Aqueous poly(vinyl acetate)-based core/shell emulsion: synthesis, morphology, properties and application
Long Bai
,
Jiyou Gu
*,
Siqi Huan
and
Zhiguo Li
*
College of Material Science and Engineering, Northeast Forestry University, Harbin 150040, PR China. E-mail: lizgmse@nefu.edu.cn; dldgujy@nefu.edu.cn; Tel: +86-451-82190635
First published on 6th June 2014
Abstract
Core/shell structured composite emulsions have become an increasingly important interdisciplinary research field and deserve more attention because of their wide and potential applications in modern and advanced materials. The development of reliable synthetic and controlling approaches to composite emulsion with well-designed morphology, composition and functionality is of scientific importance and technological interest. The generation of poly(vinyl acetate)-based core/shell emulsion with phase separation can be achieved through seeded emulsion polymerisation, as well as multi-stage emulsion polymerisation. Insights on the fabrication of poly(vinyl acetate)-based core/shell latex particles with well-defined morphology and architecture have been substantially developed by recent advances in the morphological control mechanism of thermodynamics and kinetics. The factors affecting the formation of poly(vinyl acetate)-based core/shell emulsion, including polymerisation conditions, type and nature of monomer and initiator as well as grafting structure, are discussed. Finally, the properties of the poly(vinyl acetate)-based core/shell emulsion in mechanical, film-forming and adhesive performance, and its applications and potential functionalities are also briefly described.
1 Introduction
During the past 50 years, composite polymer emulsions, which are composed of well-designed structured latex particles, have attracted considerable attention and have provided producers with a broad landscape upon which to create a rich combination of tailored properties.1,2 One of the most promising aspects of structured composite emulsion is that of regulating the internal structure of the latex particle without changing (or only slightly changing) the chemical composition of the emulsion polymer. The fabrication of structured emulsion via manipulating three-dimensional spatial structure of latex particle could be achieved by structured polymer manufacturing technique derived from emulsion polymerisation.3–5Core/shell (CS) composite emulsion is developed from structured polymer manufacturing technique and belongs to non-homogeneous emulsion. Structured latex particles create a more versatile class of materials having superior and unique colloidal and physical properties that differ from particles of random copolymers or from emulsion blends. Compared with the counterparts of random copolymers or emulsion blends, composite emulsion with CS architecture can enhance water-, heat- and creep-resistance as well as adhesive strength remarkably and reduce the minimum film-forming temperature of polymer emulsion simultaneously, and more importantly, provide possibly synergistic effect between the components.6 Thus, properties of polymer emulsion arising from either core or shell polymers can be quite diverse by changing the core to shell ratio or the constituents of core and shell. Furthermore, it is conceptually possible to produce CS latex particles with a continuous, radial gradient of chemical composition, as well as to produce surface modified particles where the outermost region of the particle has a particularly unique composition, which can endow composite emulsion with novel functionalities brought about by various morphologies.7 The investigations on CS latex featuring well-defined morphology have largely become an increasingly important interdisciplinary research field, not only because of the tremendous demand of modern material science, but also of their scientific importance in the areas of colloid and interface.8–11 Since the first CS emulsion product was marketed as K-120 by Rohm & Haas in 1957, CS emulsion has been continuously developed and widely used in a variety of everyday products, such as advanced materials,12–15 architectural coatings and adhesives,16–19 drug carrier,20,21 electronic industry22–24 and membrane separation.25 It gradually becomes the leading edge of emulsion polymerisation from both industrial and academic perspectives.
Poly(vinyl acetate) (PVAc) emulsion is environmentally friendly and exhibits superior prospects due to its waterborne nature, having lower volatile organic compounds and better film-forming property.26–28 Nevertheless, its inherent shortcomings, such as poor heat-, water- and creep-resistance, make its applications limited in specific fields.29 In order to ameliorate the drawbacks of PVAc emulsion, great effort has been made through various experimental techniques and modified approaches. Among them, it is considered that CS manufacturing technique is a promising approach to improve disadvantages of PVAc-based emulsion. The fundamental concept to obtain desirable performances through PVAc-based core/shell (PVAc-CS) emulsion could be materialised by introducing various components into the core or/and shell, thus exerting composite effect of CS structure to meet the requirements under the precondition of keeping single-component PVAc-based emulsion. Additionally, fabrication of PVAc-CS emulsion is also an innovative approach to endow PVAc-based emulsion with peculiar functionality. In general, taking advantages of CS morphology, PVAc-CS latex particle could be incorporated with polymeric or non-polymeric functional materials, such as bio-macromolecules (starch, cellulose), organic compounds (silicon, fluorine) and inorganic nanoparticles (POSS, n-MMT), to give a rich combination of functionalities.30–32
With all of the advantages to be gained from CS latex particle, and for further insights into the nature of PVAc-CS emulsion, a systemic and comprehensive overview with regard to formation, morphology and application can play a significant role in better understanding and facilitating the development of PVAc-CS emulsion both in theory and practice. Therefore, in this article, we summarise the formation and morphological control mechanism, the factors affecting the formation, as well as the properties and applications of PVAc-CS emulsion. The content of this paper will be restricted to emulsion polymerised particles with CS structure. This will exclude the significant amount published information covering interfacial polymerisations,33–35 dispersion and suspension processes leading to PVAc-CS particles.36,37 A review of the work undertaken with these points in mind, and of the results obtained, as well as an analysis of the prospects of future developments in this field is presented below.
2 Thermodynamics of core/shell latex particle
The interest in fabricating structured latex particles has grown as the theoretical knowledge has advanced. CS emulsion polymers are structured composite particles consisting of at least two different components with distinguishing thermodynamic properties, e.g. hydrophilic or hydrophobic nature of components, in which one component at the centre forms the core phase and the other in principle forms the shell phase of the particles.38 Therefore, it has been conceptualised that the formation of CS structure is the result of phase separation within the particle induced by thermodynamics during the polymerisation. Thus, the described concepts lead to the idea that thermodynamics is the primary cause for different morphologies of latex particle.39In terms of thermodynamics, latex particle morphology is basically controlled by the Gibbs free energy change, which is a calculable thermodynamic variable for describing the interfacial behaviour of systems containing various types of polymers. Thermodynamics allows us to calculate the Gibbs free energy change for morphological development of the particle during the polymerisation as a combination of terms describing interfacial energies. Sundberg et al.40 presented a thermodynamic analysis for the morphology of a variety of types of polymers which encapsulated a relatively large-sized oil dropt. It was indicated that the driving force for the development of morphology was equivalent to the Gibbs free energy change for the process. Chen et al.41 derived a mathematical model using a thermodynamic analysis to describe the free energy changes corresponding to different composite latex particle morphologies shown in Fig. 1. Their analysis also revealed that the interfacial tension of each phase was the key factor, which governed the formed morphology. The total free energy change for all types of composite latex particle shown in Fig. 1 can be expressed by eqn (1).
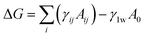 | (1) |
Subsequently, Chen et al.43 calculated the free energy change associated with the formation of a CS latex particle and resulted in an eqn (2):
| ΔG/A0 = γ12 + γ2w(1 − Φp)−2/3 − γ1w | (2) |
Based upon previous results and experimentally measured interfacial tensions, Durant et al.45 offered another approach to utilise eqn (1) to derive a free-energy surface which presents a clear and visual representation of the relative free energies of all possible equilibrium morphologies for two-component particles. Fig. 2 shows an example of such a free-energy surface where the point of minimum energy denotes a cross-sectional representation of the preferred latex particle morphology. In Fig. 2, the interfacial tension is plotted on the vertical axis and the preferred particle morphology with the lowest free energy is represented on the horizontal plane. According to Fig. 2, one can not only identify the minimum point and predict the preferred particle morphology accordingly, but also visualise the slope and trend of the surface surrounding the minimum point. When the lowest point is located within a rather flat area on the energy surface, e.g. the minimum point in Fig. 2, there are various particle morphologies which possess nearly equal energies. In such a case, very accurate values of the interfacial tensions are required to obtain predictions in agreement with experiment. Contrarily, with steep contours adjacent to the minimum point, there is little doubt that the predicted morphology should match that obtained experimentally, assuming that all the values of interfacial tensions used in the calculations are correct. Therefore, it was considered by Durant et al. that this free-energy surface could make a contribution for better understanding of the factors controlling the equilibrium morphology of latex particles.
 | ||
| Fig. 2 An example of free-energy surface for possible equilibrium morphologies45 Copyright, 1995 John Wiley & Sons, Inc. | ||
3 Formation and morphological control mechanism of PVAc-CS emulsion
The “particle design” concept that provided an important theoretical guidance for fabrication of CS emulsions was proposed by Okubo et al.46–49 Through the scientific designs at the molecule level, the formation mechanism of PVAc-CS latex particles and the relations between latex particle morphologies and reaction parameters were explored, greatly facilitating the preparation and morphological control of PVAc-CS latex particles.3.1 Formation mechanism of PVAc-CS emulsion
As mentioned above, the formation of PVAc-CS latex particle is the total result of macroscopic phase separation during the polymerisation. The degree of phase separation within latex particle is determined by various factors, including the compatibility of different polymer phases, the grafting reaction between growing chains and preformed polymer from a previous stage, the mobility of polymeric components, and the thermal history of the emulsion.50 CS structured latex particle featuring phase separation is usually designed and fabricated by a series of consecutive emulsion polymerisation sequences with different monomer types, where the second (third or more) stage monomer is polymerised in the presence of seed latex particles.51–57 This process can also be called two-stage or multi-stage seeded emulsion polymerisation. Various morphologies obtained from two-stage seeded emulsion polymerisation are presented in Fig. 3. It can be seen from Fig. 3 that possible morphologies formed are core/shell, moon-like structures, occluded structures and inverted core/shell. In addition, it is considered that various alternative structures described as confetti-, raspberry-, and sandwich-like could be the intermediates of phase separation.58 Lee et al.59 presented an extremely useful template of the types of structures that one should expect to see in two-stage latex particles. These morphologies included completely and incompletely phase separated structures, and served as a good qualitative guide. Eliseeva60 and Yuan et al.61 also investigated the relationship between phase structures and latex particle morphologies. It has been demonstrated that the particle morphologies could be determined by micro-phase separation in composite latex particle.If seed polymer could be swollen by second-stage monomer, phase separation will occur during the seeded polymerisation, leading to a variety of morphologies.62 As polystyrene (PSt) seed polymer could be swollen by acrylonitrile (AN) and its mixture, seeded emulsion polymerisation of AN within PSt seed particles was carried out by Vanderhoff et al.63 They deduced that particle formation, which was took place by polymerisation within the existing latex particles, was achieved by the mechanism that the polymerisation of second-stage monomer was in the core polymers swollen with second-stage monomers. According to particle size and thin layer chromatographic results, it was found that there existed larger size of core-PSt and P(St–AN) copolymer shell with a zone of PSt-grafted P(St–AN) copolymer in between. Obviously, phase separation was produced within larger size CS latex particle in their experiments. Meanwhile, it was also demonstrated that even phase separation occurred, grafting structure indeed existed in CS architecture.
Once seed polymer could hardly be further swollen by second-stage monomer, the second-stage monomer would be mainly polymerised at the periphery of seed particle surface during the polymerisation, and a polymer shell can be produced.64 Okubo et al.65 focused on the formation mechanism of structured emulsion prepared via seeded emulsion polymerisation of styrene onto poly(ethyl acrylate) (PEA) seed particles. It was found that the locus of second-stage monomer polymerisation tended to locate at the periphery of seed particle surface. Moreover, it was demonstrated that second-stage monomer was initiated at the surface layer, so as to make the resultant second-stage polymer also located on the surface. Therefore, it was reasonably deduced that the formation mechanism of latex particle with phase separation structure was controlled by the polymerised position of second-stage monomer.
The compatibility of PVAc and other polymers is a major cause for phase separation in PVAc-CS latex particle. By rule of thumb, the greater the incompatibility of polymer pairs, the greater the extent of phase separation within latex particle. If PVAc-CS latex particle is consisted of hydrophilic PVAc and hydrophobic polymers, incompatibility between such polymers might be enhanced due to the obviously hydrophilic difference, and then phase separation could be generated, leading to PVAc-based CS or ICS structured emulsion.66 Hergeth et al.67 explored the effect of polymer compatibility and water solubility of monomers on the formation of CS latex particle in the system of polystyrene (PSt) and poly(vinyl acetate) (PVAc). According to experimental result, it was indicated that CS structured particles of PSt/PVAc or PVAc/PSt could be produced due to the phase separation which was resulted from incompatibility of such polymers. However, the formation process of either PVAc or PSt shell polymer was different. When styrene was used as second-stage monomer, the “raspberry” structure was firstly formed due to heterocoagulation of hydrophobic PSt domains onto PVAc core surface.68 When increasing the conversion of styrene monomer, these PSt domains could gradually grow, touch each other, and build up an entire shell phase. When vinyl acetate was second-stage monomer, the shell phase was almost complete from the beginning due to its hydrophilicity.
3.2 Morphological control mechanism of PVAc-CS emulsion
Although the formation of PVAc-CS latex particle is determined by phase separation, the final morphologies are further affected by a variety of factors.69–71 The design and control of PVAc-CS latex particle morphology is crucial in order to meet the end-use requirements for these materials. In addition, a better understanding of the morphological aspects of PVAc-CS latex particle may shed some light on the utilization mechanism for other CS emulsion products. Therefore, it is of great significance to study morphological evolution of PVAc-CS latex particle for precisely controlling and manufacturing well-defined PVAc-CS latex particle. Currently, most of the factors which control composite latex particle morphology are mainly focused on two broad categories: thermodynamics and kinetics.Zhao et al.75 studied the morphological evolution of emulsion polymerisation of butyl acrylate (BA) in the presence of PVAc seed particle. Thermodynamic analysis was derived to calculate the interfacial free energy changes corresponding to various morphologies. It was predicted that ICS structure, PBA/PVAc, with minimum interfacial free energy change was the thermodynamically preferred morphology. Nevertheless, according to their experimental conclusions, CS latex particle with PBA shell could be produced at the end of the polymerisation. It was postulated that this was attributed to the restricted mobility of polymer chains closely related to high viscosity inside the particle. When such emulsion was deposited at room temperature for one year and a half, the latex particle with CS morphology was changed into ICS structure which was consistent with the thermodynamically predicted result. This was demonstrated that the ultimate equilibrium morphology of latex particle (if the aging time approached infinity) was determined by thermodynamics regardless of initial latex particle structure.
Recently, in order to accurately predict the morphologies of PVAc-CS latex particle, great effort has been devoted to simplify thermodynamic calculations or find simple thermodynamic criterion. Gonzalez et al.76 utilized minimum interfacial free energy as a thermodynamic criterion to predict equilibrium morphology of composite latex particle. Two parameters were defined as T and U for morphological prediction. T and U was calculated from corresponding interfacial tensions of γij, and a diagram representing particles morphologies was made according to the relationship between T–U and particle morphologies. Based upon this diagram, the T–U parameters of the thermodynamically preferred latex particle with CS structure were determined. The results of these calculations as a function of the following eqn (3) and (4) were presented in Fig. 4.
| T = |γ2w − γ12|/γ1w | (3) |
| U = γ12/γ2w | (4) |
 | ||
| Fig. 4 Equilibrium morphologies for V1 = 0.05 and V1 = 0 × 10 (ref. 76) Copyright, 1995 American Chemical Society. | ||
The research on CSMT applied in controlling and predicting the morphologies of PVAc-CS latex particle is highly attractive. In PVAc-CS emulsion system, CSMT predominates the equilibrium morphology of latex particle regardless of initial structures. Nevertheless, there are some limitations for CSMT. CSMT is suitable for particular systems, such as PVAc/PBA and PVAc/PSt,73 which is attributed to the difficulty in keeping a stringent balance between all factors affecting the interfacial tensions in a non-crosslinked system. Furthermore, it is almost impossible to accurately measure thermodynamic parameters separately. In addition, even if CS morphology would represent an equilibrium structure controlled by CSMT, kinetic effect might prevent the particle from attaining its equilibrium morphology. The competition between phase separation and the second-stage polymerisation kinetics can produce non-equilibrium morphologies due to the diffusional resistance caused by high local viscosity.77 In such cases, particles with occlusions or multiple surface domains may coexist. Therefore, the research on morphological evolution of latex particle throughout the polymerisation controlled by kinetics is of great necessity.
CSMK involves polymer chain initiation, growth, transfer, termination and migration as well as forming CS structure. Based on CSMK research, control mechanism of CSMK is mainly classified into two types: free radical distribution and polymer chain migration. Free radical distribution could be considered to control the locus for polymerisation initiation, and then to control the proceeding of the polymerisation. In PVAc-CS emulsion polymerisation, waterborne initiator is commonly used to initiate polymerisations in aqueous, producing oligomer chains with hydrophilic residue of initiators. In this case, the kinetic process of PVAc-CS emulsion polymerisation can be remarkably influenced by such free radicals and its distribution. The non-uniform distribution of free radical within latex particle is ordinarily originated from the hydrophilic nature of initiator residue. Delacal et al.79 simulated the morphological evolution of PVAc-CS latex particles according to free radical distribution within latex particles. A model related to free radical distribution and monomer concentration gradient was presented to simulate morphological evolution. Experimental results for latex particles with P(BA–VAc) core and PVAc shell were obtained at such conditions that the free radical concentration was higher at the outer surface of seed particle. It was pointed out that the higher radical concentration at particle surface resulted from anchoring effect of hydrophilic end-group of growing polymer chains. Therefore, it was demonstrated that PVAc-CS latex particle morphological evolution was mainly influenced by free radical distribution within latex particle.
Diffusion of free radical is also of great influence on free radical distribution. A series of experimental conditions, such as the flux of free radical, types of free radical and the nature of seed polymers, can be used to adjust and influence free radical diffusion. Jonsson et al.80 prepared two-phase latex particle by seeded emulsion polymerisation of methyl methacrylate (MMA) in the presence of PSt seed particle under various conditions, including different feed methods, rates of polymerisation, viscosity within latex particle. It was implied that the formation of CS structure was required to suppress free radical diffusion within latex particle. In this work, the occluded structured latex particles with PMMA domains in a continuous PSt matrix were produced because of high diffusion level of free radical within latex particle under all polymerisation conditions. Therefore, it was considered that the diffusion of free radical indeed played a significant role in determining CS structure. In most situations of practical interest, the successful preparation of stable CS particles must rely entirely on prohibiting free radicals generated in aqueous to transport within seed particles. Stubbs et al.81 explored the relationship between particle morphology and polymeric free radical diffusion in the systems of glassy-PMMA/PSt and soft-PMA/PSt. The equilibrium morphologies of latex particles in these systems were expected to be ICS structure with forming PSt cores. In these experiments, the diffusion extent of polymeric free radicals within latex particles was defined as penetration ratio, which was the quotient of the frequency for polymeric free radicals to diffuse and that for free radicals to terminate. As the penetration ratio was increased, it was considered that polymeric radical diffused from particle surface towards particle centre. When the penetration ratio of radical was smaller, CS structure in the system of PMMA/PSt was obtained. Whereas, when the penetration ratio was larger, the ICS structure of PSt/PMA latex particle was achieved. Therefore, it was demonstrated that the lower penetration ratio of polymeric free radical, namely restricted free radical transport, was responsible for the formation of non-equilibrium CS latex structures.
CSMK could also be controlled by polymer chain migration. Polymer chain migration determines the kinetically achievable morphology. It is considered that polymer chain migration is constantly ongoing during the formation and easy to be affected by various factors, such as polymerisation temperature, monomer feed rate, internal viscosity and hydrophilic end-group. Vanderhoff et al.82 proposed that new polymer chains formed and located on original polymer surface due to the effect of hydrophilic end-group of growing polymer chains, with which the mobility of polymer chain was restricted. Thus, polymer chain migration could be utilized to control the morphological evolution of latex particle. Moreover, it is accepted that when newly formed polymer chains are incompatible with the polymer previously produced, polymer chain migration would play a key role in controlling particle morphology. The mobility of polymer chain was explicitly demonstrated by experimental conclusions of Min et al.83 through observing PBA/PSt particle morphologies at various storage time (Fig. 5). Based on polymer chain migration, ICS latex particle with PSt core and PVAc shell was prepared by Ferguson et al.84 through an inversion process. In the experiment, styrene was firstly polymerised in the presence of PVAc seed particles, and then PSt chains migrated towards particle center, producing ICS structure. While the ICS morphology was thermodynamically favored, the conditions of polymer chain migration needed to be optimized so that it was kinetically achievable. Compared with PSt chain mobility, it was found that the mobility of PVAc chain was a more critical factor for reversal of core and shell. Therefore, it was demonstrated that the morphological evolution of ICS structure was attributed by fast mobility of PVAc chain toward particle surface layer. In order to fabricate PSt/PVAc latex particles with ICS structure, a number of methods were implemented, such as reducing the molecular weight and degree of branching of PVAc, minimising the grafting reaction of PSt radical onto PVAc chain and further increasing the hydrophilicity of PVAc.
 | ||
| Fig. 5 Electron micrographs of PBA/PSt CS latex particles (a) shortly after polymerisation (b) after 6 months (c) after 10 months (d) after 12 months83 Copyright, 1983 John Wiley & Sons, Inc. | ||
Polymer chain migration is significantly controlled by viscosity within latex particles.80,85,86 When viscosity is low, migration resistance is negligible, leading to ultimate equilibrium morphology. However, when local viscosity is high enough, polymer chain migration is restricted, and a variety of non-equilibrium morphologies are formed. The mobility of polymer chain could be adjusted by viscosity within latex particle, and then the morphological evolution is influenced by viscosity. Zhao et al.75 proved that the viscosity effect on polymer chain migration was a determinant for morphological evolution in the system of PBA/PVAc ICS emulsion. The multi-particle morphology within PVAc matrix was observed before attaining ICS structure, which was attributed to the restricted mobility of polymer chain caused by high local viscosity. Possible morphological development pathway of ICS structure with PBA core and PVAc shell is shown in Fig. 6. Winzor et al.87 investigated the morphologies of composite latex particle as a function of various viscosity. It was pointed out that viscosity within latex particle could increase dramatically when monomer conversion was high, and then entirely phase separation might become difficult to be achieved within the timeframe of the polymerisation. This would likely lead to the existence of occluded morphologies other than fully phase-separated structure, such as CS morphology. It was attributed that the higher internal viscosity restricted the mobility of polymer chain due to the predominant effect of viscous force competed with van de Waals force.
 | ||
| Fig. 6 Possible morphological development pathway of PBA/PVAc ICS latex particle75 Copyright, 2003 Elsevier Ltd. | ||
In addition to internal viscosity, polymer chain migration is also influenced by glass transition temperature (Tg). Under starved feeding conditions, when the Tg of seed polymer is higher than polymerisation temperature, seed polymer is glassy with small free volume, within which the mobility of polymer chain is blocked. Therefore, polymer chain migration could be influenced by regulating the difference between the Tg of seed polymer and polymerisation temperature. Karlsson et al.88 synthesized poly(styrene–butyl acrylate)/poly(methyl methacrylate–butyl acrylate–methacrylic acid) latex particle with heterogeneous structures, in which the Tg of seed polymer ranged from 20 to 100 °C and shell composition was constant with a Tg of 20 °C. When the Tg of seed polymer was below reaction temperature (70 °C), polymer chain mobility within seed particles was unobstructed, thus the morphological evolution was towards thermodynamic equilibrium, namely CS structure in such systems. When the Tg of seed polymer was above reaction temperature, the mobility of polymer chain was restricted, and the particle structures could diverge from CS morphology to other heterogeneous morphologies, e.g. irregular shape. Therefore, it was demonstrated that polymer chain migration within latex particle was correlated to the difference between Tg of seed polymer and polymerisation temperature.
The research on morphological evolution mechanism of PVAc-based latex particle controlled by CSMK is almost applicable to any system, especially for non-equilibrium system. It is considered that not only CSMK is a description of latex particle morphological evolution process, but also CSMK is a determinant for final latex particle morphology in some cases, namely thermodynamic non-equilibrium morphologies.89 It also provides a firm theoretical basis for morphological evolution of PVAc-CS latex particle. However, there exist some limitations for CSMK applications. First, the measurement and characterization of kinetic parameters are primary problems and remained to be resolved. Second, particle morphology controlled by kinetics is usually not consistent with thermodynamic prediction during the polymerisation and storage. In spite of this, CSMK is still a promising approach to control and synthesize PVAc-CS emulsion. Furthermore, a more comprehensive mechanism for morphological evolution is specially needed to accurately regulate and control PVAc-CS latex particle morphology.
3.3 Morphological model of PVAc-CS emulsion
CSMT dominates the ultimate equilibrium morphology of composite latex particle, while CSMK determines whether such thermodynamically preferred morphology can be achieved. In the view of thermodynamics for morphological control, it is considered that the migration resistance of resultant polymer chains is negligible once polymer chains are formed, eventually attaining thermodynamic equilibrium.40,43,44 Whereas, it is assumed by kinetics that morphological evolution will gradually stop along with the polymerisation proceeding, and polymer chains no longer move as long as formed.90 In fact, it is demonstrated that even polymer chains have been formed, it can still move due to its incompatibility with the existing polymer, but hardly achieve thermodynamic equilibrium in the end because of kinetic barrier.83,91,92 Therefore, in order to better understand the roles of thermodynamics and kinetics in controlling PVAc-CS latex particle morphology, much effort has been increasingly devoted to have an insight into the nature of morphological evolvement. Among different approaches, structural model is deemed as a promising technique to achieve this goal.In terms of thermodynamic model, many constructive reports have been focused on developing evolving models that can be used to predict the equilibrium morphologies for different systems. Based on the theory of Gibbs free energy change, Sundberg et al.40 and Chen et al.41 proposed earlier thermodynamic models and equations, eqn (1), to calculate Gibbs free energy change and to predict the equilibrium morphologies of composite latex particles. Four equilibrium morphologies calculated from such models are shown in Fig. 1. For PVAc-CS emulsion systems, Zhao et al.75 further developed a thermodynamic model to calculate the Gibbs free energy change of PVAc-CS latex particle morphology during two-stage seeded emulsion polymerisation. Based upon eqn (1), four computational formulas that corresponded to four possible equilibrium morphologies of PVAc-based latex particles shown in Fig. 1 were accurately derived. Combining the thermodynamic parameters and the conformation of latex particles, the Gibbs free energy change of these equations were calculated. According to the results, morphology with minimum free energy change, namely equilibrium morphology of PVAc-CS latex particles, was predicted. During the simulant process, it was proven that the thermodynamic equilibrium morphology predicted by such model could be partly in accordance with the experimental polymerisation results. According to the above examples, it is anticipated that thermodynamic models can be used as a guidance to provide the producer with a better understanding of the design and fabrication of PVAc-CS latex particles in practice. Whereas, the development pathway and the intermediate structures of PVAc-CS latex particles are difficult to be revealed by thermodynamic models.
During the past decades, kinetic models also attract a great deal of interest in investigating the morphological evolution process of CS latex particle. Williams et al.93,94 firstly proposed a non-uniform structural model to explain the formation process of CS structure. Subsequently, Napper95 established an improved model similar to Williams' one to study morphological evolution of CS latex particles. In his model, it was hypothesized that the center of seed particle was relatively inaccessible to free radicals due to high local viscosity, producing a polymer shell at the peripheral of seed surface where the monomer was polymerised.
In recent years, Chen et al.96 and Mills et al.97 independently developed kinetic models to illustrate the formation process of CS latex particles controlled by diffusion of growing radicals. Chern et al.98,99 also proposed a CS kinetic model for non-uniform distribution of free radicals in the latex particle during two-phase emulsion polymerisation. Their Monte Carlo simulations of the free radical distribution successfully predicted that free radical concentration was statistically greater near the particle surface, providing a possibility to in situ produce polymer shell. Delacal et al.79 presented a diffusion-controlled kinetic model to simulate polymer particle structure according to the distribution of free radical and monomer of P(VAc–BA)/PVAc emulsion system. The model was applied to the simulation of the polymerisation of VAc on a P(VAc–BA) copolymer seed. It was found that the development of the particle morphology was mainly due to the profile of concentration of radicals in the particle. In simulant process, CS morphology of PVAc-CS latex particles was developed because of the higher concentration of free radicals in an outer shell of polymer particles. According to the above conclusions, it is found that when considering the nature of VAc monomer, it might be difficult to use kinetic-controlled models to reasonably describe the development of PVAc-CS latex particles. However, in spite of this, these kinetic models can also provide some useful knowledge of the role of polymerisation kinetics to produce expected PVAc-CS latex particle morphology during the polymerisation.
Although the proposed thermodynamic or kinetic models could independently reveal the influential mechanism of morphological evolution of PVAc-CS latex particle, there still exits some unresolved problems. Therefore, a more scientific and comprehensive model that simultaneously combine thermodynamics and kinetics deserves more attentions. In addition, with the development of theoretical and practical knowledge, it is possible to creatively combine different theories and acquire accurate conclusions that provide guarantee for establishing such structural model.
Cluster migration dynamics that firstly pioneered by Gonzalez et al.76,100 was a systemic theory to creatively combine thermodynamics and kinetics in recent studies. In such model, the Gibbs free energy and cluster migration dynamics were incorporated into its framework simultaneously. It was also pointed out that the driving force for cluster migration was the balance between the van der Waals force and the viscous force. Under these simulant circumstances, various equilibrium morphologies predicted, such as CS, ICS and occluded structure, could be achieved. It was proposed that the forming process of CS latex particle was controlled by the following series-parallel steps: (a) polymer chains were formed at a given position in the polymer particle. (b) If the newly formed polymer chain was incompatible with the polymer existing in the position where it was formed, phase separation occurred. Phase separation lead to the formation of clusters. (c) In order to minimize the Gibbs free energy, the clusters migrated toward the equilibrium morphology. Subsequently, Gonzalez et al.101 improved their cluster dynamic model for describing cluster nucleation, polymer reaction, polymer chain diffusion, and cluster migration. During cluster migration, the size of cluster could increase by polymerisation of monomer inside the clusters, diffusion of polymer chains into clusters, and coagulation with other clusters. It was demonstrated that the increasing rates of cluster size depended strongly on particle internal viscosity. Interestingly, both the initial volume of cluster and the rate coefficient for the nucleation of cluster had less influence on equilibrium particle morphology.
Based on the proposed concepts, the evolution of cluster dynamic process during two-stage polymerisation can be illustrated by means of the phase diagram presented in Fig. 7. In Fig. 7, point A represents the initial state, and as monomer 1 polymerizes, the particle composition moves along the line A–B. When the system reaches point B, the polymers are no longer compatible, and phase separation occurs. If the kinetics of the phase separation is slower than that of the polymerisation, the composition of the polymer matrix evolves along the line B–C–D, reaching the point D at the end of the polymerisation. Contrarily, the polymer matrix moves from B to D along the equilibrium line. The composition of the first cluster formed is given by point E. Polymerisation of monomer 1 in the cluster may move the composition of this cluster along the trajectory E–F, namely, off of the equilibrium line. However, the composition of the new clusters is always given by the equilibrium values. For example, point G gives the composition of the clusters formed when the composition of the polymer matrix is represented by point C. This means that, at a given moment, clusters of different compositions might be present in the polymer particle.
 | ||
| Fig. 7 Phase diagram of evolution of phase separation process100 Copyright, 1996 American Chemical Society. | ||
Cluster dynamics successfully combines thermodynamics and kinetics, and explains partial experimental results. It is an effective model for morphological evolution due to its theoretical integrality in the aspects of CS latex particle formation, the effect of free energy change, polymer chain migration and interaction based upon thermodynamics and kinetics. The applicability of this model has been proven by two-phase homo-polymerisation system of PSt/PMMA100,101 and PVAc/PBA.74 Additionally, as cluster dynamics commonly limits polymer chain to be formed at a given position in polymer particle, it is more suitable for batch or swollen systems. According to the above conclusions, it is worth noting that in most PVAc-CS structural models, many of requisite parameters are difficult to be set suitably due to the fact that the polymerisation process is complicated and variable. Therefore, simplified and empirical parameters with reasonable validity are also utilized to accomplish the calculations of PVAc-based model simulation.
In general, to successfully manufacture PVAc-CS latex particles, it is specially needed to prohibit the formation of secondary particles during the polymerisation according to the nature of vinyl acetate, leading to a challenge both in theory and practice. Ferguson et al.102 developed a model for growing CS latex particles of PVAc/PSt or PSt/PVAc via exploring conditions for avoiding the formation of secondary particles. During simulant process, the model system was described as homogeneous nucleation, and the simplified kinetic description containing easily specified parameter values was used to reduce the large amount of complex mathematical calculations. Furthermore, it was demonstrated that the results obtained from simple model could give acceptable semi-quantitative accord with the predictions from more sophisticated extended model, leading to a fact that simplified parameters were usable in PVAc-CS structural model. According to the results, the occurrence of secondary nucleation could be effectively suppressed by increasing solid content, by starved feeding condition, especially by decreasing seed particle size, which was distinctly shown in Fig. 8. Thus the formation of CS latex particle with either PSt or PVAc core could be achieved by utilizing the smaller seed particle and the higher solid content. It was also simulated that if polymer cores were large PSt particles, a large amount of secondary nucleation particles would be produced due to the rapid aqueous phase propagation of vinyl acetate in combination with the low particle concentration. Interestingly, if polymer cores were large PVAc particles, there would be relatively little secondary particle, which was implied that large PSt/PVAc CS latex particles could be created by inverse CS technique. Undoubtedly, such a simple nucleation model provides an impactful and practicable approach to control the morphology and polymerisation process of PVAc-CS latex particles through inhibiting the formation of secondary particles during the polymerisation.
 | ||
| Fig. 8 Predicted new particle number as a function of seed particle radius for emulsion polymerisation of VAc in the presence of PSt seed. Results are shown for three seed particle concentrations102 Copyright, 2002 Elsevier Ltd. | ||
The research on CS morphological model has laid a foundation for formation and morphological control mechanisms of PVAc-CS latex particle. The combination of CSMT and CSMK based upon structural model is a scientific and effective way to facilitate the development of well-defined CS structured latex particles. Meanwhile, simulant results of evolution of CS latex particle morphology could help find crucial factors for manufacturing PVAc-CS emulsion, offering an improvement for experimental trials of PVAc-CS emulsion polymerisation. It should be noted that PVAc-CS structural model is commonly appointed to specific systems, such as the systems without consideration of complicated parameters,102 so generalization and simplification of various morphological models are of great necessity and significance. Furthermore, more practical factors affecting the formation of PVAc-CS emulsion should be considered to incorporate into the framework of structural models, leading to a better simulation with more stringency.
4 Factors affecting the formation of PVAc-CS emulsion
The following discussion represents a description of important variables which affect the formation of PVAc-CS emulsion. The interplay of all the variables and factors necessary for manufacturing PVAc-CS latex particles is complex, and consequently needs to be considered precisely when designing and fabricating CS structure.4.1 Effect of polymerisation condition
PVAc-CS latex particles are commonly prepared via two-stage seeded emulsion polymerisation with either PVAc core or shell.66,67,75 During seeded emulsion polymerisation, polymerisation condition is a key to prepare PVAc-CS emulsion. Therefore, it is of great importance to explore the influence of polymerisation condition on the formation of CS latex particle. One of the interesting findings about the above concept was contributed by Karlsson et al.103 through investigating the relationship between polymerisation conditions and CS morphology. Depending on a series of polymerisation conditions, it was clearly found that the morphology of two-phase particles varied from a pure CS structure, over intermediate structures in which a shell surrounded a core containing discrete phase domains, to a structure in which the entire shell was present as discrete phase domains, more or less evenly distributed.Ferguson et al.104 explored the effect of polymerisation conditions on the formation of PSt/PVAc CS latex particles without secondary nucleation during seeded polymerisation of VAc in the presence of either small- or large-PSt seed particles. A variety of polymerisation conditions, including solid content of seed latex, polymerisation temperature, addition method of VAc monomer, were implemented to produce PSt/PVAc CS latex particle. It was pointed out that the size of seed particles was a decisive factor to the formation of CS architecture. For small-PSt/PVAc system, PSt/PVAc CS latex particle could be produced without forming secondary nucleation. Nevertheless, in the system of large-PSt/PVAc, it was found that either extensive secondary nucleation particle or unstable polymerisation process was obtained.
Moreover, the likelihood of the formation of secondary nucleation particle could also be influenced by seed particle number. Ferguson et al.102 and Maxwell et al.105 demonstrated that the rate of entry of aqueous radicals into pre-existing particles, which was of great influence on secondary particle formation of PVAc-CS latex, was determined by the product of seed particle number and size. For constant particle number, entry would actually be enhanced with increasing the size of seed particle, and consequently secondary particle number was decreased. However, this effect was swamped by the much stronger effect of decreasing the number of seed particle, because, at constant solids content, the number of seed particle had an inverse cubic dependence on seed particle radius. Hence there was a strong decrease in the frequency of entry, leading to a large increase in the number of oligomers growing to nucleation, and thus there was more secondary particle formation.
Based upon various residence times of second-stage monomer during the polymerisation, there are three kinds of addition method of second-stage monomer: batch, pre-swelling and semi-continuous.30,66,75,106 The main differences among the three addition methods are briefly listed in Table 1.
| Addition method | Process |
|---|---|
| Batch | Second-stage monomer and initiator are added into seed emulsion at the beginning of the polymerisation |
| Pre-swelling | Second-stage monomer is polymerised after swelling in seed emulsion for a certain period of time |
| Semi-continuous | Second-stage monomer and initiator are added into seed emulsion dropwise at a constant speed (known as starved feeding) |
CS or other possible morphologies, such as sandwich- or snowman-like structure, as well as grafting structures can be obtained through above three methods.107 It is obvious that different addition methods can lead to different forming processes of second-stage polymer. When batch and pre-swelling are employed, relatively low local viscosity in latex particles is achieved. This enhances the polymer chain mobility and promotes the occurrence of phase separation. For semi-continuous method, the monomer concentration in the particle can be maintained at a minimal value. The extremely high local viscosity creates a kinetic barrier towards polymer chain migration. It is worthwhile to note that among them, the semi-continuous method is utilized most frequently to fabricate desired particle morphology in cases where thermodynamically non-equilibrium morphologies are desirable.108,109
PVAc-CS structured emulsion can be produced through these three addition methods, especially by semi-continuous method. The nature of VAc monomer is hydrophilic, easily grafts with other monomer and diffuses quickly in aqueous phase.84 In batch or pre-swelling of PVAc-CS systems, when VAc monomer is used as the second-stage monomer, massive amount of secondary nucleation particles will be produced, which is disadvantageous for manufacturing PVAc-CS latex particle. When PVAc is employed as seed particles, secondary particles can be suppressed. However, the lower internal viscosity and higher hydrophilicity of PVAc can promote the reversal of core and shell polymers. Thus undesired PVAc-based latex particle morphologies may be produced. In semi-continuous system, no matter VAc monomer is polymerised in either first- or second-stage, the occurrence of secondary nucleation can be reduced due to low instantaneous monomer concentration. Meanwhile, many thermodynamically non-equilibrium PVAc-CS latex particle structure, such as PVAc/PSt and PVAc/PBA, can be constructed by this method due to the restricted polymer chain migration during the polymerisation.
Additionally, polymerisation temperature is also a pivotal factor to influence the evolution of CS latex particle morphology. A series of kinetic processes which affect the ultimate morphology of latex particle, including polymerisation rate, Brownian motion of latex particle and polymer chain mobility, could be significantly affected by polymerisation temperature. It has been reported that the lower polymerisation temperature is, the easier to form CS latex particle.80 This is attributed that when polymerisation temperature was lower than the Tg of seed polymer, the seed polymer would be glassy, and during second-stage polymerisation, penetration into seed particle could be blocked. Thus an external polymer layer would be formed, which leads to the formation of CS structured latex particle.
4.2 Effect of reactive monomer
In most CS systems, the type and nature of reactive monomer are closely related to morphologies of CS latex particle. It is indicated that various monomers could be applied through different ways for constructing CS latex particles. Based upon the Tg of different polymers, the reactive monomer can be classified into two main types: soft monomer and hard monomer. Eliseeva60 investigated the relationship between latex particle morphology and physical state of seed polymer (having different Tg). It was found that the physical states of different seed polymers could exhibit different mechanisms in determining latex particle morphology under identical conditions. When Tg of seed polymer was low, a viscous-flow state was existed during the polymerisation, and as a result the mobility of both seed and second-stage polymer couldn't be hindered, leading to alternative structures. On contrary, when seed polymer was hard, the mobility of both seed and second-stage polymer might be blocked, giving rise to CS structure. In PVAc-CS system, various soft or hard monomers could be introduced through first- or second-stage polymerisation, and then improve the properties of PVAc-CS emulsion. The soft monomers could improve film flexibility and adhesion by providing plasticisation, and the hard one could increase film hardness and strength as well as improves creep-resistance. Therefore, it is of significance to further understand the role of soft or hard monomers in fabricating PVAc-CS emulsion.In addition, it should be reminded that the hydrophilicity originated from the existence of hydrophilic group in monomeric unit is also a key nature for monomer classification, which is defined as relatively hydrophilic monomer and hydrophobic monomer. In two-stage CS emulsion polymerisation, hydrophilic monomer tends to locate at or migrate towards outmost surface of particle, leading to the formation of polymer shell of CS or ICS latex particles. Contrarily, hydrophobic monomer has a trend to be wrapped into particle through forming cores or migrating to particle core centres. Therefore, based on the nature of PVAc, the final morphology of PVAc-CS particle could be significantly affected by hydrophilic–hydrophobic difference between VAc monomer and other monomers during two-stage seeded polymerisation, which has been proven by different PVAc-CS emulsion systems, such as PVAc/PSt84,104 and PVAc/PBA.75,110
Although it is challenging to obtain CS structure in two-stage emulsion polymerisation of VAc and hydrophobic monomers, much effort has been devoted to develop promising methods to achieve this goal. One of the effective methods to prepare PVAc-based emulsion with CS morphology is to introduce coupling monomer due to its moderate hydrophilicity and miscibility. Ferguson et al.84 explored the effect of coupling monomer BA on the formation of PVAc/PSt CS emulsion during two-stage seeded polymerisation of St within P(VAcBA) seed particles. It was found that PVAc/PSt latex particle with CS structure might be produced by introducing coupling monomer. Therefore, having BA as a co-monomer could promote the formation of CS morphology in such system. Coupling monomer BA, which was more hydrophobic than VAc but hydrophilic than St, was mutually compatible with St and VAc, and served as a bridge during forming process. Furthermore, the first-stage branching and second-stage grafting would be generated in the presence of PBA, as PBA was even more prone to chain transfer to polymer chain than PVAc chain.111 Improving the hydrophilicity of hydrophobic monomer is considered to be another way to suppress inverse of CS structure.112 Wang et al.113 studied the effect of sodium styrene sulphonate (NaSS) on the formation of poly(styrene–methacrylic acid)/poly(styrene–NaSS) CS latex particle through enhancing the hydrophilicity of PSt by introducing hydrophilic NaSS. It has been proved that incorporating hydrophilic NaSS would improve the hydrophilicity of PSt shell. Thus CS latex particle with more hydrophilic P(St–NaSS) shell could be produced. Consequently, it is reasonably deduced that introducing NaSS as shell monomer in preparation PVAc-CS latex particle might be a novel approach to effectively suppress the formation of ICS structure due to its higher hydrophilicity than VAc.
Additionally, according to functionalities of different monomers, it is concluded that monomers providing cross-linking or special properties are collectively referred as functional monomers, such as alkyl carboxylic acid, divinyl benzene (DVB), glycidyl methacrylate (GMA). Noteworthy, over the past several years, the fabrication of PVAc-CS emulsion with functional monomers is under rapid development.151
4.3 Effect of initiator
For CS emulsion polymerisation, the selection and usage of initiator is of great significance to influence the morphological evolution during the polymerisation.114 Generally, the initiator primarily determines the loci where the polymerisation begins, as well as the nature of the growing polymer chain. Three kinds of initiator, including waterborne initiator, oil-soluble initiator and redox initiator, are mostly utilized in CS emulsion polymerisation. According to previous studies, it is demonstrated that latex particle morphology and its evolution strongly depends on the nature of various kinds of initiators.115 When oil-soluble initiator is used, the polymerisation is initiated within particles due to its hydrophobic nature, leading to insolubility of polymer chains with oil-soluble end-group. Thus the resultant polymers are prone to be wrapped into particles and further migrate to particle centres, giving rise to alternative morphologies.116 When waterborne initiator is employed, the polymerisation is initiated in aqueous phase, and the growing polymer chains with hydrophilic initiator ionic residue are distributed at the boundary between aqueous phase and particle phase, probably forming a stable shell layer through electrostatic repulsion. Furthermore, by introducing ionic end-group that resulted from initiator residue, the electrostatic repulsion can be generated between growing polymer chains and particle surface surfactant layer, with which the growing polymer chains may be excluded out the particles. Consequently, it is the fact that latex particles with CS morphology are easily obtained by using waterborne initiator.Although waterborne initiators are popularly used in PVAc-based emulsion polymerisation,117 it is needed to understand the particular role of waterborne initiator in PVAc-CS emulsion polymerisation. Sun et al.118 investigated the effect of various initiators, e.g. potassium persulphate (KPS) and 2,2-azobis(isobutyronitrile) (AIBN), on the morphology of PVAc/PSt and PBA/PSt composite emulsions. The sensitivity and no-sensitivity of equilibrium morphology with various initiators were proposed to classify CS emulsion systems. For no-sensitivity system, it was hard to change its final morphology by adjusting initiators. It is demonstrated that PVAc/PSt composite emulsion belonged to no-sensitivity system, which meant that the equilibrium morphology could stay stable even initiator was changed. The identical equilibrium morphology with ICS structure of PSt core and PVAc shell was obtained in PVAc/PSt systems with different initiators. Whereas, for sensitivity system, final morphology might be different when changing initiators. It is proven that PBA/PSt composite emulsion was sensitive to initiator. When initiator was changed, three different equilibrium morphologies, including CS, ICS and HS, were obtained in such system with AIBN, KPS/AIBN and AIBN/KPS as initiator, respectively.
4.4 Effect of grafting structure
In CS emulsion polymerisation, grafting structure has an important effect for core or shell polymers due to high frequency of chain transfer reaction. Grafting structure can increase the compatibility of core and shell as well as participated in stabilizing CS latex particle, which was reported by Vanderhoff et al.63 Therefore, grafting structure is of considerable importance in latex particle morphology.119 Min et al.83 studied the relationship of grafting structure and latex particle morphology during seeded emulsion polymerisation of St within PBA seed particles. It was found that grafting PSt to PBA cores was mainly occurred during seeded emulsion polymerisation. It was observed that the stability of CS latex particle during storage depended on the amount of graft copolymer formed. In the experiment, upon aging, PBA core polymers migrated out latex particles with a low degree of grafting to form dumbbell-shaped particles. Whilst, PBA/PSt CS latex particles with high degree of grafting degree remained spherical because of the stabilizing effect of grafting structure. Consequently, it was demonstrated that grafting structure in CS latex particle could increase the compatibility of core and shell polymers, and prevent the transformation of latex particle morphologies, leading to a stable CS structure.During PVAc-based emulsion polymerisation, grafting reactions of PVAc chain onto other polymers120 could easily occur because electron donating groups, −OCOCH3, of VAc monomeric unit directly links to the double bond, which leads to less active of VAc monomer121 and likely occurrence of chain transfer reaction122–124 during free radical polymerisation, thus facilitating grafting reaction. In order to confirm and investigate grafting structure in PVAc-CS system, Hourston et al.125 and Hergeth et al.126 studied the thickness of grafted layer in CS latex system of PMMA and PVAc by means of differential scanning calorimetry. In the experiments, it was demonstrated that there existed grafted interfacial layer between CS structured polymers of PMMA/PVAc or PVAc/PMMA due to macromolecular diffusion and grafting reaction of PMMA and PVAc at the interface. Therefore, it is reasonably deduced that grafting structure might be of great influence on formation and stabilization of PVAc-CS emulsion. When preparing PVAc-CS emulsion with thermodynamic non-equilibrium morphology, such as PVAc/PSt CS emulsion, the high degree of grafting structure could effectively suppress the inversion of core and shell polymers due to the fact that grafting structure provides resistance to prohibit polymer chain migration towards particle centres.
In addition, it has been reported that other factors, including the type and nature of surfactant,127–131 cross-linking degree of core and/or shell polymers,132–135 could also affect the formation of PVAc-CS structured emulsion and have been widely investigated.
5 Properties and applications of PVAc-CS emulsion
Structured PVAc-CS emulsion could evidently improve the performances of PVAc-based emulsion, especially in the fields of water-, heat-resistance and film-forming property, and provides many practical applications for PVAc-based emulsion. Even so, so far as we are aware, there are few reports about the properties and applications for PVAc-CS emulsion. In this section, these properties and applications will be discussed briefly.5.1 Properties of PVAc-CS emulsion
Introducing various polymers into PVAc-based emulsion through CS structure is an effective method to improve the intrinsic drawbacks of PVAc-based emulsion. It is also expected that new performances can simultaneously be endowed by incorporating functional monomers under the premise of not impairing the excellent properties of PVAc. Grigsby et al.136 evaluated the performance of PVAc-CS emulsion with hydrophobic PSt cores and PVAc shells through exploring the effects of PSt content and the morphologies of PSt/PVAc CS particles. The mechanical properties and adhesive performance of PVAc emulsion thin film was improved through fabricating CS structure. It was shown that up to 50% PSt (by mass) could be incorporated into PVAc-CS emulsion without compromising film-forming property of PVAc emulsion. Furthermore, it is demonstrated that the mechanical properties of latex films, especially the creep-resistance, were improved at high humidity. In the experiment, PVAc-CS emulsions were synthesized through CS seeded polymerisation, ICS seeded polymerisation and blending of PSt and PVAc homopolymers, leading to CS structure, acorn morphology and just blending of homopolymers, respectively. It was found that PVAc-CS emulsion with concentric CS structure performed better than either copolymers with an acorn morphology or blending.CS latex film is commonly used as main forms to achieve the applications of CS structured emulsion.137–139 It is well known that mechanical property is one of the most effective approaches to evaluate the properties of CS latex film.140 The mechanical properties of latex film casted by CS emulsion are determined by the viscoelastic relaxation under the forces exerted by surface tension and interaction between the macromolecules at particle interface. Furthermore, the mechanical properties are also related to Tg of core or shell polymers at particle interfacial zones. Delgado et al.78 studied the dynamic mechanical properties of P(BA–VAc)/PVAc CS latex film. Two obvious Tg were observed, in which the Tg of PVAc shell shifted to lower temperature zone of P(BA–VAc). Similar results were reported in the case of PVAc/PBA CS emulsions studied by Cavaille et al.141 It was emphasized that the existence of an interfacial transition layer between core and shell could strengthen the binding of the two phases. Therefore, a buffer zone resulted from transition layer leads an increase in inter chain interaction and helps to scatter or transmit stress, in which the performance of PVAc/PBA CS latex film was significant enhanced. In order to improve the mechanical properties of CS latex film, cross-linking or ionic bonding provided by introducing modified agents at interfacial layer is considerable. Aguiar et al.142 investigated the mechanical performance of PSt/PBA CS latex film prepared by introducing itaconic acid into shell polymer. It was shown that mechanical properties of CS latex film, such as tensile strength and dynamic mechanical property, could be significantly improved by introducing itaconic acid units. It was attributed that the itaconic acid unit provided cross-linked structures at the interfacial layer, and promoted the coalescence of interfacial polymer phase.143
The minimum film-forming temperature (MFT) is a key to evaluate the property and utilization performance of latex polymer film.144 Cao et al.145 systematically studied the relationship between the MFT of polymer latexes and the morphology of latex particles. It was demonstrated that the MFTs of polymer emulsions were seriously affected by latex particle morphology. The MFT of polymer latex featuring hard core and soft shell is lower than the counterpart with same chemical composition. This is interpreted that film-forming property of CS latex particle is governed solely by soft polymer shells,146–149 and accordingly the film-forming process from CS latex is illustrated as Fig. 9. Many other properties of CS polymer latex film, such as optical properties, peel strength and tensile strength, were thoroughly investigated by Okubo et al.150 It was indicated that these physical properties were influenced by the variations in morphologies of various phases in the film caused by temperature effect. It was concluded that this “temperature-sensitive” property could be controlled by regulating the content and the Tg of core or shell polymers.
 | ||
| Fig. 9 Schematic diagram showing film-forming process from a CS particle latex (white as core polymer, black as shell polymer) in which the shells create a continuous matrix and the cores are embedded as discrete phases149 Copyright, 2009 Royal Society of Chemistry. | ||
5.2 Applications of PVAc-CS emulsion
The inherent shortcomings of PVAc-based emulsion in end-use applications could be improved by fabricating PVAc-CS structure through incorporating different constituents into either core or shell. Lu et al.151 synthesized cross-linkable PVAc-CS emulsion with poly(vinyl acetate-co-vinyl versatate) P(VAc-co-VeoVa) core and poly(glycidyl methacrylic) (PGMA) shell through seeded emulsion polymerisation of GMA in the presence of P(VAc-co-VeoVa) seed particles. PVAc-CS morphology was facilitated by using starved feeding of GMA monomer during second-stage polymerisation, which is clearly shown in Fig. 10. The cross-linkable and curing characteristics of resultant PVAc-CS emulsion were investigated using real-time FTIR spectroscopy. It was found that the cure rate was slow at ambient temperature, which was attributed to the restricted diffusion of crosslinking agent to the crosslinking sites at lower temperature. It was further indicated that the cure rate could be accelerated by elevating temperature, resulting in complete curing about 4 h at 30 °C and 1.5 h at 40 °C, respectively. Consequently, it was demonstrated that the cross-linkable PVAc-CS emulsion could retain better film-forming property of PVAc-based emulsion, leaving PGMA shells to provide crosslinking sites for post-crosslinking, and then the water-, heat- and creep-resistance of PVAc emulsion could be improved. The cross-linkable PVAc-CS emulsion might be utilized as single-component adhesive to substitute PVAc/isocyanate bi-component cross-linkable wood adhesives because of its similar performance and convenient implementing conditions. | ||
| Fig. 10 Effect of feed rate of GMA monomer on the morphology of particles (a) add all GMA once; (b) 40 g h−1; (c) 20 g h−1; (d) 10 g h−1 (ref. 151) Copyright, 2012 John Wiley & Sons, Inc. | ||
Pusch et al.152 reported a novel approach to synthesize three layers transparent PVAc-CS structural emulsion containing poly(divinyl benzene)/polystyrene (PDVB/PSt) cores and PVAc shells via two-stage emulsion polymerisation. In the experiment, a layer of PSt shell could be polymerised onto the surface of PDVB seed particles before seeded polymerisation of VAc monomers. It was attributed that a mass of vinyl groups on the surface of the PDVB seed particles could inhibit the conversion of VAc monomers. Thus the PSt shell layer was used to cover the vinyl groups for increasing conversion. It was demonstrated that this was an effective way to fabricate the three layers transparent CS emulsion. In this system, it was indicated that a densely cross-linked PDVB core supplied the material with stability and a hydrophilic PVAc shell was considered to be biocompatible. Therefore, it might be interested in the field of biomaterials because it allowed a possible use inside the human body. Meanwhile, it could also be considered as bio-optical devices due to its transparency and biocompatibility.
PVAc-CS products could be further endowed novel properties by incorporating functional components. Ying et al.153 synthesized hydrogel microspheres with stimuli-responsive recognition by fabricating PVAc-graft-calcium alginate CS structural particles with bovine serum albumin as molecular template. The rebinding and swelling property of PVAc-CS hydrogel microspheres exhibited responsiveness toward ionic strength, temperature, and pH. Furthermore, the separation factor of PVAc-CS microspheres has been increased because of the hydrophobic effect and covalent cross-linking resulting from PVAc segments.
6 Summary
Composite polymer emulsion with well-defined morphology, ingenious design and unique performance will become the leading edge and hotspot in composite emulsion research from both academic and industrial interests. In this review, the progress on PVAc-based emulsion with core/shell morphology from synthesis, morphological control, properties and applications over the past decades has been summarised.First, an overview of formation mechanism of PVAc-CS latex particle was represented. The formation of PVAc-CS latex particle is mainly controlled by phase separation process during the polymerisation and storage process. Seeded or multi-stage emulsion polymerisation is used for generating phase separation. Obviously, phase separation is influenced by the compatibility of different polymer phases, the grafting reactions between growing chains and preformed polymer, the mobility of polymeric components, and the thermal history of the emulsion. Among these factors, the incompatibility resulted from hydrophilic difference and grafting structures between core and shell polymers are the key for formation and stabilization of PVAc-CS structured latex particle.
The morphological control of PVAc-CS structured latex particle has been greatly developed from an interdisciplinary angle. The final morphology and morphological evolution of PVAc-CS latex particles are controlled by CSMT and CSMK. From CSMT, the final equilibrium morphology of PVAc-CS latex particles is determined by CSMT regardless of initial structures. It is concluded that the thermodynamically preferred morphology is the one with minimum interfacial free energy change. In order to predict the equilibrium morphology rapidly and accurately, simplified predicted approaches, including thermodynamic models, the diagrams of various equilibrium morphologies, etc., are proposed for PVAc-CS emulsion systems. Compared with CSMT, CSMK determines the morphological evolution of PVAc-CS latex particles throughout polymerisation, especially for the morphology in thermodynamic non-equilibrium emulsion systems. For CSMK, free radical distribution and polymer chain migration are main factors to control morphological evolution of PVAc-CS latex particles. Free radical distribution of PVAc-CS emulsion systems is affected by anchoring effect of initiator hydrophilic end-groups and diffusion of free radicals within particles. Polymer chain migration of PVAc-CS emulsion is commonly influenced by the effect of viscosity within latex particles and Tg of different polymers.
The combination of CSMT and CSMK is fascinating and strongly demanded because it could optimize shortages of both theories for PVAc-CS emulsion systems. It is believed that structural models can provide scientific and effective approaches to realize the combination of CSMT and CSMK. Based upon structural models, the simulant results of PVAc-CS latex can give a better understanding of morphological evolution of PVAc-CS latex particle, reveal the formation mechanism of PVAc-CS emulsion, and provide crucial factors for preparation of PVAc-CS emulsion. During the polymerisation, the formation of PVAc-CS latex particle is synergistically affected by adjustable factors, such as polymerisation conditions, the type and nature of monomer and initiator, grafting structures, etc.
The properties and applications of PVAc-CS emulsion have been summarized briefly. The properties of PVAc-CS emulsion are mainly studied in the term of latex film. The performance of PVAc-CS latex film is significantly increased by a buffer zone resulted from interfacial transition layer, which can help to scatter or transmit stress. PVAc-CS emulsion is applied to improve the inherent drawbacks of PVAc-based products, such as water-, heat- and creep-resistance, etc. The potential applications endowed by composite effect of CS structure are exhibited by novel performances from interdisciplinary perspective, such as the application for bio-optical devices.
Further research emphasis should be put on quantitative relationship between morphological controlling mechanisms of PVAc-CS emulsion and influencing factors. More attentions should be paid for potential applications in high-performance adhesives, environmental friendly coatings and multi-functional modifiers, because PVAc-CS emulsion can meet the utilization performances while reducing the industrial cost. The subject of academic and industrial importance deserves more fundamental research effort.
Abbreviations
| AN | Acrylonitrile |
| AIBN | 2,2-Azobis(isobutylronitrile) |
| BA | Butyl acrylate |
| CS | Core/shell |
| DVB | Divinyl benzene |
| GMA | Glycidyl methacrylate |
| HS | Hemisphere |
| ICS | Inverted core/shell |
| IP | Individual particles |
| KPS | Potassium persulfate |
| n-MMT | Nanometer montmorillonite |
| MA | Methyl acrylate |
| MAA | Methacrylic acid |
| MFT | Minimum film-forming temperature |
| MMA | Methyl methacrylate |
| NaSS | Sodium styrene sulphonate |
| PBA | Poly(butyl acrylate) |
| PDVB | Poly(divinyl benzene) |
| PEA | Poly(ethyl acrylate) |
| PGMA | Poly(glycidyl methacrylate) |
| PMA | Poly(methyl acrylate) |
| PMMA | Poly(methyl methacrylate) |
| POSS | Polyhedral oligomeric silsesquioxane |
| PSt | Polystyrene |
| PVAc | Poly(vinyl acetate) |
| PVAc-CS | PVAc-based core/shell |
| St | Styrene |
| Tg | Glass transition temperature |
| VAc | Vinyl acetate |
| VeoVa | Vinyl versatate |
Acknowledgements
Financial support was given by the Fundamental Research Funds for the Central Universities (DL12EB02-01), the National Natural Science Foundation of China (31000270 and 31270609) and Youths Science Foundation of Heilongjiang Province of China (QC2011C010).References
- V. A. M. Herk, Chemistry and Technology of Emulsion Polymerisation, John Wiley and Sons, Inc., New York, 2005, pp. 79–110 Search PubMed
.
- P. A. Lovell and M. S. El-Aasser, Emulsion Polymerisation and Emulsion Polymers, John Wiley and Sons, Inc., New York, 1997, pp. 293–323 Search PubMed
.
- C. H. Choi, D. A. Weitz and C. S. Lee, Adv. Mater., 2013, 25, 2536–2541 CrossRef CAS PubMed
.
- R. A. Ramli, W. A. Laftah and S. Hashim, RSC Adv., 2013, 3, 15543–15565 RSC
.
- Y. D. Jeong, D. Patra, A. Sanyal and V. M. Rotello, Curr. Org. Chem., 2013, 17, 49–57 CrossRef CAS
.
- Q. Wang, S. K. Fu and T. Y. Yu, Prog. Polym. Sci., 1994, 19, 703–753 CrossRef CAS
.
- M. Ratanajanchai, D. H. Lee, P. Sunintaboon and S. G. Yang, J. Colloid Interface Sci., 2014, 415, 70–76 CrossRef CAS PubMed
.
- A. R. Mahdavian, M. Ashjari and H. S. Mobarakeh, J. Appl. Polym. Sci., 2008, 110, 1242–1249 CrossRef CAS
.
- M. H. Kin, W. Y. Li, H. W. Chun and P. Li, Colloid Polym. Sci., 2010, 288, 1503–1523 Search PubMed
.
- R. G. Chaudhuri and S. Paria, Chem. Rev., 2012, 112, 2373–2433 CrossRef PubMed
.
- Y. S. Lu, Y. Xia and R. C. Larock, Prog. Org. Coat., 2011, 71, 336–342 CrossRef CAS PubMed
.
- F. Caruso, A. S. Susha, M. Giersig and H. Möhwald, Adv. Mater., 1999, 11, 950–953 CrossRef CAS
.
- W. J. Zhang, L. Y. Mu, Y. Wang, S. H. Lin and Q. M. Pan, J. Appl. Polym. Sci., 2014 DOI:10.1002/app.39991
.
- S. Park, Y. Lee, Y. Kim, H. M. Lee, J. H. Kim, I. W. Cheong and W. G. Koh, Colloids and Surfaces A: Physicochem. Eng. Aspects, 2014, 450, 46–51 CrossRef CAS PubMed
.
- J. H. Chang, L. Q. Wang, Y. Shin, B. Jeong, J. C. Birnbaum and G. J. Exarhos, Adv. Mater., 2002, 14, 378–382 CrossRef CAS
.
- W. J. Ye, M. F. Leung, J. Xin, T. L. Kwong, D. K. L. Lee and P. Li, Polymer, 2005, 46, 10538–10543 CrossRef CAS PubMed
.
- S. H. Fu, A. L. Tian, C. S. Du and C. X. Wang, J. Appl. Polym. Sci., 2013, 127, 2678–2683 CrossRef CAS
.
- A. K. Khana, B. C. Raya and S. K. Dolui, Prog. Org. Coat., 2008, 62, 65–70 CrossRef PubMed
.
- S. F. Zhang, R. M. Wang, Y. F. He, P. F. Song and Z. M. Wu, Prog. Org. Coat., 2013, 7, 6729–6735 Search PubMed
.
- J. M. Chan, L. F. Zhang, K. P. Yuet, G. Liao, J. W. Rheed, R. Langerc and O. C. Farokhzad, Biomaterials, 2009, 30, 1627–1634 CrossRef CAS PubMed
.
- C. Hazra, D. Kundu, A. Chatterjee, A. Chaudhari and S. Mishra, Colloids and Surfaces A: Physicochem. Eng. Aspects, 2014, 449, 96–113 CrossRef CAS PubMed
.
- F. M. Huijsa, F. F. Vercauteren, B. de Ruiter, D. Kalicharan and G. Hadziioannou, Synth. Met., 1999, 102, 1151–1152 CrossRef
.
- M. A. Khan and S. P. Armes, Adv. Mater., 2000, 12, 671–674 CrossRef CAS
.
- J. M. Lee, D. G. Lee, S. J. Lee and J. H. Kim, Macromolecules, 2009, 42, 4511–4519 CrossRef CAS
.
- G. D. Fu, G. L. Li, K. G. Neoh and E. T. Kang, Prog. Polym. Sci., 2011, 36, 127–167 CrossRef CAS PubMed
.
- C. D. Anderson and E. S. Daniels, Emulsion Polymerisation and Applications of Latex (Rapra Review Reports), Rapra Technology Limited, United Kingdom, 2003, pp. 25–29 Search PubMed
.
- F. Chiozza, F. Toniolo and B. Pizzo, J. Appl. Polym. Sci., 2013, 129, 1157–1169 CrossRef CAS
.
- J. Lu, A. J. Easteal and N. R. Edmonds, Pigm. Resin Technol., 2011, 40, 161–168 CrossRef CAS
.
- A. Kaboorani and B. Riedl, Int. J. Adhes. Adhes., 2011, 31, 605–611 CrossRef CAS PubMed
.
- J. Z. Ma, Y. H. Liu, Y. Bao, J. L. Liu and J. Zhang, Adv. Colloid Interface Sci., 2013, 197–198, 118–131 CrossRef CAS PubMed
.
- A. R. Unnithan, N. A. M. Barakat, M. F. Abadir, A. Yousef and H. Y. Kim, J. Mol. Catal. A: Chem., 2012, 363–364, 186–194 CrossRef CAS PubMed
.
- L. Quaroni and G. Chumanov, J. Am. Chem. Soc., 1999, 121, 10642–10643 CrossRef CAS
.
- S. L. Wang, K. Yue, L. Y. Liu and W. T. Yang, J. Colloid Interface Sci., 2013, 389, 126–133 CrossRef CAS PubMed
.
- L. W. Zhang, S. H. Piao and H. J. Choi, J. Colloid Interface Sci., 2013, 402, 100–106 CrossRef PubMed
.
- C. L. Lin, W. Y. Chiu and C. F. Lee, Polymer, 2005, 46, 10092–10101 CrossRef CAS PubMed
.
- C. L. Lin, W. Y. Chiu and C. F. Lee, J. Colloid Interface Sci., 2005, 290, 397–405 CrossRef CAS PubMed
.
- F. Caruso, Adv. Mater, 2001, 13, 11–22 CrossRef CAS
.
- J. W. Ha, I. J. Park, S. B. Lee and D. K. Kim, Macromolecules, 2002, 35, 6811–6818 CrossRef CAS
.
- C. J. McDonald and M. J. Devon, Adv. Colloid Interface Sci., 2002, 99, 181–213 CrossRef CAS
.
- D. C. Sundberg, A. P. Casassa, J. Pantazopoulos, M. R. Muscato, B. Kronberg and J. Berg, J. Appl. Polym. Sci., 1990, 41, 1425–1442 CrossRef CAS
.
- Y. C. Chen, V. Dimonie and M. S. El-Aasser, J. Appl. Polym. Sci., 1991, 42, 1049–1063 CrossRef CAS
.
- J. Berg, D. C. Sundberg and B. Kronberg, Polym. Mater. Sci. Eng., 1986, 54, 367–369 CAS
.
- Y. C. Chen, V. Dimonie and M. S. El-Aasser, Macromolecules, 1991, 24, 3779–3787 CrossRef CAS
.
- E. J. Sundberg and D. C. Sundberg, J. Appl. Polym. Sci., 1993, 47, 1277–1294 CrossRef CAS
.
- Y. G. Durant and D. C. Sundberg, J. Appl. Polym. Sci., 1995, 58, 1607–1618 CrossRef CAS
.
- M. Okubo, Y. Katsuta, A. Yamada and T. Matsumoto, Kobunshi Ronbunshu, 1979, 36, 459–464 CrossRef CAS
.
- M. Okubo, Y. Katsuta and T. Matsumoto, J. Polym. Sci., Polym. Lett. Ed., 1982, 20, 45–51 CrossRef CAS
.
- M. Okubo, M. Ando, A. Yamada, Y. Katsuta and T. Matsumoto, J. Polym. Sci., Polym. Lett. Ed., 1981, 19, 143–147 CrossRef CAS
.
- M. Okubo, A. Yamada and T. Matsumoto, J. Polym. Sci., Polym. Lett. Ed., 1980, 16, 3219–3228 CrossRef
.
- K. O'Couner and S. L. Tsavr, J. Appl. Polym. Sci., 1987, 33, 2007–2027 CrossRef
.
- K. F. Ni, G. R. Shan, Z. X. Weng, N. Sheibat-Othman, G. Fevotte, F. Lefebvre and E. Bourgeat-Lami, Macromolecules, 2005, 38, 7321–7329 CrossRef CAS
.
- N. V. Dziomkina, M. A. Hempenius and G. Vancso, Eur. Polym. J., 2006, 42, 81–91 CrossRef CAS PubMed
.
- L. J. Borthakur, T. Jana and S. K. Dolui, J. Coat. Technol. Res., 2010, 7, 765–772 CrossRef CAS
.
- T. Wang, S. Shi, F. Yang, L. M. Zhou and S. Kuroda, J. Mater. Sci., 2010, 45, 3392–3395 CrossRef CAS PubMed
.
- Y. Lu and M. Ballauff, Prog. Polym. Sci., 2011, 36, 767–792 CrossRef CAS PubMed
.
- X. F. Li, J. F. Zhang, X. P. Zhou, C. Lu, X. T. Tong, G. X. He and X. Q. Wang, J. Appl. Polym. Sci., 2012, 126, 110–115 CrossRef CAS
.
- D. L. Chicoma, V. Carranza and R. Giudici, Macromol. Symp., 2013, 324, 124–133 CrossRef CAS
.
- Y. C. Mu, T. Qiu, X. C. Li, Y. D. Guan, S. W. Zhang and X. Y. Li, Langmuir, 2011, 27, 4968–4978 CrossRef CAS PubMed
.
- D. I. Lee and T. Ishikawa, J. Polym. Sci., Polym. Chem. Ed., 1983, 21, 147–154 CrossRef CAS
.
- V. I. Eliseeva, Prog. Org. Coat., 1985, 13, 195–221 CrossRef CAS
.
- X. Y. Yuan, D. X. Huo and Q. W. Qian, J. Colloid Interface Sci., 2010, 346, 72–78 CrossRef CAS PubMed
.
- M. Okubo, Y. Katsuta and T. Matsumoto, J. Polym. Sci., Polym. Chem. Ed., 1980, 18, 481–486 CrossRef CAS
.
- V. Dimonie, M. S. EL-aasser, A. Klein and J. W. Vanderhoff, J. Polym. Sci., Polym. Chem. Ed., 1984, 22, 2197–2215 CrossRef CAS
.
- D. R. Stutman, A. Klein, M. S. El-Aasser and J. W. Vanderhoff, Ind. Eng. Chem. Prod. Res. Dev., 1985, 24, 404–412 CrossRef CAS
.
- T. Matsumoto, M. Okubo and Y. Katsuta, Jpn. J. Polym. Sci. Technol., 1976, 33, 575–583 CAS
.
- W. D. Hergeth, H. J. Bittrich, F. Eichhornt, S. Schlenker, K. Schmutzler and U. J. Steinau, Polymer, 1989, 60, 1913–1917 CrossRef
.
- W. D. Hergeth and K. Schmutzler, Acta Polym., 1985, 36, 472–474 CrossRef CAS
.
- L. Zhang, E. S. Daniels, V. L. Dimonie and A. Klein, J. Appl. Polym. Sci., 2014 DOI:10.1002/app.40124
.
- J. M. Stubbs and D. C. Sundberg, J. Coat. Technol. Res., 2008, 5, 169–180 CrossRef CAS
.
- E. P. Pedraza and M. D. Soucek, Polymer, 2005, 46, 11174–11185 CrossRef CAS PubMed
.
- C. S. Chern, Prog. Polym. Sci., 2006, 31, 443–486 CrossRef CAS PubMed
.
- D. C. Sundberg and Y. G. Durant, Polym. React. Eng., 2003, 11, 379–432 CrossRef CAS PubMed
.
- K. Zhao, P. Q. Sun, D. Z. Liu and L. J. Wang, J. Appl. Polym. Sci., 2004, 92, 3144–3152 CrossRef CAS
.
- P. Q. Sun, K. Zhao, D. Z. Liu and D. X. Huo, J. Appl. Polym. Sci., 2002, 83, 2930–2937 CrossRef CAS
.
- K. Zhao, P. Q. Sun, D. Z. Liu and G. Dai, Eur. Polym. J., 2004, 40, 89–96 CrossRef CAS PubMed
.
- L. J. Gonzalez-Ortizt and J. M. Asua, Macromolecules, 1995, 28, 3135–3145 CrossRef
.
- S. C. Thickett and R. G. Gilbert, Polymer, 2007, 48, 6965–6991 CrossRef CAS PubMed
.
- J. Delgado, M. S. El-aasser and J. W. Vanderhoff, J. Polym. Sci., Part A: Polym. Chem., 1986, 24, 861–874 CrossRef CAS
.
- J. C. Delacal, R. Urzay, A. Zamora, J. Forcada and J. M. Asua, J. Polym. Sci., Part A: Polym. Chem., 1990, 28, 1011–1031 CrossRef CAS
.
- J. E. Jonsson, H. Hassander, L. H. Jansson and B. Tornell, Macromolecules, 1991, 24, 126–131 CrossRef
.
- J. Stubbs, O. Karlsson, J. E. Jonsson, E. Sundberg, Y. Durant and D. Sundberg, Colloids Surf., A, 1999, 153, 255–270 CrossRef CAS
.
- S. C. Misra, C. Pichot, M. S. El-aasser and J. W. Vanderhoff, J. Polym. Sci., Polym. Chem. Ed., 1983, 21, 2383–2396 CrossRef CAS
.
- T. I. Min, A. Klein, M. S. EL-aasser and J. W. Vanderhoff, J. Polym. Sci., Polym. Chem. Ed., 1983, 21, 2845–2861 CrossRef CAS
.
- C. J. Ferguson, G. T. Russell and R. G. Gilbert, Polymer, 2003, 44, 2607–2619 CrossRef CAS
.
- E. S. Daniels, V. L. Dimonie, M. S. El-Aasser and J. W. Vanderhoff, J. Appl. Polym. Sci., 1990, 41, 246324–246377 CrossRef
.
- Y. C. Chen, V. L. Dimonie, O. L. Shaffer and M. S. El-Aasser, Polym. Int., 1993, 30, 185–194 CrossRef CAS
.
- C. L. Winzor and D. C. Sundberg, Polymer, 1992, 33, 3797–3810 CrossRef CAS
.
- O. J. Karlsson, H. Hassander and D. Colombini, C. R. Chim., 2003, 6, 1233–1244 CrossRef CAS PubMed
.
- L. E. Karlsson, O. J. Karlsson and D. C. Sundberg, J. Appl. Polym. Sci., 2003, 90, 905–915 CrossRef CAS
.
- C. A. Croxton, M. F. Mills, R. G. Gilbert and D. H. Napper, Macromolecules, 1993, 26, 3563–3571 CrossRef CAS
.
- M. R. Muscato and D. C. Sundberg, J. Polym. Sci., Part B: Polym. Phys., 1991, 29, 1021–1024 CrossRef CAS
.
- O. J. Karlsson, J. M. Stubbs, R. H. Carrier and D. C. Sundberg, Polym. React. Eng., 2003, 11, 589–625 CrossRef CAS PubMed
.
- M. R. Grancio and D. J. Williams, J. Polym. Sci., Part A-1: Polym. Chem., 1970, 8, 2617–2629 CrossRef CAS
.
- M. R. Grancio and D. J. Williams, J. Polym. Sci., Part A-1: Polym. Chem., 1970, 8, 2733–2745 CrossRef CAS
.
- D. H. Napper, J. Polym. Sci., Part A-1: Polym. Chem., 1972, 9, 2089–2091 CrossRef
.
- S. A. Chen and H. S. Chang, J. Polym. Sci., Part A: Polym. Chem., 1985, 23, 2615–2630 CrossRef CAS
.
- M. F. Mills, R. G. Gilbert and D. H. Napper, Macromolecules, 1990, 23, 4247–4257 CrossRef CAS
.
- C. S. Chern and G. W. Poehlein, J. Polym. Sci., Part A: Polym. Chem., 1987, 25, 617–635 CrossRef CAS
.
- C. S. Chern and G. W. Poehlein, J. Polym. Sci., Part A: Polym. Chem., 1990, 28, 3055–3071 CrossRef CAS
.
- L. J. Gonzalez-Ortiz and J. M. Asua, Macromolecules, 1996, 29, 383–389 CrossRef CAS
.
- L. J. Gonzalez-Ortiz and J. M. Asua, Macromolecules, 1996, 29, 4520–4527 CrossRef CAS
.
- C. J. Ferguson, G. T. Russell and R. G. Gilbert, Polymer, 2002, 43, 4557–4570 CrossRef CAS
.
- J. E. Jonsson, O. J. Karlsson, H. Hassander and B. Tornell, Eur. Polym. J., 2007, 43, 1322–1332 CrossRef PubMed
.
- C. J. Ferguson, G. T. Russell and R. G. Gilbert, Polymer, 2002, 43, 6371–6382 CrossRef CAS
.
- I. A. Maxwell, B. R. Morrison, D. H. Napper and R. G. Gilbert, Macromolecules, 1991, 24, 1629–1640 CrossRef CAS
.
- M. Duan, T. Qiu, C. Huang, G. Y. Xu, L. H. Guo and X. Y. Li, Prog. Org. Coat., 2013, 76, 216–223 CrossRef CAS PubMed
.
- S. L. Chai, M. M. Jin and H. M. Tan, Eur. Polym. J., 2008, 44, 3306–3313 CrossRef CAS PubMed
.
- R. L. Sherman Jr and W. T. Ford, Ind. Eng. Chem. Res., 2005, 44, 8538–8541 CrossRef
.
- L. M. Gugliotta, M. Arotcarena, J. R. Leiza and J. M. Asua, Polymer, 1995, 36, 2019–2023 CrossRef CAS
.
- M. S. Dehghan, N. S. Sanjani and N. Naderi, J. Appl. Polym. Sci., 2006, 100, 2409–2414 CrossRef
.
- N. M. Ahmad, F. Heatley and P. A. Lovell, Macromolecules, 1998, 31, 2822–2827 CrossRef CAS
.
- J. H. Kim, M. S. EL-Aasser, A. Klein and J. W. Vanderhoff, J. Appl. Polym. Sci., 1988, 35, 2117–2131 CrossRef CAS
.
- J. S. Wang, W. Deng, Y. S. Chen and C. Y. Kan, Chin. Chem. Lett., 2010, 21, 1259–1262 CrossRef CAS PubMed
.
- J. E. Jonsson, H. Hassander and B. Tdrnell, Macromolecules, 1994, 27, 1932–1937 CrossRef
.
- J. E. Jonsson, O. J. Karlsson, H. Hassander and B. Tdrnell, Macromolecules, 2001, 34, 1512–1514 CrossRef
.
- I. Cho and K. W. Lee, J. Appl. Polym. Sci., 1985, 30, 1903–1926 CrossRef CAS
.
- H. D. Bruyn and R. G. Gilbert, Polymer, 2001, 42, 7999–8005 CrossRef
.
- P. Q. Sun, Y. Li, D. Z. Liu, S. H. Sun and K. Zhao, Polym. Bull., 2005, 55, 323–332 CrossRef CAS
.
- D. Colombini, N. Ljungberg, H. Hassander and O. J. Karlsson, Polymer, 2005, 46, 1295–1308 CrossRef CAS PubMed
.
- J. Gao and A. Penlidis, Prog. Polym. Sci., 2002, 27, 403–535 CrossRef CAS
.
- F. R. Mayo, C. Walling, F. M. Lewis and W. F. Hulse, J. Am. Chem. Soc., 1948, 70, 1523–1525 CrossRef CAS
.
- X. B. Zhou, J. Zhu, M. Y. Xing, Z. B. Zhang, Z. P. Cheng, N. C. Zhou and X. L. Zhu, Eur. Polym. J., 2011, 47, 1912–1922 CrossRef CAS PubMed
.
- A. Debuigne, J. R. Caille, C. Detrembleur and R. Jerome, Angew. Chem., Int. Ed., 2005, 44, 3439–3442 CrossRef CAS PubMed
.
- D. Britton, F. Heatley and P. A. Lovell, Macromolecules, 1998, 31, 2828–2837 CrossRef CAS
.
- D. J. Hourston, H. X. Zhang, M. Song, M. Pollock and A. Hammiche, Thermochim. Acta, 1997, 294, 23–31 CrossRef CAS
.
- W. D. Hergeth, U. J. Steinau, H. J. Bittrich and H. Tanneberger, Colloid Polym. Sci., 1990, 268, 991–994 CAS
.
- H. Hoffmann, Adv. Colloid Interface Sci., 2012, 178, 21–33 CrossRef CAS PubMed
.
- M. L. Wang, Z. J. Ma, D. Zhu, D. Y. Zhang and W. Yin, J. Appl. Polym. Sci., 2013, 128, 4224–4230 CrossRef CAS
.
- X. Y. Wang, W. J. Liu, X. B. Wang, Z. C. Zhang and H. R. Liu, Mater. Lett., 2007, 61, 4478–4481 CrossRef CAS PubMed
.
- Y. C. Chen, V. Dimonie and M. S. EL-aasser, J. Appl. Polym. Sci., 1992, 45, 487–499 CrossRef CAS
.
- M. G. Dufour and A. Guyot, Colloid Polym. Sci., 2003, 281, 105–112 CAS
.
- Y. G. Durant and D. C. Sundberg, Macromolecules, 1996, 29, 8466–8472 CrossRef CAS
.
- Y. G. Durant, E. J. Sundberg and D. C. Sundberg, Macromolecules, 1997, 30, 1028–1032 CrossRef CAS
.
- C. Xu, T. Qiu, J. Q. Deng, Y. Meng, L. F. He and X. Y. Li, Prog. Org. Coat., 2012, 74, 233–239 CrossRef CAS PubMed
.
- Y. C. Mu, T. Qiu and X. Y. Li, Mater. Lett., 2009, 63, 1614–1617 CrossRef CAS PubMed
.
- W. J. Grigsby, C. J. Ferguson, R. A. Franich and G. T. Russell, Int. J. Adhes. Adhes., 2005, 25, 127–137 CrossRef CAS PubMed
.
- L. A. Pérez-Carrillo, M. Puca, M. Rabelero, K. E. Meza, J. E. Puig, E. Mendizábal, F. López-Serrano and R. G. López, Polymer, 2007, 48, 1212–1218 CrossRef PubMed
.
- C. Schellenberg, K. Tauer and M. Antonietti, Macromol. Symp., 2000, 151, 465–471 CrossRef CAS
.
- S. Bas and M. D. Soucek, React. Funct. Polym., 2013, 73, 291–302 CrossRef CAS PubMed
.
- F. D. D. Santos and L. Leibler, J. Polym. Sci., Part B: Polym. Phys., 2003, 41, 224–234 CrossRef
.
- J. Y. Cavaille, C. Jourdan, X. Z. Kong and J. Perez, Polymer, 1986, 27, 693–702 CrossRef CAS
.
- A. Aguiar, S. G. L. Villegas, M. Rabelero, E. Mendizábal and J. E. Puig, Macromolecules, 1999, 32, 6767–6771 CrossRef CAS
.
- D. Ik Lee, Polymer, 2005, 46, 1287–1293 CrossRef PubMed
.
- K. J. O'Callaghan, A. J. Paine and A. Rudin, J. Polym. Sci., Part A: Polym. Chem., 1995, 33, 1849–1857 CrossRef
.
- T. Y. Cao, Y. S. Xu, Y. C. Su, F. Long, X. Li and Y. H. Hu, J. Appl. Polym. Sci., 1990, 41, 1965–1972 CrossRef CAS
.
- D. Juhue and J. Lang, Macromolecules, 1995, 28, 1306–1308 CrossRef CAS
.
- O. Kalinina and E. Kumacheva, Macromolecules, 1999, 32, 4122–4129 CrossRef CAS
.
- A. B. Foster, P. A. Lovell and M. A. Rabjohns, Polymer, 2009, 50, 1654–1670 CrossRef CAS PubMed
.
- F. Deplace, M. A. Rabjohns, T. Yamaguchi, A. B. Foster, C. Carelli, C. H. Lei, K. Ouzine, J. L. Keddie, P. A. Lovell and C. Creton, Soft Matter, 2009, 5, 1440–1447 RSC
.
- M. Okubo, M. Seike and T. Matsumoto, J. Appl. Polym. Sci., 1983, 28, 383–390 CrossRef CAS
.
- J. Lu, A. J. Easteal and N. Edmond, J. Appl. Polym. Sci., 2012, 126, 172–181 CrossRef
.
- J. Pusch and A. M. van Herk, Macromolecules, 2005, 38, 6909–6914 CrossRef CAS
.
- X. G. Ying, L. L. Qi, X. Li, W. Y. Zhang and G. X. Cheng, J. Appl. Polym. Sci., 2013, 127, 3898–3909 CrossRef CAS
.
| This journal is © The Royal Society of Chemistry 2014 |






