N-methylpyrrolidine]. | ||
Results and discussion
The reaction of 2-chloropyrimidine and p-cresol to give 2-(p-tolyloxy)pyrimidine 1 was used as a typical example, in order to carry out an initial investigation into the optimum choice of solvent and base (Table 1). We limited our solvent selection only to those that are generally accepted as ‘green’.20| Entry | Base | Solvent | Temp/°C | Yield/% |
|---|---|---|---|---|
| a All reactions were performed using 2-chloropyrimidine (1.75 mmol) and p-cresol (1.75 mmol), with the specified base (3.50 mmol) and solvent (1 mL), at the given temperature for 17 h.b Yield given as a conversion% as measured from 1H NMR integration. | ||||
| 1 | KF | 2-Methyl THF | 80 | — |
| 2 | KF | 1-Butanol | 80 | 19b |
| 3 | KF | Cyclopentylmethylether | 80 | — |
| 4 | KF | H2O | 80 | 24b |
| 5 | KF | DMSO | 80 | 40b |
| 6 | K3PO4 | H2O | 100 | 60b |
| 7 | K2CO3 | H2O | 100 | 54b |
| 8 | DBU | DMSO | 100 | 33b |
| 9 | K3PO4 | DMSO/H2O | 100 | 64 (72b) |
| 10 | K2CO3 | DMSO/H2O | 100 | 66 (71b) |
| 11 | K3PO4 | DMSO | 100 | 70 (78b) |
| 12 | K2CO3 | DMSO | 100 | 73 (78b) |
| 13 | Cs2CO3 | DMSO | 100 | 65 (69b) |
| 14 | K2CO3 | Propylene carbonate | 100 | 60b |
| 15 | K2CO3 | 2-Methyl THF | 80 | 64b |
| 16 | K2CO3 | 1-Butanol | 80 | 59b |
Reactions in 2-methyltetrahydrofuran, 1-butanol and cyclopentylmethylether (entries 1–3) gave poor recoveries, as did reactions in water using potassium fluoride as the base (entry 4), although these could be improved when switching to potassium carbonate and tribasic potassium phosphate (entries 6 and 7). Combinations of DMSO and water gave improved yields (entries 9 and 10) whilst the highest yields were obtained using DMSO, which also made the reaction mixtures much cleaner and easier to extract and purify. For extractions, isopropyl acetate was chosen since this is easier to recycle and recover on larger scale.
In terms of the base, inorganic reagents proved more effective than an organic base such as DBU (entry 8). Potassium fluoride (entries 1–5) gave poor recoveries whilst potassium and caesium carbonate bases and tribasic potassium phosphate gave good to excellent yields. Propylene carbonate (entry 14) was also tested as an alternative to DMSO, however not only was the yield lowered but removal of the propylene carbonate at the end of the process was difficult. On the other hand, 2-methyl-THF and 1-butanol performed relatively well (entries 15 and 16), and in the latter case there was no sign of any butoxypyrimidine showing the lack of reactivity of primary alcohols under these conditions (see below). It was found that by using two equivalents of potassium carbonate in DMSO at 100 °C for 17 h, the highest yield of 73% of 2-(p-tolyloxy)pyrimidine 1 was achieved (entry 9). Indeed this result was superior to the literature yield of 66% using copper catalysis under more forcing conditions.13
With the optimum conditions established, the reactivities of 2-chloropyrimidine and 2-chloropyrazine were tested against various phenols and primary alcohols (Table 2) to judge how the methodology compared to the metal-catalysed literature protocols shown in Scheme 1. The results show that reactions involving phenols (entries 1–6, 9–11) gave the highest yields. For the reactions with 2-chloropyrimidine (entries 1, 4–7) the products were obtained in directly comparable yields to those repeated in the literature; p-cresol for instance gave a yield of 73% compared to 66% under copper catalysis (Scheme 1).13
| Entry | Heteroaryl halide | Alcohol/phenol | Product | Yield/%b |
|---|---|---|---|---|
| a All reactions were performed using 2-chloropyrimidine or -pyrazine (1 eq.), alcohol (1 eq.) and K2CO3 (2 eq.) in DMSO at 100 °C for 17 h.b Yields in square brackets are for metal-catalysed literature couplings shown in Scheme 1. | ||||
| 1 |  |
 |
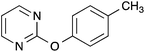 |
73 [66] |
| 2 |  |
 |
 |
63 |
| 3 |  |
 |
 |
72 |
| 4 |  |
 |
 |
67 [71] |
| 5 | 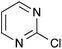 |
 |
 |
67 [76] |
| 6 |  |
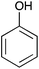 |
 |
65 [65, 97, 99] |
| 7 |  |
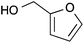 |
 |
51 |
| 8 |  |
 |
 |
35 |
| 9 |  |
 |
 |
93 |
| 10 |  |
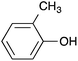 |
 |
91 [87] |
| 11 |  |
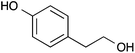 |
 |
87 [83] |
Reactions with simple primary and secondary alkyl alcohols, such as 2-(4-methoxyphenyl)ethanol, 3-octyn-1-ol or cyclohexanol, were examined but without success. Changing the base from potassium carbonate to potassium hydroxide or KHMDS (in THF) made no difference. Substitutions involving primary benzylic alcohols (entries 7 and 8) gave lower yields; 51% and 33% for furfuryl alcohol and benzyl alcohol respectively.
Interestingly, substitution reactions with 2-chloropyrazine (entries 9–11) gave superior yields than those with 2-chloropyrimidine (entries 1 and 2); for instance the coupling of 2-chloropyrazine with p-cresol gave an excellent yield of 93%. However in our previous amination chemistry,19 2-chloropyrimidine was found to generally give better yields as it is 102 times more reactive to nucleophilic substitution.21 Again, the greater reactivity of phenols over primary alcohols under these conditions is seen with the reaction of 2-(4-hydroxyphenyl)ethanol (entry 11).
Not only do the couplings have comparable and often superior yields to their metal-catalysed counterparts described in the literature that generally require the use of use temperatures up to 120 °C for 24 h (Scheme 1), compared to 100 °C for 17 h. Hence these conditions are not only greener in terms of solvent,20 cheaper in that potassium carbonate is less expensive than palladium or copper catalysts (plus complex ligands in some cases), but also more energy efficient. The potassium carbonate used was assayed by ICP-MS, and found to contain less than 20 and 75 ppb of palladium and copper respectively. Hence we are wary of claiming that our reactions are palladium- or copper-free, mindful of previous “palladium-free” Suzuki biaryl couplings, where the commercial sodium carbonate that contained 50 ppb palladium was possibly responsible for the biaryl formation,22 but nevertheless they proceed well in the absence of added transition-metal or ligands.
Conclusions
An efficient, green method to access aryl C–O bonds, without the need for added transition-metal catalysis is described. Results show that optimum yields were obtained using only potassium carbonate in DMSO at 100 °C. These conditions were tested on several phenols and alcohols. The best results were seen with phenols, which gave high yields comparable to those seen in the literature involving palladium and copper catalysis.Experimental
General experimental details
Commercially available reagents were used throughout without purification unless otherwise stated. Light petroleum refers to the fraction with bp 40–60 °C.Analytical thin layer chromatography was carried out on aluminium backed plates coated with silica gel, and visualized under UV light at 254 and/or 360 nm and/or by chemical staining. Flash chromatography was carried out using silica gel, with the eluent specified.
Infrared spectra were recorded using an FT-IR spectrometer over the range 4000–600 cm−1. NMR spectra were recorded at 400 or 500 MHz (1H frequency, 100 or 125 MHz 13C frequency). Chemical shifts are quoted in parts per million (ppm), and are referenced to residual H in the deuterated solvent as the internal standard. Coupling constants, J, are quoted in Hz. In the 13C NMR spectra, signals corresponding to CH, CH2, or CH3 groups are assigned from DEPT. Mass spectra were recorded on a time-of-flight mass spectrometer using electrospray ionization (ESI), or an EI magnetic sector instrument.
General procedure
To a Reacti-vial (Thermo Scientific) was added aryl halide (1 eq., 1.75 mmol), alcohol (1 eq., 1.75 mmol), reagent grade potassium carbonate (2 eq., 3.50 mmol) in dimethylsulfoxide (1 mL) and the resulting mixture heated to 100 °C for 17 h on a heating block. After cooling, the mixture was quenched with water (20 mL) and saturated potassium carbonate solution (10 mL), and extracted with isopropyl acetate (2 × 20 mL). The combined organic layers were then washed with brine (30 mL), dried over sodium sulfate and the solvent evaporated under reduced pressure. If necessary the residue was purified by chromatography on silica gel.2-(4-Methylphenoxy)pyrimidine 1
Following the general procedure, 2-chloropyrimidine (200 mg, 1.75 mmol), p-cresol (184 μL, 1.75 mmol) and potassium carbonate (483 mg, 3.50 mmol) in DMSO (1 mL) at 100 °C for 17 h gave after purification by chromatography (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 ethyl acetate/light petroleum) the title compound as a yellow oil (237 mg, 73%); (lit.,13 mp 70–71 °C); (found; M+, 187.0872. C11H11N2O requires 187.0866); δH (400 MHz; CDCl3) 8.58 (2H, d, J 4.8, 4H, 6H), 7.25 (2H, d, J 8.3, ArH), 7.11 (2H, d, J 8.3, ArH), 7.03 (1H, t, J 4.8, 5H), 2.39 (3H, s, CH3); δC (100 MHz; CDCl3) 165.6 (C), 159.7 (CH), 150.6 (C), 135.2 (C), 130.3 (CH), 121.4 (CH), 116.0 (CH), 21.0 (CH3). Data recorded matches literature.13
9 ethyl acetate/light petroleum) the title compound as a yellow oil (237 mg, 73%); (lit.,13 mp 70–71 °C); (found; M+, 187.0872. C11H11N2O requires 187.0866); δH (400 MHz; CDCl3) 8.58 (2H, d, J 4.8, 4H, 6H), 7.25 (2H, d, J 8.3, ArH), 7.11 (2H, d, J 8.3, ArH), 7.03 (1H, t, J 4.8, 5H), 2.39 (3H, s, CH3); δC (100 MHz; CDCl3) 165.6 (C), 159.7 (CH), 150.6 (C), 135.2 (C), 130.3 (CH), 121.4 (CH), 116.0 (CH), 21.0 (CH3). Data recorded matches literature.13
2-(2-Methylphenoxy)pyrimidine 2
Following the general procedure, 2-chloropyrimidine (200 mg, 1.75 mmol), o-cresol (180 μL, 1.75 mmol) and potassium carbonate (483 mg, 3.50 mmol) in DMSO (1 mL) at 100 °C for 17 h gave after purification by chromatography (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 ethyl acetate/light petroleum) the title compound as a yellow solid (238 mg, 67%); mp 76–78 °C (lit.,13 mp 77–78 °C); (found; M+, 187.0875. C11H11N2O requires 187.0866); νmax (CHCl3)/cm−1 3004, 1573, 1495, 1409, 1320, 1287; δH (400 MHz; CDCl3) 8.58 (2H, d, J 4.5, 4H, 6H), 7.32–7.27 (2H, m, ArH), 7.22–7.19 (1H, m, ArH), 7.15–7.13 (1H, m, ArH), 7.05–7.02 (1H, m, ArH), 7.03 (1H, t, J 4.5, 5H), 2.21 (3H, s, CH3); δC (100 MHz; CDCl3) 165.2 (C), 159.8 (CH), 151.3 (C), 131.4 (CH), 130.6 (C), 127.1 (CH), 125.9 (CH), 121.9 (CH), 116.0 (CH), 16.3 (CH3). Data recorded matches literature.13
9 ethyl acetate/light petroleum) the title compound as a yellow solid (238 mg, 67%); mp 76–78 °C (lit.,13 mp 77–78 °C); (found; M+, 187.0875. C11H11N2O requires 187.0866); νmax (CHCl3)/cm−1 3004, 1573, 1495, 1409, 1320, 1287; δH (400 MHz; CDCl3) 8.58 (2H, d, J 4.5, 4H, 6H), 7.32–7.27 (2H, m, ArH), 7.22–7.19 (1H, m, ArH), 7.15–7.13 (1H, m, ArH), 7.05–7.02 (1H, m, ArH), 7.03 (1H, t, J 4.5, 5H), 2.21 (3H, s, CH3); δC (100 MHz; CDCl3) 165.2 (C), 159.8 (CH), 151.3 (C), 131.4 (CH), 130.6 (C), 127.1 (CH), 125.9 (CH), 121.9 (CH), 116.0 (CH), 16.3 (CH3). Data recorded matches literature.13
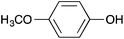
2-(4-Methoxyphenoxy)pyrimidine 3
Following the general procedure, 2-chloropyrimidine (200 mg, 1.75 mmol), 4-methoxyphenol (217 mg, 1.75 mmol) and potassium carbonate (483 mg, 3.50 mmol) in DMSO (1 mL) at 100 °C for 17 h gave after purification by chromatography (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 ethyl acetate/light petroleum) the title compound as a colourless solid (253 mg, 72%); mp 57–59 °C; (found; M+, 225.0640. C11H11N2O2Na+ requires 225.0640); νmax (CHCl3)/cm−1 3006, 2838, 1612, 1571, 1507, 1465; δH (400 MHz; CDCl3) 8.58 (2H, d, J 4.7, 4H, 6H), 7.15 (2H, d, J 9.2, ArH), 7.03 (1H, t, J 4.7, 5H), 6.97 (2H, d, J 9.2, ArH), 3.84 (3H, s, CH3); δC (100 MHz; CDCl3) 165.7 (C), 159.7 (CH), 157.1 (C), 146.3 (C), 122.5 (CH), 116.0 (CH), 114.7 (CH), 55.6 (CH3).
4 ethyl acetate/light petroleum) the title compound as a colourless solid (253 mg, 72%); mp 57–59 °C; (found; M+, 225.0640. C11H11N2O2Na+ requires 225.0640); νmax (CHCl3)/cm−1 3006, 2838, 1612, 1571, 1507, 1465; δH (400 MHz; CDCl3) 8.58 (2H, d, J 4.7, 4H, 6H), 7.15 (2H, d, J 9.2, ArH), 7.03 (1H, t, J 4.7, 5H), 6.97 (2H, d, J 9.2, ArH), 3.84 (3H, s, CH3); δC (100 MHz; CDCl3) 165.7 (C), 159.7 (CH), 157.1 (C), 146.3 (C), 122.5 (CH), 116.0 (CH), 114.7 (CH), 55.6 (CH3).
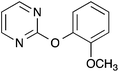
2-(2-Methoxyphenoxy)pyrimidine 4
Following the general procedure, 2-chloropyrimidine (200 mg, 1.75 mmol), 2-methoxyphenol (195 μL, 1.75 mmol) and potassium carbonate (483 mg, 3.50 mmol) in DMSO (1 mL) at 100 °C for 17 h gave after purification by chromatography (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 ethyl acetate/light petroleum) the title compound as a yellow solid (238 mg, 67%); mp 118–120 °C (lit.,13 mp 121–122 °C); (found; M+, 203.0822. C11H11N2O2 requires 203.0815); νmax (CHCl3)/cm−1 3009, 1607, 1573, 1501, 1465, 1439; δH (400 MHz; CDCl3) 8.57 (2H, d, J 4.7, 4H, 6H), 7.27–7.26 (1H, m, ArH), 7.25–7.24 (1H, m, ArH), 7.23–7.22 (1H, m, ArH), 7.06–7.04 (1H, m, ArH), 7.03 (1H, t, J 4.7, 5H), 3.77 (3H, s, OCH3); δC (100 MHz; CDCl3) 165.3 (C), 159.6 (CH), 151.6 (C), 141.9 (C), 126.6 (CH), 122.8 (CH), 121.0 (CH), 115.9 (CH), 112.8 (CH), 55.9 (CH3). Data recorded matches literature.13
4 ethyl acetate/light petroleum) the title compound as a yellow solid (238 mg, 67%); mp 118–120 °C (lit.,13 mp 121–122 °C); (found; M+, 203.0822. C11H11N2O2 requires 203.0815); νmax (CHCl3)/cm−1 3009, 1607, 1573, 1501, 1465, 1439; δH (400 MHz; CDCl3) 8.57 (2H, d, J 4.7, 4H, 6H), 7.27–7.26 (1H, m, ArH), 7.25–7.24 (1H, m, ArH), 7.23–7.22 (1H, m, ArH), 7.06–7.04 (1H, m, ArH), 7.03 (1H, t, J 4.7, 5H), 3.77 (3H, s, OCH3); δC (100 MHz; CDCl3) 165.3 (C), 159.6 (CH), 151.6 (C), 141.9 (C), 126.6 (CH), 122.8 (CH), 121.0 (CH), 115.9 (CH), 112.8 (CH), 55.9 (CH3). Data recorded matches literature.13
2-(3,4-Dimethylphenoxy)pyrimidine 5
Following the general procedure, 2-chloropyrimidine (200 mg, 1.75 mmol), 3,4-dimethylphenol (214 mg, 1.75 mmol) and potassium carbonate (483 mg, 3.50 mmol) in DMSO (1 mL) at 100 °C for 17 h gave after purification by chromatography (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 ethyl acetate/light petroleum) the title compound as a yellow oil (235 mg, 67%); (lit.,13 mp 49–51 °C); (found; M+, 201.1028. C12H13N2O requires 201.1022); νmax (CHCl3)/cm−1 3004, 1614, 1571, 1500, 1450, 1410; δH (400 MHz; CDCl3) 8.58 (2H, m, ArH, 4H, 6H), 7.20 (1H, d, J 7.8, ArH), 7.04–7.00 (2H, m, ArH), 6.98 (1H, d, J 7.8, ArH), 2.30 (3H, s, CH3), 2.29 (3H, s, CH3); δC (100 MHz; CDCl3) 165.7 (C), 159.7 (CH), 150.8 (C), 138.2 (C), 133.9 (C), 130.7 (CH), 122.6 (CH), 118.8 (CH), 115.9 (CH), 20.0 (CH3), 19.3 (CH3). Data recorded matches literature.13
4 ethyl acetate/light petroleum) the title compound as a yellow oil (235 mg, 67%); (lit.,13 mp 49–51 °C); (found; M+, 201.1028. C12H13N2O requires 201.1022); νmax (CHCl3)/cm−1 3004, 1614, 1571, 1500, 1450, 1410; δH (400 MHz; CDCl3) 8.58 (2H, m, ArH, 4H, 6H), 7.20 (1H, d, J 7.8, ArH), 7.04–7.00 (2H, m, ArH), 6.98 (1H, d, J 7.8, ArH), 2.30 (3H, s, CH3), 2.29 (3H, s, CH3); δC (100 MHz; CDCl3) 165.7 (C), 159.7 (CH), 150.8 (C), 138.2 (C), 133.9 (C), 130.7 (CH), 122.6 (CH), 118.8 (CH), 115.9 (CH), 20.0 (CH3), 19.3 (CH3). Data recorded matches literature.13
2-Phenoxypyrimidine 6
Following the general procedure, 2-chloropyrimidine (200 mg, 1.75 mmol), phenol (165 mg, 1.75 mmol) and potassium carbonate (483 mg, 3.50 mmol) in DMSO (1 mL) at 100 °C for 17 h gave after purification by chromatography (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 ethyl acetate/light petroleum) the title compound as a yellow crystalline solid (195 mg, 65%); mp 84–86 °C (lit.,13 mp 85–86 °C); νmax (CHCl3)/cm−1 3062, 1592, 1568, 1489, 1453, 1401; δH (400 MHz; CDCl3) 8.58 (2H, d, J 4.8, 4H, 6H), 7.46 (1H, t, J 7.7, ArH), 7.28 (1H, t, J 7.8, ArH), 7.25–7.21 (2H, m, ArH), 7.05 (1H, t, J 4.8, 5H), 6.89 (1H, d, J 7.8, ArH); δC (100 MHz; CDCl3) 165.3 (C), 159.8 (CH), 152.3 (C), 129.8 (CH), 125.7 (CH), 121.7 (CH), 155.6 (CH), 111.6 (CH). Data recorded matches literature.13
9 ethyl acetate/light petroleum) the title compound as a yellow crystalline solid (195 mg, 65%); mp 84–86 °C (lit.,13 mp 85–86 °C); νmax (CHCl3)/cm−1 3062, 1592, 1568, 1489, 1453, 1401; δH (400 MHz; CDCl3) 8.58 (2H, d, J 4.8, 4H, 6H), 7.46 (1H, t, J 7.7, ArH), 7.28 (1H, t, J 7.8, ArH), 7.25–7.21 (2H, m, ArH), 7.05 (1H, t, J 4.8, 5H), 6.89 (1H, d, J 7.8, ArH); δC (100 MHz; CDCl3) 165.3 (C), 159.8 (CH), 152.3 (C), 129.8 (CH), 125.7 (CH), 121.7 (CH), 155.6 (CH), 111.6 (CH). Data recorded matches literature.13
2-(2-Furylmethoxy)pyrimidine 7
Following the general procedure, 2-chloropyrimidine (200 mg, 1.75 mmol), furfuryl alcohol (156 μL, 1.75 mmol) and potassium carbonate (483 mg, 3.50 mmol) in DMSO (1 mL) at 100 °C for 17 h gave the title compound as a brown oil (156 mg, 51%); (found; M+, 199.0987. C9H9N2O2Na+ requires 199.0483); νmax (CHCl3)/cm−1 3009, 1578, 1568, 1502, 1449, 1425; δH (400 MHz; CDCl3) 8.55 (2H, d, J 4.8, 4H, 6H), 7.44 (1H, m, ArH), 6.97 (1H, t, J 4.8, 5H), 6.50–6.49 (1H, m, ArH), 6.38 (1H, m, ArH), 5.41 (2H, s, CH2); δC (100 MHz; CDCl3) 165.5 (C), 159.3 (CH), 149.9 (C), 143.0 (CH), 115.3 (CH), 110.5 (CH), 107.4 (CH), 61.1 (CH2).2-Benzyloxypyrimidine 8
Following the general procedure, 2-chloropyrazine (156 μL, 1.75 mmol), benzyl alcohol (181 μL, 1.75 mmol) and potassium carbonate (483 mg, 3.50 mmol) in DMSO (1 mL) at 100 °C for 17 h gave evaporation of the remaining benzyl alcohol the title compound as a colourless oil (115 mg, 35%); (found; M+, 187.0882. C11H10N2O requires 187.0866); νmax (CHCl3)/cm−1 3010, 1579, 1567, 1497, 1458, 1424; δH (400 MHz; CDCl3) 8.56 (2H, d, J 4.8, 4H, 6H), 7.52 (1H, dd, J 7.2, 1.5, ArH), 7.39 (2H, dt, J 7.2, 1.5, ArH), 7.35–7.31 (1H, m, ArH), 6.97 (1H, t, J 4.8, 5H), 5.48 (2H, s, CH2); δC (100 MHz; CDCl3) 165.0 (C), 159.3 (CH), 136.5 (C), 128.4 (CH), 127.9 (CH), 127.9 (CH), 115.1 (CH), 69.0 (CH2).2-(4-Methylphenoxy)pyrazine 9
Following the general procedure, 2-chloropyrazine (156 μL, 1.75 mmol), p-cresol (184 μL, 1.75 mmol) and potassium carbonate (483 mg, 3.50 mmol) in DMSO (1 mL) at 100 °C for 17 h gave the title compound as a beige crystalline solid (302 mg, 93%); mp 38–40 °C; (found; M+, 187.0875. C11H10N2O requires 187.0866); νmax (CHCl3)/cm−1 3061, 1580, 1532, 1504, 1465, 1401; δH (400 MHz; CDCl3) 8.43 (1H, d, J 1.1, 3H), 8.25 (1H, d, J 2.7, 6H), 8.11 (1H, dd, J 2.7, 1.1, 5H), 7.24 (2H, d, J 8.5, ArH), 7.07 (2H, d, J 8.5, ArH), 2.39 (3H, s, CH3); δC (100 MHz; CDCl3) 160.5 (C), 150.7 (C), 141.1 (CH), 138.3 (CH), 135.8 (CH), 135.1 (C), 130.3 (CH), 121.1 (CH), 20.9 (CH3).2-(2-Methylphenoxy)pyrazine 10
Following the general procedure, 2-chloropyrazine (156 μL, 1.75 mmol), o-cresol (180 μL, 1.75 mmol) and potassium carbonate (483 mg, 3.50 mmol) in DMSO (1 mL) at 100 °C for 17 h gave the title compound as a yellow solid (295 mg, 91%); mp 40–42 °C; (found; M+, 187.0868. C11H10N2O requires 187.0866); νmax (CHCl3)/cm−1 2915, 1578, 1531, 1485, 1466, 1397; δH (400 MHz; CDCl3) 8.45 (1H, d, J 1.2, 3H), 8.27 (1H, d, J 2.8, 6H), 8.11 (1H, dd, J 2.8, 1.2, 5H), 7.34–7.27 (2H, m, ArH), 7.22 (1H, dt, J 7.5, 1.5, ArH), 7.09 (1H, dd, J 7.8, 1.5, CH3); δC (100 MHz; CDCl3) 160.2 (C), 151.3 (C), 141.3 (CH), 138.1 (CH), 135.3 (CH), 131.6 (CH), 130.7 (C), 127.3 (CH), 125.9 (CH), 121.7 (CH), 16.3 (CH3). Data recorded matches literature.112-(4-(Pyrazin-2-yloxy)phenyl)ethanol 11
Following the general procedure, 2-chloropyrazine (156 μL, 1.75 mmol), 2-(4-hydroxyphenyl)ethanol (242 mg, 1.75 mmol) and potassium carbonate (483 mg, 3.50 mmol) in DMSO (1 mL) at 100 °C for 17 h gave after purification by chromatography (ethyl acetate) the title compound as a colourless crystalline solid (311 mg, 87%); mp 65–67 °C (lit.,14 mp 75 °C); (found; M+, 239.0779. C12H12N2O2 requires 239.0797); νmax (CHCl3)/cm−1 3619, 3441, 3066, 3008, 2951, 1581; δH (400 MHz; CDCl3) 8.42 (1H, d, J 1.2, 3H), 8.25 (1H, d, J 2.6, 5H), 8.09 (1H, dd, J 2.6, 1.2, 6H), 7.30 (2H, d, J 8.5, ArH), 7.12 (2H, d, J 8.5, ArH), 3.88 (2H, t, J 6.5, CH2), 2.90 (2H, t, J 6.5, CH2); δC (100 MHz; CDCl3) 160.3 (C), 151.5 (C), 141.1 (CH), 138.4 (CH), 135.9 (CH), 135.9 (CH), 130.4 (CH), 121.3 (CH), 63.5 (CH2), 38.6 (CH2). Data recorded matches literature.14ICP-MS analysis
Approximately 100 mg of potassium carbonate sample material was dissolved and diluted to 50 mL of sample matrix solution (5% hydrochloric acid, 1% hydrogen peroxide aqueous solution), and analysed by ICP-MS (ICP-MS X-SERIES 2 Thermo analyser). The instrument limit of quantitation was determined by 10 times injection of blank matrix solution as 21 ppb and 7 ppb for Cu and Pd respectively.| Cu (ppb) | Pd (ppb) | |
|---|---|---|
| Sample A | 88.7 | 21.9 |
| Sample B | 59.9 | 16.7 |
| Mean | 74 | 19 |
Acknowledgements
We thank the Engineering and Physical Sciences Research Council and GlaxoSmithKline for an Industrial CASE award (to K. W) and Ben Pointer-Gleadhill for the ICP-MS data.References
- S. Sinha and J. K. Chen, Nat. Chem. Biol., 2006, 2, 29–30 CrossRef CAS PubMed.
- K. Jabran, Ehsanullah, M. Hussain, M. Farooq, M. Babar, M. N. Dogan and D.-J. Lee, Weed Biol. Manage., 2012, 12, 136–145 CrossRef CAS PubMed.
- K. E. Torraca, X. H. Huang, C. A. Parrish and S. L. Buchwald, J. Am. Chem. Soc., 2001, 123, 10770–10771 CrossRef CAS.
- C. A. Parrish and S. L. Buchwald, J. Org. Chem., 2001, 66, 2498–2500 CrossRef CAS.
- A. V. Vorogushin, X. H. Huang and S. L. Buchwald, J. Am. Chem. Soc., 2005, 127, 8146–8149 CrossRef CAS PubMed.
- J. F. Marcoux, S. Doye and S. L. Buchwald, J. Am. Chem. Soc., 1997, 119, 10539–10540 CrossRef CAS.
- D. W. Ma and Q. Cai, Org. Lett., 2003, 5, 3799–3802 CrossRef CAS PubMed.
- H. J. Cristau, P. P. Cellier, S. Hamada, J. F. Spindler and M. Taillefer, Org. Lett., 2004, 6, 913–916 CrossRef CAS PubMed.
- G. Evano, N. Blanchard and M. Toumi, Chem. Rev., 2008, 108, 3054–3131 CrossRef CAS PubMed.
- J. A. Joule and K. Mills, Heterocyclic Chemistry, John Wiley & Sons, 5th edn, 2010 Search PubMed.
- C. H. Burgos, T. E. Barder, X. Huang and S. L. Buchwald, Angew. Chem., Int. Ed., 2006, 45, 4321–4326 CrossRef CAS PubMed.
- Z. Chen, Y. Wu, Y. Liu, S. Yang, Y. Chen and L. Lai, J. Med. Chem., 2011, 54, 3650–3660 CrossRef CAS PubMed.
- S. Gu, C. Chen and W. Chen, J. Org. Chem., 2009, 74, 7203–7206 CrossRef CAS PubMed.
- D. Maiti, Chem. Commun., 2011, 47, 8340–8342 RSC.
- N. D. D'Angelo, J. J. Peterson, S. K. Booker, I. Fellows, C. Dominguez, R. Hungate, P. J. Reider and T.-S. Kim, Tetrahedron Lett., 2006, 47, 5045–5048 CrossRef PubMed.
- M. Platon, L. Cui, S. Mom, P. Richard, M. Saeys and J.-C. Hierso, Adv. Synth. Catal., 2011, 353, 3403–3414 CrossRef CAS.
- N. Shah Bakhtiar, Z. Abdullah and S. W. Ng, Acta Crystallogr., Sect. E: Struct. Rep. Online, 2009, 65, o114 CAS.
- C. W. Am Ende, S. E. Knudson, N. Liu, J. Childs, T. J. Sullivan, M. Boyne, H. Xu, Y. Gegina, D. L. Knudson, F. Johnson, C. A. Peloquin, R. A. Slayden and P. J. Tonge, Bioorg. Med. Chem. Lett., 2008, 18, 3029–3033 CrossRef CAS PubMed.
- K. Walsh, H. F. Sneddon and C. J. Moody, ChemSusChem, 2013, 6, 1455–1460 CrossRef CAS PubMed.
- R. K. Henderson, C. Jimenez-Gonzalez, D. J. C. Constable, S. R. Alston, G. G. A. Inglis, G. Fisher, J. Sherwood, S. P. Binks and A. D. Curzons, Green Chem., 2011, 13, 854–862 RSC.
- T. L. Chan and J. Miller, Aust. J. Chem., 1967, 20, 1595–1600 CrossRef CAS.
- R. K. Arvela, N. E. Leadbeater, M. S. Sangi, V. A. Williams, P. Granados and R. D. Singer, J. Org. Chem., 2005, 70, 161–168 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Copies of NMR spectra. See DOI: 10.1039/c4ra04851b |
| This journal is © The Royal Society of Chemistry 2014 |



