Cascade OFF–ON–OFF fluorescent probe: dual detection of trivalent ions and phosphate ions†
Xuejuan Wan‡
*ab,
Tianqi Liu‡a,
Haiyang Liub,
Liqiang Gub and
Youwei Yao*b
aShenzhen Key Laboratory of Special Functional Materials, College of Materials Science and Engineering, Shenzhen University, Shenzhen 518060, PR China. E-mail: wanxj@szu.edu.cn
bAdvanced Materials Institute, Graduate School at Shenzhen, Tsinghua University, Shenzhen 518055, PR China. E-mail: yaoyw@sz.tsinghua.edu.cn
First published on 16th June 2014
Abstract
A new rhodamine B-based fluorescent probe was developed for the selective cascade signaling of trivalent cations (Fe3+, Al3+, Cr3+) and phosphate anion (PO43−). Non-fluorescent rhodamine derivatives can selectively detect trivalent cations over some other metal ions in CH3CN–Tris buffer (1/1, v/v, pH 7.0) solutions, leading to prominent fluorescence OFF–ON switching. The obtained probe–cation complex can subsequently serve as a sensitive and selective chemosensor for PO43−, exhibiting complete signal quenching (fluorescence ON–OFF switching).
Introduction
The fluorogenic sensing of different metal ions has become particularly attractive because of its simplicity, high sensitivity, and instantaneous response.1–6 Among them, triple-charged metal cation detection is of significant importance because of its crucial influence in a wide range of environmental and human health areas.7–17 For example, Fe3+ ion is an essential element in the human body and acts as a cofactor in several enzymatic reactions;18–20 whereas the widely existing Al3+ ion in the environment is considered to be toxic in biological activities, and responsible for the Parkinson's disease;21,22 for Cr3+, which is also an effective nutrient for the human body, a deviation of its concentration from normal levels can increase the risk of diabetes and cardiovascular diseases.40 Simultaneously, the detection of anions through fluorescence emission readouts has also attracted significant attention considering the recognized biological roles played by anions.23 However, it is difficult to recognize anions in water or in a water mixture media compared with most of the metal cations because anions usually experience competition from the solvent for sensor binding sites.24 Nevertheless, a metal complex may resolve this problem via a simple displacement approach, causing a signal change in the UV-vis or fluorescence spectrum.25–28 Phosphate anions (PO43−), both inorganic and organic, have a unique position in nature because they participate in almost all the metabolic processes.29,30 Moreover, phosphorylated biomolecules play an important role in signal transduction for various biological processes. Human serum contains 0.80–1.45 mM phosphate but higher phosphate levels are directly connected to cardiovascular disease and acute renal failure.31Up till now, even though a large number of fluorescence chemosensors for divalent metal ions and phosphate anions have been reported in the literature, relatively few studies have been devoted to the development of fluorescence sensors which are sensitive to trivalent ions41–43 and phosphate ions.39 In view of achieving a rapid qualitative analysis of the pollutants, it is highly desirable to design a probe which is capable of simultaneous discrimination of trivalent ions and phosphate ions from other ions.
Herein, we report a simple cascade “OFF–ON–OFF” fluorescent chemosensor for trivalent and phosphate ions based on the equilibrium between spirolactam (non-fluorescent) to the ring-open amide (fluorescent) form of rhodamine chromophore.4,9,32 In general, the chemosensor showed a specific selectivity toward trivalent ions over other common metal ions, leading to prominent fluorescence OFF–ON switching. The resultant sensor–cation complex could selectively recognize PO43− over other anions, exhibiting complete ON–OFF signal quenching.
Experimental
Materials and methods
Rhodamine B was obtained from Aladdin. Phthalic anhydride and hydrazine hydrate were purchased from Sinopharm Chemical Reagent. The solution of metal ions was prepared from their chloride salts of analytical grade. The solvents were used as-received without further purification. Deionized water was used throughout the experiment.UV-vis spectra were recorded using a SCINCO S-4100. A HORIBA Jobin Yvon SPEX spectrofluorimeter was used for fluorescence measurements. The excitation wavelength was 520 nm and the spectra were recorded in the range of 540–700 nm. Both 1H NMR (400 MHz) and 13C NMR spectra (100 MHz) of the compounds were recorded on a Brurker Dpx spectrometer in CDCl3 with tetramethylsilane as the internal standard. The high-resolution mass spectra were recorded on a Bruker APEX IV.
Synthetic methodology
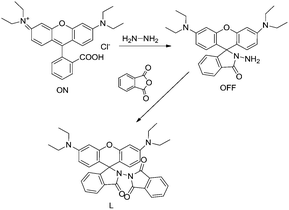
Compound L was synthesized in two steps, as shown in Scheme 1. Rhodamine B hydrazide was first synthesized following literature procedures.32 Phthalic anhydride (100 mg, 0.67 mmol) was added to a solution of rhodamine B hydrazide (300 mg, 0.66 mmol), which was dissolved in 40 mL anhydrous toluene. The reaction mixture was heated under microwave irradiation for 30 minutes and allowed to cool to room temperature. After that, the solvent was removed under reduced pressure, and the crude product was purified with a silica gel column using a mixture of petroleum ether and ethyl acetate (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) as an eluent to afford a yellow solid in a 30% yield. 1H NMR (Fig. S1†) (400 MHz, CDCl3) δ 8.02 (d, J = 7.4 Hz, 1H), 7.75–7.65 (m, 4H), 7.62 (t, J = 7.2 Hz, 1H), 7.56 (t, J = 7.2 Hz, 1H), 7.25 (d, J = 7.5 Hz, 1H), 6.83 (d, J = 8.9 Hz, 2H), 6.40 (d, J = 6.4 Hz, 2H), 6.26 (d, J = 2.3 Hz, 2H), 3.42–3.26 (m, 8H), 1.16 (t, J = 7.0 Hz, 12H). 13C NMR (Fig. S2†) (100 MHz, CDCl3) δ (ppm) 164.07, 153.68, 153.06, 149.47, 148.82, 133.97, 133.40, 130.88, 130.09, 128.32, 124.45, 123.64, 123.40, 107.52, 104.23, 97.01, 73.37, 44.18, 12.47. HRMS (ESI) (Fig. S3†): calcd for C36H35N4O4 [M + H+]+: 587.2658; found: 587.2652.
1, v/v) as an eluent to afford a yellow solid in a 30% yield. 1H NMR (Fig. S1†) (400 MHz, CDCl3) δ 8.02 (d, J = 7.4 Hz, 1H), 7.75–7.65 (m, 4H), 7.62 (t, J = 7.2 Hz, 1H), 7.56 (t, J = 7.2 Hz, 1H), 7.25 (d, J = 7.5 Hz, 1H), 6.83 (d, J = 8.9 Hz, 2H), 6.40 (d, J = 6.4 Hz, 2H), 6.26 (d, J = 2.3 Hz, 2H), 3.42–3.26 (m, 8H), 1.16 (t, J = 7.0 Hz, 12H). 13C NMR (Fig. S2†) (100 MHz, CDCl3) δ (ppm) 164.07, 153.68, 153.06, 149.47, 148.82, 133.97, 133.40, 130.88, 130.09, 128.32, 124.45, 123.64, 123.40, 107.52, 104.23, 97.01, 73.37, 44.18, 12.47. HRMS (ESI) (Fig. S3†): calcd for C36H35N4O4 [M + H+]+: 587.2658; found: 587.2652.
Results and discussion
The equilibrium between the non-fluorescent spirolactam form and the fluorescent ring-open amide form of rhodamine derivatives has been proven to be an efficient platform for the construction of fluorescent sensors for numerous heavy metal cations (such as Hg2+, Pb2+ and Cu2+) due to its large absorption coefficient and high fluorescence quantum yield.33–35 In the current work, this typical ion-recognition mechanism was utilized for the successive detection of trivalent ions and then phosphate ions via fluorescence OFF–ON–OFF switching.Fluorescence OFF–ON sensing for Fe3+, Al3+ and Cr3+
The detection selectivity of probe L (10 μM) toward Fe3+ ions was investigated by monitoring the fluorescence changes upon the addition of 8 equiv. chloride salts of a wide range of cations in CH3CN–Tris-buffer (1/1, v/v, pH 7.0) media, including Na+, K+, Mg2+, Cu2+, Hg2+, Cd2+, Fe2+, Pb2+, Zn2+, Mn2+, Ca2+, Fe3+, Al3+ and Cr3+. The fluorescence emission spectra of L were measured in 1.0 cm quartz cells, and the excitation and emission slits were set to 2.0 and 2.0 nm, respectively. As shown in Fig. 1, the sensor itself was almost non-fluorescent due to its ring-closed spirolactam structure. After the addition of Fe3+, probe L exhibited a color change from colorless to pink as well as a bright red fluorescence. A strong emission band centered at 586 nm was observed with an extreme fluorescence enhancement compared to the metal-free L. This result indicated that the probe L exhibited a high selectivity toward Fe3+ over the monovalent and divalent cations in CH3CN–Tris-buffer (1/1, v/v, pH 7.0) media.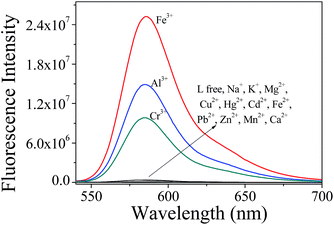 | ||
| Fig. 1 Fluorescence spectra of L (10 μM) in the presence of various metal ions (80 μM each) in CH3CN–Tris-buffer (1/1, v/v, pH 7.0), λex = 520 nm. | ||
To further explore the selectivity of the sensor, competitive ions were first added to the detection solution, and then Fe3+ ions were added after 20 minutes. As shown in Fig. 2, most of the detection systems exhibited minimum interference in the detection of Fe3+; Al3+ and Cr3+ (80 μM each) also did not induce any obvious interference in the fluorescence sensing of Fe3+, indicating that L has a relatively higher binding affinity to Fe3+ than Al3+ and Cr3+. In addition, the association constants were 6.13 × 104 for L–Fe3+, 3.14 × 104 for L–Al3+ and 2.26 × 104 for L–Cr3+, according to the reported work,44,45 respectively. A similar study was carried out with Al3+ and Cr3+, and there were no obvious interferences in the presence of Na+, K+, Mg2+, Cu2+, Hg2+, Cd2+, Fe2+, Pb2+, Zn2+, Mn2+ and Ca2+.
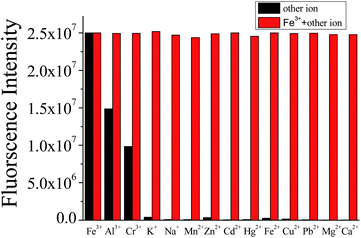 | ||
| Fig. 2 Change in fluorescence intensity of L (10 μM) in various mixtures of metal ions (the concentration of Fe3+ and other metal ions were all 80 μM). | ||
The sensing property of L was investigated in detail utilizing the Fe3+ cation. Fig. 3 displays the absorption spectrum changes of L upon Fe3+ addition. Sensor L exhibited almost no absorption peak in the visible wavelength due to the spirolactam form of molecule L. However, a new band centered at about 562 nm emerged upon the gradual addition of Fe3+, indicating the sensor L–Fe3+ complex formation and Fe3+-induced spirolactam ring-opening processes. Moreover, the titration solution displayed a characteristic color change of rhodamine derivatives from colorless to pink, indicating that probe L could serve as a “naked-eye” indicator for Fe3+ in CH3CN–Tris-buffer (1/1, v/v) media. With increasing amounts of Fe3+, the absorbance increased proportionally and levelled off when the concentration of Fe3+ reached 250 μM. When UV-vis titration measurements were carried out with Al3+ (Fig. S4†) and Cr3+ (Fig. S5†), similar changes were found to those of Fe3+, i.e., absorbance increased at first and then levelled off.
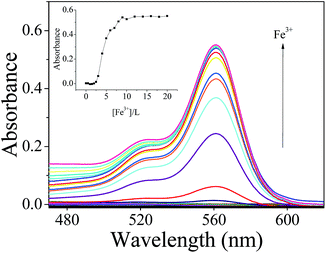 | ||
| Fig. 3 UV-vis spectra and (inset) absorbance changes (564 nm) recorded for L (25 μM) in CH3CN–Tris (1/1, v/v) with various amounts of Fe3+ ions. | ||
Appropriate pH conditions for the successful operation of the fluorescence sensing were evaluated. Without the addition of the Fe3+ ion, the ring opening of the RhB-based sensor L occurred under acidic conditions (pH < 5.0) due to protonation, while no fluorescence change was observed with pH values over 5 (Fig. 4). The gradual addition of Fe3+, however, led to an obvious fluorescence enhancement over a wide pH range from 5.0 to 8.0, which was attributed to the similar opening of the spirolactam structure.36 Because the most remarkable Fe3+-induced OFF–ON fluorescence changes occurred under the physiological pH window, all the fluorescence measurements were conducted at pH 7.0. Similar phenomenon can also be observed in the fluorescence sensing of Al3+ and Cr3+, as shown in Fig. S6.†
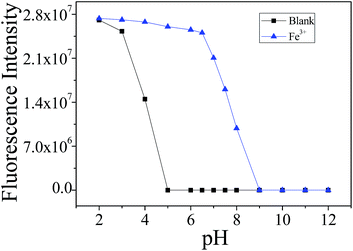 | ||
Fig. 4 Fluorescence intensities recorded for L (10 μM, CH3CN–H2O 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) at various pH values in the (a) absence and (b) presence of 8 equiv. Fe3+ (λex = 520 nm, λem = 586 nm). 1, v/v) at various pH values in the (a) absence and (b) presence of 8 equiv. Fe3+ (λex = 520 nm, λem = 586 nm). | ||
The relative affinities of Fe3+ toward sensor L were evaluated from fluorescence spectroscopic titration experiments in CH3CN–Tris-buffer (1/1, v/v) medium, as shown in Fig. 5. The concentration of L was maintained at 10 μM, while the concentration of Fe3+ was varied between 0 and 250 μM. The fluorescence spectra were recorded at an excitation wavelength of 520 nm and emission wavelength of 540–700 nm. For free L, no obvious characteristic emission of rhodamine derivatives was observed. With increasing concentrations of Fe3+, the fluorometric titration reaction curve showed a steady and smooth enhancement, which was used as the basis of Fe3+-sensing. The recognition interaction was completed immediately after the addition of Fe3+, i.e., within 1 min. When the concentration of Fe3+ was greater than 200 μM, the fluorescence intensity did not increase any further and a plateau was reached. The inset in Fig. 5 shows the dependence of fluorescence intensity at 586 nm on Fe3+ concentration. Plotting fluorescence intensity versus Fe3+ concentration (1–4 equiv.) afforded a good linear relationship (R = 0.9844) (Fig. S7†). The detection limit of Fe3+ was calculated from the equation DL = (3Sb1 − I)/S instead of DL = 3Sb1/S (ref. 37 and 38) because the intercept is not negligible, where Sb1 is the standard deviation of the blank solution, I is the intercept and S is the slope of the calibration curve. The detection limit of L for Fe3+ was 1.10 × 10−5 M. And the detection limits of L were 3.20 × 10−7 M (Fig. S8 and S9†) for Al3+ and 2.55 × 10−5 M for Cr3+ (Fig S10 and S11†).
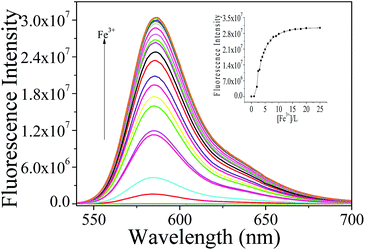 | ||
| Fig. 5 Fluorescence titrations of L (10 μM) with Fe3+ ions in CH3CN–Tris (1/1, v/v, pH 7.0). Inset: fluorescence emission intensity changes with increasing Fe3+ ions (λex = 520 nm, λem = 586 nm). | ||
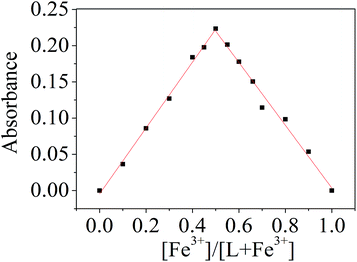
To determine the interaction stoichiometry between sensor L and Fe3+, Job's method was employed using an absorbance intensity at 562 nm as a function of molar fraction of L because the total concentration of L and Fe3+ ion was located at 50 μM. The maximum absorbance was observed when the molar fraction of L reached 0.50 (Fig. 6), which was indicative of a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry complexation between L and Fe3+. This result was further confirmed by high-resolution mass spectrum (HR-MS Fig. S12†), in which the peak at m/z 786.0831 (calcd = 786.0811) corresponding to [L + Fe3+ + 2H+ + 4Cl−]+ was clearly observed when 5 equiv. of FeCl3 was added to probe L. Similarly, the stoichiometric ratios were 1
1 stoichiometry complexation between L and Fe3+. This result was further confirmed by high-resolution mass spectrum (HR-MS Fig. S12†), in which the peak at m/z 786.0831 (calcd = 786.0811) corresponding to [L + Fe3+ + 2H+ + 4Cl−]+ was clearly observed when 5 equiv. of FeCl3 was added to probe L. Similarly, the stoichiometric ratios were 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 for Al3+ (Fig. S13†) and Cr3+ (Fig. S14†). As inferred by FTIR (Fig. S15†), the structure of the L–M3+ complex was proposed, as shown in Fig. S16.† When probe L was treated with Fe3+, the characteristic amide carbonyl absorption peak at 1631 cm−1 shifted to 1608 cm−1, indicating that the amide carbonyl O of rhodamine B unit was actually involved in the recognition of Fe3+, which also agreed with the previously reported work.46,47
1 for Al3+ (Fig. S13†) and Cr3+ (Fig. S14†). As inferred by FTIR (Fig. S15†), the structure of the L–M3+ complex was proposed, as shown in Fig. S16.† When probe L was treated with Fe3+, the characteristic amide carbonyl absorption peak at 1631 cm−1 shifted to 1608 cm−1, indicating that the amide carbonyl O of rhodamine B unit was actually involved in the recognition of Fe3+, which also agreed with the previously reported work.46,47
Fluorescence ON–OFF sensing for PO43−
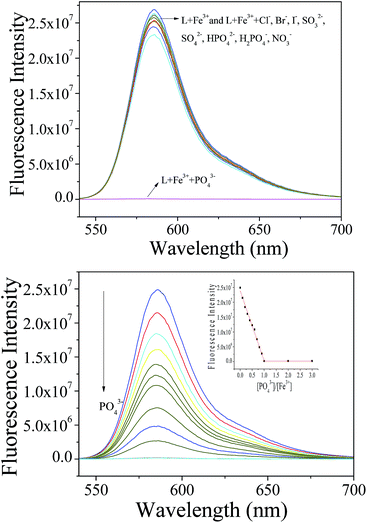
Interestingly, when the L–Fe3+ complex was treated with a sodium salt of PO43−, the color of the solution changed from pink to colorless, and the emission was completely quenched within 1 min (Fig. 7a), while other anions, such as Cl−, Br−, I−, SO42−, SO32−, HPO42−, H2PO4−, NO3−, did not show any interference to the detection. This observation suggested that the probe L could not only act as a sensor for Fe3+ but also for PO43− in a successive manner. Once the Fe3+ ion interacted with the sensor L, the spirolactum ring was opened, yielding a high fluorescence emission. However, when treated with the PO43− anion, the metal ion was abstracted and the spirolactum ring closed, leading to the absence of fluorescence. The PO43− anion-sensing capability of the obtained complex was further investigated in detail with fluorescence titration analysis (Fig. 7b). The titration curve showed an excellent linear decrease, and about 1.0 equiv. of PO43− (compared with Fe3+) was required to obtain absolute fluorescence quenching. The detection limit was calculated to be 0.20 μM (Fig. S17†) according to the fluorescence titration curve, which reveals a high sensitivity for the analysis of phosphate ion using the L-Fe3+ complex. The Al3+ complex can also serve as a sensor for PO43− via a simple displacement approach because of the strong affinity of PO43− to Al3+. Moreover, the cascade fluorescence OFF–ON–OFF response can be observed with the alternate addition of Fe3+ cation and PO43− anion into the detection solution (Fig. S18†), suggesting that the obtained fluorescence probe L can respond to both the cation and anion even when they co-exist in the solution. EDTA was further used to investigate the reversibility of the Fe3+ sensing process (Fig. S19†). The results indicate that EDTA could also cause an “ON–OFF” response of the L–Fe3+ complex via chelation. After the addition of excess Fe3+ to the detection solution, the recovered fluorescence emission can be clearly observed, suggesting that the probe exhibits good detection reversibility.Conclusions
In summary, we report the synthesis and characterization of a new probe for the cascade fluorogenic detection of trivalent ions and phosphate ions. The probe L showed excellent “OFF–ON” fluorescence signals with high sensitivity and selectivity in the presence of trivalent ions, whereas it remained silent in the presence of monovalent and divalent cations such as Na+, K+, Mg2+, Cu2+, Hg2+, Cd2+, Fe2+, Pb2+, Zn2+, Mn2+, Ca2+. A successive “ON–OFF” fluorescence switching was then observed in the presence of PO43− in CH3CN–Tris-buffer (1/1, v/v) media.Acknowledgements
The financial support from National Natural Scientific Foundation of China (NNSFC) Project (21204042), China Postdoctoral Science Foundation (2013T60099), National High Technology Research and Development Program of China (863 Program, 2012AA030312), Fundamental Research Project of Shenzhen (JCYJ20120830152316443) and Technology Innovation Program of Shenzhen (CXZZ20130322101824104) are gratefully acknowledged.Notes and references
- Y. Yang, Q. Zhao, W. Feng and F. Li, Chem. Rev., 2012, 113, 192–270 CrossRef PubMed.
- H. N. Kim, W. X. Ren, J. S. Kim and J. Yoon, Chem. Soc. Rev., 2012, 41, 3210–3244 RSC.
- L. Yuan, W. Lin, K. Zheng, L. He and W. Huang, Chem. Soc. Rev., 2013, 42, 622–661 RSC.
- X. Chen, T. Pradhan, F. Wang, J. S. Kim and J. Yoon, Chem. Rev., 2011, 112, 1910–1956 CrossRef PubMed.
- X. Wan, S. Yao, H. Liu and Y. Yao, J. Mater. Chem. A, 2013, 1, 10505–10512 CAS.
- J.-T. Hou, B.-Y. Liu, K. Li, K.-K. Yu, M.-B. Wu and X.-Q. Yu, Talanta, 2013, 116, 434–440 CrossRef CAS PubMed.
- Y. Lu, S. Huang, Y. Liu, S. He, L. Zhao and X. Zeng, Org. Lett., 2011, 13, 5274–5277 CrossRef CAS PubMed.
- A. Sahana, A. Banerjee, S. Lohar, B. Sarkar, S. K. Mukhopadhyay and D. Das, Inorg. Chem., 2013, 52, 3627–3633 CrossRef CAS PubMed.
- J. Mao, L. Wang, W. Dou, X. Tang, Y. Yan and W. Liu, Org. Lett., 2007, 9, 4567–4570 CrossRef CAS PubMed.
- S. H. Kim, H. S. Choi, J. Kim, S. J. Lee, D. T. Quang and J. S. Kim, Org. Lett., 2009, 12, 560–563 CrossRef PubMed.
- S. K. Sahoo, D. Sharma, R. K. Bera, G. Crisponi and J. F. Callan, Chem. Soc. Rev., 2012, 41, 7195–7227 RSC.
- K. Huang, H. Yang, Z. Zhou, M. Yu, F. Li, X. Gao, T. Yi and C. Huang, Org. Lett., 2008, 10, 2557–2560 CrossRef CAS PubMed.
- D. Maity and T. Govindaraju, Inorg. Chem., 2010, 49, 7229–7231 CrossRef CAS PubMed.
- W. Lin, L. Yuan, J. Feng and X. Cao, Eur. J. Org. Chem., 2008, 2008, 2689–2692 CrossRef.
- S. Goswami, A. Manna, S. Paul, K. Aich, A. K. Das and S. Chakraborty, Dalton Trans., 2013, 42, 8078–8085 RSC.
- P. Calvo, J. L. Barrio-Manso, F. C. García, J. L. Pablos, T. Torroba and J. M. García, Polym. Chem., 2013, 4, 4256–4264 RSC.
- J. Mao, Q. He and W. Liu, Talanta, 2010, 80, 2093–2098 CrossRef CAS PubMed.
- L. Yang, W. Yang, D. Xu, Z. Zhang and A. Liu, Dyes Pigm., 2013, 97, 168–174 CrossRef CAS PubMed.
- A. J. Weerasinghe, F. A. Abebe and E. Sinn, Tetrahedron Lett., 2011, 52, 5648–5651 CrossRef CAS PubMed.
- Y. Li, Z. Csók, P. Szuroczki, L. Kollár, L. Kiss and S. Kunsági-Máté, Anal. Chim. Acta, 2013, 799, 51–56 CrossRef CAS PubMed.
- Z.-C. Liao, Z.-Y. Yang, Y. Li, B.-D. Wang and Q.-X. Zhou, Dyes Pigm., 2013, 97, 124–128 CrossRef CAS PubMed.
- M. Yu, R. Yuan, C. Shi, W. Zhou, L. Wei and Z. Li, Dyes Pigm., 2013, 99, 887–894 CrossRef CAS PubMed.
- P. A. Gale, N. Busschaert, C. J. Haynes, L. E. Karagiannidis and I. L. Kirby, Chem. Soc. Rev., 2014, 43, 205–241 RSC.
- P. Saluja, N. Kaur, N. Singh and D. O. Jang, Tetrahedron Lett., 2012, 53, 3292–3295 CrossRef CAS PubMed.
- P. Saluja, N. Kaur, N. Singh and D. O. Jang, Tetrahedron Lett., 2012, 53, 3292–3295 CrossRef CAS PubMed.
- S. B. Maity and P. K. Bharadwaj, Inorg. Chem., 2013, 52, 1161–1163 CrossRef CAS PubMed.
- Y. Sun and S. Wang, Inorg. Chem., 2010, 49, 4394–4404 CrossRef CAS PubMed.
- S. He, S. T. Iacono, S. M. Budy, A. E. Dennis, D. W. Smith and R. C. Smith, J. Mater. Chem., 2008, 18, 1970–1976 RSC.
- G. V. Zyryanov, M. A. Palacios and P. Anzenbacher, Angew. Chem., Int. Ed., 2007, 46, 7849–7852 CrossRef CAS PubMed.
- X. Peng, Y. Xu, S. Sun, Y. Wu and J. Fan, Org. Biomol. Chem., 2007, 5, 226–228 CAS.
- S. Khatua, S. H. Choi, J. Lee, K. Kim, Y. Do and D. G. Churchill, Inorg. Chem., 2009, 48, 2993–2999 CrossRef CAS PubMed.
- V. Dujols, F. Ford and A. W. Czarnik, J. Am. Chem. Soc., 1997, 119, 7386–7387 CrossRef CAS.
- Z. Hu, C. Lin, X. Wang, L. Ding, C. Cui, S. Liu and H. Lu, Chem. Commun., 2010, 46, 3765 RSC.
- E. M. Nolan and S. J. Lippard, Chem. Rev., 2008, 108, 3443–3480 CrossRef CAS PubMed.
- X. Fang, S. Zhang, G. Zhao, W. Zhang, J. Xu, A. Ren, C. Wu and W. Yang, Dyes Pigm., 2014, 101, 58–66 CrossRef CAS PubMed.
- H.-S. Lv, S.-Y. Huang, B.-X. Zhao and J.-Y. Miao, Anal. Chim. Acta, 2013, 788, 177–182 CrossRef CAS PubMed.
- H. Wang, F. He, R. Yan, X. Wang, X. Zhu and L. Li, ACS Appl. Mater. Interfaces, 2013, 5, 8254–8259 CAS.
- C. R. Lohani, J.-M. Kim, S.-Y. Chung, J. Yoon and K.-H. Lee, Analyst, 2010, 135, 2079–2084 RSC.
- G. Zhang, B. Lu and Y. Wen, Sens. Actuators, B, 2012, 171, 786–794 CrossRef PubMed.
- Y. Zhou, J. Zhang, L. Zhang, Q. Zhang, T. Ma and J. Niu, Dyes Pigm., 2013, 97(1), 148–154 CrossRef CAS PubMed.
- S. Goswami, K. Aich, A. K. Das, A. Manna and S. Das, RSC Adv., 2013, 3(7), 2412–2416 RSC.
- A. Barba-Bon, A. M. Costero, S. Gil, M. Parra, J. Soto, R. Martínez-Máñez and F. Sancenón, Chem. Commun., 2012, 48(24), 3000–3002 RSC.
- A. KumaráDas and T. KumaráMondal, Chem. Commun., 2013, 49(91), 10739–10741 RSC.
- A. K. Mandal, M. Suresh, P. Das, E. Suresh, M. Baidya, S. K. Ghosh and A. Das, Org. Lett., 2012, 14(12), 2980–2983 CrossRef CAS PubMed.
- A. Liu, L. Yang, Z. Zhang, Z. Zhang and D. Xu, Dyes Pigm., 2013, 99(2), 472–479 CrossRef CAS PubMed.
- K. Tiwari, M. Mishra and V. P. Singh, RSC Adv., 2013, 3(30), 12124–12132 RSC.
- H. Liu, X. Wan, T. Liu, Y. Li and Y. Yao, Sens. Actuators, B, 2014, 200, 191–197 CrossRef CAS PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra03339f |
| ‡ These authors contributed equally to the work. |
| This journal is © The Royal Society of Chemistry 2014 |