Metal organic framework-laden composite polymer electrolytes for efficient and durable all-solid-state-lithium batteries
R. Senthil Kumara,
M. Rajab,
M. Anbu Kulandainathana and
A. Manuel Stephan*b
aElectro Organic Division, CSIR – Central Electrochemical Research Institute, Karaikudi, 630 006, India
bElectrochemical Power Sources Division, CSIR – Central Electrochemical Research Institute, Karaikudi, 630 006, India. E-mail: arulmanuel@gmail.com
First published on 28th May 2014
Abstract
A copper benzene dicarboxylate metal organic framework (Cu-BDC MOF) was synthesized and successfully incorporated in a poly(ethylene oxide) (PEO) and lithium bis(trifluoromethanesulfonylimide) (LiTFSI) complex. The incorporation of Cu-BDC MOF was found to significantly enhance the ionic conductivity, compatibility and thermal stability of the composite polymer electrolyte (CPE). An all-solid-state-lithium cell composed of Li/CPE/LiFePO4 was assembled, and its cycling profile has been analysed for different C-rates at 70 °C. The appreciable ionic conductivity, thermal stability and cycling ability qualify these membranes as electrolytes for all-solid-state-lithium batteries used in elevated temperature applications.
Introduction
Declining fossil fuel resources and global warming have motivated researchers to identify alternative energy sources.1 Although lithium-ion batteries remain the standard in portable electronic devices such as laptop computers, mobile phones etc., their safety is limited due to the use of non-aqueous liquid electrolytes with poor thermal stability, flammable reaction products and the leakage of electrolyte causing internal short-circuits.2 These problems can be circumvented by replacing the non-aqueous liquid electrolytes with solid polymer electrolytes, which possess several advantages including high energy density, no electrolyte leakage, flame resistance and flexible geometry.3 The development of polymer electrolytes has proceeded in two main directions: (i) dry solid polymer electrolytes; and (ii) gel and composite polymer electrolytes. The use of dry solid polymer electrolytes composed of a polar polymer host and a lithium salt is hampered by their poor ionic conductivity and rate capability at ambient temperatures. Gel polymer electrolytes, in contrast, exhibit appreciable ionic conductivities (of the order of 10−3 S cm−1 at 30 °C) and transport numbers. Upon plasticization, however, they lose their mechanical integrity, leading to a poor interface with the lithium metal anode.4 Recently, Zhu et al. intensively analysed gel polymer electrolytes based on nonwoven fabrics, glass fibre mats and PVdF/polyborate/PVdF trilayer membranes; these materials exhibited good mechanical strength and enhanced electrochemical properties along with being safe and low-cost.5–7 Xiao and co-workers introduced a novel environmentally friendly and less expensive cellulose-based gel polymer electrolyte for lithium-ion batteries that offered a higher lithium-ion transference number than the commercial separator.8 Saito et al.9 demonstrated a gel polymer electrolyte with incorporated Lewis acid ionic groups. The cycling performance of a calcium carbonate hard template-assisted three-dimensional macroporous polymer electrolyte was recently reported by Liu and co-workers.10 These studies reveal that composite polymer electrolytes alone can create safe and reliable lithium batteries.11 Generally, inorganic fillers (e.g., TiO2, SiO2, Al2O3) are incorporated in polymeric matrices to enhance the ionic conductivity and improve the thermal and mechanical properties. These fillers also substantially improve the interfacial properties between the electrolyte and the lithium metal anode. Numerous reports are available on the physical and electrochemical properties of composite polymer electrolytes for lithium batteries.12–16 Metal organic frameworks (MOFs), which are micro-porous solids comprising an infinite network of metal centres (or inorganic clusters) bridged by simple organic linkers through metal–ligand bonds, have attracted considerable attention from researchers.17–19 MOFs are widely used in catalysis, sensors, ion exchange, and gas storage, purification, separation and sequestration, and they are also widely employed to promote both electronic and proton conductivity.20 Generally, the incorporation of ceramic fillers grafted with organic groups with hybrid properties improves the miscibility with PEO to promote the ionic conductivity and mechanical integrity of the system21. Reports on MOF-laden composite polymer electrolytes for lithium battery applications are rare. In the present study, Cu-BDC MOF is successfully synthesized by an electrochemical method and is suitably incorporated in a PEO + LiTFSI complex. In addition, its physical and electrochemical properties are described.Experimental procedure
The synthetic procedure and structural characterization of Cu-BDC MOF have been reported elsewhere.22 PEO (Aldrich, USA) and lithium bistrifluorosulfonylimide (LiTFSI; Merck, Germany) were dried under vacuum for 2 days at 50 and 100 °C, respectively. Cu-BDC MOF was also dried under vacuum at 50 °C for 5 days before use. Composite polymer electrolytes were prepared by dispersing the appropriate amounts of Cu-BDC MOF in PEO–LiN(CF3SO2)2 (as shown in Table 1) and hot-pressing into films as described elsewhere.23 The composite electrolyte films had an average thickness of 30–50 μm. This procedure yielded homogeneous and mechanically strong membranes, which were dried under vacuum for 24 h at 50 °C for further characterization. The ionic conductivity of the membranes sandwiched between two stainless steel blocking electrodes (1 cm2 area) was measured using an electrochemical impedance analyser (IM6, Bio Analytical Systems) in the frequency range from 50 mHz and 100 kHz at various temperatures (0, 15, 30, 45, 60 and 70 °C). Symmetrical non-blocking Li/CPE/Li cells were assembled for compatibility, which was investigated by studying the time dependence of system impedance under an open-circuit potential at 70 °C. The lithium transference number was calculated by the method proposed by Vincent and co-workers:24
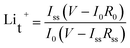 | (1) |
| S. no. | Sample Code | PEO (wt%) | Cu-BDC MOF (wt%) | LiTFSI (wt%) |
|---|---|---|---|---|
| 1 | S1 | 95 | 0 | 5 |
| 2 | S2 | 93 | 2 | 5 |
| 3 | S3 | 85 | 10 | 5 |
| 4 | S4 | 80 | 10 | 10 |
| 5 | S5 | 75 | 10 | 15 |
DSC and TG-DTA measurements were performed in a N2 atmosphere at a heating rate of 10 °C min−1 in the temperature ranges of −100 to +100 °C and 20 and 650 °C, respectively.
The LiFePO4/C cathode material was synthesized in the form of a nanosized powder via a mild hydrothermal procedure described by Meligrana et al.25 The composite cathode was prepared in the form of a film (average thickness approximately 70 μm) by thoroughly mixing 10 wt% of poly(vinylidene fluoride) binder (SolvaySolef 6020), 20 wt% of acetylene black (Shawinigan Black AB50, Chevron Corp., USA) electronic conductivity enhancer and 70 wt% of LiFePO4/C active material in 1-methyl-2-pyrrolidone (Aldrich, USA). The slurry was then coated onto an aluminium foil current collector. All preparations were performed in an argon-filled glovebox (MBraunLabstar, Germany) with a humidity content below 1 ppm. Lithium metal was used as the anode. The cycling of the cell was performed at 70 °C by an Arbin Instrument Testing System mode BT-2000 using cut-off voltages of 2.50–4.00 V vs. Li/Li+. The charge–discharge cycles were set to different current rates as reported previously.25
Results and discussion
Thermal analyses
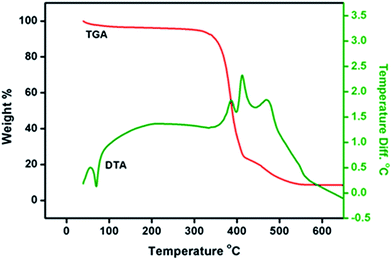
The DSC thermogram of the composite polymer electrolyte (sample S5 as this sample was found to be optimal in terms of ionic conductivity) is illustrated in Fig. 1.The glass transition temperature of PEO + LiTFSI increased (towards the positive side) from −54 to −50 °C upon addition of Cu-BDC MOF to the polymeric matrix. This observed increase in the value of Tg is attributed to (i) the effect of dispersed Cu-BDC MOF and (ii) the confinement of the intercalated/exfoliated polymer chains within the filler galleries, which resists the segmental motion of the polymer chains and indicates the plasticization effect that arises as a result of the mildly retarding effect that the Cu-BDC MOF filler has on crystallization.26 Fig. 2 depicts the TG-DTA traces of sample S5 (75% PEO + 10% Cu-BDC MOF + 15% LiTFSI).
Generally, the heating process creates a lot of changes in the composite electrolytes, finally leaving behind inert residues. A weight loss of approximately 3% attributed to the removal of moisture absorbed at the time of loading the sample was observed around 50 °C. The irreversible degradation of PEO typically begins at 190 °C,27 while the degradation of PEO + LiTFSI + Cu-BDC MOF starts at approximately 310 °C. The enhanced thermal stability of the Cu-BDC MOF-added composite electrolytes may be attributed to the intercalation/exfoliation of the polymer matrix with MOF particles, resulting in a strong barrier effect that prevents thermal degradation to a certain extent. This observation indicates that PEO + LiTFSI + Cu-BDC MOF is stable up to a temperature of 300 °C in a nitrogen atmosphere.28
Tensile strength
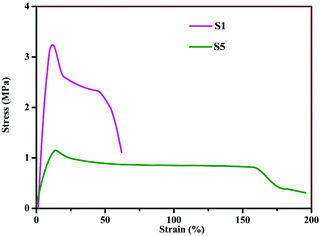
The stress–strain traces of sample S1 (95% PEO + 5% LiTFSI) and S5 (75% PEO + 10% Cu-BDC MOF + 15% LiTFSI) are displayed in Fig. 3. The tensile strength of sample S1 is 3.15 Mpa with an elongation-at-break value of 63%. Upon addition of 10% Cu-BDC MOF in the PEO + LiTFSI complexes, the elongation-at-break increased to 162%, while the mechanical strength decreased to 1.15 MPa. This reduction in mechanical strength arises from the plasticization of the PEO matrix caused by Cu-BDC MOF. A similar observation about the mechanical properties of succinonitrile-added PEO + LiTFSI complexes was reported by Fan and Maier29; however, the tensile strength of this composite polymer electrolyte is higher than that of MMT- modified carbon nanotube-incorporated PEO-based electrolytes.30Ionic conductivity and charge–discharge studies
The ionic conductivities of the composite polymer electrolytes with different proportions of PEO, LiTFSI and Cu-BDC MOF are depicted in Fig. 4 in an Arrhenius plot. Ionic conductivity increases with increasing temperature or Cu-BDC MOF content (samples S1–S5). The ionic conductivity varies from 10−6 to 10−4 S cm−1 for the Cu-BDC MOF-free sample (S1). In contrast, it varies from 10−6 to 10−3 S cm−1 with a 10% Cu-BDC MOF content. An increase in ionic conductivity of one order of magnitude was observed at 30 °C, while an increase of nearly two orders of magnitude was seen above 50 °C. The increase in ionic conductivity for the Cu-BDC MOF-added membrane (sample S5) was observed at 0 °C, which is far from the glass transition temperature of PEO.Interestingly, a sharp change in the conductivity pattern is observed above 50 °C: the knee disappears, suggesting that the melting point and reduction in the degree of crystallinity of PEO promotes salt dissolution.2 As commonly found in composite materials, the ionic conductivity is not a linear function of filler concentration. At low filler concentrations, the diffusion effect, which tends to depress conductivity, is effectively opposed by the specific interactions of the ceramic surfaces, which promote fast ion transport. At high filler concentrations, the dilution effect predominates, and conductivity is reduced.31
According to the NMR studies of Scrosati and co-workers,32 Lewis acid groups of the added inert filler may compete with the Lewis acid lithium cations as well as the anions of the added lithium salt for the formation of complexes with the alkoxide of PEO chains. Consequently, structural modifications to the filler surfaces occur, arising from specific actions of the polar surface groups of the inorganic filler. The Lewis acid–base interaction centres on the electrolytic species thus lowering, the ionic coupling and promoting the salt dissolution by the formation of a sort of “ion–filler complex”. In the present study, Cu-BDC MOF (filler), which has a Lewis acid centre, can react with the anions of the lithium salt, leading to a reduction in the crystallinity of the polymer host. Based on this type of Lewis acid–base interaction, Zheng et al. recently demonstrated the performance enhancement of lithium sulfur batteries by incorporating nickel II-MOF in a polysulfide base.33 Fig. 5 illustrates the interaction of PEO chains with Cu-BDC MOF and LiTFSI.
The lithium transference number, Lit+, is critical in the performance and rate capability of lithium batteries for high-power applications such as hybrid electric vehicles. In the present study, lithium transference number was calculated using eqn (1).
Fig. 6a and b show the chronoamperometric curves of samples S1 and S5, respectively. The insets show the Nyquist plots before and after perturbation. It can be seen that there is not much difference between the Nyquist plots (before and after perturbation), further confirming the stability of the lithium electrode with the Cu-BDC MOF-incorporated CPE. The value of Lit+ was calculated as 0.1 and 0.41 for the samples S1 and S5, respectively; the Lit+ value of sample S5 (0.41) is sufficient for low C-rate applications.34
 | ||
| Fig. 6 Chronoamperometric measurements for the samples (a) S1 and (b) S5. Insets show impedance spectra before and after perturbation. | ||
In order to ascertain the usefulness of this CPE, cycling studies were carried out in a 2032-type coin cell. Fig. 7 depicts the discharge capacity versus the cycle number of the Li/CPE/LiFePO4 cell at 70 °C. In the present study, LiFePO4 was chosen as the cathode material because of its appealing properties, including non-toxicity, thermal stability and environmental friendliness.
LiFePO4 is commonly the ultimate choice of cathode material for nanocomposite polymer electrolyte systems as it shows a flat operating voltage of 3.45 V vs. Li.25 The experimental cell delivered an initial discharge capacity of 132 mA h g−1 at C/20-rate and 126 mA h g−1 at C/10-rate without much fade in capacity. At 1 C-rate, the cell delivered 120 mA h g−1 with 98% columbic efficiency.
The cell is able to deliver a specific capacity of 31 mA h g−1 even at 5 C-rate. The reduction discharge capacity at higher C-rates is a typical characteristic of LiFePO4 that is attributed to its low electronic conductivity and the limited diffusion of Li+ ions into its structure, causing electrode polarization and the formation of a solid electrolyte interface.35
The declining discharge capacity at higher C-rates may also be due to the solid electrolyte interface (SEI) film formation with electrolyte decomposition. A recent study revealed that the increase in interfacial resistance originating from parameters related to the electrode's design, such as thickness and density, can cause capacity to fade at higher rates.36 It is also apparent from Fig. 7 that the specific capacity of a cell is restored at 1 C-rate by its 40th cycle, indicating retention of structural stability by the cathode material.
Conclusions
The incorporation of Cu-BDC MOF in a PEO + LiTFSI matrix is an effective way to enhance the ionic conductivity and thermal stability of composite polymer electrolytes. It also promotes the elongation-at-break of polymer electrolytes. A better cyclability has been achieved due to the higher ionic conductivity of the composite polymer electrolytes. The unique advantage of all-solid-state-lithium polymer cells over their liquid counterparts lies in their ability to function at higher temperatures without any safety issues. The Li/CPE/LiFePO4 cell is able to be cycled even at 70 °C, which is superior to liquid electrolytes and guarantees its safety.References
- J. M. Tarascon and M. Armand, Nature, 2001, 414, 359 CrossRef CAS PubMed.
- R. Bouchet, S. Maria, R. Meziane, A. Aboulaich, L. Lienafa, J. P. Bonnet, T. N. T. Phan, D. Bertin, D. Gigmes, D. Devaux, R. Denoyel and M. Armand, Nat. Mater., 2013, 12, 452 CrossRef CAS PubMed.
- A. Hommanmi, N. Raymond and M. Armand, Nature, 2003, 424, 635 CrossRef PubMed.
- A. Manuel Stephan, Eur. Polym. J., 2006, 42, 21 CrossRef CAS PubMed.
- Y. Zhu, F. Wang, L. Liu, S. Xiao, Y. Yang and Y. Wu, Sci. Rep., 2013, 3, 3187 Search PubMed.
- Y. Zhu, F. Wang, L. Liu, S. Xiao, Z. Chang and Y. Wu, Energy Environ. Sci., 2013, 6, 618 CAS.
- Y. Zhu, S. Xiao, Y. Shi, Y. Yang and Y. Wu, J. Mater. Chem. A, 2013, 1, 7790 CAS.
- S. Xiao, F. Wang, Y. Yang, Z. Chang and Y. Wu, RSC Adv., 2014, 4, 76 RSC.
- Y. Saito, M. Okano, T. Sakai and T. Kamada, J. Phys. Chem. C, 2014, 118, 6064 CAS.
- H. Y. Liu, L. L. Liu, C. L. Yang, Z. H. Li, Q. Z. Xiao, G. T. Lei and Y. H. Ding, Electrochim. Acta, 2014, 121, 328 CrossRef CAS PubMed.
- F. Croce, G. B. Appetecchi, L. Persi and B. Scrosati, Nature, 1998, 394, 456 CrossRef CAS PubMed.
- A. Manuel Stephan and K. S. Nahm, Polymer, 2006, 47, 5952 CrossRef PubMed.
- N. Angulakshmi, J. R. Nair, C. Gerbaldi, R. Bongiovanni, N. Pennazi and A. Manuel Stephan, Electrochim. Acta, 2013, 90, 179 CrossRef CAS PubMed.
- B. Kumar and S. J. Rodrigues, J. Electrochem. Soc., 2001, 148, A1336 CrossRef CAS PubMed.
- B. Kumar, L. Scanlon, R. Marsh, R. L. Mason, R. Higgins and R. Baldwin, Electrochim. Acta, 2001, 46, 1515 CrossRef CAS.
- J. H. Shin, F. Alessandrini and S. Passerini, J. Electrochem. Soc., 2005, 152, A283 CrossRef CAS PubMed.
- H. Hosseini, H. Ahmar, A. Dehghani, A. Bagheri, A. Tadjarodi and A. R. Fakhari, Biosens. Bioelectron., 2013, 42, 426 CrossRef CAS PubMed.
- J. Y. Yang, A. Grazeb, F. M. Mulder and T. J. Dinglmars, Microporous Mesoporous Mater., 2013, 171, 65 CrossRef CAS PubMed.
- L. J. Murray, M. Dinca and J. R. Long, Chem. Soc. Rev., 2009, 38, 1294 RSC.
- J. R. Li, J. Sculley and H. C. Zhou, Chem. Rev., 2012, 112, 869 CrossRef CAS PubMed.
- Y. X. Jiang, J. M. Xu, Q. C. Zhuang, L. Y. Jin and S. G. Sun, J. Solid State Electrochem., 2008, 12, 352 CrossRef PubMed.
- R. Senthil Kumar, S. Senthil Kumar and M. Anbu Kulandainathan, Electrochem. Commun., 2012, 25, 70 CrossRef CAS PubMed.
- A. Manuel Stephan, T. Prem Kumar, M. Anbu Kulandainathan and N. Angulakshmi, J. Phys. Chem. B, 2009, 113, 1963 CrossRef PubMed.
- J. Evans, C. A. Vincent and P. G. Bruce, Polymer, 1987, 28, 2324 CrossRef CAS.
- G. Meligrana, C. Gerbaldi, A. Tuel, S. Bodoardo and N. Pennazi, J. Power Sources, 2006, 160, 516 CrossRef CAS PubMed.
- Z. Guo, Z. Fang, L. Tong and Z. Xu, Polym. Degrad. Stab., 2007, 92, 545 CrossRef CAS PubMed.
- T. Shodai, B. B. Owens, M. Ostsuka and J. Yamaki, J. Electrochem. Soc., 1994, 141, 2978 CrossRef CAS PubMed.
- S. P. Thomas, S. Thomas and S. Bandyopadhyay, J. Phys. Chem. C, 2009, 113, 97 CAS.
- L. Z. Fan and J. Maier, Electrochem. Commun., 2006, 8, 1753 CrossRef CAS PubMed.
- C. Tang, K. Hackenberg, Q. Fu, P. M. Ajayan and H. Ardebili, Nano Lett., 2012, 12, 1152 CrossRef CAS PubMed.
- C. Yuan, F. Li, P. Han, Z. Zhang and J. Liu, J. Power Sources, 2013, 240, 653 CrossRef CAS PubMed.
- F. Crose, L. Perci, B. Scrosati, F. S. Fiory, E. Plichta and M. A. Hendrickson, Electrochim. Acta, 2001, 46, 2457 CrossRef.
- J. Zheng, J. Tian, D. Wu, M. Gu, W. Xu, C. Wang, F. Gao, M. Engelhard, J. G. Zhang, J. Liu and J. Xiao, Nano Lett., 2014, 14, 2345 CrossRef CAS PubMed.
- A. Fernicola, F. Croce, B. Scrosati, T. Watanabe and H. Ohno, J. Power Sources, 2007, 174, 342 CrossRef CAS PubMed.
- S. Brutti, J. Hassoun, B. Scrosati, C. Y. Lin, H. Wu and H. W. Hsieh, J. Power Sources, 2012, 217, 72 CrossRef CAS PubMed.
- J. H. Kim, S. C. Woo, M. S. Park, K. J. Kim, T. Yim, J. S. Kim and Y. J. Kim, J. Power Sources, 2013, 229, 190 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2014 |