Grafting of a chiral Mn(III) complex on graphene oxide nanosheets and its catalytic activity for alkene epoxidation†
Mohammad Ali Nasseri*,
Ali Allahresani and
Heidar Raissi
Department of Chemistry, Faculty of Science, University of Birjand, P. O. Box 97175-615, Birjand, Iran. E-mail: manaseri@birjand.ac.ir; Fax: +98-561-2502065; Tel: +98-561-2502065
First published on 3rd June 2014
Abstract
A chiral Mn(III) complex supported by a covalent grafting method on modified graphene oxide was synthesized using 3-chloropropyltrimethoxysilane as a reactive surface modifier. The heterogeneous catalyst was characterized by X-ray diffraction, Fourier transform infrared spectra, thermogravimetric analysis, ultraviolet-visible spectra, nitrogen adsorption–desorption isotherms, transmission electron microscopy and atomic absorption spectroscopy. The catalytic potential of the heterogeneous catalyst and comparison with its homogeneous counterpart were studied for enantioselective epoxidation of various alkenes using m-chloroperbenzoic acid as an oxidant and it showed high selectivity and comparable catalytic reactivity with its homogeneous analogue. In addition, higher enantioselectivity and higher yield were observed in the presence of 4-methylmorpholine N-oxide and pyridine N-oxide, respectively. The catalyst was reused for several runs without significant loss of activity and selectivity.
Introduction
The asymmetric epoxidation of alkenes into unique labile three-membered ether rings, which are useful organic building blocks for the synthesis of pharmaceuticals, agrochemicals and fine chemicals, is one of the fundamental organic transformations.1–4 From a synthesis point of view, the selective insertion of one oxygen atom into organic compounds, under mild conditions, remains a difficult challenge in chemical catalysis. Chiral Mn(III) salen complexes, first reported by Jacobsen and Katsuki groups, as excellent catalysts have emerged as extremely efficient systems for the asymmetric epoxidation of unfunctionalized olefins.5,6 Currently, much attention is focused on the heterogenization of Mn(III) complexes from drawbacks such as difficult recovery and recycling of the catalysts, product contamination and deactivation of homogeneous catalysts through the formation of dimeric peroxo and m-oxo species.7 To overcome these drawbacks, heterogeneous Mn(III) complexes prepared by immobilizing the homogeneous analogue onto insoluble solid supports such as mesoporous materials, zeolites, polymer supports, organic polymer-inorganic hybrid materials, clays, MCM-41, SBA-15 and activated silica have been developed.8–17 Unfortunately, despite their excellent performance in separation and reuse, immobilization often decreased enantioselectivities or efficiencies of heterogeneous Mn(III) complexes than homogenous counterparts.18 Therefore, novel and effective heterogeneous chiral Mn(III) catalysts for asymmetric epoxidation of various olefins are highly desirable.Compared to mesoporous silicates such as MCM-41 and SBA-15, graphene oxide (GO) has unique nanostructure (monolayer) and hydrophilic nature owing to the wide range of oxygen carrying functional groups on its basal planes and edges.19 These functional groups allow GO to be readily functionalized with various surfactants, polymeric materials, and nanoparticles in order to provide enormous potential for applications in materials science and engineering. In addition, some studies have demonstrated that immobilization of transition metal nanoparticles on GO nanosheets produced efficient catalysts.20–23 In this study, we report design and characterization of the covalent attachment of homogeneous Mn(III) complex onto GO and its catalytic properties in the epoxidation of alkenes using m-CPBA as an oxidant.
Results and discussion
Characterization of the heterogeneous catalyst
Synthesis of the heterogeneous Mn(III) complex and the intermediate steps were examined and monitored by XRD, FT-IR, TEM, TGA, UV-vis, AAS and nitrogen adsorption at room temperature.3-Chloropropylterimethoxysilane which plays the role of coupling agent, merely providing a large surface to anchor the active complex and increase the efficiency of heterogeneous catalyst was used for anchoring the homogeneous Mn(III) catalyst onto GO.24–26
FT-IR analysis
The FT-IR spectrum of GO (Fig. 1a) shows a broad peak appeared at 3446 cm−1, assigned to the stretching mode of an O–H bond, reveals the abundance of hydroxyl groups in GO. The strong band at 1716, 1619, 1224 and 1059 cm−1 revealed the presence of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, C
O, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, C–OH and C–O (epoxy) functional groups in GO. Fig. 1b which is assigned to CPGO after reaction with 3-chloropropyltrimethylsilane shows an obvious decrease in intensities of peaks such as 3446, 1728 and 1395 cm−1 which are corresponding to the OH, C
C, C–OH and C–O (epoxy) functional groups in GO. Fig. 1b which is assigned to CPGO after reaction with 3-chloropropyltrimethylsilane shows an obvious decrease in intensities of peaks such as 3446, 1728 and 1395 cm−1 which are corresponding to the OH, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibration and O–H deformation peak, respectively. Also, the bands at 2880 and 2920 cm−1 are corresponding to the aliphatic C–H bonds. The IR spectra of the ligand salen showed a strong band at 1614 cm−1 which is assigned to stretching vibration of C
O stretching vibration and O–H deformation peak, respectively. Also, the bands at 2880 and 2920 cm−1 are corresponding to the aliphatic C–H bonds. The IR spectra of the ligand salen showed a strong band at 1614 cm−1 which is assigned to stretching vibration of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N group (Fig. 1e). This band shifts to lower position (30 cm−1) in homogeneous Mn(III) complex (B) that suggests coordination of Mn through C
N group (Fig. 1e). This band shifts to lower position (30 cm−1) in homogeneous Mn(III) complex (B) that suggests coordination of Mn through C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N functional groups (Fig. 1d). The new stretching vibration bands at 2930, 2880, 1580 and 1190 cm−1 suggest the grafting of homogeneous Mn(III) complex on the CPGO (Fig. 1c).
N functional groups (Fig. 1d). The new stretching vibration bands at 2930, 2880, 1580 and 1190 cm−1 suggest the grafting of homogeneous Mn(III) complex on the CPGO (Fig. 1c).
 | ||
| Fig. 1 The FT-IR spectra of (a) GO (b) CPGO (c) heterogeneous catalyst (d) Mn(III) complex and (e) salen. | ||
XRD analysis
Fig. 2 shows XRD patterns of GO, CPGO and heterogeneous catalyst. Upon oxidation of graphite, the basal reflection (002) peak appeared at 2θ = 26.6° (d spacing = 0.335 nm)27 shifts to a lower angle (2θ = 11.79°, d spacing = 0.79 nm) Fig. 2(a). The increase in d spacing is due to the intercalation of water molecules and the formation of oxygen-containing functional groups between the layers of the graphite. By immobilization of CPGO, a broad peak appeared at 2θ = 15–27° depending on amorphous silica (Fig. 2b). The decrease in the intensity of 002 reflection peak of GO and the appearance of a broad peak appeared at 2θ = 15–27° in the CPGO indicated the immobilization of 3-chloropropyltrimethoxysilane on the GO. By immobilization of the homogeneous Mn(III) complex on graphene oxide, 002 reflections peak of GO was completely disappeared (Fig. 2c).Thermogravimetric analysis
The TGA curve of pure GO, CPGO and heterogeneous catalyst are shown in Fig. 3. A small mass loss (3 wt%) at ca. 100 °C is attributed to the loss of adsorbed water in the TGA curve of GO (Fig. 3a). Two more degradation steps were observed in GO; the first commencing at 165–250 °C, due to the loss of hydroxyl, epoxy functional groups and remaining water molecules and the second degradation step (250–650 °C) depend on the pyrolysis of the remaining oxygen-containing groups as well as the burning of ring carbon. In the TGA curve of CPGO a weight loss over a wide range of temperature (170–600 °C) was observed (Fig. 3b). In the TGA curve of heterogeneous catalyst three stepwise weight losses are observed. The first step with a weight loss of 4.59% at 60–170 °C is attributed to the loss of the surface or interstitial water. In the temperature range of 170–600 °C, a weight loss of 33% is corresponded to the decomposition of the appended organic groups. In comparison, a 42.5% weight loss is experienced by pure GO up to 600 °C. The thermogram of the heterogeneous catalyst shows that the heterogeneous catalyst is stable.27,28 The loading of Mn(III) complex is 0.31 mmol g−1 based on the difference in the weight losses for GO and heterogeneous catalyst. The molar ratio of ligand to metal in homogeneous Mn(III) catalyst is 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 in immobilized complex catalyst, indicating that the molar ratio of Mn element is 0.31 mmol g−1. The content of Mn is 0.35 mmol g−1 estimated by AAS, respectively. Moreover, these results suggest that metal complex has been anchored onto CPGO.
1 in immobilized complex catalyst, indicating that the molar ratio of Mn element is 0.31 mmol g−1. The content of Mn is 0.35 mmol g−1 estimated by AAS, respectively. Moreover, these results suggest that metal complex has been anchored onto CPGO.
UV-vis analysis
The UV-vis spectrum of GO shows a strong absorption peak at 230 nm and a shoulder at about 300 nm, which are attributed to the π → π* transition of graphitic C–C bonds and to the n → π* transitions of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds, respectively (Fig. 4a). For CPGO, all absorption peaks are disappeared and a new absorption peak appeared at 280 nm which is attributed to Si (Fig. 4b). The UV-vis spectrum of heterogeneous catalyst (Fig. 4c) can provide evidence for the immobilization of Mn(III) complex onto the support. All the characteristic bands appeared in its spectrum (325, 380 and 490 nm) indicated that an interaction took place between the homogeneous Mn(III) complex and the CPGO.
O bonds, respectively (Fig. 4a). For CPGO, all absorption peaks are disappeared and a new absorption peak appeared at 280 nm which is attributed to Si (Fig. 4b). The UV-vis spectrum of heterogeneous catalyst (Fig. 4c) can provide evidence for the immobilization of Mn(III) complex onto the support. All the characteristic bands appeared in its spectrum (325, 380 and 490 nm) indicated that an interaction took place between the homogeneous Mn(III) complex and the CPGO.
TEM analysis
The high magnification TEM image of GO exhibits a completely amorphous and disordered structure (Fig. 5). Fig. 6 shows the nanoscopic features of the heterogeneous Mn(III) complex with amorphous nature.The BET specific surface area of the heterogeneous catalyst which is determined at room temperature by nitrogen physisorption was 18.6 m2 g−1. The loading of homogeneous Mn(III) complex (B) in the heterogeneous catalyst was 0.35 mmol g−1 based on Mn element analysis by AAS.
Catalyst activity
The catalytic activity of heterogeneous chiral Mn(III) complex was studied for the oxidation of alkenes by using m-CPBA as the oxygen source. The catalytic tests for the heterogeneous chiral Mn(III) complex was compared with homogeneous Mn(III) complex under identical reaction conditions. In this content, α-methylstyrene was chosen as model substrate to investigate the activity of heterogeneous chiral Mn(III) complex. Various olefins such as cyclic and linear terminal olefins were tested by this catalyst and in all cases high catalytic activity and selectivity were observed. To optimize the amount of catalyst, the reaction was carried out in the presence of different amounts of heterogeneous chiral Mn(III) complex (0.5–3 mol% based on Mn element) at 0 °C. It was found that by increasing the amount of catalyst from 0.5 to 2 mol% the yields were increased from 30 to 93% and additional increasing the amount of catalyst did not show any significant effect on the conversion of the products (Table 1, entry 4). For comparison, the homogeneous chiral Mn(III) complex (B) was also investigated under the same reaction conditions, but shortening the reaction time to 3 h (Table 1, entry 8). Blank experiment showed that the heterogeneous chiral Mn(III) complex alone is inactive towards epoxidation of α-methylstyrene (Table 1, entry 7).| Entry | Catalyst mol% | Yieldb (%) |
|---|---|---|
| a Reaction conditions: α-methylstyrene (1 mmol), m-CPBA (2 mmol) in 3 ml EtOAC. The reactions were run at 0 °C for 6 h except for entry 7 which was run for 12 h. The reaction was sonicated for 20 min before addition of oxidant.b Determined by GC with a CBP1 column (Shimadzu 30 m × 0.32 mm × 0.25 mm). | ||
| 1 | 0.5 | 30 |
| 2 | 1 | 45 |
| 3 | 1.5 | 70 |
| 4 | 2 | 93 |
| 5 | 2.5 | 94 |
| 6 | 3 | 96 |
| 7 | No catalyst | 8 |
| 8 | Homogeneous chiral Mn(III) complex | 96 |
The effect of different solvents in the epoxidation of α-methylstyrene
The choice of solvent is crucial in the asymmetric epoxidation of olefins. In this study, a systematic examination of the solvent nature was performed in different solvents, namely acetonitrile, ethyl acetate, tetrahydrofuran, dichloromethane, chloroform, methanol, ethanol, ethanol–water and water, using 2 mol% of heterogeneous Mn catalyst (Fig. 7). The best enantioselectivity and moderate yield were obtained in ethyl acetate at 0 °C. | ||
| Fig. 7 The effect of different solvents on the epoxidation of α-methylstyrene (1 mmol) using 2 mmol m-CPBA in 3 ml EtOAC catalyzed by heterogeneous Mn(III) complex (2 mol% based on Mn) at 0 °C. | ||
The effect of different oxidants and axial additives on the asymmetric epoxidation of α-methylstyrene
The effect of various oxidants such as H2O2, NaIO4, t-BuOOH, m-CPBA, UHP, PhIO, PhI(OAC)2 and oxone are investigated in the asymmetric epoxidation of α-methylstyrene. To choose the best oxygen source, suitable axial additive (PNO) was added to the reaction. As the results show in Fig. 8, m-CPBA was the best oxygen source for alkenes oxidation in the presence of heterogeneous chiral Mn(III) complex. | ||
| Fig. 8 The effect of different oxidants on the epoxidation of α-methylstyrene (1 mmol) using 2 mmol oxidant in 3 ml EtOAC catalyzed by heterogeneous Mn(III) complex (2 mol% based on Mn) at 0 °C. | ||
Then, the effect of different additives was studied (Fig. 8). In the epoxidation reactions, despite the conversion increase in the presence of PNO, higher ee was observed in the presence of MNO as an additive (Fig. 9). When NH4OAC is used in EtOAC, low yield (15%) with only 30% ee was obtained (Fig. 9).
 | ||
| Fig. 9 The effect of different additives on the epoxidation of α-methylstyrene (1 mmol) using 2 mmol m-CPBA in 3 ml EtOAC catalyzed by heterogeneous Mn(III) complex (2 mol% based on Mn) at 0 °C. | ||
In addition, different amounts of oxidants were tested, and the results showed that the higher catalytic activity was obtained with 2 equivalents of the oxidant (Fig. 10). Consequently, the optimum molar ratio of olefin to oxidant is 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.
2.
 | ||
| Fig. 10 The effect of different amount of m-CPBA on the epoxidation of α-methylstyrene (1 mmol) in 3 ml EtOAC catalyzed by heterogeneous Mn(III) complex (2 mol% based on Mn) at 0 °C. | ||
We also performed the model reaction at different temperatures in EtOAC in the presence of heterogeneous catalyst (−78, 25, 40 and 60 °C) (Table 2, entries 1 and 3–5). Temperature plays an important role in the yield and ee of the reaction. There was a decrease in the yield at −78 °C and no significant change in the ee values (Table 2, entry 1). The reaction proceeded faster (4 h) at 25 °C compared with 0 °C and gave higher TOF (11.5–12.1 h−1). It is notable that a drop in the selectivity was not observed when the reaction carried out at 25 °C. However, when temperature is higher than room temperature (40 and 60 °C), an obvious decreasing in enantioselectivity was observed (Table 2, entries 4 and 5).
| Entry | T (°C) | Yieldb (%) | ee (%) |
|---|---|---|---|
| a Reaction conditions: α-methylstyrene (1 mmol), m-CPBA (2 mmol), catalyst (2 mol% based on Mn) in 3 ml EtOAC. The reactions were run for 6 h for entries 1 and 2 and 4 h for entries 3–5.b Determined by GC with a CBP1 column (Shimadzu 30 m × 0.32 mm × 0.25 mm). | |||
| 1 | −78 | 55 | 92 |
| 2 | 0 | 93 | 90 |
| 3 | 25 | 95 | 84.6 |
| 4 | 40 | 97 | 65 |
| 5 | 60 | 99 | 40 |
Under optimized conditions, the catalytic activity of heterogeneous chiral Mn(III) complex was investigated for the asymmetric epoxidation of various olefins using m-CPBA. Table 2 shows that the heterogeneous chiral Mn(III) complexes effectively catalyze the epoxidation of various olefins. Conditions used: injector temperature, 220 °C; detector temperature, 250 °C, injection volume, 0.2 μl, and column, isothermal 90 °C for styrene and 80 °C for α-methyl styrene. The following formulas were used for calculation of TON, TOF and % ee: TON = mmol of converted alkene per mmol Mn; TOF = TON per time of reaction; and % ee = [A (major enantiomer) − A (minor enantiomer)] × 100/[A (major enantiomer) + A (minor enantiomer)] where A stands for the area of the chromatographic peak.
The performed study on the catalytic transformation of alkenes by m-CPBA in the presence of heterogeneous chiral Mn(III) complex showed that the system with the heterogeneous catalyst exhibits high catalytic efficiency with TON (42.5–46.5). Also, the catalytic activity of heterogeneous chiral Mn(III) complex showed good catalytic performance reaching a TOF of 7.1–7.8 h−1 at conversion of 85–95% and selectivity higher than 98% in the epoxidation of different alkenes (Table 3). The catalytic activity of heterogeneous chiral Mn(III) complex was also investigated for the asymmetric epoxidation of various olefins using m-CPBA at 25 °C. In all the cases, the oxidation reactions were done faster (4 h) at 25 °C compared with 0 °C and gave higher TOF (11.5–12.1 h−1) (Table 3).
| Entry | Alkene | T (°C) | Time (h) | Selectivityb (%) | Yieldc (%) | eed (%) | TON | TOF (h−1) |
|---|---|---|---|---|---|---|---|---|
| a Reaction conditions: substrate (1 mmol), DCM (3 ml), oxidant (2 mmol) and catalyst (2 mol% based on Mn).b Selectivity based on alkenes conversion.c Determined by GC with a Shimadzu CBP5 column (30 m × 0.32 mm × 0.25 mm).d ee% was performed by GC with a Agilent HP column (19091G-B213, 30 m × 0.25 mm × 0.25 μm). | ||||||||
| 1 | 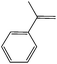 |
0 | 6 | 99 | 93 | 90 | 46.5 | 7.75 |
| 25 | 4 | 98 | 95 | 84.6 | 48 | 12.1 | ||
| 2 | 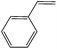 |
0 | 6 | 98 | 90 | 65 | 45 | 7.5 |
| 25 | 4 | 97 | 93 | 61.5 | 46.5 | 11.6 | ||
| 3 | 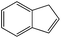 |
0 | 6 | 99 | 92 | 85.1 | 46 | 7.66 |
| 25 | 4 | 99 | 95 | 80.2 | 47.5 | 11.87 | ||
| 4 | 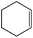 |
0 | 6 | 99 | 90 | 55.6 | 46 | 7.66 |
| 25 | 4 | 98 | 94 | 55 | 47 | 7.8 | ||
| 5 | 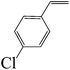 |
0 | 6 | 99 | 92 | 60.9 | 46 | 7.66 |
| 25 | 4 | 98 | 96 | 58.6 | 48 | 12.0 | ||
| 6 | 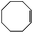 |
0 | 6 | 98 | 85 | 53 | 42.5 | 7.1 |
| 25 | 4 | 98 | 90 | 50 | 46 | 11.5 | ||
| 7 | 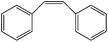 |
0 | 6 | 99 | 94 | 86 | 48 | 8.0 |
| 25 | 4 | 99 | 95 | 80 | 48.5 | 12.1 | ||
| 8 | 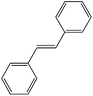 |
0 | 6 | 99 | 95 | 88 | 44 | 7.33 |
| 25 | 4 | 99 | 96 | 84.7 | 48 | 12.0 | ||
| 9 |  |
0 | 6 | 99 | 90 | 62 | 45 | 7.5 |
| 25 | 4 | 98 | 93 | 60 | 46.5 | 11.62 | ||
Reusability of the catalyst
To access the recyclability of the heterogeneous chiral Mn(III) complex, the heterogeneous catalyst was completely washed by ethanol and dichloromethane and used in repeated epoxidation reactions with α-methylstyrene by adding fresh reactants. In each run, no manganese was detected in the filtrates by AAS. As shown in Fig. 11, it is obvious that the catalyst worked well for up to five cycles with no considerable decrease in reactivity and enantioselectivity (conversion: from 93% to 88%; ee: from excess 90% to 87%).Conclusions
Chiral Mn(III) salen complex was anchored onto CPGO and characterized by XRD, FT-IR, UV-vis and nitrogen adsorption at room temperature. The catalyst exhibited high catalytic activity and selectivity towards the epoxidation of olefins. Catalytic tests showed that the heterogeneous Mn(III) complex is the most active with m-CPBA as the oxidant in EtOAC. Moreover, the heterogeneous Mn(III) complex showed higher yield and enantioselectivity with PNO and MNO, respectively. The catalyst can be recovered and reused five runs without any significant decrease in reactivity.Experimental
Materials
Tetraethoxysilane (TEOS), FeCl3·6H2O, FeCl2·4H2O, styrene, 4-chlorostyrene, cyclohexene, α-methyl styrene, indene, cis & trans stilbene, m-chloroperbenzoic acid (m-CPBA), pyridine N-oxide (PNO), pyridine (Py), 4-methylmorpholine N-oxide (MNO), 1-methyl imidazole (MI), imidazole (IM), dichloromethane (DCM), ethyl acetate (EtOAC), ethanol (EtOH), H2O2, NaIO4, NH4OAC, t-BOOH, PhI(OAC)2 and oxone were purchased from Merck Company and used without purification. Graphite powder was obtained from Aldrich. The resulting GO and heterogeneous chiral Mn(III) complex (C) were characterized by X-ray diffraction (XRD), Fourier transform infrared spectra (FT-IR), thermogravimetric analysis (TGA), ultraviolet-visible spectra (UV-vis), transmission electron microscopy (TEM), nitrogen adsorption–desorption isotherm and Atomic absorption spectroscopy (AAS). The FT-IR experiments were carried out on a Bruker Vector-22 FT-IR spectrometer (AVATR-370) in a KBr pellet. The XRD measurements were carried out by using a Bruker D8-advance X-ray diffractometer with Cu Kα radiation (k = 1.5406 Å). The TEM measurements were obtained using Philips CM10 instrument. The UV-vis spectra were carried out by using a Shimadzu UV-160A spectrophotometer. The surface area was carried out by N2 adsorption using Quanta chrome (Chem. Bet-3000). TGA analysis was performed by heating the samples in an Argon flow at a rate of 100 ml min−1 using a Perkin-Elmer Diamond TG/DTA thermal analyzer with a heating rate of 10 °C min−1. The Mn content of the catalysts was determined by Shimadzu AA-6300 Atomic absorption spectroscopy (AAS). The yields were determined by Shimadzu GC-17A gas chromatograph (GC) equipped with a flame ionization detector with a capillary column (CBP-1, 30 m × 0.25 mm × 0.25 μm), using helium as carrier gas. The enantioselectivity was determined in a chiral GC column (HP 19091G-B213, 30 m × 0.25 mm × 0.25 μm).Synthesis of 3-chloropropyltrimethoxysilane functionalized GO (CPGO, A)
GO was prepared as described in the literature.27,30 Brown powder of GO (1 g) was dispersed in 70 ml of dry toluene for 20 min followed by dropwise addition of 3 ml of 3-chloropropyltrimethoxysilane, which was diluted in 20 ml dry toluene.29 The resulting mixture was heated to reflux with continuous stirring for 24 h under nitrogen atmosphere (Fig. 12). After cooling the mixture, the black powdery sample was filtered, washed with toluene four times and then washed with EtOH. The solid was dried at 70 °C under vacuum for 6 h.Synthesis of Mn(III) complex immobilized on CPGO
(R,R)-N,N′-Bis(2,4-di-hydroxybenzaldehyde)-1,2-cyclohexanediamine31 and Mn(III) salen complex32 were prepared as described in the literature. The synthesized Mn(III) complex (B) was chemically anchored to the CPGO (A) as described in Fig. 1.19 Briefly, CPGO (2 g) was dispersed in 140 ml dry toluene for 20 min. Then, 0.6 g of homogeneous Mn(III) complex was added to the stirring mixture. The resulting suspension was refluxed for 24 h under inert atmosphere. After completion of the reaction, the immobilized catalyst was washed with dry toluene, EtOH and extracted repeatedly on a Soxhlet extractor with methanol and dichloromethane until the washing becomes colorless. The solid was dried at 70 °C under vacuum for 6 h. The immobilized chiral Mn(III) complex was characterized by XRD, TEM, UV-vis, FT-IR, AAS, TGA and nitrogen adsorption at room temperature.Catalytic measurements
In a typical procedure, catalyst (2 mol% based on Mn element) was dispersed in 3 ml of ethyl acetate for 20 min and cooled to 0 °C followed by addition of α-methyl styrene as substrate (1 mmol), toluene (internal standard, 40 μl), m-CPBA (2 mmol) as oxidant and MNO (0.5 mmol) as additive. The mixture was magnetically stirred at 0 °C for appropriate time. The progress of the reaction was monitored by TLC. After completion of the reaction, the catalyst was separated by centrifugation. The supernatant was washed with NaOH (1 M, 8 ml), brine (8 ml), dried over MgSO4 and concentrated to 1 ml. Finally, the conversion and enantioselectivity values of products were determined by GC. The catalyst was washed twice with ethanol and reused.Acknowledgements
The authors are grateful to the University of Birjand for financial support.References
- Q. H. Fan, Y. M. Li and A. S. C. Chan, Chem. Rev., 2002, 102, 3385–3465 CrossRef CAS PubMed.
- L. Canali and D. C. Sherrington, Chem. Soc. Rev., 1999, 28, 85–93 RSC.
- W. Zhang, J. L. Loebach, S. R. Wilson and E. N. Jacobsen, J. Am. Chem. Soc., 1990, 112, 2801–2803 CrossRef CAS.
- W. Zhang and E. N. Jacobsen, J. Org. Chem., 1991, 56, 2296–2298 CrossRef CAS.
- W. Zhang, J. L. Loebach, S. R. Wilson and E. N. Jacobsen, J. Am. Chem. Soc., 1990, 112, 2801–2803 CrossRef CAS.
- R. Irie, K. Noda, Y. Ito, N. Matsumoto and T. Katsuki, Tetrahedron Lett., 1990, 31, 7345–7348 CrossRef CAS.
- K. J. Balkus Jr, A. Khanmamedova, K. Dixon and F. Bedioui, Appl. Catal., A, 1996, 143, 159–173 CrossRef.
- H. Zhang, Y. M. Wang, L. Zhang, G. Gerritsen, H. C. L. Abbenhuis, R. A. Van Santen and C. Li, J. Catal., 2008, 256, 226–236 CrossRef CAS PubMed.
- R. I. Kureshy, I. Ahmad, N. H. Khan, S. H. R. Abdi, K. Pathak and R. V. Jasra, J. Catal., 2006, 238, 134–141 CrossRef CAS PubMed.
- R. I. Kureshy, I. Ahmad, N. H. Khan, S. H. R. Abdi, K. Pathak and R. V. Jasra, Tetrahedron: Asymmetry, 2005, 16, 3562–3569 CrossRef CAS PubMed.
- H. Zhang, S. Xiang, J. Xiao and C. Li, J. Mol. Catal. A: Chem., 2005, 238, 175–184 CrossRef CAS PubMed.
- V. Mirkhani, M. Moghadam, S. Tangestaninejad, B. Bahramian and A. Mallekpoor-Shalamzari, Appl. Catal., A, 2007, 321, 49–57 CrossRef CAS PubMed.
- K. Smith and C. H. Liu, Chem. Commun., 2002, 886–887 RSC.
- X. C. Zou, X. K. Fu, Y. D. Li, X. B. Tu, S. D. Fu, Y. F. Luo and X. J. Wu, Adv. Synth. Catal., 2010, 352, 163–170 CrossRef CAS.
- I. Kuzniarska-Biernacka, C. Pereira, A. P. Carvalho, J. Pires and C. Freire, Appl. Clay Sci., 2011, 53, 195–203 CrossRef CAS PubMed.
- K. Yu, Z. C. Gu, R. N. Ji, L. L. Lou and S. X. Liu, Tetrahedron, 2009, 65, 305–311 CrossRef CAS PubMed.
- S. Sahoo, P. Kumar, F. Lefebvre and S. B. Halligudi, Appl. Catal., A, 2009, 354, 17–25 CrossRef CAS PubMed.
- D. A. Annis and E. N. Jacobsen, J. Am. Chem. Soc., 1999, 121, 4147–4154 CrossRef CAS.
- T. Szabo, E. Tombacz, E. Illes and I. Dekany, Carbon, 2006, 44, 537–545 CrossRef CAS PubMed.
- M. Zhou, A. Zhang, Z. Dai, C. Zhang and Y. P. Feng, J. Chem. Phys., 2010, 132, 194704 CrossRef PubMed.
- M. Stein, J. Wieland, P. Steurer, F. Tolle, R. Mulhaupt and B. Breit, Adv. Synth. Catal., 2011, 353, 523–527 CrossRef CAS.
- S. Donner, H. W. Li, E. S. Yeung and M. D. Porter, Anal. Chem., 2006, 78, 2816–2822 CrossRef CAS PubMed.
- S. Pei and H.-M. Cheng, Carbon, 2012, 50, 3210–3228 CrossRef CAS PubMed.
- M. Lazghab, K. Saleh and P. Guigon, Chem. Eng. Res. Des., 2010, 88, 686–692 CrossRef CAS PubMed.
- C. Baleizão, B. Gigante, M. J. Sabater and H. Garcia, Appl. Catal., A, 2002, 228, 279–288 CrossRef.
- B. Jarrais, C. Pereira, A. Rosa Silva, A. P. Carvalho, J. Pires and C. Freire, Polyhedron, 2009, 28, 994–1000 CrossRef CAS PubMed.
- F. Liu, J. Sun, L. Zhu, X. Meng, C. Qi and F.-S. Xiao, J. Mater. Chem., 2012, 22, 5495–5502 RSC.
- M. R. Maunja, K. Kumar, S. J. J. Titinchi, H. S. Abbo and S. Chad, Catal. Lett., 2003, 86, 97–105 CrossRef.
- H. P. Mungse, S. Verma, N. Kumar, B. Sain and O. P. Khatri, J. Mater. Chem., 2012, 22, 5427–5433 RSC.
- W. S. Hummers and R. E. Offeman, J. Am. Chem. Soc., 1958, 80, 1339 CrossRef CAS.
- J. Lopez, S. Liang and X. R. Bu, Tetrahedron Lett., 1998, 39, 4199–4202 CrossRef CAS.
- H. Vezin, E. Lamour and J. P. Catteau, J. Inorg. Biochem., 2002, 92, 177–182 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra03047h |
| This journal is © The Royal Society of Chemistry 2014 |







