Synthesis of monoglycosides, bisglycosides and open-chain ketoacetals from sugar derived exocyclic enol ethers†
Soumendra Nath Dasa,
Praveen Krishna Kb and
Sukhendu Bikas Mandal*a
aChemistry Division, CSIR-Indian Institute of Chemical Biology, 4, Raja S. C. Mullick Road, Jadavpur, Kolkata 700 032, India. E-mail: sbmandal@iicb.res.in
bNational Institute of Pharmaceutical Education and Research, 4, Raja S. C. Mullick Road, Jadavpur, Kolkata 700 032, India
First published on 23rd June 2014
Abstract
Under acidic conditions, the reaction of pent-4-enofuranosides with various nucleophiles of mostly alcohol origin furnished bisglycosylated products along with open-chain ketoacetals and monoglycosylated products depending upon the reagents and reaction conditions. The reactions appear to proceed via nucleophilic attack at C-1 and/or C-4 of the reaction intermediates.
Exocyclic vinyl ether derivatives of sugar1 are formed as intermediates in various biosynthetic pathways, for example during the enzymatic hydrolysis of S-adenosylhomocysteine to adenosine,2 enzymatic synthesis of 2-deoxy-scyllo-inosose from D-glucose-6-phosphate,3 formation of dehydroquinate in the shikimic pathway,4 the synthesis of nucleoside antibiotic angustmycin A,5 and the biomimetic rearrangement to carbocycles6 that are key precursors of nucleoside-based natural products neplanocin A and aristeromycin.7 Several synthetic reports for these vinyl ethers via dehydrohalogenation of 5-halogeno-furanose derivatives,8 Bamford–Stevens rearrangement of 5-tosylhydrazono-furanose derivatives9 and Wittig reaction of aldehydes with sugar-based triphenylphosphonium salts10 have been reported in the literature. Attempts to elaborate the synthetic utility of these ethers in producing glycosides,11 polyethers,12 spiroketals13 and related compounds14 including heterocycles,15 carbocyles,16 and nucleosides17 using coupling reactions,18 radical processes19 and cycloaddition reactions20 have generated molecules of diverse nature. Nevertheless, the chemistry of these systems is less developed as compared to endocyclic vinyl ethers that allow regio- and stereoselective transformation, glycosylation as well as C–C and C-heteroatom bond formations at the anomeric centre.21 However, the methods, which take advantage of the nucleophilic character of these ethers for the direct formation of C–C and C–O bonds during organic synthesis are limited to dimerization,22 and opening of epi-sulphonium ion23 and iodonium ion24 intermediates by nucleophiles. As functionalized tetrahydrofurans are integral components of many biologically significant compounds, the development of more general processes using exocyclic vinyl ethers could find broad application in the synthesis of bioactive molecules.
For a long time, we have been interested in the preparation of various glycoside derivatives via open-chain acetals used as chiral auxiliaries25 towards asymmetric synthesis of natural products or products useful in drug research.26 As a part of that programme, we initiated the present work taking a pent-4-enofuranoside derivative (exocyclic enol ether) as the precursor that could deliver the desired products via nucleophilic attack by alcohols at C-1 and/or C-4 under acidic conditions. The oxocarbenium ions formed at C-1 and C-4 are stabilized, providing scope of nucleophilic attack at these two positions as shown (Fig. 1). We herein report the results of the work.
To begin with, the key precursor pent-4-enofuranose derivative 1a,15a easily prepared from 3-O-benzyl-1,2-O-isopropylidene-5-O-mesyl-xylofuranose,27 was exposed to 5% conc. HCl in methanol for 15 min to afford the monoglycosylated product (MGP) 2a in 45% yield (Scheme 1). Though there was no appreciable change in product formation up to 1 h, continuation of the reaction for 6 h yielded the open-chain acetal (OCA) 3a (65%). Similar results were obtained when 1a was treated with 5% conc. HCl in 2-methoxyethanol furnishing the MGP 2b (42%) and the OCA 3b (65%). But treatment with 5% conc. HCl in benzyl thiol provided the MGP 2c (45%) in 15 min and the bisglycosylated product (BGP) 4a (47%) after 1 h. No OCA product however resulted from this reaction. Reaction of the same starting material with 5% conc. HCl in allyl alcohol, propargyl alcohol, isopropanol or 2-chloroethanol, surprisingly, did not provide any identifiable product in 15 min–1 h. But all of these reactions afforded the OCAs 3c–3f in 52–67% yields after 6 h. In contrast, addition of 5% conc. HCl in ethanol or n-butanol to 1a did not result in any appreciable change in 15 min. However, it afforded the BGPs 4b (53%) and 4c (52%) respectively after 1 h and produced their OCAs 3g (60%) and 3h (61%) after 6 h. Further, reaction of the same precursor with 5% conc. HCl in allyltrimethylsilane, pent-4-ene-1-ol, or n-propanol did not indicate any appreciable change after 15 min, but furnished the BGPs 4d–4f in 52–58% yields when the reaction time was 1 h. Similar reactions of 1b28 with 5% conc. HCl in ethanol, n-butanol, or n-propanol furnished BGPs 4g–4i as the major products in 46–55% yields after 1 h. The product 4i was isolated as its acetate derivative. However, these reactions produced intractable mixture of products after 5–6 h.
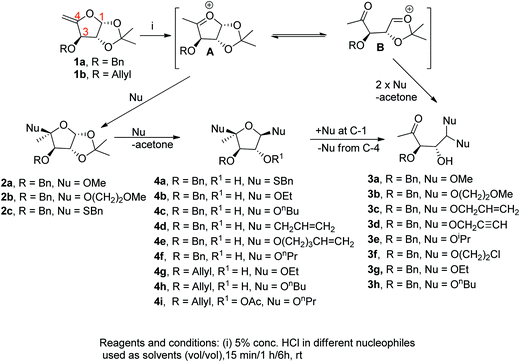 | ||
| Scheme 1 Conversion of pent-4-enofuranose to glycosides and open-chain ketoacetals via the proposed pathway. | ||
It is apparent from the results that the nucleophiles (also solvents) can solvate the oxocarbenium ion intermediates A and B, and lower their potential energy. The intermediate B can be more solvated, possibly due to smaller in size and difficult for it to escape from the solvents furnishing less reactivity to the nucleophiles and take longer time (6 h) for the reactions to occur in these solvents (Table 1). 2-Chloroethanol afforded lower yield of its OCA product in comparison to 2-methoxyethanol due to the lesser nucleophilicity of the latter (due to electron-withdrawing inductive effect of Cl). On the other hand, the relatively more hindered intermediate A appeared to be less solvated and thus attack can even take place by allyltrimethylsilane and pent-4-ene-1-ol having comparatively low nucleophilicity affording their BGP within 1 h of the reactions. The reactions in the solvents like ethanol, n-propanol and n-butanol produced their BGPs through this intermediate also. The MGPs formed in methanol, benzyl thiol and 2-methoxyethanol solvents after 15 min are expected to be going through this oxocarbenium ion intermediate.
| Entry | Substrate | Reagent: 5% conc. HCl (vol/vol) in | Products (yield %) after | ||
|---|---|---|---|---|---|
| 15 min | 1 h | 6 h | |||
| a All reactions carried out at rt.b Isolated as acetate derivative. | |||||
| 1 | 1a | CH3OH | 2a (45) | 3a (65) | |
| 2 | 1a | MeO(CH2)2OH | 2b (42) | 3b (65) | |
| 3 | 1a | PhCH2SH | 2c (45) | 4a (47) | |
| 4 | 1a | CH2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CHCH2OH CHCH2OH |
3c (63) | ||
| 5 | 1a | CH![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CCH2OH CCH2OH |
3d (67) | ||
| 6 | 1a | (CH3)2CHOH | 3e (64) | ||
| 7 | 1a | Cl(CH2)2OH | 3f (52) | ||
| 8 | 1a | CH3CH2OH | 4b (53) | 3g (60) | |
| 9 | 1a | CH3(CH2)3OH | 4c (52) | 3h (61) | |
| 10 | 1a | CH2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CHCH2SiMe3 CHCH2SiMe3 |
4d (58) | ||
| 11 | 1a | CH2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH(CH2)3OH CH(CH2)3OH |
4e (52) | ||
| 12 | 1a | CH3(CH2)2OH | 4f (52) | ||
| 13 | 1b | CH3CH2OH | 4g (50) | ||
| 14 | 1b | CH3(CH2)3OH | 4h (55) | ||
| 15 | 1b | CH3(CH2)2OH | 4i (46)b | ||
The structures of all the products were confirmed by 1H and 13C NMR and MS analyses. Anomeric purity of the bisglycosides was provided by their 1H NMR spectra. The resonance corresponding to the anomeric proton appeared at δ ∼ 5.0 as a doublet (J = ∼5.0 Hz) indicating the presence of only the β-anomer.29 The absence of any detectable amount of the α-anomer was not surprising due to the acid catalyzed attack by nucleophiles at the anomeric carbon from β-face removing the isopropylidene moiety. The relative syn-disposition of the incoming nucleophiles at C-1 and C-4 of BGPs was deduced from the NOESY correlation between one of the methylene protons (2 × multiplets at δ 3.86–3.90 and δ 3.65–3.68) of each ethyl group at C-1 and C-4 of 4g. The methylene carbons (δ 56.8 and 64.2) of the ethyl groups have been identified by HSQC spectrum. The carbon signal at δ 56.8 showed HMBC correlation with the signal at δ 1.40 for the methyl protons at C-4 indicating their propinquity.
Regarding the mechanism of product formation in the reaction, initial protonation of the sugar-derived exocyclic vinyl ether 1a/1b must have furnished the oxocarbenium intermediates. Attack by nucleophiles at C-4 of the intermediate produced the C-4 MGPs. Apparently, because of the steric hindrance induced by the acetonide group of the intermediate the addition occurred across the double bond from the opposite face.15a Further, under acidic condition the cleavage of the isopropylidene group of the MGPs leading to an oxocarbenium ion and concomitant attack by a variety of nucleophiles at C-1 furnished the BGPs. The other intermediate, upon nucleophilic addition, afforded the OCAs. The OCAs, however, appeared to be generated also from the corresponding BGPs during the progress of the reaction.
In conclusion, sugar-derived exocyclic vinyl ether derivatives are important biosynthetic intermediates in various metabolic pathways in biological processes and could also be regarded as synthetic intermediates for the synthesis of important bioactive molecules. We have demonstrated that the 4-methylidenefuranoside derivative, the key precursor of the reaction, can be utilized to synthesize mono- and bis-glycosides of diverse structures through nucleophilic attack at C-1 and C-4 under acidic condition. Many of these reactions produced open-chain γ-keto acetals as the sole products when the solvents were miscible with conc. HCl. These acetals could be used as chiral auxiliaries in asymmetric synthesis of appropriate organic molecules. On the other hand, immiscible and partially miscible solvents furnished bisglycosides as the major products.
Experimental
General experimental
Melting points were taken in open capillaries and are uncorrected. 1H and 13C NMR spectra were recorded in CDCl3 as solvent using TMS as internal standard. Mass spectra were recorded using ESI mode. Specific rotations were measured at 589 nm. Pre-coated plates (0.25 mm, silica gel 60 F254) were used for TLC. All the solvents were distilled and purified as necessary.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19) was added to 1a (150 mg, 0.57 mmol) and stirred at room temperature for 15 min. The reaction mixture was neutralized by slow addition of a saturated NaHCO3 solution, the solvent was evaporated and the product was extracted by DCM (3 × 10 mL). The solvent was washed with water (2 × 5 mL), dried (Na2SO4) and concentrated to an oil, which was purified on a silica gel (230–400 mesh) column using petroleum ether–EtOAc (23
19) was added to 1a (150 mg, 0.57 mmol) and stirred at room temperature for 15 min. The reaction mixture was neutralized by slow addition of a saturated NaHCO3 solution, the solvent was evaporated and the product was extracted by DCM (3 × 10 mL). The solvent was washed with water (2 × 5 mL), dried (Na2SO4) and concentrated to an oil, which was purified on a silica gel (230–400 mesh) column using petroleum ether–EtOAc (23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) as eluent to furnish 2a (75 mg, 45%) as colourless liquid. [α]25D + 195.6 (c 0.5 in CHCl3); 1H NMR (600 MHz, CDCl3): δ 1.41 (s, 3H), 1.42 (s, 3H), 1.46 (s, 3H), 3.32 (s, 3H), 3.78 (d, 1H, J = 3.0 Hz), 4.61 (d, 1H, J = 12.0 Hz), 4.78 (t, 1H, J = 3.6 Hz), 4.84 (d, 1H, J = 12.0 Hz), 5.75 (d, 1H, J = 4.8 Hz), 7.29–7.40 (m, 5H); 13C NMR (150 MHz, CDCl3): δ 19.9 (CH3), 27.8 (CH3), 28.0 (CH3), 48.6 (CH3), 72.2 (CH2), 86.2 (CH), 88.2 (CH), 103.6 (CH), 105.8 (C), 114.5 (C), 127.9–128.4 (5 × CH), 137.2 (C); ESIMS, m/z: 317 (M + Na)+.
2) as eluent to furnish 2a (75 mg, 45%) as colourless liquid. [α]25D + 195.6 (c 0.5 in CHCl3); 1H NMR (600 MHz, CDCl3): δ 1.41 (s, 3H), 1.42 (s, 3H), 1.46 (s, 3H), 3.32 (s, 3H), 3.78 (d, 1H, J = 3.0 Hz), 4.61 (d, 1H, J = 12.0 Hz), 4.78 (t, 1H, J = 3.6 Hz), 4.84 (d, 1H, J = 12.0 Hz), 5.75 (d, 1H, J = 4.8 Hz), 7.29–7.40 (m, 5H); 13C NMR (150 MHz, CDCl3): δ 19.9 (CH3), 27.8 (CH3), 28.0 (CH3), 48.6 (CH3), 72.2 (CH2), 86.2 (CH), 88.2 (CH), 103.6 (CH), 105.8 (C), 114.5 (C), 127.9–128.4 (5 × CH), 137.2 (C); ESIMS, m/z: 317 (M + Na)+.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19) solution (10 mL) at room temperature for 15 min. Work up and purification on a silica gel (230–400 mesh) column using a mixture of petroleum ether–EtOAc (23
19) solution (10 mL) at room temperature for 15 min. Work up and purification on a silica gel (230–400 mesh) column using a mixture of petroleum ether–EtOAc (23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) as eluent furnished 2b (80 mg, 42%) as colourless liquid. [α]25D − 175.9 (c 0.34 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 1.30 (s, 3H), 1.49 (s, 3H), 1.54 (s, 3H), 3.36 (s, 3H), 3.48–3.61 (m, 2H), 3.67–3.80 (m, 2H), 3.97 (s, 1H), 4.56 (d, 1H, J = 11.7 Hz), 4.64 (d, 1H, J = 4.8 Hz), 4.70 (d, 1H, J = 11.7 Hz), 5.94 (d, 1H, J = 4.8 Hz), 7.27–7.38 (m, 5H); 13C NMR (75 MHz, CDCl3): δ 18.3 (CH3), 26.1 (CH3), 26.4 (CH3), 58.9 (CH3), 60.8 (CH2), 71.8 (CH2), 72.1 (CH2), 83.9 (CH), 87.5 (CH), 106.0 (CH), 112.2 (C), 112.5 (C), 127.5 (2 × CH), 127.8 (CH), 128.4 (2 × CH), 137.5 (C); HRMS (ESI, positive ion) calcd for C18H26NaO6, m/z 361.1627, found 361.1642.
2) as eluent furnished 2b (80 mg, 42%) as colourless liquid. [α]25D − 175.9 (c 0.34 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 1.30 (s, 3H), 1.49 (s, 3H), 1.54 (s, 3H), 3.36 (s, 3H), 3.48–3.61 (m, 2H), 3.67–3.80 (m, 2H), 3.97 (s, 1H), 4.56 (d, 1H, J = 11.7 Hz), 4.64 (d, 1H, J = 4.8 Hz), 4.70 (d, 1H, J = 11.7 Hz), 5.94 (d, 1H, J = 4.8 Hz), 7.27–7.38 (m, 5H); 13C NMR (75 MHz, CDCl3): δ 18.3 (CH3), 26.1 (CH3), 26.4 (CH3), 58.9 (CH3), 60.8 (CH2), 71.8 (CH2), 72.1 (CH2), 83.9 (CH), 87.5 (CH), 106.0 (CH), 112.2 (C), 112.5 (C), 127.5 (2 × CH), 127.8 (CH), 128.4 (2 × CH), 137.5 (C); HRMS (ESI, positive ion) calcd for C18H26NaO6, m/z 361.1627, found 361.1642.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19, 10 mL) was added to 1a (150 mg, 0.57 mmol) and the mixture was stirred at room temperature for 15 min. Usual work up and purification on a silica gel (230–400 mesh) column using a mixture of petroleum ether–EtOAc (93
19, 10 mL) was added to 1a (150 mg, 0.57 mmol) and the mixture was stirred at room temperature for 15 min. Usual work up and purification on a silica gel (230–400 mesh) column using a mixture of petroleum ether–EtOAc (93![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7) as eluent furnished 2c (100 mg, 45%) as yellowish liquid. [α]25D + 195.6 (c 0.5 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 1.43 (s, 3H), 1.52 (s, 3H), 1.74 (s, 3H), 3.84 (brs, 2H), 3.93 (d, 1H, J = 3.0 Hz), 4.66 (d, 1H, J = 12.0 Hz), 4.75–4.79 (m, 2H), 5.86 (d, 1H, J = 4.5 Hz), 7.20–7.39 (m, 10H); 13C NMR (75 MHz, CDCl3): δ 27.7 (CH3), 27.9 (2 × CH3), 31.4 (CH2), 72.4 (CH2), 86.8 (CH), 90.9 (CH), 93.1 (C), 104.2 (CH), 114.8 (C), 126.8–129.3 (10 × CH), 137.1 (C), 138.5 (C); HRMS (ESI, positive ion) calcd for C22H26NaO4S, m/z 409.1449, found 409.1465.
7) as eluent furnished 2c (100 mg, 45%) as yellowish liquid. [α]25D + 195.6 (c 0.5 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 1.43 (s, 3H), 1.52 (s, 3H), 1.74 (s, 3H), 3.84 (brs, 2H), 3.93 (d, 1H, J = 3.0 Hz), 4.66 (d, 1H, J = 12.0 Hz), 4.75–4.79 (m, 2H), 5.86 (d, 1H, J = 4.5 Hz), 7.20–7.39 (m, 10H); 13C NMR (75 MHz, CDCl3): δ 27.7 (CH3), 27.9 (2 × CH3), 31.4 (CH2), 72.4 (CH2), 86.8 (CH), 90.9 (CH), 93.1 (C), 104.2 (CH), 114.8 (C), 126.8–129.3 (10 × CH), 137.1 (C), 138.5 (C); HRMS (ESI, positive ion) calcd for C22H26NaO4S, m/z 409.1449, found 409.1465.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). [α]25D + 26.3 (c 0.24 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 2.24 (s, 3H), 2.40 (brs, 1H), 3.29 (s, 3H), 3.42 (s, 3H), 3.88 (dd, 1H, J = 2.1, 6.6 Hz), 3.99 (d, 1H, J = 2.7 Hz), 4.40 (d, 1H, J = 6.9 Hz), 4.46 (d, 1H, J = 11.4 Hz), 4.72 (d, 1H, J = 11.4 Hz), 7.32–7.38 (m, 5H); 13C NMR (75 MHz, CDCl3): δ 27.3 (CH3), 54.1 (CH3), 55.3 (CH3), 72.0 (CH), 73.4 (CH2), 83.7 (CH), 103.5 (CH), 128.2 (CH), 128.4 (2 × CH), 128.5 (2 × CH), 136.9 (C), 209.8 (C); HRMS (ESI, positive ion) calcd for C14H20NaO5, m/z 291.1208, found 291.1236.
1). [α]25D + 26.3 (c 0.24 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 2.24 (s, 3H), 2.40 (brs, 1H), 3.29 (s, 3H), 3.42 (s, 3H), 3.88 (dd, 1H, J = 2.1, 6.6 Hz), 3.99 (d, 1H, J = 2.7 Hz), 4.40 (d, 1H, J = 6.9 Hz), 4.46 (d, 1H, J = 11.4 Hz), 4.72 (d, 1H, J = 11.4 Hz), 7.32–7.38 (m, 5H); 13C NMR (75 MHz, CDCl3): δ 27.3 (CH3), 54.1 (CH3), 55.3 (CH3), 72.0 (CH), 73.4 (CH2), 83.7 (CH), 103.5 (CH), 128.2 (CH), 128.4 (2 × CH), 128.5 (2 × CH), 136.9 (C), 209.8 (C); HRMS (ESI, positive ion) calcd for C14H20NaO5, m/z 291.1208, found 291.1236.Preparation of 2a and 3a (scale up to 20 fold)
To 1a (3.0 g, 11.4 mmol) was added a mixture of conc. HCl–MeOH (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19) (200 mL) and stirred at room temperature for 15 min. Usual work up and purification yielded 2a (1.6 g, 48%). This reaction was repeated and after 6 h it afforded 3a (3.20 g, 63%) upon work up and purification.
19) (200 mL) and stirred at room temperature for 15 min. Usual work up and purification yielded 2a (1.6 g, 48%). This reaction was repeated and after 6 h it afforded 3a (3.20 g, 63%) upon work up and purification.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19) solution (20 mL) at room temperature for 6 h, following the procedure as described in the preparation of 2a. Usual work up and purification by column chromatography over silica gel (230–400 mesh) using petroleum ether–EtOAc (23
19) solution (20 mL) at room temperature for 6 h, following the procedure as described in the preparation of 2a. Usual work up and purification by column chromatography over silica gel (230–400 mesh) using petroleum ether–EtOAc (23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) as eluent afforded 3b (219 mg, 65%) as colourless liquid. [α]25D + 57.1 (c 0.36 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 2.25 (s, 3H), 3.32 (s, 3H), 3.37 (s, 3H), 3.40–3.42 (m, 3H), 3.53 (t, 3H, J = 4.2 Hz), 3.62–3.69 (m, 1H), 3.72–3.80 (m, 1H), 3.87–3.94 (m, 2H), 4.02 (d, 1H, J = 2.1 Hz), 4.47 (d, 1H, J = 11.4 Hz), 4.65 (d, 1H, J = 7.5 Hz), 4.71 (d, 1H, J = 11.4 Hz), 7.32–7.36 (m, 5H); 13C NMR (75 MHz, CDCl3): δ 27.5 (CH3), 58.9 (2 × CH3), 65.4 (CH2), 67.1 (CH2), 71.5 (CH2), 71.9 (CH2), 72.2 (CH), 73.5 (CH2), 84.2 (CH), 102.8 (CH), 128.0 (CH), 128.4 (2 × CH), 128.4 (2 × CH), 137.3 (C), 210.7 (C); ESIMS, m/z: 379 (M + Na)+.
2) as eluent afforded 3b (219 mg, 65%) as colourless liquid. [α]25D + 57.1 (c 0.36 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 2.25 (s, 3H), 3.32 (s, 3H), 3.37 (s, 3H), 3.40–3.42 (m, 3H), 3.53 (t, 3H, J = 4.2 Hz), 3.62–3.69 (m, 1H), 3.72–3.80 (m, 1H), 3.87–3.94 (m, 2H), 4.02 (d, 1H, J = 2.1 Hz), 4.47 (d, 1H, J = 11.4 Hz), 4.65 (d, 1H, J = 7.5 Hz), 4.71 (d, 1H, J = 11.4 Hz), 7.32–7.36 (m, 5H); 13C NMR (75 MHz, CDCl3): δ 27.5 (CH3), 58.9 (2 × CH3), 65.4 (CH2), 67.1 (CH2), 71.5 (CH2), 71.9 (CH2), 72.2 (CH), 73.5 (CH2), 84.2 (CH), 102.8 (CH), 128.0 (CH), 128.4 (2 × CH), 128.4 (2 × CH), 137.3 (C), 210.7 (C); ESIMS, m/z: 379 (M + Na)+.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19) solution (20 mL) at room temperature for 6 h, following the procedure as described in the preparation of 2a. Usual work up and purification by column chromatography over silica gel (230–400 mesh) using petroleum ether–EtOAc (19
19) solution (20 mL) at room temperature for 6 h, following the procedure as described in the preparation of 2a. Usual work up and purification by column chromatography over silica gel (230–400 mesh) using petroleum ether–EtOAc (19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as eluent furnished 3c (190 mg, 63%) as colourless liquid. [α]25D + 70.1 (c 0.23 in CHCl3); 1H NMR (600 MHz, CDCl3): δ 2.24 (s, 3H), 3.89 (dd, 1H, J = 5.4, 12.6 Hz), 3.93 (m, 1H), 4.04 (d, 1H, J = 2.4 Hz), 4.11–4.20 (m, 3H), 4.47 (d, 1H, J = 11.4 Hz), 4.65 (d, 1H, J = 6.6 Hz), 4.71 (d, 1H, J = 11.4 Hz), 5.15 (dd, 1H, J = 1.2, 10.2 Hz), 5.19 (dd, 1H, J = 1.2, 10.2 Hz), 5.24 (dd, 1H, J = 1.2, 16.8 Hz), 5.30 (dd, 1H, J = 1.2, 16.8 Hz), 5.78–5.84 (m, 1H), 5.89–5.95 (m, 1H), 7.32–7.36 (m, 5H); 13C NMR (150 MHz, CDCl3): δ 27.4 (CH3), 68.0 (CH2), 68.8 (CH2), 72.6 (CH), 73.5 (CH2), 83.7 (CH), 101.2 (CH), 117.4 (2 × CH2), 128.2–128.5 (5 × CH), 133.9 (CH), 134.1 (CH), 137.0 (C), 209.8 (C); HRMS (ESI, positive ion) calcd for C18H24NaO5, m/z 343.1521, found 343.1507.
1) as eluent furnished 3c (190 mg, 63%) as colourless liquid. [α]25D + 70.1 (c 0.23 in CHCl3); 1H NMR (600 MHz, CDCl3): δ 2.24 (s, 3H), 3.89 (dd, 1H, J = 5.4, 12.6 Hz), 3.93 (m, 1H), 4.04 (d, 1H, J = 2.4 Hz), 4.11–4.20 (m, 3H), 4.47 (d, 1H, J = 11.4 Hz), 4.65 (d, 1H, J = 6.6 Hz), 4.71 (d, 1H, J = 11.4 Hz), 5.15 (dd, 1H, J = 1.2, 10.2 Hz), 5.19 (dd, 1H, J = 1.2, 10.2 Hz), 5.24 (dd, 1H, J = 1.2, 16.8 Hz), 5.30 (dd, 1H, J = 1.2, 16.8 Hz), 5.78–5.84 (m, 1H), 5.89–5.95 (m, 1H), 7.32–7.36 (m, 5H); 13C NMR (150 MHz, CDCl3): δ 27.4 (CH3), 68.0 (CH2), 68.8 (CH2), 72.6 (CH), 73.5 (CH2), 83.7 (CH), 101.2 (CH), 117.4 (2 × CH2), 128.2–128.5 (5 × CH), 133.9 (CH), 134.1 (CH), 137.0 (C), 209.8 (C); HRMS (ESI, positive ion) calcd for C18H24NaO5, m/z 343.1521, found 343.1507.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19) solution (20 mL) and the mixture was stirred at room temperature for 6 h, following the procedure as described in the preparation of 2a. Usual work up and purification by column chromatography over silica gel (230–400 mesh) using petroleum ether–EtOAc (19
19) solution (20 mL) and the mixture was stirred at room temperature for 6 h, following the procedure as described in the preparation of 2a. Usual work up and purification by column chromatography over silica gel (230–400 mesh) using petroleum ether–EtOAc (19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as eluent afforded 3d (200 mg, 67%) as colourless liquid. [α]25D + 36.0 (c 0.31 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 2.22 (s, 3H), 2.50 (brs, 2H), 3.94–4.00 (m, 1H), 4.08 (d, 1H, J = 2.4 Hz), 4.23–4.41 (m, 4H), 4.56 (d, 1H, J = 11.1 Hz), 4.70 (d, 1H, J = 11.1 Hz), 4.92 (d, 1H, J = 6.6 Hz), 7.35–7.61 (m, 5H); 13C NMR (75 MHz, CDCl3): δ 27.4 (CH3), 54.8 (CH2), 55.0 (CH2), 72.6 (CH), 73.6 (CH2), 75.2 (2 × CH), 78.9 (C), 79.3 (C), 83.4 (CH), 100.2 (CH), 128.2–128.5 (5 × CH), 136.8 (C), 209.9 (C); HRMS (ESI, positive ion) calcd for C18H20NaO5, m/z 339.1208, found 339.1227.
1) as eluent afforded 3d (200 mg, 67%) as colourless liquid. [α]25D + 36.0 (c 0.31 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 2.22 (s, 3H), 2.50 (brs, 2H), 3.94–4.00 (m, 1H), 4.08 (d, 1H, J = 2.4 Hz), 4.23–4.41 (m, 4H), 4.56 (d, 1H, J = 11.1 Hz), 4.70 (d, 1H, J = 11.1 Hz), 4.92 (d, 1H, J = 6.6 Hz), 7.35–7.61 (m, 5H); 13C NMR (75 MHz, CDCl3): δ 27.4 (CH3), 54.8 (CH2), 55.0 (CH2), 72.6 (CH), 73.6 (CH2), 75.2 (2 × CH), 78.9 (C), 79.3 (C), 83.4 (CH), 100.2 (CH), 128.2–128.5 (5 × CH), 136.8 (C), 209.9 (C); HRMS (ESI, positive ion) calcd for C18H20NaO5, m/z 339.1208, found 339.1227.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19, 20 mL) at room temperature for 6 h, following the procedure as described in the preparation of 2a. Usual work up and purification by column chromatography over silica gel (230–400 mesh) using petroleum ether–EtOAc (19
19, 20 mL) at room temperature for 6 h, following the procedure as described in the preparation of 2a. Usual work up and purification by column chromatography over silica gel (230–400 mesh) using petroleum ether–EtOAc (19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as eluent gave 3e (195 mg, 64%) as colourless liquid. [α]25D + 16.5 (c 0.27 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 1.14–1.25 (m, 12H), 2.24 (s, 3H), 2.48 (d, 1H, J = 5.7 Hz), 3.77–3.97 (m, 3H), 4.06 (d, 1H, J = 3.0 Hz), 4.52 (d, 1H, J = 11.4 Hz), 4.66 (d, 1H, J = 11.4 Hz), 4.71 (d, 1H, J = 6.0 Hz), 7.32–7.41 (m, 5H); 13C NMR (75 MHz, CDCl3): δ 21.9 (CH3), 22.8 (CH3), 23.2 (CH3), 23.4 (CH3), 27.4 (CH3), 69.1 (CH), 70.1 (CH), 73.3 (CH, CH2), 83.7 (CH), 98.8 (CH), 128.0–128.6 (5 × CH), 137.2 (C), 209.9 (C); HRMS (ESI, positive ion) calcd for C18H28NaO5, m/z 347.1834, found 347.1851.
1) as eluent gave 3e (195 mg, 64%) as colourless liquid. [α]25D + 16.5 (c 0.27 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 1.14–1.25 (m, 12H), 2.24 (s, 3H), 2.48 (d, 1H, J = 5.7 Hz), 3.77–3.97 (m, 3H), 4.06 (d, 1H, J = 3.0 Hz), 4.52 (d, 1H, J = 11.4 Hz), 4.66 (d, 1H, J = 11.4 Hz), 4.71 (d, 1H, J = 6.0 Hz), 7.32–7.41 (m, 5H); 13C NMR (75 MHz, CDCl3): δ 21.9 (CH3), 22.8 (CH3), 23.2 (CH3), 23.4 (CH3), 27.4 (CH3), 69.1 (CH), 70.1 (CH), 73.3 (CH, CH2), 83.7 (CH), 98.8 (CH), 128.0–128.6 (5 × CH), 137.2 (C), 209.9 (C); HRMS (ESI, positive ion) calcd for C18H28NaO5, m/z 347.1834, found 347.1851.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19, 20 mL) was added to 1a (250 mg, 0.95 mmol) and the mixture was stirred at room temperature for 6 h, following the procedure as described in the preparation of 2a. Usual work up and purification by column chromatography over silica gel (230–400 mesh) using petroleum ether–EtOAc (19
19, 20 mL) was added to 1a (250 mg, 0.95 mmol) and the mixture was stirred at room temperature for 6 h, following the procedure as described in the preparation of 2a. Usual work up and purification by column chromatography over silica gel (230–400 mesh) using petroleum ether–EtOAc (19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as eluent produced 3f (220 mg, 64%) as colourless liquid. [α]25D + 107.1 (c 0.16 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 2.25 (s, 3H), 2.52 (brs, 1H), 3.50–3.57 (m, 2H), 3.66 (t, 3H, J = 5.4 Hz), 3.81–3.99 (m, 4H), 4.47 (d, 1H, J = 11.4 Hz), 4.65 (d, 1H, J = 7.2 Hz), 4.75 (d, 1H, J = 11.4 Hz), 7.36 (s, 5H); 13C NMR (75 MHz, CDCl3): δ 27.4 (CH3), 42.8 (CH2), 43.2 (CH2), 66.3 (CH2), 67.7 (CH2), 72.0 (CH), 73.3 (CH2), 83.4 (CH), 101.8 (CH), 128.2 (CH), 128.4 (2 × CH), 128.5 (2 × CH), 136.8 (C), 209.9 (C); HRMS (ESI, positive ion) calcd for C16H22Cl2NaO5, m/z 387.0742, found 387.0760.
1) as eluent produced 3f (220 mg, 64%) as colourless liquid. [α]25D + 107.1 (c 0.16 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 2.25 (s, 3H), 2.52 (brs, 1H), 3.50–3.57 (m, 2H), 3.66 (t, 3H, J = 5.4 Hz), 3.81–3.99 (m, 4H), 4.47 (d, 1H, J = 11.4 Hz), 4.65 (d, 1H, J = 7.2 Hz), 4.75 (d, 1H, J = 11.4 Hz), 7.36 (s, 5H); 13C NMR (75 MHz, CDCl3): δ 27.4 (CH3), 42.8 (CH2), 43.2 (CH2), 66.3 (CH2), 67.7 (CH2), 72.0 (CH), 73.3 (CH2), 83.4 (CH), 101.8 (CH), 128.2 (CH), 128.4 (2 × CH), 128.5 (2 × CH), 136.8 (C), 209.9 (C); HRMS (ESI, positive ion) calcd for C16H22Cl2NaO5, m/z 387.0742, found 387.0760.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as eluent afforded 3g (170 mg, 60%) as colourless liquid. [α]25D + 153.1 (c 0.31 in CHCl3); 1H NMR (600 MHz, CDCl3): δ 1.12 (t, 3H, J = 7.2 Hz), 1.22 (t, 3H, J = 7.2 Hz), 2.25 (s, 3H), 2.40 (d, 1H, J = 4.2 Hz), 3.34 (quint, 1H, J = 7.2 Hz), 3.59–3.65 (m, 1H), 3.66–3.76 (m, 2H), 3.85 (brt, 1H, J = 3.0 Hz), 4.01 (d, 1H, J = 2.4 Hz), 4.46 (d, 1H, J = 11.4 Hz), 4.53 (d, 1H, J = 6.6 Hz), 4.72 (d, 1H, J = 12.0 Hz), 7.29–7.36 (m, 5H); 13C NMR (150 MHz, CDCl3): δ 15.2 (CH3), 15.4 (CH3), 27.4 (CH3), 63.1 (CH2), 63.7 (CH2), 72.6 (CH), 73.4 (CH2), 83.8 (CH), 102.0 (CH), 127.9, 128.1, 128.2, 128.4, and 128.5 (5 × CH), 137.0 (C), 209.8 (C); HRMS (ESI, positive ion) calcd for C16H24NaO5, m/z 319.1521, found 319.1493.
1) as eluent afforded 3g (170 mg, 60%) as colourless liquid. [α]25D + 153.1 (c 0.31 in CHCl3); 1H NMR (600 MHz, CDCl3): δ 1.12 (t, 3H, J = 7.2 Hz), 1.22 (t, 3H, J = 7.2 Hz), 2.25 (s, 3H), 2.40 (d, 1H, J = 4.2 Hz), 3.34 (quint, 1H, J = 7.2 Hz), 3.59–3.65 (m, 1H), 3.66–3.76 (m, 2H), 3.85 (brt, 1H, J = 3.0 Hz), 4.01 (d, 1H, J = 2.4 Hz), 4.46 (d, 1H, J = 11.4 Hz), 4.53 (d, 1H, J = 6.6 Hz), 4.72 (d, 1H, J = 12.0 Hz), 7.29–7.36 (m, 5H); 13C NMR (150 MHz, CDCl3): δ 15.2 (CH3), 15.4 (CH3), 27.4 (CH3), 63.1 (CH2), 63.7 (CH2), 72.6 (CH), 73.4 (CH2), 83.8 (CH), 102.0 (CH), 127.9, 128.1, 128.2, 128.4, and 128.5 (5 × CH), 137.0 (C), 209.8 (C); HRMS (ESI, positive ion) calcd for C16H24NaO5, m/z 319.1521, found 319.1493.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19) solution (20 mL) at room temperature for 6 h, following the procedure as described in the preparation of 2a. Work up and purification by column chromatography over silica gel (230–400 mesh) using petroleum ether–EtOAc (93
19) solution (20 mL) at room temperature for 6 h, following the procedure as described in the preparation of 2a. Work up and purification by column chromatography over silica gel (230–400 mesh) using petroleum ether–EtOAc (93![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7) as eluent gave 3h (205 mg, 61%) as colourless liquid. [α]25D + 153.1 (c 0.31 in CHCl3); 1H NMR (600 MHz, CDCl3): δ 0.90 (t, 3H, J = 7.8 Hz), 0.92 (t, 3H, J = 7.8 Hz), 1.30–1.39 (m, 4H), 1.44–1.49 (m, 2H), 1.55–1.59 (m, 2H), 2.25 (s, 3H), 3.28 (dd, 1H, J = 6.6, 15.6 Hz), 3.54 (dd, 1H, J = 6.6, 15.6 Hz), 3.61–3.68 (m, 2H), 3.87 (dd, 1H, J = 3.0, 7.2 Hz), 4.04 (d, 1H, J = 2.4 Hz), 4.46 (d, 1H, J = 11.4 Hz), 4.53 (d, 1H, J = 6.6 Hz), 4.70 (d, 1H, J = 11.4 Hz), 7.28–7.36 (m, 5H); 13C NMR (150 MHz, CDCl3): δ 13.8 (2 × CH3), 19.3 (2 × CH2), 27.4 (CH3), 31.8 (CH2), 31.9 (CH2), 67.1 (CH2), 67.9 (CH2), 72.6 (CH), 73.4 (CH2), 83.8 (CH), 102.0 (CH), 128.3 (CH), 128.4 (2 × CH), 128.5 (2 × CH), 137.1 (C), 210.0 (C); HRMS (ESI, positive ion) calcd for C20H32NaO5, m/z 375.2147, found 375.2125.
7) as eluent gave 3h (205 mg, 61%) as colourless liquid. [α]25D + 153.1 (c 0.31 in CHCl3); 1H NMR (600 MHz, CDCl3): δ 0.90 (t, 3H, J = 7.8 Hz), 0.92 (t, 3H, J = 7.8 Hz), 1.30–1.39 (m, 4H), 1.44–1.49 (m, 2H), 1.55–1.59 (m, 2H), 2.25 (s, 3H), 3.28 (dd, 1H, J = 6.6, 15.6 Hz), 3.54 (dd, 1H, J = 6.6, 15.6 Hz), 3.61–3.68 (m, 2H), 3.87 (dd, 1H, J = 3.0, 7.2 Hz), 4.04 (d, 1H, J = 2.4 Hz), 4.46 (d, 1H, J = 11.4 Hz), 4.53 (d, 1H, J = 6.6 Hz), 4.70 (d, 1H, J = 11.4 Hz), 7.28–7.36 (m, 5H); 13C NMR (150 MHz, CDCl3): δ 13.8 (2 × CH3), 19.3 (2 × CH2), 27.4 (CH3), 31.8 (CH2), 31.9 (CH2), 67.1 (CH2), 67.9 (CH2), 72.6 (CH), 73.4 (CH2), 83.8 (CH), 102.0 (CH), 128.3 (CH), 128.4 (2 × CH), 128.5 (2 × CH), 137.1 (C), 210.0 (C); HRMS (ESI, positive ion) calcd for C20H32NaO5, m/z 375.2147, found 375.2125.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19) solution (20 mL) at room temperature for 1 h, following the procedure as described in the preparation of 2a. Subsequent work up and purification by column chromatography over silica gel (230–400 mesh) using petroleum ether–EtOAc (23
19) solution (20 mL) at room temperature for 1 h, following the procedure as described in the preparation of 2a. Subsequent work up and purification by column chromatography over silica gel (230–400 mesh) using petroleum ether–EtOAc (23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) as eluent afforded 4a (200 mg, 47%) as yellowish liquid; [α]25D − 16.5 (c 0.12 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 1.73 (s, 3H), 2.54 (brd, 1H J = 7.5 Hz), 3.71 (d, 1H, J = 3.3 Hz), 3.77–3.91 (m, 4H), 4.06–4.11 (m, 1H), 4.60 (d, 1H, J = 12.0 Hz), 4.66 (d, 1H, J = 12.0 Hz), 4.83 (d, 1H, J = 6.0 Hz), 7.26–7.36 (m, 15H); 13C NMR (150 MHz, CDCl3): δ 24.2 (CH3), 33.0 (CH2), 35.1 (CH2), 72.2 (CH2), 80.6 (CH), 86.9 (CH), 89.1 (CH), 93.5 (C), 127.1–129.0 (15 × CH), 137.4 (C), 137.5 (C), 137.8 (C); HRMS (ESI, positive ion) calcd for C26H28NaO3S2, m/z 475.1378, found 475.1366.
2) as eluent afforded 4a (200 mg, 47%) as yellowish liquid; [α]25D − 16.5 (c 0.12 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 1.73 (s, 3H), 2.54 (brd, 1H J = 7.5 Hz), 3.71 (d, 1H, J = 3.3 Hz), 3.77–3.91 (m, 4H), 4.06–4.11 (m, 1H), 4.60 (d, 1H, J = 12.0 Hz), 4.66 (d, 1H, J = 12.0 Hz), 4.83 (d, 1H, J = 6.0 Hz), 7.26–7.36 (m, 15H); 13C NMR (150 MHz, CDCl3): δ 24.2 (CH3), 33.0 (CH2), 35.1 (CH2), 72.2 (CH2), 80.6 (CH), 86.9 (CH), 89.1 (CH), 93.5 (C), 127.1–129.0 (15 × CH), 137.4 (C), 137.5 (C), 137.8 (C); HRMS (ESI, positive ion) calcd for C26H28NaO3S2, m/z 475.1378, found 475.1366.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19) solution (20 mL) and the reaction mixture was stirred at room temperature for 1 h. Work up and purification on a silica gel (230–400 mesh) column using a mixture of petroleum ether–EtOAc (19
19) solution (20 mL) and the reaction mixture was stirred at room temperature for 1 h. Work up and purification on a silica gel (230–400 mesh) column using a mixture of petroleum ether–EtOAc (19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as eluent to furnish 4b (150 mg, 53%) as colourless liquid. [α]25D + 72.7 (c 0.33 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 1.21 (t, 3H, J = 6.9 Hz), 1.41–1.46 (m, 6H), 3.53–3.61 (m, 2H), 3.64–3.74 (m, 2H), 3.76 (d, 1H, J = 3.6 Hz), 4.62 (d, 1H, J = 12.0 Hz), 4.79 (t, 1H, J = 4.2 Hz), 4.84 (d, 1H, J = 12.0 Hz), 5.77 (d, 1H, J = 4.5 Hz), 7.28–7.41 (m, 5H); 13C NMR (75 MHz, CDCl3): δ 15.7 (2 × CH3) 21.1 (CH3), 56.8 (CH2), 63.7 (CH2), 72.0 (CH2), 86.3 (CH), 88.2 (CH), 103.6 (CH), 105.9 (C), 127.8–128.3 (5 × CH), 137.37 (C); HRMS (ESI, positive ion) calcd for C16H24NaO5, m/z 319.1521, found 319.1509.
1) as eluent to furnish 4b (150 mg, 53%) as colourless liquid. [α]25D + 72.7 (c 0.33 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 1.21 (t, 3H, J = 6.9 Hz), 1.41–1.46 (m, 6H), 3.53–3.61 (m, 2H), 3.64–3.74 (m, 2H), 3.76 (d, 1H, J = 3.6 Hz), 4.62 (d, 1H, J = 12.0 Hz), 4.79 (t, 1H, J = 4.2 Hz), 4.84 (d, 1H, J = 12.0 Hz), 5.77 (d, 1H, J = 4.5 Hz), 7.28–7.41 (m, 5H); 13C NMR (75 MHz, CDCl3): δ 15.7 (2 × CH3) 21.1 (CH3), 56.8 (CH2), 63.7 (CH2), 72.0 (CH2), 86.3 (CH), 88.2 (CH), 103.6 (CH), 105.9 (C), 127.8–128.3 (5 × CH), 137.37 (C); HRMS (ESI, positive ion) calcd for C16H24NaO5, m/z 319.1521, found 319.1509.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19) solution (20 mL) at room temperature for 1 h, following the procedure as described in the preparation of 2a and subsequent work up and purification by column chromatography over silica gel (230–400 mesh) using petroleum ether–EtOAc (19
19) solution (20 mL) at room temperature for 1 h, following the procedure as described in the preparation of 2a and subsequent work up and purification by column chromatography over silica gel (230–400 mesh) using petroleum ether–EtOAc (19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as eluent furnished 4c (175 mg, 52%) as colourless liquid. [α]25D + 14.5 (c 0.22 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 0.89–0.95 (m, 6H), 1.30–1.65 (m, 11H, including a ‘s, 3H’ at δ 1.42), 3.44–3.65 (m, 3H), 3.67–3.85 (m, 1H), 3.91 (d, 1H, J = 5.7 Hz), 4.02–4.16 (m, 1H), 4.67 (d, 1H, J = 12.0 Hz), 4.72 (d, 1H, J = 11.7 Hz), 4.98 (d, 1H, J = 4.8 Hz), 7.28–7.41 (m, 5H); 13C NMR (75 MHz, CDCl3): δ 13.9 (2 × CH3), 19.3 (CH2), 19.4 (CH2), 20.2 (CH3), 31.5 (CH2), 32.1 (CH2), 61.1 (CH2), 68.6 (CH2), 72.0 (CH2), 75.7 (CH), 87.1 (CH), 100.4 (CH), 107.9 (C), 127.5–128.3 (5 × CH), 138.0 (C); HRMS (ESI, positive ion) calcd for C20H32NaO5, m/z 375.2147, found 375.2125.
1) as eluent furnished 4c (175 mg, 52%) as colourless liquid. [α]25D + 14.5 (c 0.22 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 0.89–0.95 (m, 6H), 1.30–1.65 (m, 11H, including a ‘s, 3H’ at δ 1.42), 3.44–3.65 (m, 3H), 3.67–3.85 (m, 1H), 3.91 (d, 1H, J = 5.7 Hz), 4.02–4.16 (m, 1H), 4.67 (d, 1H, J = 12.0 Hz), 4.72 (d, 1H, J = 11.7 Hz), 4.98 (d, 1H, J = 4.8 Hz), 7.28–7.41 (m, 5H); 13C NMR (75 MHz, CDCl3): δ 13.9 (2 × CH3), 19.3 (CH2), 19.4 (CH2), 20.2 (CH3), 31.5 (CH2), 32.1 (CH2), 61.1 (CH2), 68.6 (CH2), 72.0 (CH2), 75.7 (CH), 87.1 (CH), 100.4 (CH), 107.9 (C), 127.5–128.3 (5 × CH), 138.0 (C); HRMS (ESI, positive ion) calcd for C20H32NaO5, m/z 375.2147, found 375.2125.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as eluent afforded 4d (95 mg, 58%) as colourless liquid. [α]25D + 27.8 (c 0.43 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 1.22 (s, 3H), 2.27–2.48 (m, 4H), 3.66–3.69 (m, 1H), 3.99–4.17 (m, 2H), 4.54–4.71 (m, 2H), 4.98–5.23 (m, 4H), 5.74–5.94 (m, 2H), 7.29–7.36 (m, 5H); 13C NMR (75 MHz, CDCl3): δ 20.2 (CH3), 33.6 (CH2), 45.6 (CH2), 72.4 (CH2), 77.3 (CH), 77.4 (CH), 82.9 (C), 89.2 (CH), 117.1 (CH2), 118.5 (CH2), 127.3–128.6 (5 × CH), 134.0 (CH), 134.7 (CH), 138.2 (C); ESIMS, m/z: 311 (M + Na)+.
1) as eluent afforded 4d (95 mg, 58%) as colourless liquid. [α]25D + 27.8 (c 0.43 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 1.22 (s, 3H), 2.27–2.48 (m, 4H), 3.66–3.69 (m, 1H), 3.99–4.17 (m, 2H), 4.54–4.71 (m, 2H), 4.98–5.23 (m, 4H), 5.74–5.94 (m, 2H), 7.29–7.36 (m, 5H); 13C NMR (75 MHz, CDCl3): δ 20.2 (CH3), 33.6 (CH2), 45.6 (CH2), 72.4 (CH2), 77.3 (CH), 77.4 (CH), 82.9 (C), 89.2 (CH), 117.1 (CH2), 118.5 (CH2), 127.3–128.6 (5 × CH), 134.0 (CH), 134.7 (CH), 138.2 (C); ESIMS, m/z: 311 (M + Na)+.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19) solution (20 mL) and stirring at room temperature for 1 h, following the procedure as described in the preparation of 2a. Work up and purification by column chromatography over silica gel (230–400 mesh) using petroleum ether–EtOAc (19
19) solution (20 mL) and stirring at room temperature for 1 h, following the procedure as described in the preparation of 2a. Work up and purification by column chromatography over silica gel (230–400 mesh) using petroleum ether–EtOAc (19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as eluent afforded 4e (185 mg, 52%) as colourless liquid. [α]25D + 34.5 (c 0.21 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 1.42 (s, 3H), 1.60–1.77 (m, 4H), 2.09–2.25 (m, 4H), 3.45–3.92 (m, 5H), 4.03–4.13 (m, 1H), 4.67 (d, 1H, J = 12.0 Hz), 4.72 (d, 1H, J = 12.0 Hz), 4.96–5.05 (m, 5H), 5.74–5.86 (m, 2H), 7.27–7.45 (m, 5H); 13C NMR (75 MHz, CDCl3): δ 20.2 (CH3), 28.6 (CH2), 29.1 (CH2), 30.2 (CH2), 30.3 (CH2), 60.8 (CH2), 68.2 (CH2), 72.0 (CH2), 75.8 (CH), 87.1 (CH), 100.4 (CH), 107.9 (C), 114.7 (CH2), 114.9 (CH2), 127.5–128.3 (5 × CH), 137.9 (CH), 138.0 (C), 138.2 (CH); HRMS (ESI, positive ion) calcd for C22H32NaO5, m/z 399.2147, found 399.2150.
1) as eluent afforded 4e (185 mg, 52%) as colourless liquid. [α]25D + 34.5 (c 0.21 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 1.42 (s, 3H), 1.60–1.77 (m, 4H), 2.09–2.25 (m, 4H), 3.45–3.92 (m, 5H), 4.03–4.13 (m, 1H), 4.67 (d, 1H, J = 12.0 Hz), 4.72 (d, 1H, J = 12.0 Hz), 4.96–5.05 (m, 5H), 5.74–5.86 (m, 2H), 7.27–7.45 (m, 5H); 13C NMR (75 MHz, CDCl3): δ 20.2 (CH3), 28.6 (CH2), 29.1 (CH2), 30.2 (CH2), 30.3 (CH2), 60.8 (CH2), 68.2 (CH2), 72.0 (CH2), 75.8 (CH), 87.1 (CH), 100.4 (CH), 107.9 (C), 114.7 (CH2), 114.9 (CH2), 127.5–128.3 (5 × CH), 137.9 (CH), 138.0 (C), 138.2 (CH); HRMS (ESI, positive ion) calcd for C22H32NaO5, m/z 399.2147, found 399.2150.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19) solution (20 mL), at room temperature for 60 min, following the procedure as described in the preparation of 2a, followed by work up and purification by column chromatography over silica gel (230–400 mesh) using petroleum ether–EtOAc (19
19) solution (20 mL), at room temperature for 60 min, following the procedure as described in the preparation of 2a, followed by work up and purification by column chromatography over silica gel (230–400 mesh) using petroleum ether–EtOAc (19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as eluent furnished 4f (160 mg, 52%) as colourless liquid. [α]25D + 86.7 (c 0.26 in CHCl3); 1H NMR (600 MHz, CDCl3): δ 0.91 (t, 3H, J = 7.2 Hz), 0.94 (t, 3H, J = 7.2 Hz), 1.42 (s, 3H), 1.53–1.59 (m, 2H), 1.60–1.66 (m, 2H), 2.73 (d, 1H, J = 10.8 Hz), 3.42–3.48 (m, 2H), 3.49–3.52 (m, 1H), 3.75–3.79 (m, 1H), 3.92 (d, 1H, J = 5.4 Hz), 4.12 (quint, 1H, J = 4.8 Hz), 4.68 (d, 1H, J = 12.0 Hz), 4.72 (d, 1H, J = 12.0 Hz), 4.99 (d, 1H, J = 5.4 Hz), 7.27–7.36 (m, 5H); 13C NMR (150 MHz, CDCl3): δ 10.6 (CH3), 10.6 (CH3), 20.3 (CH3), 22.7 (CH2), 23.2 (CH2), 63.0 (CH2), 70.4 (CH2), 72.0 (CH2), 75.8 (CH), 87.2 (CH), 100.4 (CH), 107.9 (C), 127.5 (2 × CH), 127.6 (CH), 128.3 (2 × CH), 138.1 (C); HRMS (ESI, positive ion) calcd for C18H28NaO5, m/z 347.1834, found 347.1881.
1) as eluent furnished 4f (160 mg, 52%) as colourless liquid. [α]25D + 86.7 (c 0.26 in CHCl3); 1H NMR (600 MHz, CDCl3): δ 0.91 (t, 3H, J = 7.2 Hz), 0.94 (t, 3H, J = 7.2 Hz), 1.42 (s, 3H), 1.53–1.59 (m, 2H), 1.60–1.66 (m, 2H), 2.73 (d, 1H, J = 10.8 Hz), 3.42–3.48 (m, 2H), 3.49–3.52 (m, 1H), 3.75–3.79 (m, 1H), 3.92 (d, 1H, J = 5.4 Hz), 4.12 (quint, 1H, J = 4.8 Hz), 4.68 (d, 1H, J = 12.0 Hz), 4.72 (d, 1H, J = 12.0 Hz), 4.99 (d, 1H, J = 5.4 Hz), 7.27–7.36 (m, 5H); 13C NMR (150 MHz, CDCl3): δ 10.6 (CH3), 10.6 (CH3), 20.3 (CH3), 22.7 (CH2), 23.2 (CH2), 63.0 (CH2), 70.4 (CH2), 72.0 (CH2), 75.8 (CH), 87.2 (CH), 100.4 (CH), 107.9 (C), 127.5 (2 × CH), 127.6 (CH), 128.3 (2 × CH), 138.1 (C); HRMS (ESI, positive ion) calcd for C18H28NaO5, m/z 347.1834, found 347.1881.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19) at room temperature for 1 h, following the procedure as described in the preparation of 2a followed by work up and purification by column chromatography over silica gel (230–400 mesh) using petroleum ether–EtOAc (23
19) at room temperature for 1 h, following the procedure as described in the preparation of 2a followed by work up and purification by column chromatography over silica gel (230–400 mesh) using petroleum ether–EtOAc (23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) as eluent furnished 4g (145 mg, 50%) as colourless liquid. [α]25D + 160.3 (c 0.18 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 1.18 (t, 3H, J = 6.9 Hz), 1.26 (t, 3H, J = 6.9 Hz), 1.38 (s, 3H), 2.76 (d, 1H, J = 10.5 Hz), 3.51–3.70 (m, 3H), 3.82–3.93 (m, 2H), 4.03–4.11 (m, 1H), 4.15 (t, 2H, J = 5.4 Hz), 5.00 (d, 1H, J = 5.1 Hz), 5.19 (d, 1H, J = 10.5 Hz), 5.32 (d, 1H, J = 17.4 Hz), 5.86–5.99 (m, 1H); 13C NMR (75 MHz, CDCl3): δ 15.1 (CH3), 15.6 (CH3), 20.0 (CH3), 56.8 (CH2), 64.2 (CH2), 71.1 (CH2), 75.9 (CH), 87.3 (CH), 100.3 (CH), 108.0 (C), 117.0 (CH2), 134.4 (CH); HRMS (ESI, positive ion) calcd for C12H22NaO5, m/z 269.1365, found 269.1382.
2) as eluent furnished 4g (145 mg, 50%) as colourless liquid. [α]25D + 160.3 (c 0.18 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 1.18 (t, 3H, J = 6.9 Hz), 1.26 (t, 3H, J = 6.9 Hz), 1.38 (s, 3H), 2.76 (d, 1H, J = 10.5 Hz), 3.51–3.70 (m, 3H), 3.82–3.93 (m, 2H), 4.03–4.11 (m, 1H), 4.15 (t, 2H, J = 5.4 Hz), 5.00 (d, 1H, J = 5.1 Hz), 5.19 (d, 1H, J = 10.5 Hz), 5.32 (d, 1H, J = 17.4 Hz), 5.86–5.99 (m, 1H); 13C NMR (75 MHz, CDCl3): δ 15.1 (CH3), 15.6 (CH3), 20.0 (CH3), 56.8 (CH2), 64.2 (CH2), 71.1 (CH2), 75.9 (CH), 87.3 (CH), 100.3 (CH), 108.0 (C), 117.0 (CH2), 134.4 (CH); HRMS (ESI, positive ion) calcd for C12H22NaO5, m/z 269.1365, found 269.1382.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19) was added to 1b (250 mg, 1.18 mmol) and the mixture was stirred at room temperature for 1 h, following the procedure as described in the preparation of 2a. Work up and purification by column chromatography over silica gel (230–400 mesh) using petroleum ether–EtOAc (19
19) was added to 1b (250 mg, 1.18 mmol) and the mixture was stirred at room temperature for 1 h, following the procedure as described in the preparation of 2a. Work up and purification by column chromatography over silica gel (230–400 mesh) using petroleum ether–EtOAc (19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as eluent furnished 4h (195 mg, 55%) as colourless liquid. [α]25D + 21.3 (c 0.37 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 0.92 (t, 3H, J = 7.5 Hz), 0.95 (t, 3H, J = 7.2 Hz), 1.32 (s, 3H), 1.13–1.42 (m, 4H), 1.47–1.66 (m, 4H), 3.45–3.75 (m, 3H), 3.78–3.84 (m, 2H), 4.01–4.10 (m, 1H), 4.14–4.16 (m, 2H), 4.96 (d, 1H, J = 5.1 Hz), 5.20 (d, 1H, J = 10.5 Hz), 5.32 (dd, 1H, J = 1.5, 17.1 Hz), 5.86–5.99 (m, 1H); 13C NMR (75 MHz, CDCl3): δ 13.9 (CH3), 14.0 (CH3), 19.3 (CH2), 19.4 (CH2), 20.2 (CH3), 31.6 (CH2), 32.1 (CH2), 61.2 (CH2), 68.6 (CH2), 71.1 (CH2), 75.8 (CH), 87.0 (CH), 100.4 (CH), 107.9 (C), 117.0 (CH2), 134.4 (CH). HRMS (ESI, positive ion) calcd for C16H30NaO5, m/z 325.1991, found 325.1980.
1) as eluent furnished 4h (195 mg, 55%) as colourless liquid. [α]25D + 21.3 (c 0.37 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 0.92 (t, 3H, J = 7.5 Hz), 0.95 (t, 3H, J = 7.2 Hz), 1.32 (s, 3H), 1.13–1.42 (m, 4H), 1.47–1.66 (m, 4H), 3.45–3.75 (m, 3H), 3.78–3.84 (m, 2H), 4.01–4.10 (m, 1H), 4.14–4.16 (m, 2H), 4.96 (d, 1H, J = 5.1 Hz), 5.20 (d, 1H, J = 10.5 Hz), 5.32 (dd, 1H, J = 1.5, 17.1 Hz), 5.86–5.99 (m, 1H); 13C NMR (75 MHz, CDCl3): δ 13.9 (CH3), 14.0 (CH3), 19.3 (CH2), 19.4 (CH2), 20.2 (CH3), 31.6 (CH2), 32.1 (CH2), 61.2 (CH2), 68.6 (CH2), 71.1 (CH2), 75.8 (CH), 87.0 (CH), 100.4 (CH), 107.9 (C), 117.0 (CH2), 134.4 (CH). HRMS (ESI, positive ion) calcd for C16H30NaO5, m/z 325.1991, found 325.1980.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19) (20 mL) at room temperature for 1 h, following the procedure as described in the preparation of 2a and subsequent work up and purification led to an inseparable mixture (230 mg). To a solution of the mixture in pyridine (8 mL) at 0 °C, was added Ac2O (0.17 mL, 1.8 mmol) and the solution was stirred at 0 °C for 60 min and then at 25 °C for 6 h. The solvent was removed, crushed ice was added to the residue and the crude product was extracted with DCM (3 × 20 mL). The solvent was successively washed with 1(N) HCl (10 mL), a saturated NaHCO3 solution (2 × 15 mL), brine (2 × 15 mL), dried (Na2SO4), and evaporated to a residue, which was purified by column chromatography on silica gel (230–400 mesh) using petroleum ether–EtOAc (24
19) (20 mL) at room temperature for 1 h, following the procedure as described in the preparation of 2a and subsequent work up and purification led to an inseparable mixture (230 mg). To a solution of the mixture in pyridine (8 mL) at 0 °C, was added Ac2O (0.17 mL, 1.8 mmol) and the solution was stirred at 0 °C for 60 min and then at 25 °C for 6 h. The solvent was removed, crushed ice was added to the residue and the crude product was extracted with DCM (3 × 20 mL). The solvent was successively washed with 1(N) HCl (10 mL), a saturated NaHCO3 solution (2 × 15 mL), brine (2 × 15 mL), dried (Na2SO4), and evaporated to a residue, which was purified by column chromatography on silica gel (230–400 mesh) using petroleum ether–EtOAc (24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as eluent to afford 4i (170 mg, 46%) as colourless liquid. [α]25D + 30.5 (c 0.28 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 0.86–0.96 (m, 6H), 1.39 (s, 3H), 1.57 (q-like, 4H, J = 6.9 Hz), 2.11 (s, 3H), 3.45–3.57 (m, 3H), 3.64–3.73 (m, 2H), 4.06–4.22 (m, 2H), 4.99 (dd-like, 1H, J = 1.8, 4.8 Hz), 5.13 (d, 1H, J = 4.8 Hz), 5.16–5.34 (m, 2H), 5.82–5.97 (m, 1H). 13C NMR (75 MHz, CDCl3): δ 10.5 (CH3), 10.6 (CH3), 20.7 (CH3), 21.5 (CH3), 22.6 (CH2), 23.3 (CH2), 63.3 (CH2), 70.2 (CH2), 71.3 (CH2), 77.0 (CH), 83.1 (CH), 97.9 (CH), 106.5 (C), 117.1 (CH2), 134.2 (CH), 170.6 (C). HRMS (ESI, positive ion) calcd for C16H28NaO6, m/z 339.1784, found 339.1809.
1) as eluent to afford 4i (170 mg, 46%) as colourless liquid. [α]25D + 30.5 (c 0.28 in CHCl3); 1H NMR (300 MHz, CDCl3): δ 0.86–0.96 (m, 6H), 1.39 (s, 3H), 1.57 (q-like, 4H, J = 6.9 Hz), 2.11 (s, 3H), 3.45–3.57 (m, 3H), 3.64–3.73 (m, 2H), 4.06–4.22 (m, 2H), 4.99 (dd-like, 1H, J = 1.8, 4.8 Hz), 5.13 (d, 1H, J = 4.8 Hz), 5.16–5.34 (m, 2H), 5.82–5.97 (m, 1H). 13C NMR (75 MHz, CDCl3): δ 10.5 (CH3), 10.6 (CH3), 20.7 (CH3), 21.5 (CH3), 22.6 (CH2), 23.3 (CH2), 63.3 (CH2), 70.2 (CH2), 71.3 (CH2), 77.0 (CH), 83.1 (CH), 97.9 (CH), 106.5 (C), 117.1 (CH2), 134.2 (CH), 170.6 (C). HRMS (ESI, positive ion) calcd for C16H28NaO6, m/z 339.1784, found 339.1809.Acknowledgements
S N D thanks CSIR, Govt. of India for providing a Senior Research Fellowship to him and P K K, a student of NIPER-Kolkata, is grateful to Indian Institute of Chemical Biology for giving him laboratory facility.References
- Y. Bechor and A. Albeck, Tetrahedron, 2008, 64, 2080–2089 CrossRef CAS PubMed.
- X. D. Yang, Y. B. Hu, D. H. Yin, M. A. Turner, M. Wang, R. T. Borchardt, P. L. Howell, K. Kuczera and R. L. Schowen, Biochemistry, 2003, 42, 1900–1909 CrossRef CAS PubMed.
- N. Yamauchi and K. Kakinuma, J. Org. Chem., 1995, 60, 5614–5619 CrossRef CAS.
- (a) S. L. Bender, T. Widlanski and J. R. Knowles, Biochemistry, 1989, 28, 7560–7572 CrossRef CAS; (b) T. Widlanski, S. L. Bender and J. R. Knowles, Biochemistry, 1989, 28, 7572–7582 CrossRef CAS.
- U. Chiacchio, L. Borrello, D. Iannazzo, P. Merino, A. Piperno, A. Rescifina, B. Richichib and G. Romeob, Tetrahedron: Asymmetry, 2003, 14, 2419–2425 CrossRef CAS.
- P. A. Bartlett and K. Satake, J. Am. Chem. Soc., 1988, 110, 1628–1630 CrossRef CAS.
- (a) R. J. Parry, V. Bornemann and R. Subramanian, J. Am. Chem. Soc., 1989, 111, 5819–5824 CrossRef CAS; (b) J. M. Hill, G. N. Jenkins, C. P. Rush, N. J. Turner, A. J. Willetts, A. D. Buss, M. J. Dawson and B. A. M. Rudd, J. Am. Chem. Soc., 1995, 117, 5391–5392 CrossRef CAS.
- (a) L. Hough and B. Otter, Chem. Commun., 1966, 173–174 RSC; (b) R. Robles, I. Izquierdo, C. Rodríguez, M. Antonio, J. Mota and L. Cienfuegos, Tetrahedron: Asymmetry, 2002, 13, 399–405 CrossRef CAS.
- S. Chandrasekhar, S. Mohapatra and S. Lakshman, Chem. Lett., 1996, 211–212 CrossRef CAS.
- P. A. Colinas, V. Jäger, A. Lieberknechta and R. D. Bravoa, Tetrahedron Lett., 2003, 44, 1071–1074 CrossRef CAS.
- (a) A. D. Campbell, D. E. Paterson, R. J. K. Taylor and T. M. Raynham, Chem. Commun., 1999, 1599–1600 RSC; (b) A. M. Gómez, A. Pedregosa, S. Valverde and J. C. López, Chem. Commun., 2002, 2022–2023 RSC.
- K. C. Nicolaou, K. P. Cole, M. O. Frederick, R. J. Aversa and R. M. Denton, Angew. Chem., Int. Ed., 2007, 46, 8875–8879 CrossRef CAS PubMed.
- G. Zhou, D. Zheng, S. Da, Z. Xie and Y. Li, Tetrahedron Lett., 2006, 47, 3349–3352 CrossRef CAS PubMed.
- (a) C. Taillefumier and Y. Chapleur, Chem. Rev., 2004, 104, 263–292 CrossRef CAS PubMed; (b) C.-H. Lin, H. C. Lin and W. B. Yang, Curr. Top. Med. Chem., 2005, 5, 1431–1457 CrossRef CAS; (c) G. Liang, L. J. Bateman and N. I. Totah, Chem. Commun., 2009, 6457–6459 RSC.
- (a) J. K. Gallos, V. C. Sarli, C. I. Stathakis, T. V. Koftis, V. R. Nachmia and E. Coutouli-Argyropoulou, Tetrahedron, 2002, 58, 9351–9357 CrossRef CAS; (b) J. K. Gallos and T. V. Koftis, J. Chem. Soc., Perkin Trans. 1, 2001, 415–423 RSC.
- J. Zhou, G. Wang, L. H. Zhang and X. S. Ye, Curr. Org. Chem., 2006, 10, 625–642 CrossRef CAS.
- Y. Kubota, Y. Kaneda, K. Haraguchi, M. Mizuno, H. Abe, S. Shuto, T. Hamasaki, M. Baba and H. Tanaka, Tetrahedron, 2013, 69, 10884–10892 CrossRef CAS PubMed.
- S. Ciblat, J. Kim, C. A. Stewart, J. Wang, P. Forgione, D. Clyne and L. A. Paquette, Org. Lett., 2007, 9, 719–722 CrossRef CAS PubMed.
- B. Vauzeilles and P. Sinay, Tetrahedron Lett., 2001, 42, 7269–7272 CrossRef CAS.
- P. Z. Zhang, X. L. Li, H. Chen, Y. N. Li and R. Wang, Tetrahedron Lett., 2007, 48, 7813–7816 CrossRef CAS PubMed.
- W. Priebe, I. Fokt and G. Grynkiewicz, in Glycosciences, ed. B. Fraser-Reid, K. Tatsuta and J Thiem, Springer-Verlag Berlin, Heidelberg, 2008, pp. 699–735 Search PubMed.
- G. Fráter and F. Schröder, J. Org. Chem., 2007, 72, 1112–1120 CrossRef PubMed.
- H. Liu, I. P. Smoliakova and L. N. Koikov, Org. Lett., 2002, 4, 3895–3898 CrossRef CAS.
- R. Rafael Robles, I. Izquierdo, C. Rodríguez, M. T. Plaza, A. J. Mota and L. A. Cienfuegos, Tetrahedron: Asymmetry, 2002, 13, 399–405 CrossRef.
- (a) M. Ohwa, T. Kogura and E. L. Eliel, J. Org. Chem., 1986, 51, 2599–2601 CrossRef CAS; (b) M. Asami and T. Mukaiyama, Chem. Lett., 1983, 93–96 CrossRef CAS; (c) I. L. Lysenko, H. S. Oh and J. K. Cha, J. Org. Chem., 2007, 72, 7903–7908 CrossRef CAS PubMed; (d) H. Tsukada, Y. Mukaeda and S. Hosokawa, Org. Lett., 2013, 15, 678–681 CrossRef CAS PubMed; (e) K. M. Akhoon and D. C. Myles, J. Org. Chem., 1997, 62, 6041–6045 CrossRef CAS.
- G. Grynkiewicz, W. Szeja and J. Boryski, Acta Pol. Pharm., 2008, 65, 655–676 CAS.
- G. W. J. Fleet and D. R. Witty, Tetrahedron: Asymmetry, 1990, 1, 119–136 CrossRef CAS.
- J. Sengupta, R. Mukhopadhyay, A. Bhattacharjya, M. M. Bhadbhade and G. V. Bhosekar, J. Org. Chem., 2005, 70, 8579–8582 CrossRef CAS PubMed.
- E. Simon, K. Cook, M. R. Pritchard, W. Stripe, M. Bruch and K. Bendinskas, J. Chem. Educ., 2010, 87, 739–741 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: 1H and 13C-NMR spectra of all new compounds; NOESY, HSQC and HMBC spectra of 4g. See DOI: 10.1039/c4ra03340j |
| This journal is © The Royal Society of Chemistry 2014 |

