The hydrogen bond – practice and QTAIM theory
M. Rozenberg*
Institute of Chemistry, The Hebrew University of Jerusalem, Jerusalem, 91904, Israel. E-mail: mark.rozenberg1@mail.huji.ac.il
First published on 9th June 2014
Abstract
The direct proportionality between the energy (enthalpy) of the hydrogen bond and the intensification of the infrared stretching mode of the AH group on formation of the H-complex A–H⋯B [A. V. Iogansen, Spectrochim. Acta, Part A, 1999, 55, 1585] is compared empirically with the linear correlation of stabilization energies, in the range of 1–70 kJ mol−1, with the electron density of the H-bond critical point, which follows from the published computational results of the quantum theory of atoms in molecules (QTAIM). Very close similarity is seen between the increase in IR intensity on H-bond formation as ΔA1/2 ≡ AH1/2 − A01/2 (subscripts H and 0 relate to bound and free AH groups, respectively) and the electron density, ρ, in their relevance to the energy. Despite the fact that both parameters, ρ and ΔA1/2, are defined by the same wave functions of the electronic and vibrational states of a system, the direct interrelation between two parameters of different dimensionalities is not obvious. The suggestion is made that IR intensities in the spectra of H-complexes, in addition to the results of diffraction methods, can be a source of electron-density data as ρ(rc) (e a−3) = 10−2(ΔA1/2) (A, 10−4 cm mmol−1).
The quantum theory of atoms in molecules (QTAIM) is now accepted as a powerful instrument in studies of intermolecular interactions including those involved in hydrogen bonding.1 The topological properties of electron density at the critical point of the H-bond have been used to obtain information on the nature of this interaction. Electron density (ρ(rc)) and the Laplacian of the electron density (∇2ρ(rc)) at the critical point of a hydrogen bond have been used as the criteria for quantifying the H-bonding interaction. It was found that both topological parameters are linearly related to the stabilization energy of the H-bond, i.e. with H-bond strength.1 It is remarkable that the energy of interaction is governed by only one parameter – ρ(rc) – over the whole range of interaction energies, from the very weak (van der Waals) level up to that of strong hydrogen bonds. This approach is the opposite of the usual description of hydrogen bonds as a sum of different components – electrostatic, polarization, delocalization, charge-transfer – whose contribution varies as the energy of interaction and the whole range of H-bonding cannot, obviously, depend on one parameter.
Besides the theoretical approach of QTAIM, quite a long time ago the experimental direct proportionality was suggested, which also described the whole range of H-bond energies with only one parameter – the intensification of the IR stretching mode νs of the acidic group AH on H-complex formation.2 Taking into account that both – theoretical and experimental – approaches are formally very similar, the comparison of the two is attempted here on the basis of published in literature data. The rationale for a such comparison is justified because the electron density ρ(rc) and IR intensity Ai∞(∂μ/∂Qi)2 (or the square root of the intensity of a fundamental IR band (Ai1/2)), which in the harmonic approximation, correspond to the derivative of the dipole moment μ with respect to the normal coordinate Qi of the particular (nondegenerate) vibration, are defined by the same wave functions of electronic and vibrational states of a system.
There are many studies of H-bond complexes of separate systems with the QTAIM theory (see f.e. ref. 1). Here, several reports were chosen, in which a large number of the model intermolecular binary complexes was systematically studied3–21,23–28 – from the weak van der Waals to the typical H-bonds – with the H-bond strength from almost zero to approximately 70 kJ mol−1. The results of the chosen studies, which include a comprehensive number of systems, are presented in the Table 1.
| No. | a | b | n | References |
|---|---|---|---|---|
| 1 | −6.5 | 1671 | 34 | 4–6 |
| 2 | −6.5 | 1222 | 4 | 7 |
| 3 | 0.78 | 986 | 14 | 8 |
| 4 | −2.9 | 967 | 7 | 9 |
| 5 | −10.7 | 1416 | 19 | 10 |
| 6 | −2.8 | 1002 | 20 | 11 |
| 7 | −6.65 | 1515 | 15 | 12 |
| 8 | −16.2 | 2152 | 14 | 13 |
| 9 | 0.5 | 690 | 8 | 14 |
| 10 | −1.1 | 878 | 28 | 3 and 15 |
| 11 | −14.3 | 1791 | 9 | 16 |
| 12 | −5.4 | 996 | 24 | 17 |
| 13 | 4.4 | 820 | 11 | 18 |
| 14 | 1.8 | 786 | 11 | |
| 15 | −27.8 | 1781 | 4 | 19 |
| 16 | 1.9 | 703 | 22 | 20 |
| 17 | 6.6 | 1151 | 15 | 21 |
| 18 | −0.09 | 850 | 23 | 23 |
| 19 | −10.6 | 732 | 30 | 24 |
| 20 | −16.4 | 991 | 30 | |
| 21 | −10.0 | 1629 | 6 | 25 |
| 22 | −22.3 | 2112 | 6 | 26 |
| 23 | −2.11 | 1107 | 11 | 27 |
| 24 | −7.6 | 1220 | 14 | 28 |
| Mean values | ||||
| −6.6 (s = 8.0) | 1215 (s = 440) | 377 | ||
The most recent data on more complex H-bonded cyclic molecules are added to those found in ref. 15. All these studies, which were mainly calculated on the MP/2 level of the theory with some variations, result in the linear relations E = a + bρ(rc) between the calculated stabilization energy E and the critical-point electron density ρ(rc) with a correlation coefficient R very close to unity. The results obtained by different authors are close (the slopes differ by not more than 30%). The deviations resulted possibly because of differences in the computational techniques and complexity of computed systems. The small intercepts (as compared to the whole energy range) which are about ±6 kJ mol−1, make these relationships closely related to the straight proportionalities. Being summarized these separate results can be expressed as a general relationship (with standard deviations shown in parentheses)
| E (kJ mol−1) = −6.6(8.0) + 1215(440)ρ (e a−3) | (1) |
From the data presented in the Table 1, it follows that the QTAIM theoretical approach indeed predicts the existence of linear correlation, close to proportionality, between the energy of hydrogen bonding (the energy of stabilization) and its topological property – the electron density ρ(rc).
The proportionality of the increase in intensity of the νs stretching mode of the acidic AH group on H-bond formation,2 which is compared with the theoretical approach above, was initiated by the well known experimental fact: “…the integrated absorption coefficient of the fundamental νs increases many-fold when a H-bond is formed…”.29 It was later confirmed experimentally and published in a final form as the direct proportionality of the intensification, ΔA1/2, of the ν1 band and hydrogen-bond enthalpy, ΔH.
| −ΔH (kJ mol−1) = 12.2ΔA1/2 | (2) |
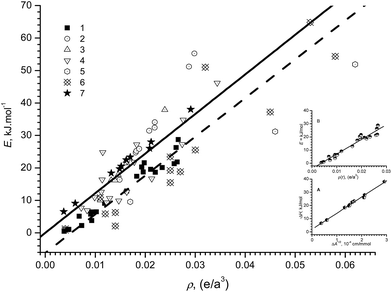
The parallelism of the two correlations (1) and (2) is striking and shows the direct connection of the effective charge A1/2 ∝ (∂μ/∂r) with the electron density ρ(rc) in the H-bond. The mean value of the computed proportionality coefficient b in relation (1) (taking into account the constant factor (102) in the abscissa) agrees well – within 1% – with the experimental value of relation (2). The comparison of both relations is shown in the Fig. 1.
The illustrative example of the similarity of the theoretical (from ref. 3) with experimental correlations (from Table 1 in ref. 31) is shown in the Fig. 1 inset. The data for very weak binary complexes with Xe are practically in the origin of the graph on the Fig. 1.35
Thus it can be concluded that the mutual interconnection between both theoretical ρ(rc) and experimental ΔA1/2 parameters does exist. It is also clear from the Fig. 1 that the two bold lines – solid and dashed – are very close. It should be pointed out that experimental correlation pertains to the association enthalpies (−ΔH), whereas theoretical calculation yields bond energy values. If it is taken into account that the difference between the two entities may be estimated not to exceed RT ≈ 2.5 kJ mol−1, then at room temperature and for freely translating molecules, the two correlations are practically coincident.
In conclusion, it can be stated that the empirical comparison reveals the correlation of the QTAIM electron-density parameter ρ(rc) with the experimentally measurable spectral parameter ΔA1/2 as
| ρ(rc) (e a−3) = 10−2(ΔA1/2) (A, 10−4 cm mmol−1) | (3) |
The empirical correlation −ΔH = 12.2 ΔA1/2 may possibly receive solid theoretical grounds for the first time and can serve as proof of the theory. From the practical point of view, the experiment, which is applicable to many different acid–base H-complexes in condensed phases (in diluted solutions and pure solvents), that cannot be computed because of their complexity, can be used as an instrument for a probe of the theory.
The possibility of measurement of electron density with eqn (3) can be important for theoretical and practical construction of the of H-bond acidity and basicity scales.
It is of note, that practically in all the literature devoted to the theory of the H-bond, the red shift of the νs band is indicated as one of the crucial characteristics of H-bond formation, (see f.e. ref. 21 and 22), despite the fact that its relative change from the free state does not exceed 30% for strong H-bonds, which are those usually discussed. However, the integral intensity changes by a factor of a hundred and is obviously the most impressive and fundamental characteristic of the H-bond. Up to now, the work of Iogansen31 and those which were done before it, remain unique in the field of hydrogen bonding. Furthermore, these IR-intensity studies, which cover a very large body of H-bond complexes, were possibly the first experimental evidence for the covalency of the hydrogen bond. These, study of together with other experimental results, such as an NMR spin–spin coupling36 and X-ray scattering,37 which relate to discrete systems, showed the inconsistency of the classical picture of H-bond interaction, as a sum of different components – electrostatic, polarization, delocalization, charge-transfer – and were not compatible with the “partial or non-covalent” nature of the H-bond.38
From very general point of view the H-bond can always be considered a covalent chemical bond – it is directional, short-range and, in general, saturated. The correlation between the energy of the H-bond and its force constant, which was found to be the same as those for the first-row hydrides,39 also supported this conclusion.
The only property, for which the H-bond differs from the usual chemical bond with energy of 400 kJ mol−1, is its weakness – the limits of energy, 1–60 kJ mol−1, with the strongest not exceeding 150 kJ mol−1. As a result, it follows that the low-frequency intermolecular deformational modes are excited at room temperature and this affects the width of the infrared bands of the proton modes, at least in condensed phase. If other causes of broadening, such as intermolecular vibrational interaction or crystal disorder, are removed, the bands become at low temperature narrow as usual in molecular spectra.40
References
- R. Parthasarathi and V. Subramanian, in Hydrogen Bonding-New Insight, ed. S. J. Grabowski, Springer, 2006, p. 1–50 Search PubMed.
- A. V. Iogansen, Dokl. Akad. Nauk SSSRR, 1965, 164, 610 CAS.
- R. Parthasarathi, V. Subramanian and N. Sathyamurthy, J. Phys. Chem., 2006, 110, 3349–3351 CrossRef CAS PubMed.
- R. J. Boyd and S. C. Choi, Chem. Phys. Lett., 1985, 120(1), 80–85 CrossRef CAS.
- R. J. Boyd and S. C. Choi, Chem. Phys. Lett., 1986, 129(1), 62–65 CrossRef CAS.
- R. D. Parra and X. C. Zeng, J. Chem. Phys., 1999, 110(13), 6329–6338 CrossRef CAS PubMed.
- M. T. Carroll and R. F. W. Bader, Mol. Phys., 1988, 65(3), 695–722 CrossRef CAS.
- S. J. Grabowski, J. Phys. Chem. A, 2001, 105, 10739–10746 CrossRef CAS.
- S. J. Grabowski, Chem. Phys. Lett., 2001, 338, 361–366 CrossRef CAS.
- S. J. Grabowski, J. Mol. Struct., 2002, 615, 239–245 CrossRef CAS.
- J. Poater, X. Fradera, M. Sola, M. Duran and S. Simon, Chem. Phys. Lett., 2003, 369, 248–255 CrossRef CAS.
- P. Kolandaivel and V. Nirmala, J. Mol. Struct., 2004, 694, 33–38 CrossRef CAS PubMed.
- F.-Q. Shi, J.-Y. An and J.-Y. Yu, Chin. J. Chem., 2005, 23, 400–403 CrossRef CAS.
- B. G. Oliveira, F. S. Pereira, R. C. M. U. de Araujo and M. N. Ramos, Chem. Phys. Lett., 2006, 427, 181–184 CrossRef CAS PubMed.
- B. G. Oliveira, R. C. M. U. Araujo, A. B. Carvalho and M. N. Ramos, J. Mol. Model., 2009, 15, 123–131 CrossRef CAS PubMed.
- M. Ziolkowski, S. J. Grabowski and J. Leszczynski, J. Phys. Chem. A, 2006, 110, 6514–6521 CrossRef CAS PubMed.
- D. Hugas, S. Simon and M. Duran, J. Phys. Chem. A, 2007, 111, 4506–4512 CrossRef CAS PubMed.
- A. M. Navarrete-Lopez, J. Garza and R. Vargas, J. Phys. Chem. A, 2007, 111, 11147–11152 CrossRef CAS PubMed.
- A. Zabardasti, S. Amani, M. Solimannejad and M. Salehnassaj, Struct. Chem., 2009, 20, 1087–1092 CrossRef CAS.
- S. J. Grabowski and P. Lipkowski, J. Phys. Chem. A, 2011, 115, 4765–4773 CrossRef CAS PubMed.
- F. Weinhold and R. A. Klein, Mol. Phys., 2012, 110(9–10), 565–579 CrossRef CAS.
- B. G. de Oliveira, Phys. Chem. Chem. Phys., 2013, 15, 37–79 RSC.
- D. Hugas, L. Guillumes, M. Duran and S. Simon, Comput. Theor. Chem., 2012, 998, 113–119 CrossRef CAS PubMed.
- H. Raissi, M. Yoosefian and F. Mollania, Int. J. Quantum Chem., 2012, 112, 2782–2786 CrossRef CAS.
- S. J. Grabowski, Phys. Chem. Chem. Phys., 2013, 15, 7249–7259 RSC.
- S. J. Grabowski, J. Mol. Model., 2013, 19, 4713–4721 CrossRef CAS PubMed.
- P. Durlak, K. Mierzwicki and Z. Latajka, J. Phys. Chem. B, 2013, 117, 5430–544023 CrossRef CAS PubMed.
- X. Zhang, X. Li, Y. Zeng, L. Meng and S. Zheng, Int. J. Quantum Chem., 2014, 114, 400–408 CrossRef CAS.
- G. C. Pimentel and A. L. McClellan, The Hydrogen Bond, W. H. Freeman and Co., San Francisco and London, 1960, p. 70 Search PubMed.
- A. V. Iogansen, in Vodorodnaya Svyaz (The Hydrogen Bond), ed. N. D. Sokolov, Izd. Nauka, Moscow, 1981, p. 112–155, Russian Search PubMed.
- A. V. Iogansen, Spectrochim. Acta, Part A, 1999, 55, 1585–1612 CrossRef.
- R. F. W. Bader, A. Larouche, C. Gatti, M. T. Carrol, P. J. MacDougall and K. B. Wiberg, Chem. Phys., 1987, 87(2), 1142–1152 CrossRef CAS PubMed.
- T. A. Keith, in The Quantum Theory of Atoms in Molecules, ed. C. F. Matta and R. J. Boyd, 2007, Wiley-VCH, p. 61–94 Search PubMed.
- A. F. Silva, W. E. Richter, L. A. Terrabuio, R. L. A. Haiduke and R. E. Bruns, J. Chem. Phys., 2014, 140, 084306 CrossRef PubMed.
- D. Gaszowski and M. Ilczyzyn, Int. J. Quantum Chem., 2014, 114, 473–480 CrossRef CAS.
- N. S. Golubev, I. G. Shenderovich, S. N. Smirnov, G. S. Denisov and H.-H. Limbach, Chem. – Eur. J., 1999, 5(2), 492–497 CrossRef CAS.
- E. D. Isaacs, A. Shukla, P. M. Platzman, D. R. Hamann, B. Barbiellini and C. A. Tulk, Phys. Rev. Lett., 1999, 82(3), 600–603 CrossRef CAS.
- S. J. Grabowski, Chem. Rev., 2011, 111, 2597–2625 CrossRef CAS PubMed.
- A. V. Iogansen and M. S. Rozenberg, Dokl. Akad. Nauk SSSR, 1971, 197(1), 117 CAS.
- A. V. Iogansen, Izv. Akad. Nauk SSSR, Ser. Fiz., 1989, 53(9), 1741–1745 CAS.
| This journal is © The Royal Society of Chemistry 2014 |