Synthesis and characterization of magnetic bromochromate hybrid nanomaterials with triphenylphosphine surface-modified iron oxide nanoparticles and their catalytic application in multicomponent reactions
Abstract
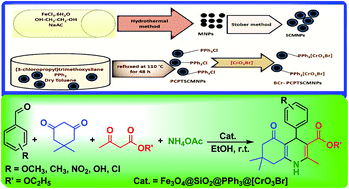
In this work, the preparation of a new magnetic hybrid core–shell nanomaterial is reported in four steps. The magnetic Fe3O4 nanoparticles were synthesized via a simple hydrothermal system. Tetraethyl orthosilicate was used as a source of SiO2 for the creation of Fe3O4@SiO2 core–shell nanoparticles. The subsequent functionalization of Fe3O4@SiO2 was achieved by using (3-chloropropyl)trimethoxysilane and triphenylphosphine. Finally, immobilization of the anionic part including the [CrO3Br]− was carried out to obtain Fe3O4@SiO2@PPh3@[CrO3Br]. The structure and morphology of the prepared nanocatalyst was characterized by using elemental analysis, scanning electron microscopy (SEM), transmission electron microscopy (TEM), X-ray fluorescence (XRF), X-ray diffraction (XRD), Fourier transform infrared (FT-IR) and a vibrating sample magnetometer (VSM). The prepared catalyst showed efficient activity as an heterogeneous catalyst in the synthesis of 1,4-dihydropyridine derivatives at room temperature. Moreover, the catalyst can be readily recovered by simple magnetic decantation and can be recycled several times with no significant loss of catalytic activity.

 Please wait while we load your content...
Please wait while we load your content...