Camptothecin delivery into hepatoma cell line by galactose-appended fluorescent drug delivery system†
Sun Dongbanga,
Hyun Mi Jeonb,
Min Hee Leea,
Weon Sup Shina,
Joon Kook Kwonc,
Chulhun Kang*b and
Jong Seung Kim*a
aDepartment of Chemistry, Korea University, Seoul 136-701, Korea. E-mail: jongskim@korea.ac.kr; Fax: +82-2-3290-3121
bThe School of East-West Medical Science, Kyung Hee University, Yongin, 446-701, Korea. E-mail: kangch@khu.ac.kr
cProtected Horticulture Research Station, National Institute of Horticultural and Herbal Science, Rural Development Administration, Busan 618-800, Korea
First published on 9th April 2014
Abstract
A galactose-appended camptothecin prodrug was newly synthesized. The prodrug preferentially entered a hepatoma cell line due to the galactose unit and the drug release triggered by disulfide-bond cleavage via reaction with glutathione was visualized on the basis of fluorescence changes of the naphthalimide moiety. The prodrug entered the cells via receptor-mediated endocytosis and released drug molecules into lysosomes, however, the released drug molecules failed to show significant anticancer activity, probably due to lysosomal hydrolysis of their lactone ring converting them to an inactive carboxylate form.
Selective delivery to cancer cells over normal cells is preferred to minimize unwanted side effects in cancer chemotherapy.1 Accordingly, there have been increasing reports of utilization of various drug delivery systems (DDSs) for cancer cells.2–4 Recently, galactose-appended chemicals capable of targeting human hepatoma were described,5 which might be valuable tools for sensing or delivery to liver cells.6 In fact, a galactose-appended fluorescent probe has been introduced for thiol imaging of liver cells.7a Therefore, a DDS containing a galactose unit, which fluoresces upon drug release, would provide information on the details of intracellular drug delivery, including elucidating the delivery pathway, cellular location, and cell selectivity. However, an example of monitable drug deliveries based upon a galactose-appended DDS has not been demonstrated so far.
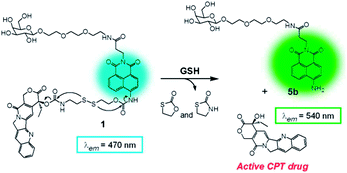
Herein, we describe a galactose-appended DDS with a camptothecin (CPT) drug, 1, to visually examine the site of drug release where fluorescence is detected upon release of CPT by the disulfide bond cleavage reaction7b with glutathione (GSH) in hepatoma cells. For this purpose, we designed 1 composed of four parts: (i) a galactose unit for selective targeting to hepatoma cells via asialoglycoprotein receptor (ASGP-R), (ii) a naphthalimide moiety for reporting CPT release in fluorescence emission, (iii) a disulfide bond for drug release in the cells upon its cleavage by GSH, and (iv) the anticancer drug CPT.8 A presumed drug release mechanism is suggested in Scheme 1.
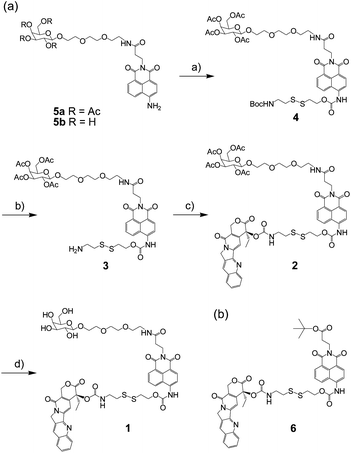
The synthesis of compound 1 consists of three parts: synthesis of the galactose unit; preparation of the disulfide bond-containing linker; and combining the galactose, naphthalimide, linker, and drug. First, the galactose unit was synthesized by previously reported methods (Scheme S1†).9 Subsequently, the naphthalimide portion was attached to the galactose unit to obtain 5a, as described previously.7 Compound 5a was linked to the disulfide bond linker 7 as seen in Scheme S2.†10 Compound 4 was obtained through a phosgene reaction in 18% yield. The Boc group protecting amine group was deprotected with TFA to afford 3 in 78% yield. The CPT molecule was attached to the amine end by a phosgene reaction to obtain 2 in 13% yield. Deprotection of the acetate groups was carried out using NaOMe in MeOH and exchanging sodium ions with protons by using a resin exchange to afford the target final compound 1 in 59% yield (Scheme 2). The identities of 1–4 were confirmed by 1H NMR, 13C NMR, and ESI-MS spectrometry (ESI, Fig. S1–S8).
The photophysical properties of 1 were studied to confirm that 1 reacts with GSH as demonstrated in Scheme 1. GSH is a thiol, which has been reported to be present in a large concentration in cancer cells than in normal cells.11 The strongest absorption intensity was observed at 370 nm, which was shifted to 430 nm upon addition of GSH (Fig. 1a). The new absorption spectrum is presumed to be attributed to 5b, after the disulfide bond was cleaved by the –SH nucleophilic unit of GSH. Fluorescence spectra were obtained under an excitation wavelength of 370 or 430 nm (Fig. 1b and c, respectively). As shown in Fig. 1b, upon excitation at 370 nm, the fluorescence spectrum of 1 showed a maximum at around 470 nm, whereas 5b showed little fluorescence with the same irradiation. Upon the addition of GSH to the solution of 1, a new fluorescence emission appeared at 445 nm. These results indicate that, when excited at 370 nm, the emission was contributed by the CPT released from 1. This finding is further supported by the fact that the excitation spectra of 1 observed at 445 nm emission wavelength was similar to that of CPT regardless of the presence of GSH. The corresponding spectra of 1, CPT, and 1 + GSH at 540 nm emission showed a slight excitation peak at 370 nm (Fig. S10†). However, when excited at 430 nm, fluorescence peak for 1 or CPT was not observed. The strong fluorescence emission appears at 540 nm upon GSH addition, which is consistent with that of 5b (Fig. 1c). Taken together, these results reveal that the CPT is released through the mechanism suggested in Scheme 1. Fluorescence spectra of 1 with various equivalents of GSH were investigated using an excitation wavelength at 430 nm to determine a linear relationship between the amount of GSH and the fluorescence intensity (Fig. 1d).
Further experiments to confirm the mechanism suggested in Scheme 1 were carried out using mass spectrometry and thin-layer chromatography (Fig. S11†). Mass spectra of 1 showed a mass peak that corresponds to the exact mass number (M + H+). After adding an excess amount of GSH at pH 7.4, the mass peak appeared at the mass number of compound 5b (M + Na+). Compound 2 with and without GSH, 5a, and CPT were compared on a thin-layer chromatograph. When GSH was added to 2, two new spots were observed, which are consistent with the retention value (Rf) of CPT and 5a. HPLC analysis also supports the suggested reaction mechanism followed by the CPT delivery from probe 1 upon reaction with GSH as depicted in Fig. S12.†
Fluorescence intensity of compound 1 remains constant throughout whole pH range. Upon addition of GSH, the fluorescence intensity marked enhanced at pH over 5 as seen in Fig. 2a. pH values of tumoral tissues were estimated to be higher than 6.4.11 Therefore, the probe is capable of drug delivery even in cancer cells. In order to characterize the selectivity of 1 toward to GSH, the putative fluorescence changes with various other biological species were measured. Impressively, with the addition of various amino acids and subsequent incubation, no significant changes in the fluorescence spectra were detected upon excitation at 430 nm (Fig. 2b). However, other thiol species such as cysteine (Cys), dithiothreitol (DTT), and homocysteine (Hcy) showed fluorescence changes similar to that of GSH (Fig. S13†). Considering that DTT is not present and that GSH is the most abundant thiol in the cells as compared to other thiols, (1–10 mM, 20–40 μM for GSH, and other thiols),12 the fluorescence change of 1 can be assumed to be selective for GSH.
To determine whether the fluorescence emission in the cells indicates the extent of drug release, time courses of the drug release from 1 and its fluorescence intensity in the presence of GSH were compared. Each time-course was measured at a 340 nm excitation wavelength and a 420 nm emission wavelength for CPT, and a 420 nm excitation wavelength and a 540 nm emission wavelength for 5a (Fig. S14†). The Ex 340 nm/Em 420 nm for fluorescence measurement for CPT was selected to minimize the fluorescence emitted from 1, as shown in the results of Fig. 1. The result in Fig. S14† clearly indicates that the disulfide bond cleavage is parallel to the CPT release into the solution; therefore, we can assume that the fluorescence intensity is simply proportional to the CPT release, presumably even in the cells.
For validation of 1 in the bio-applicability to the cells, it was added to a human hepatoma cell line (HepG2) where 6, a derivative with a tert-butyl moiety instead of the galactose unit in 1 is substituted, was characterized for comparison. Compounds 1 and 6 were incubated with HepG2, A549, HeLa, and KB cells with little expression of ASGP-R. In Fig. 3, HepG2 was found to be accumulated 1 inside the cells, whereas the other cell lines showed scarce amounts of fluorescence intensity. In contrast, 6 accumulated within all types of cells as demonstrated in Fig. 3. These data indicate that 1 is selectively taken up into the hepatic cells over the other cells, depending on the presence of the galactose moiety.
To obtain further support for the proposed ASGP-R-mediated endocytosis process, the cell uptake of 1 was tested in the presence of okadaic acid, a well-known inhibitor of endocytosis, in HepG2 cells.13 As shown in Fig. 4, the fluorescence intensity of 1 distinctly decreased in the cells pretreated with 15 nM of okadaic acid, which was seen even after 60 min incubation. In the case of the parallel experiment using 6, the fluorescence intensity was found to be unaffected by the presence of okadaic acid in cells. This contrasting result is reasonably consistent with the notion that ASGP-R-mediated endocytosis is involved in the cellular uptake of 1, but not the control system 2, into HepG2 cells through the selective interaction of galactose moiety of 1 with ASGP-R.
To determine whether the fluorescence changes observed with 1 and 6 resulted from the disulfide cleavage reaction shown in Scheme 1, HepG2 were treated with NEM (N-ethylmaleimide). NEM is known to react with thiol groups. In the presence of this agent, fluorescence of cells treated with 1 was expected to be relatively decreased (Fig. S15†). Thus, based on the results shown in Fig. 3 and 4 in conjunction with the results shown in Fig. S15,† we conclude that the selective uptake of 1 into HepG2 cells through ASGP-R-mediated endocytosis gives rise to a fluorescence signature by thiol-induced disulfide bond cleavage.
To get insight into the final location of CPT release from 1 in the HepG2 cells, a colocalization experiment was carried out with various commercially available subcellular trackers, i.e., MitoTracker, ERTracker, and LysoTracker (Fig. 5). The fluorescence image of 1 does not seem to overlap either MitoTracker or ERTracker to any extent, as shown in Fig. 5a and b, respectively, whereas it has remarkable overlap with LysoTracker as demonstrated in Fig. 5c. These results strongly implicate that probe 1 is localized to the lysosome and release CPT. Since lysosome has acidic environment ranging from pH 4.5 to 5,14 how the fluorogenic reaction occurs in it is of interest. The mechanism may be due to the reaction by GSH even though it is significantly slower than those at higher pH or by Trx-like activity in the lysosome, as similarly shown in a Trx-dependent disulfide cleavage in our previous report.7c
To evaluate the anticancer effects of the drug delivery systems, HepG2 cells were treated with CPT, the prodrugs, 1 and 6 and the cell viability data are presented in Fig. S16.† The cell viability under CPT and hydrophobic delivery system, 6, decreased in dose-dependent manner. However, the prodrug 1 unexpectedly failed to show a drug effect even at 50 μM although the system was delivered into cells, which is evident by the clear fluorescent intensities associated with the prodrug 1 in Fig. 3 and 4. The result is in contrast to our previous publication on biotin-mediated CPT delivery where CPT molecules were released into the endoplasmic reticulum of the cells with its strong anticancer activity.15 The reason for the futile delivery of 1 into the cells is probably due to conversion of the released CPT to the inactive carboxylate derivative through hydrolysis of its lactone ring by rich lysosomal hydrolytic enzymes.16 In fact, a similar inactivation of CPT by blood hydrolytic enzymes has been pointed as a significant weak point of this drug and enhancing its stability from unwanted hydrolysis is a main topic in medicinal chemistry.17 The results in Fig. S16† make us realize that one of the key facts to be considered for successful drug delivery is the final location of drug delivery although the lysosomal delivery of CPT drug susceptible to hydrolysis failed to reveal the drug activity.
In conclusion, a galactose-appended camptothecin prodrug 1 capable of reporting drug release via fluorescence change was synthesized and the drug delivery into cells was characterized. The CPT delivery did not interfere with other biologically active amino acids and functions in physiological conditions. Moreover, 1 can enter selectively into ASGP-R-expressing HepG2 cells via endocytosis. Upon cleavage of its disulfide bonds by GSH, the CPT is released into the lysosomes with concomitant strong fluorescence emission at 540 nm. Compound 1 provides a tool to evaluate drug delivery into cells and encourages us to visually characterize DDS at a single molecular level. However, the CPT was released into lysosome, which it failed to show an anticancer activity probably due to degradation to an inactive carboxylate derivative by lysosomal enzymes.
Acknowledgements
This work was supported by CRI project (no. 2009-0081566) (JSK) from the National Research Foundation of Korea grant and by Cooperative Research Program for agriculture Science & Technology Development (Project no. PJ008959) Rural Development Administration, Republic of Korea.Notes and references
- P. G. Corrie and G. Pippa, Medicine, 2008, 36, 24 CrossRef PubMed.
- M. C. Garnett, Adv. Drug Delivery Rev., 2001, 53, 171 CrossRef CAS.
- V. Dixit, J. Van den Bossche, D. M. Sherman, D. H. Thompson and R. P. Andres, Bioconjugate Chem., 2006, 17, 603 CrossRef CAS PubMed.
- H. P. Lesch, M. U. Kaikkonen, J. T. Pikkarainen and S. Ylä-Herttuala, Expert Opin. Drug Delivery, 2010, 7, 551 CrossRef CAS PubMed.
- (a) H. W. Kao, C. L. Chen, W. Y. Chang, J. T. Chen, W. J. Lin, R. S. Liu and H. E. Wang, Bioorg. Med. Chem., 2013, 21, 912 CrossRef CAS PubMed; (b) F. Lv, X. He, L. Wu and T. Liu, Bioorg. Med. Chem. Lett., 2013, 23, 1878 CrossRef CAS PubMed.
- (a) J. R. Merwin, G. S. Noell, W. L. Thomas, H. C. Chiou and M. E. DeRome, Bioconjugate Chem., 1994, 5, 612 CrossRef CAS; (b) R. Kikkeri, B. Lepenies, A. Adibekian, P. Laurino and P. H. Seeberger, J. Am. Chem. Soc., 2009, 131, 2110 CrossRef CAS PubMed.
- (a) M. H. Lee, J. H. Han, P. S. Kwon, S. Bhuniya, J. Y. Kim, J. L. Sessler, C. Kang and J. S. Kim, J. Am. Chem. Soc., 2012, 134, 1316 CrossRef CAS PubMed; (b) M. H. Lee, J. H. Han, J. Y. Kim, S. Bhuniya, J. L. Sessler, C. Kang and J. S. Kim, J. Am. Chem. Soc., 2012, 134, 12668 CrossRef CAS PubMed; (c) M. H. Lee, J. H. Han, J.-H. Lee, H. G. Choi, C. Kang and J. S. Kim, J. Am. Chem. Soc., 2012, 134, 17317 Search PubMed; (d) M. H. Lee, Z. Yang, J. H. Han, Y. H. Lee, S. Dongbang, C. Kang and J. S. Kim, Chem. Rev., 2013, 113, 5071 CrossRef CAS PubMed; (e) Z. Yang, J. H. Lee, H. M. Jeon, J. H. Han, N. Park, Y. He, H. Lee, K. S. Hong, C. Kang and J. S. Kim, J. Am. Chem. Soc., 2013, 135, 11657 CrossRef CAS PubMed; (f) D. Jung, S. Maiti, J. H. Lee, J. H. Lee and J. S. Kim, Chem. Commun., 2014, 50, 3044 RSC.
- Y. H. Hsiang, R. Hertzberg, S. Hecht and L. F. Liu, J. Biol. Chem., 1985, 260, 14873 CAS.
- H. Sato, E. Hayashi, N. Yamada, M. Yatagai and Y. Takahara, Bioconjugate Chem., 2001, 12, 701 CrossRef CAS PubMed.
- M. M. Pires and J. Chmielewski, Org. Lett., 2008, 10, 837 CrossRef CAS PubMed.
- O. W. Griffith, Free Radical Biol. Med., 1999, 27, 922 CrossRef CAS.
- C. Huang, A. J. Sinskey and H. F. Lodish, Science, 1992, 257, 1496 Search PubMed.
- I. Holen, P. B. Gordon, P. E. Stromhaug, T. O. Berg, M. Fengsrud, A. Brech, N. Roos, T. Bergs and P. O. Seglen, Biochem. J., 1995, 311, 317 CAS.
- J. A. Mindell, Annu. Rev. Physiol., 2012, 74, 69 CrossRef CAS PubMed.
- S. Maiti, N. Park, J. H. Han, H. M. Jeon, J. H. Lee, S. Bhuniya, C. Kang and J. S. Kim, J. Am. Chem. Soc., 2013, 135, 4567 CrossRef CAS PubMed.
- O. Lavergne, D. Demarquay, P. G. Kasprzyk and D. C. Bigg, Ann. N. Y. Acad. Sci., 2000, 922, 100 CrossRef CAS.
- (a) Q. Huang, L. Wang and W. Lu, Eur. J. Med. Chem., 2013, 63, 746 CrossRef CAS PubMed; (b) D. J. Adams, M. W. Dewhirst, J. L. Flowers, M. P. Gamcsik, O. M. Colvin, G. Manikumar, M. C. Wani and M. E. Wall, Cancer Chemother. Pharmacol., 2000, 46, 263 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra02588a |
| This journal is © The Royal Society of Chemistry 2014 |