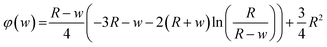Assembly of thin-walled, cell-laden hydrogel conduits inflated with perfluorocarbon†
A. Blaeser*,
D. F. Duarte Campos,
M. Köpf,
M. Weber and
H. Fischer
Department of Dental Materials and Biomaterial Research, RWTH Aachen University Hospital, Pauwelsstrasse 30, 52074 Aachen, Germany. E-mail: ablaeser@ukaachen.de; Fax: +49 241 8082027; Tel: +49 241 8088265
First published on 10th September 2014
Abstract
Autologous vessel grafts are considered the best option for coronary artery bypass surgery. However, their restricted availability and the poor patency of allografts as well as synthetic polymer grafts, make the development of biofunctional, small-calibre blood vessel substitutes of great interest. We propose a novel free-form manufacturing technique for the extrusion of cell-laden hydrogel tubes with adjustable wall thickness. Alginate and an alginate–fibrinogen blend were coextruded with high-density perfluorocarbon, which served as a liquid-supporting core. Hydrogel conduits measuring several centimetres in length and 2 mm in diameter were fabricated. By varying the volume flow ratio of gel and perfluorocarbon the wall thickness could be adjusted precisely in a range from 0.2 to 0.3 mm. In order to adjust the operating parameters of the technique, a theoretical model of the bioextrusion process based on the gelling kinetics of alginate was developed. Experimental validation revealed a high level of accordance with this model (90–100%). Burst-pressure tests indicated high mechanical stability of the extruded conduits (>100 kPa). Fibroblasts encapsulated in the hydrogel tubes were cultured for 7 days and exhibited a high degree of cell viability (>90%) and proliferation over the first week of culture. The proposed technique holds great promise for the fully automated manufacturing of cell-laden hydrogel conduits to be used for tissue engineering purposes or as tubing in bio-artificial, medical devices such as dialysers or liver support systems, in the future.
1. Introduction
The repair or replacement of stenotic or occluded arteries is a commonly occurring clinical issue. Occluded arteries can either be opened using such procedures as balloon or stent angioplasty, or completely replaced in coronary artery bypass surgery.1–5 In the latter case, vascular autografts,6 allografts,7,8 or synthetic blood vessel substitutes have been investigated.8–10 Autografts of blood vessels harvested, for example, from the patient's saphenous vein,5 internal-thoracic-artery,11 or internal mammary artery12 are predominantly used. However, the limited disposability, risk of donor site morbidity,6,13 and often insufficient quality in certain cases require the implantation of allografts, blood vessels from a human donor.12,14 Those, in turn, have shown only short-term patency and high re-occlusion rates and should therefore only be applied when no alternative autograft is available.7,11,15 The same applies for synthetic small-calibre vessel grafts, such as polytetrafluoroethylene conduits, which promote thrombus formation and intimal hyperplasia.16To overcome these hurdles, researchers are attempting to create biological blood vessel substitutes using tissue engineering (TE) approaches.17,18 In the past decades, various strategies for the biofabrication of blood vessel substitutes have evolved. Those strategies have involved techniques such as hydrogel moulding,19–22 cell seeding on de-cellularized matrices11,14,23–25 or tubular biomaterial scaffolds,26–31 wrapping of cohesive cellular sheets,32,33 in-body tissue architecture-induced grafts,34 or combinations of these (i.e. graft and moulding,35 graft and cell sheet36). Although these TE techniques have delivered excellent results in terms of blood vessel anatomy, cell biology, and mechanical integrity, they lack geometrical versatility and are difficult to incorporate into an automated process chain. Application of free-form biofabrication techniques such as bioprinting is one means towards the automated generation of biofunctional tissue that has recently emerged.37–46 As an example, Norotte and co-workers applied a bioprinting technique where multicellular spheroids were placed one by one according to a predefined, arbitrary geometry resulting in a scaffold-free vascular structure that was subsequently conditioned in a bioreactor.47
Here, we propose a novel fabrication technique for the rapid and fully automated manufacture of thin-walled, cell-laden hydrogel tubes with adjustable diameters and arbitrary length. In a technique similar to blow moulding in plastics engineering, we extrude hydrogel–cell suspension through the circumferential part of a multichannel orifice while supporting fluid is pressed through a centred opening, thus inflating the hydrogel tube. Perfluorocarbon (PFC), a chemically inert and immiscible high-density liquid is applied as the inflating fluid. Its capacity to dissolve high ratios of respiratory gases, such as oxygen and carbon dioxide, as well as its bioinertness lend it to use in tissue engineering applications.48–52 Lately, PFCs have also been used as liquid support material in bioprinting applications in order to improve the mechanical integrity of high-aspect ratio hydrogel constructs.53,54 We hypothesize that using this bioextrusion technique tubes with adjustable wall thickness can be fabricated by varying the volume flow rates of hydrogel and supporting liquid. This work presents the results of the bioextrusion of (cell-laden) alginate tubes, as well as the development and experimental validation of a diffusion model describing the complex process of alginate tube formation.
2. Materials and methods
2.1. Extrusion system
The extruder mainly comprised two disposable syringes (Terumo Syringe SS*05SE1 without needle, Terumo, Tokyo, Japan) kept within a heatable copper housing (Fig. 1). Syringes with volumes of 1, 2, or 5 millilitres could be loaded into the housing. The syringe containing the alginic acid solution (cell suspension) was connected to a channel made of stainless steel which led the liquid to the circumferential part of the extruder nozzle. A needle fixed at the tip of the second syringe was stuck through the central opening of the nozzle allowing perfluorocarbon (PFC) as supporting liquid to be squeezed into the core of the hydrogel strand. PFC from 3M (Fluorinert™ Electronic Liquids FC-43, 3M, St. Paul/Minnesota, USA) was applied. The extruder nozzle was made of PEEK (polyether ether ketone). A nozzles with a 2 mm inner diameter and a G21 needle (0.8 mm outer diameter) for the supporting liquid where applied. A linear drive based on a stepping motor (no. 82920001 Zwei Phasen Schrittmotor, Crouzet, Hilden, Germany) and an M4 threaded rod were used to move the plungers in and out of the syringes.2.2. Hydrogel preparation
Extrusion experiments were conducted with alginate (alginic acid sodium salt from brown algae, Sigma, St. Louis/Missouri, USA), an ionically cross-linkable hydrogel. Stock solutions of 3% m/v (30 mg ml−1), 2% m/v (20 mg ml−1), and 1% m/v (10 mg ml−1) alginate dissolved in de-ionized water were prepared. Alginate used throughout this study comprised 61 m% β-D-mannuronic and 39 m% α-L-guluronic units.2.3. Bioextrusion of alginate hydrogels
Alginate (3% m/v) and PFC were co-extruded into calcium-chloride solution (0.45 mol l−1) with a 2 mm outer diameter PEEK nozzle. To prevent clogging of the nozzle the extrusion process was started right after the extruder tip was dipped into the cross-linker bath. The extrusion process encompassed three phases: (1) immediate sol–gel transition of the surface of a freshly formed alginate bubble, where the PFC sank and displaced the remaining, non-cross-linked alginate solution by buoyant lift (Fig. 2A); (2) the PFC-containing alginate bowl was pulled away from the extruder nozzle by gravity, and the tube elongation caused the upward-squeezed, non-cross-linked alginate to be exposed to the calcium chloride solution (Fig. 2B); and (3) the formation of thin-walled alginate tubes (Fig. 2C). To successfully extrude alginate tubes, the hydrogel and PFC volume flows had to be adjusted with great precision. In order to optimize this, a theoretical model describing the alginate extrusion process was developed. The ideal PFC volume flow required to completely fill the lumen of the hydrogel tube could be calculated as a function of the alginate volume flow (![[V with combining dot above]](https://www.rsc.org/images/entities/i_char_0056_0307.gif) alginate), the tube diameter, and the wall thickness of the tube (eqn (1)). Due to rapid gelation of the alginate tube's surface when exposed to calcium chloride, the initial tube outer diameter was estimated to equal the nozzle diameter (DO).
alginate), the tube diameter, and the wall thickness of the tube (eqn (1)). Due to rapid gelation of the alginate tube's surface when exposed to calcium chloride, the initial tube outer diameter was estimated to equal the nozzle diameter (DO).
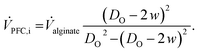 | (1) |
In order to determine the wall thickness (w) of the extruded alginate strand, the diffusion of calcium chloride into alginate had to be modelled. We adopted the calcium ion transfer rate equation and the equation for the alginate consumption for a spherical model from Kim and transferred it to a cylindrical model resembling the alginate strand (eqn (2), Fig. 2D and E).55 The gelling time required to achieve an alginate strand with a radius R and a wall thickness w can be expressed as a function of the effective calcium chloride diffusion coefficient (De), the moles of calcium ion required for complete gelation based on the alginate mass (q), the alginate concentration (p), the initial calcium chloride concentration (C0), and the annular surface to be jellified (φ).
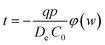 | (2) |
The gelling time can further be derived from the alginate extrusion speed ve (the speed at which the alginate strand leaves the nozzle) and the needle protrusion length LN (eqn (3)). Alongside the needle, the alginate interacts with the calcium chloride solution. The longer the alginate takes to surpass this zone, the deeper the calcium ions diffuse toward the centre of the tube, and hence, the thicker the tube's wall grows. The reaction time ends at the PFC inlet point, where all non-cross-linked gel is displaced by PFC. The extrusion speed can be derived from the alginate volume flow and the nozzle diameter (DO).
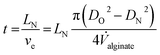 | (3) |
When the ideal PFC flow is applied (eqn (1)), the alginate volume flow required to extrude a tube with a specified wall thickness w can be derived (eqn (4)).
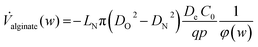 | (4) |
The ratio of the PFC volume flow (![[V with combining dot above]](https://www.rsc.org/images/entities/i_char_0056_0307.gif) PFC) actually applied and the calculated ideal volume flow (
PFC) actually applied and the calculated ideal volume flow (![[V with combining dot above]](https://www.rsc.org/images/entities/i_char_0056_0307.gif) PFC,i) provides a measure for the displacement of non-cross-linked alginate inside the tube (ε).
PFC,i) provides a measure for the displacement of non-cross-linked alginate inside the tube (ε).
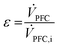 | (5) |
According to the gel displacement ratio (ε), three different states are distinguished:
ε < εL, alginate that was not cross-linked during the gelation time t remains in the tube and gels over time. As a result, the wall thickness increases beyond that calculated. If the PFC volume is below a certain threshold, an alginate cylinder containing PFC bubbles is formed instead of a tube (video S1†).
εL < ε < εH, complete displacement of non-cross-linked alginate, resulting in a homogeneously formed, thin-walled tube (video S2†).
ε > εH, The PFC flow pressed into the tube exceeds the tube's core volume. Excess PFC rises into the circumferential part of the extruder nozzle and leaks out, tearing the tube open (video S3†).
The lower (εL) and higher end (εH) of the gel displacement ratio range, where tube extrusion is still feasible have to be determined experimentally.
2.4. Estimation of the effective diffusion coefficient of calcium chloride in alginate
The effective diffusion coefficient of calcium chloride in alginate (De) was experimentally derived for three different CaCl2 concentrations (0.09 mol l−1, 0.18 mol l−1, and 0.45 mol l−1). Using a syringe, 3% m/v alginate strands (non-hollow) with an outer diameter of 2 mm were extruded into the calcium chloride solution. The thickness of the gelled alginate mantle was measured following immersion in the CaCl2 solution for 10, 30, 60, and 90 seconds using a travelling microscope. The effective diffusion coefficient could thus be derived from the alginate gelling model by solving eqn (2) for De.2.5. Validation of the alginate tube extrusion model
To verify the reliability of the alginate bioextrusion model, the displacement ratio range which enabled tube extrusion was validated. Experimentally derived data were compared to the theoretical model. Using eqn (4), the alginate flow needed to extrude a tube with a wall thickness of 0.24 mm was calculated to be 530 μl min−1. Different gel displacement ratios (ε) were tested varying the PFC flow while maintaining the calculated alginate flow. In a second experiment, eqn (4) was used to calculate the alginate flows required to obtain different wall thicknesses. The PFC flow was adjusted to keep an ideal displacement ratio (ε ≈ 1).2.6. Measurement of the wall thickness and outer diameter of extruded alginate tubes
The alginate tube extrusion process was recorded with a digital camera (EOS 7D, Canon, Tokyo, Japan) and single frames of the video were exported and analyzed using imageJ software (http://rsb.info.nih.gov/ij/). Three specimens were analyzed for each measurement. The wall thickness was measured at three distinct points on the left side of the tube and three points on the right. The outer diameter was measured at three distinct points along the tube.2.7. Dynamic viscosity measurements
Dynamic viscosity of 1, 2, and 3% m/v alginate-solutions was measured with a rotary rheometer (Kinexus, Malvern Instruments Ltd, Worcestershire, UK). Alginate solution (1.2 ml) was placed in between a plate and a 0.15 mm distant 4°-cone. The measurements were conducted in steady-state mode over a shear rate range from 0.1–1000 s−1. The dynamic viscosity was determined by measuring the zero-shear viscosity.2.8. Burst pressure measurements
The burst pressure of 1% m/v, 2% m/v and 3% m/v alginate tubes extruded into 0.45 mol l−1 CaCl2 was measured with a custom built device. One end of a freshly formed tube was connected to a barbed adaptor (1.0 mm, PEEK, Kinesis Ltd, St. Neots, Cambridgeshire, UK) and clamped with parafilm. The other end was kept shut by hand. Using a syringe connected to the adaptor and a pressure sensor (I2C, DRMOD-I2C-R1B6, from B & B Thermotechnik, Donaueschingen, Germany) tap water was pressed into the alginate tube. The pressure was manually increased until the tube burst. The increase in pressure was monitored and the peak pressure determined.2.9. Extrusion of cell-laden hydrogel tubes
L929 mouse fibroblasts (CCL-1, American Type Culture Collection) were cultured in low glucose DMEM supplemented with 10% FCS and 0.4% gentamycin (10 mg ml−1) and then re-suspended in 600 μl low glucose DMEM, 600 μl DMEM (10×) from Biochrom (F0455 DMEM (10×), Biochrom AG, Berlin, Germany) and 60 μl antibiotic-antimycotic reagent (100× Gibco antibiotic-antimycotic, Invitrogen, Burlington, Canada). Two different cell–hydrogel compositions were tested, cells suspended in pure alginate (−Fib) and cells mixed with an alginate–fibrinogen blend (+Fib). For the −Fib experiments pure alginate (30 mg ml−1) was mixed in equal parts with cell suspension resulting in a final cell concentration of 2.1 × 106 cells ml−1 and a final alginate concentration of 15 mg ml−1. For the +Fib experiments cell suspension was mixed with 4 ml alginate (30 mg ml−1) and 1.5 ml fibrinogen (5 mg ml−1) resulting in a final cell concentration of 4.6 × 106 cells ml−1. The final hydrogel solution comprised 17.7 mg ml−1 alginate and 1.1 mg ml−1 fibrinogen. The hydrogel–cell suspension was coextruded with PFC using the 2 mm outer diameter nozzle into a bath of 0.45 mol l−1 calcium chloride solution. The tubular structures were statically cultured in a six-well plate filled with 2 ml low glucose DMEM for up to 7 days. During culture, the coextruded PFC was kept in the lumen of the extruded hydrogel tubes.2.10. Cell viability, proliferation and morphology study
Live/dead staining was performed after 1, 3 (only +Fib), 4 (only −Fib), and 7 days of culture. Staining solution from 0.083 mg ml−1 propidium iodide (P4170-10116, Sigma, Taufkirchen, Germany) and 0.083 mg ml−1 fluorescein diacetate (F7378-10G, Sigma, Taufkirchen, Germany) in Ringer's solution was prepared and dripped onto the hydrogel tubes. An inverted microscope (DMI6000B, Leica Microsystems, Wetzlar, Germany) was used to record fluorescence images. Due to the high initial cell concentration and subsequently high cell proliferation, large cell clusters were formed from day 3 on and individual, living cells could no longer be distinguished. Thus, the number of living cells (Ncell) was estimated from the number of clusters (Ncluster), the cluster diameter (Dcluster), and the average diameter of a single cell (Dcell) derived from the cell images taken after 24 hours using eqn (6).
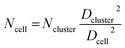 | (6) |
In addition, single cell clusters were analyzed in greater detail using fluorescence microscopy at 20× magnification. Live/dead staining as well as 2 μg ml−1 Hoechst staining (Hoechst 33342, trihydrochloride, trihydrate, Invitrogen, Burlington, Canada) were applied one after another.
2.11. Statistical analysis
The data presented in this work represent the mean ± standard deviation. The replicate number n is listed in the respective figure caption. Statistical significance was calculated using one-tailed t-test in Microsoft Excel 2010. Significance levels were p < 0.05 (*), and p < 0.005 (***).3. Results
3.1. Estimation of the effective calcium chloride diffusion coefficient
The thickness of the gelled surface of an alginate strand immersed in CaCl2 solution as a function of gelling time was investigated (Fig. 3). The effective diffusion coefficient was estimated for each CaCl2 concentration by matching the theoretical gelling curve calculated using eqn (4) to the experimental results. The diffusion coefficient dropped for decreasing CaCl2 concentrations from 2.66 × 10−6 cm2 s−1 (0.45 mol l−1) to 6.73 × 10−7 cm2 s−1 (0.09 mol l−1).3.2. Bioextrusion of alginate tubes
To verify the reliability of the alginate bioextrusion model, theoretically derived data were compared to experimental observations. According to the applied gel displacement ratio (ε), three results were observed: bubble formation (Fig. 4A), tube formation (Fig. 4B), and bursting (Fig. 4C-C2). For 0.7 < ε < 1.5, homogeneously formed hollow tubes could be extruded (Table 1). For ε < 0.7, discrete PFC bubbles formed inside an alginate strand. If the displacement ratio was higher than 1.5, PFC leaked out breaking the tube wall. By applying gel displacement ratios close to 1.0, extremely thin walled alginate tubes (w < 0.5 mm) of arbitrary length (>10 cm) could be manufactured in a few seconds (Fig. 5). For alginate tubes extruded with gel displacement ratios close to the ideal, the theoretically estimated wall thickness was compared to the actual measurements (Table 2). Experimental validation revealed high accordance with the theoretical model. For alginate volume flows from 260 to 1026 μl s−1, the theoretically calculated wall thickness matched the experimental results with 90 to 100% precision.![[V with combining dot above]](https://www.rsc.org/images/entities/i_char_0056_0307.gif) alginate (μl min−1) alginate (μl min−1) |
![[V with combining dot above]](https://www.rsc.org/images/entities/i_char_0056_0307.gif) PFC (μl min−1) PFC (μl min−1) |
ε [−] | Observation |
|---|---|---|---|
| 530 | 55 | 0.07 | Bubble formation |
| 115 | 0.15 | Bubble formation | |
| 255 | 0.34 | Bubble formation | |
| 530 | 0.71 | Tube extrusion | |
| 1102 | 1.48 | Tube extrusion | |
| 2454 | 3.28 | Bursting | |
| 5103 | 6.83 | Bursting |
 | ||
| Fig. 5 Bioextrusion of alginate with adjusted gel displacement ratio (ε ≈ 1), several centimetre-long, thin-walled alginate tubes are manufactured. (A) Scale bar = 2 cm, (B) scale bar = 2 mm. | ||
![[V with combining dot above]](https://www.rsc.org/images/entities/i_char_0056_0307.gif) alginate (μl min−1) alginate (μl min−1) |
![[V with combining dot above]](https://www.rsc.org/images/entities/i_char_0056_0307.gif) PFC (μl min−1) PFC (μl min−1) |
ε [−] | wtheory (mm) | wexperiment (mm) |
|---|---|---|---|---|
| 260 | 260 | 1.02 | 0.30 | 0.30 ± 0.02 |
| 530 | 530 | 0.71 | 0.24 | 0.24 ± 0.01 |
| 530 | 1102 | 1.48 | 0.24 | 0.22 ± 0.01 |
| 1026 | 2133 | 1.09 | 0.19 | 0.21 ± 0.01 |
3.3. Impact of alginate concentration on viscosity, tube diameter and burst pressure
Extrusion of alginate tubes with different hydrogel concentration and hence different viscosities was studied. Varying the alginate concentration from 1% m/v to 3% m/v the dynamic viscosity increased from approximately 100 mPa s to more than 3000 mPa s (Table 3). The theoretical extrusion model was applied to estimate the ideal volume flows of PFC and alginate for each alginate concentration and with an estimated tube outer diameter of 2 mm (ε*). The results indicate that the tube outer diameter (OD) is influenced by the alginate composition. The OD increased from 1.3 mm for the 1% m/v alginate tube to 2 mm for the 3% m/v alginate tube (Table 3, Fig. 6) although the same nozzle was applied. After the experiment the actual gel displacement ratio was calculated using the real tube ODs (ε). In addition burst pressure experiments were conducted with three specimen of each alginate concentration. The maximum pressure before bursting rose from 16.7 kPa (1% m/v) to over 100 kPa (3% m/v). Since the burst pressure sensor could only be applied up to 100 kPa, the exact value of the 3% m/v samples could not be identified.![[V with combining dot above]](https://www.rsc.org/images/entities/i_char_0056_0307.gif) alginate and
alginate and ![[V with combining dot above]](https://www.rsc.org/images/entities/i_char_0056_0307.gif) PFC) were adjusted in order to operate closely to the optimal gel displacement ratio. Before the experiment the gel displacement ratio was calculated for an estimated outer diameter of 2 mm (ε*). The actual displacement ratio (ε) was calculated with the real tube diameter accessed after the experiment
PFC) were adjusted in order to operate closely to the optimal gel displacement ratio. Before the experiment the gel displacement ratio was calculated for an estimated outer diameter of 2 mm (ε*). The actual displacement ratio (ε) was calculated with the real tube diameter accessed after the experiment
| 1% m/v | 2% m/v | 3% m/v | ||||
|---|---|---|---|---|---|---|
| η (mPa s) | 100.9 ± 6.52 | 815.3 ± 65.19 | 3289.0 ± 113.12 | |||
| OD (mm) | 1.3 ± 0.03 | 1.8 ± 0.09 | 1.97 ± 0.06 | |||
| pburst (kPa) | 16.7 ± 3.60 | 37.2 ± 6.60 | >100 | |||
![[V with combining dot above]](https://www.rsc.org/images/entities/i_char_0056_0307.gif) PFC (ml min−1) PFC (ml min−1) |
4.02 | 1.80 | 0.26 | |||
![[V with combining dot above]](https://www.rsc.org/images/entities/i_char_0056_0307.gif) alginate (ml min−1) alginate (ml min−1) |
2.60 | 1.23 | 0.26 | |||
| ε* | 1.00 | 0.99 | 1.02 | |||
| ε | 0.87 | 0.98 | 1.01 |
3.4. Biofabrication of cell-laden alginate tubes
Fluorescence images of cells embedded in the tube wall indicated high viability over 7 days of culture for cells in pure alginate (−Fib) as well as for those embedded in alginate–fibrinogen blend (+Fib), Fig. 7A. During the first week of culture cell viability (ratio of living to dead cells) was ≥90% and thus as high as the cell viability in the control group before the extrusion process. The total number of cell clusters decreased slightly from 386 ± 10 (+Fib, day 1), 262 ± 21 (−Fib, day 1) to 269 ± 17 (+Fib, day 7) and 148 ± 2 (−Fib, day 7), Fig. 7B. In the same time, the average cell cluster diameter increased significantly from 13 μm at day 1 to 29 μm (+Fib), and to 40 μm (−Fib) after 7 days of culture (Fig. 7C). The total number of living cells per volume was calculated from the number of clusters and the average cluster diameter using eqn (6). Due to the strong increase in the average cluster diameter the total number of living cells increased significantly from 4.6 × 106 ml−1 (+Fib), and 2.1 × 106 ml−1 (−Fib) to 23.1 × 106 ml−1 (+Fib), and 24.2 × 106 ml−1 (−Fib) after seven days of culture (Fig. 7D). Over the complete culture period the number of dead cells remained as high as in the control (0.2–0.78 × 106 ml−1).In order to outline the shape of a cell-laden tube and the cell distribution within its walls, a volume scan using fluorescence microscopy was performed on an alginate tube cultured for three days (Fig. 8A, video S4†). A cross-sectional image of fluorescently marked cells encapsulated in the tube reveal the tube's thin wall thickness, which is almost in the single cell range (Fig. 8B). Hoechst staining along with live/dead staining was applied to visualize cell clusters that formed after seven days of culture (Fig. 8C and D).
4. Discussion
Bioextrusion of alginate conduits is a complex manufacturing process that requires extensive knowledge about the material's gelling characteristics. Only a few theoretical approaches for modelling the gelation kinetics of alginate exist in the literature.56–58 Most of the published work focuses on ion diffusion into spherical alginate beads.59,60 Complex numerical models describing the gelation of alginate fibres are also available.61 However, in our work the rather simple, and therefore elegant, diffusion approach described by Kim was applied as a basis to develop an alginate tube extrusion model.55 An ideal PFC flow rate that enables tube formation and corresponds to a specific alginate flow rate was calculated. The gel displacement ratio (ε), the quotient of the actually applied and the calculated ideal PFC flow, was introduced. Experiments revealed that for alginate tubes with 2 mm diameter, tube formation was only possible in a narrow operating range (0.6 < ε < 1.7). For 3% m/v alginate, experimental validation revealed high accordance with the model in estimating the tube wall thickness as a function of alginate volume flow. Within the optimal process window (ε ≈ 1) the extrusion process is very stable. Hydrogel tubes measuring more than 10 cm in length could easily be fabricated with high repeatability (Fig. 5). However, the initial assumption that the tube outer diameter equals the inner diameter of the applied nozzle (eqn (1)) is not generally valid. Additional extrusion experiments revealed that the tube's outer diameter depends on the applied alginate concentration (Table 3). This could chiefly be ascribed to the concentration dependent change in the alginate's viscosity. Low viscosity alginate filled with high density PFC is pulled out of the extrusion nozzle by gravity easier than alginate with higher viscosity. This leads to a circumferential contraction which can be observed by the decrease in the tube's diameter. The outer diameter decreased from 2 mm for 3% m/v alginate with a viscosity higher than 3000 mPa s to 1.3 mm for 1% m/v alginate with a viscosity of 100 mPa s. Applying the theoretical model using the wrong outer diameter induces a certain error in the calculation of the ideally applicable PFC and alginate volume flows. The error can be quantified as the difference between the assumed (ε*) and the real gel displacement ratio (ε), Table 3. To prevent this error, the real outer diameter should be determined experimentally in preliminary tests. Alternatively, the theoretical model could be modified in the future. Besides the gelation kinetics, the influence of the hydrogel's rheological properties and the associated flow behaviour could be incorporated. Apart from geometrical and rheological aspects, the mechanical stability of freshly formed tubes comprising 1, 2, and 3% m/v alginate was tested. The maximum burst pressure strongly depended on the alginate concentration. While tubes made of low concentrated alginate (1% m/v) ruptured already at 16.7 kPa, 3% m/v alginate tubes withstood pressures higher than 100 kPa (Table 3). In comparison, native saphenous veins have a burst pressure greater than 220 kPa (>1700 mmHg).18 Cell incorporation and prolonged incubation induce alginate degradation, which might reduce the maximum burst pressure by time even further. Thus, future work should focus on enhancing the mechanical load capacity of extruded alginate tubes, e.g. by mixing it with an additional polymer and exploiting further crosslinking mechanisms.62,63To verify the cytocompatibility of the extrusion process a viability and proliferation study on L929-mouse fibroblast laden alginate tubes was conducted. Cells embedded in tubes made of pure alginate (15 mg ml−1) as well as in tubes comprising alginate (17.7 mg ml−1) blended with fibrinogen (1.1 mg ml−1) were studied. The bioextrusion process which involved immersion of cells into 0.45 mol l−1 calcium chloride was shown to be cytocompatible (Fig. 7). Cells exposed to the extrusion procedure showed high cell viability as well as proliferation potential immediately following the extrusion and for seven days in culture (Fig. 7A). The total number of separate cell clusters decreased from day 1 to 7 (Fig. 7B). However, the average cluster diameter of the remaining cell clusters grew (Fig. 7C). For this reason, the sum of cells embodied in all clusters cumulated steadily (Fig. 7D). The drop in the number of separate cell clusters by time (Fig. 7 B) can mainly be ascribed to the absence of adequate focal adhesion points for mammalian cells in non-modified alginate.64–67 Thus, cells can only be encapsulated in alginate but neither attach nor spread within the biomaterial, which was also observed in our experiments. Cells had a spherical morphology, indicating that they did not attach to the surrounding environment. Hence, single cells and small cell clusters might have been washed out of the slowly disintegrating alginate tube.71 Furthermore, cells that did not bind to the alginate and were not fully enclosed in gel might have migrated into the medium. On the other hand, cells which were kept inside the tube wall proliferated and showed signs of clonal growth (Fig. 7A and 8). The increase in the average diameter of those cell clusters which prevailed contributes to this conclusion. It is striking, that addition of fibrinogen, which exhibits natural RGD-binding sites, did not significantly affect the cell response. It neither induced cell spreading nor improved cell growth. In fact, after 7 days of culture the average cluster diameter of cells in pure alginate even exceeded the size of those in the alginate–fibrinogen blend, albeit not significantly (Fig. 7C). Restricted expansion space might have outranged the impact of fibrinogen and disabled cell spreading. The pore size of calcium-alginate hydrogels is reported to be in the range of 5–200 nm,60 which is only a fraction of the measured cell-cluster size (up to 40 μm). To improve cell adhesion and enable cell spreading, which is seen to be a crucial factor for cell survival,69,70 alginates with enhanced porosity as well as biologically functionalized alginates, i.e. alginates with RGD-sequences, could be examined in the future.65,68,72–75
Compared to former methods for the manufacture of biological blood vessel substitutes, bioextrusion offers the possibility of generating fully biological, cell-containing hydrogel tubes without the use of casting moulds and cores,19–21,34 synthetic scaffolds,11,26–31 decellularized tissue,14,23–25 or excessive handwork.32,36 The proposed technique offers a certain amount of flexibility in the geometry of the extruded tube. Depending on the operating parameters, most prominently the volume flows of hydrogel and perfluorocarbon, the tube's wall thickness could be adjusted. The geometric versatility of other free-form fabrication techniques, such as bioprinting, is certainly higher.27,37–39,41,42,44,47 However, bioextrusion is a more specialized technique with a primary focus on the manufacture of cell-encapsulating, tubular tissue engineering constructs. In this niche, it surpasses other free-form fabrication techniques especially with respect to production time, reliability, tube length, and the minimal achievable wall thickness.
5. Conclusion
This work proves the general feasibility of bioextrusion as a tissue engineering tool and confirms the hypothesis that the tube's wall thickness could be adjusted by varying the volume flows of hydrogel and PFC. A theoretical model for the extrusion of alginate tubes was developed and successfully validated. Bioextrusion was shown to be a useful tool for the fabrication of small calibre, cell-laden hydrogel tubes. In contrast to other free-form fabrication techniques, it is focused solely on tubular geometries. Despite its limited geometric freedom, the proposed technique enables fast and fully automatable production of long hydrogel conduits with high repeat accuracy. Studying cell viability and growth of mouse fibroblasts, the extrusion process was shown to be cytocompatible. In order to apply the proposed technique for the manufacturing of tissue engineering scaffolds, e.g. for blood vessels or other tubular tissues (i.e. ureters or oviducts), the usage of more sensitive cell types such as endothelial cells and pericytes needs to be explored in future studies. Besides tissue engineering scaffolds, fabrication of cell-encapsulating tubes to be used in bio-artificial dialysers or liver-support devices could be an alternative field of application.However, the focus of upcoming work will be the advanced examination of the tube's mechanical properties such as degradation, swelling rate, and load capacity over prolonged culture periods, implementation of a bioreactor-based culture system, and modification of the extruder in order to achieve multi-layered tubes with different cell types and biomaterials.
Acknowledgements
The authors are grateful to Roswitha Davtalab for her assistance in preparing the cell culture experiments, and Darius Hinüber for his help in conducting bioextrusion experiments, both Department of Dental Materials and Biomaterials Research, RWTH Aachen University Hospital.References
- P. W. Serruys, F. Unger, J. E. Sousa, A. Jatene, H. J. Bonnier, J. P. Schönberger, N. Buller, R. Bonser, M. J. van den Brand, L. A. van Herwerden, M. A. Morel and B. A. van Hout, N. Engl. J. Med., 2001, 344, 1117–1124 CrossRef CAS PubMed.
- S. B. King, N. J. Lembo, W. S. Weintraub, A. S. Kosinski, H. X. Barnhart, M. H. Kutner, N. P. Alazraki, R. A. Guyton and X. Q. Zhao, N. Engl. J. Med., 1994, 331, 1044–1050 CrossRef PubMed.
- D. L. Fischman, M. B. Leon, D. S. Baim, R. A. Schatz, M. P. Savage, I. Penn, K. Detre, L. Veltri, D. Ricci and M. Nobuyoshi, N. Engl. J. Med., 1994, 331, 496–501 CrossRef CAS PubMed.
- I. Sim, M. Gupta, K. McDonald, M. G. Bourassa and M. A. Hlatky, Am. J. Cardiol., 1995, 76, 1025–1029 CrossRef CAS.
- S. J. Pocock, R. a. Henderson, a. F. Rickards, J. R. Hampton, S. B. King, C. W. Hamm, J. Puel, W. Hueb, J. J. Goy and a. Rodriguez, Lancet, 1995, 346, 1184–1189 CrossRef CAS.
- L. D. Berrizbeitia, S. Tessler, I. J. Jacobowitz, P. Kaplan, L. Budzilowicz and J. N. Cunningham, Chest, 1989, 96, 873–876 CrossRef CAS.
- U. Bortolotti, D. Casarotto, C. Frugoni, P. De Mozzi, G. Thiene and V. Gallucci, Cardiovasc. Dis., 1981, 8, 250–258 Search PubMed.
- G. W. Laub, S. Muralidharan, R. Clancy, W. J. Eldredge, C. Chen, M. S. Adkins, J. Fernandez, W. A. Anderson and L. B. McGrath, Ann. Thorac. Surg., 1992, 54, 826–831 CrossRef CAS.
- O. E. Teebken and A. Haverich, Eur. J. Vasc. Endovasc. Surg., 2002, 23, 475–485 CrossRef PubMed.
- S. Li, D. Sengupta and S. Chien, Wiley Interdiscip. Rev.: Syst. Biol. Med., 2013, 6, 61–76 CrossRef PubMed.
- H. R. Laube, J. Duwe, W. Rutsch and W. Konertz, J. Thorac. Cardiovasc. Surg., 2000, 120, 134–141 CrossRef CAS PubMed.
- M. S. Conte, FASEB J., 1998, 12, 43–45 CAS.
- D. Hurlbut, M. L. Myers, M. Lefcoe and M. Goldbach, Ann. Thorac. Surg., 1990, 50, 959–964 CrossRef CAS.
- P. Lamm, G. Juchem, S. Milz, M. Schuffenhauer and B. Reichart, Circulation, 2001, 104, I–108–I–114 CrossRef CAS.
- R. a. Iaffaldano, B. E. Lewis, S. a. Johnson, R. Piffare and T. L. McKiernan, Chest, 1995, 108, 725–729 CrossRef CAS.
- R. Y. Kannan, H. J. Salacinski, P. E. Butler, G. Hamilton and A. M. Seifalian, J. Biomed. Mater. Res., Part B, 2005, 74, 570–581 CrossRef PubMed.
- L. Xue and H. P. Greisler, J. Vasc. Surg., 2003, 37, 472–480 CrossRef PubMed.
- D. G. Seifu, A. Purnama, K. Mequanint and D. Mantovani, Nat. Rev. Cardiol., 2013, 10, 410–421 CrossRef CAS PubMed.
- C. B. Weinberg and E. Bell, Science, 1986, 231, 397–400 CAS.
- Z. H. Syedain, L. a. Meier, J. W. Bjork, A. Lee and R. T. Tranquillo, Biomaterials, 2011, 32, 714–722 CrossRef CAS PubMed.
- L. Gui, M. J. Boyle, Y. M. Kamin and A. H. Huang, Tissue Eng., Part A, 2013, 1–34 Search PubMed.
- L.-Y. Jiang and Y. Luo, Soft Matter, 2013, 9, 1113 RSC.
- W. Zhai, H. Zhang, C. Wu, J. Zhang, X. Sun, H. Zhang, Z. Zhu and J. Chang, J. Biomed. Mater. Res., Part B, 2014, 1–9 Search PubMed.
- Y. Zhao, S. Zhang, J. Zhou, J. Wang, M. Zhen, Y. Liu, J. Chen and Z. Qi, Biomaterials, 2010, 31, 296–307 CrossRef CAS PubMed.
- M. Bertanha, A. Moroz, R. Almeida, F. C. Alves, M. J. Acorci Valério, R. Moura, M. A. C. Domingues, M. L. Sobreira and E. Deffune, J. Vasc. Surg., 2013, 1–9 Search PubMed.
- L. E. Niklason, Science., 1999, 284, 489–493 CrossRef CAS.
- A. Nieponice, L. Soletti, J. Guan, B. M. Deasy, J. Huard, W. R. Wagner and D. a. Vorp, Biomaterials, 2008, 29, 825–833 CrossRef CAS PubMed.
- J. D. Roh, G. N. Nelson, M. P. Brennan, T. L. Mirensky, T. Yi, T. F. Hazlett, G. Tellides, A. J. Sinusas, J. S. Pober, W. M. Saltzman, T. R. Kyriakides and C. K. Breuer, Biomaterials, 2008, 29, 1454–1463 CrossRef CAS PubMed.
- X. Zhang, X. Wang, V. Keshav, X. Wang, J. T. Johanas, G. G. Leisk and D. L. Kaplan, Biomaterials, 2009, 30, 3213–3223 CrossRef CAS PubMed.
- A. Atala, Curr. Opin. Biotechnol., 2009, 20, 575–592 CrossRef CAS PubMed.
- Y. Wang, H. Shi, J. Qiao, Y. Tian, M. Wu, W. Zhang, Y. Lin, Z. Niu and Y. Huang, ACS Appl. Mater. Interfaces, 2014, 6, 2958–2962 CAS.
- N. L'Heureux, S. Pâquet, R. Labbé, L. Germain and F. a. Auger, FASEB J., 1998, 12, 47–56 Search PubMed.
- H. Kerdjoudj, F. Boulmedais, N. Berthelemy, H. Mjahed, H. Louis, P. Schaaf, J. C. Voegel and P. Menu, Soft Matter, 2011, 7, 3621 RSC.
- T. Watanabe, K. Kanda, M. Yamanami, H. Ishibashi-Ueda, H. Yaku and Y. Nakayama, J. Biomed. Mater. Res., Part B, 2011, 98, 120–126 CrossRef PubMed.
- S. Koch, T. C. Flanagan, J. S. Sachweh, F. Tanios, H. Schnoering, T. Deichmann, V. Ellä, M. Kellomäki, N. Gronloh, T. Gries, R. Tolba, T. Schmitz-Rode and S. Jockenhoevel, Biomaterials, 2010, 31, 4731–4739 CrossRef CAS PubMed.
- S. Rayatpisheh, D. E. Heath, A. Shakouri, P.-O. Rujitanaroj, S. Y. Chew and M. B. Chan-Park, Biomaterials, 2014, 35, 2713–2719 CrossRef CAS PubMed.
- W. C. Wilson and T. Boland, Anat. Rec., Part A, 2003, 272, 491–496 CrossRef PubMed.
- V. Mironov, G. Prestwich and G. Forgacs, J. Mater. Chem., 2007, 17, 2054 RSC.
- B. da Graca and G. Filardo, Am. J. Cardiol., 2011, 107, 141–142 CrossRef PubMed.
- S. Khetan and J. a. Burdick, Soft Matter, 2011, 7, 830 RSC.
- F. Marga, K. Jakab, C. Khatiwala, B. Shepherd, S. Dorfman, B. Hubbard, S. Colbert and F. Gabor, Biofabrication, 2012, 4, 022001 CrossRef PubMed.
- T. Billiet, M. Vandenhaute, J. Schelfhout, S. Van Vlierberghe and P. Dubruel, Biomaterials, 2012, 33, 6020–6041 CrossRef CAS PubMed.
- Y.-J. Seol, T.-Y. Kang and D.-W. Cho, Soft Matter, 2012, 8, 1730 RSC.
- S. Tasoglu and U. Demirci, Trends Biotechnol., 2013, 31, 10–19 CrossRef CAS PubMed.
- A. Faulkner-Jones, S. Greenhough, J. A. King, J. Gardner, A. Courtney and W. Shu, Biofabrication, 2013, 5, 015013 CrossRef PubMed.
- K. P. Lee V, G. Singh, J. P. Trasatti, C. Bjornsson, X. Xu, T. N. Tran, S. S. Yoo and G. Dai, Tissue Eng., Part C, 2014, 473–484 CrossRef PubMed.
- C. Norotte, F. S. Marga, L. E. Niklason and G. Forgacs, Biomaterials, 2009, 30, 5910–5917 CrossRef CAS PubMed.
- K. C. Lowe, M. R. Davey and J. B. Power, Trends Biotechnol., 1998, 16, 272–277 CrossRef CAS.
- K. C. Lowe, J. Fluorine Chem., 2002, 118, 19–26 CrossRef CAS.
- K. C. Lowe, Tissue Eng., 2003, 9, 389–399 CrossRef CAS PubMed.
- M. Radisic, H. Park, F. Chen, J. E. Salazar-Lazzaro, Y. Wang, R. Dennis, R. Langer, L. E. Freed and G. Vunjak-Novakovic, Tissue Eng., 2006, 12, 2077–2091 CrossRef CAS PubMed.
- M. Radisic, A. Marsano, R. Maidhof, Y. Wang and G. Vunjak-Novakovic, Nat. Protoc., 2008, 3, 719–738 CrossRef CAS PubMed.
- D. F. Duarte Campos, A. Blaeser, M. Weber, J. Jäkel, S. Neuss, W. Jahnen-Dechent and H. Fischer, Biofabrication, 2013, 5, 015003 CrossRef PubMed.
- A. Blaeser, D. F. Duarte Campos, M. Weber, S. Neuss, B. Theek, H. Fischer and W. Jahnen-Dechent, BioRes. Open Access, 2013, 2, 374–384 CrossRef CAS PubMed.
- H. Kim, Korean J. Chem. Eng., 1990, 7, 1–6 CrossRef CAS.
- K. I. Draget, O. Smidsrød and G. Skjåk-Bræk, in Biopolymers Online, Wiley-VCH Verlag GmbH & Co. KGaA, 2005 Search PubMed.
- J. Chrastil, J. Agric. Food Chem., 1991, 39, 874–876 CrossRef CAS.
- E. Secchi, T. Roversi, S. Buzzaccaro, L. Piazza and R. Piazza, Soft Matter, 2013, 9, 3931 RSC.
- A. Blandino, M. Macías and D. Cantero, J. Biosci. Bioeng., 1999, 88, 686–689 CrossRef CAS.
- O. Smidsrød and G. Skjåk-Braek, Trends Biotechnol., 1990, 8, 71–78 CrossRef.
- A. Mikkelsen and A. Elgsaeter, Biopolymers, 1995, 36, 17–41 CrossRef CAS.
- H. Chen, W. Ouyang, B. Lawuyi and S. Prakash, Biomacromolecules, 2006, 7, 2091–2098 CrossRef CAS PubMed.
- F. Topuz, A. Henke, W. Richtering and J. Groll, Soft Matter, 2012, 8, 4877 RSC.
- J. a. Rowley, G. Madlambayan and D. J. Mooney, Biomaterials, 1999, 20, 45–53 CrossRef CAS.
- E. Alsberg, K. W. Anderson, a. Albeiruti, R. T. Franceschi and D. J. Mooney, J. Dent. Res., 2001, 80, 2025–2029 CrossRef CAS PubMed.
- A. D. Augst, H. J. Kong and D. J. Mooney, Macromol. Biosci., 2006, 6, 623–633 CrossRef CAS PubMed.
- K. Smetana, Biomaterials, 1993, 14, 1046–1050 CrossRef CAS.
- U. Hersel, C. Dahmen and H. Kessler, Biomaterials, 2003, 24, 4385–4415 CrossRef CAS.
- C. S. Chen, M. Mrksich, S. Huang, G. M. Whitesides and D. E. Ingber, Science, 1997, 276, 1425–1428 CrossRef CAS.
- L. E. Dike, C. S. Chen, M. Mrksich, J. Tien, G. M. Whitesides and D. E. Ingber, In Vitro Cell. Dev. Biol.: Anim., 1999, 35, 441–448 CrossRef CAS PubMed.
- L. Wang, R. M. Shelton, P. R. Cooper, M. Lawson, J. T. Triffitt and J. E. Barralet, Biomaterials, 2003, 24, 3475–3481 CrossRef CAS.
- H. Shin, S. Jo and A. G. Mikos, Biomaterials, 2003, 24, 4353–4364 CrossRef CAS.
- L. Perlin, S. MacNeil and S. Rimmer, Soft Matter, 2008, 4, 2331 RSC.
- S. L. Bellis, Biomaterials, 2011, 32, 4205–4210 CrossRef CAS PubMed.
- G. Cheng, V. Castelletto, R. R. Jones, C. J. Connon and I. W. Hamley, Soft Matter, 2011, 7, 1326 RSC.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra04135f |
| This journal is © The Royal Society of Chemistry 2014 |