Ultra-low friction achieved by diluted lactic acid solutions
Yuan Gao,
Liran Ma,
Dan Guo and
Jianbin Luo*
State Key Laboratory of Tribology, Tsinghua University, Beijing, 100084, China. E-mail: luojb@mail.tsinghua.edu.cn; Fax: +86-1062781379; Tel: +86-1062781385
First published on 3rd June 2014
Abstract
Notable lubrication performance was found using lactic acid solutions between two solid surfaces, silica glass against silicon nitride. The sliding friction coefficient was as low as 0.02 under a Hertzian contact pressure up to 400 MPa with a quite low concentration of 0.085 mol L−1. The results in the present work also exhibit the significant effect of concentration on the running-in process. A shortened running-in period, which could be contributed to the effective adsorption of the lactic acid molecules on the solid substrates, could be obtained by increasing the concentration of the lactic acid solution. In addition, surface hydroxyl groups gained by piranha solution were suggested to effectively reduce the friction. The work suggests that the excellent lubricity of lactic acid can be mostly contributed to the surface adsorption combined with the surface hydroxylation.
1. Introduction
Attempts to reduce friction have been reported since antiquity.1 Nowadays, new research interests in the tribology field have been devoted to the lubrication performance of biomaterials.2,3Typical biolubricants have been extracted from living organisms, such as the synovial and tear fluids, which have been artificially reproduced in both tribology and the medicine domain, with numerous reports on the lubrication performances of various components.4–7 Typically, hyaluronic acid, extensively existing in organisms, has been gaining popularity due to its distinguished water-holding capacity and outstanding biocompatibility.8–10 The indication is that the contributive role for reducing the friction coefficient of hyaluronic acid in aqueous solution comes from its super hydrophilicity.9,10 Phospholipids, consisting of hydrophobic tails and a hydrophilic head, found in rich quantities in living joints, have also been found to act as a kind of effective lubricant, protecting the cartilage from damage.11–13 Recently, many efforts have focused on the surface-attached molecules in aqueous liquids.14,15 Hakala et al.16 reported that the formation of a protein layer significantly reduced friction and wear. Raviv and Chen et al.17,18 showed that polyelectrolyte brushes attached to sliding solid surfaces can provide a friction coefficient remarkably similar to those found in healthy joints. Meanwhile, effective components extracted from plants have also shed new light on research for environmental friendly lubricants. Exceptional lubrication performance was reported by Arad et al.19 using polysaccharides extracted from red algae, although the mechanism is still unclear. The mucilage of both Brasenia schreberi and aloe has good lubricity between two rubbing surfaces.20,21
There is strong interest in exploring the lubrication mechanism between two contact surfaces across such bio-aqueous media. In particular, silicon nitride ceramic, one of the major wear-resistant materials that possess high flexural strength, high hardness and good fracture resistance, can obtain an excellent lubrication condition.22,23 During the process of lubricating the molecular origins, a common running-in period has emerged together with the reduction of friction in the nano/micro gap between the sliding and the contacted surfaces.19–23 The behavior and changes of both surfaces and lubricants during the running-in process will be crucial for revealing the fundamental mechanism of friction reduction, which requires great efforts to be thoroughly understood.
Lactic acid, a simple compound with the chemical formula CH3C(OH)COOH, has a hydroxyl group adjacent to the carboxyl group, making it an alpha-hydroxy acid. It can be constantly produced in the living body and also exists in some dairy foods, but has rarely been studied as a kind of lubricant. Some related investigations have been reported. Kim and Langer24 evaluated the lubrication performance of the biodegradable polyether networks that were copolymerized with oligomers of lactic acid. McKinney et al.25 measured the Knoop hardness and pin-and-disc-wear of a commercial silver-sintered glass-ionomer cement that had been preconditioned in lactic acid, decreasing the hardness through chemical dissolution. Yoshida et al.26 obtained a super low coefficient of friction (0.01) at the end of a sliding test of tetrahedral amorphous carbon (ta-C) lubricated with lactic acid. This study also indicates that lactic acid can efficiently reduce the sliding friction of glass surfaces in aqueous solution. Through the study of the lubricating characteristics of a lactic acid solution, our work aims to explore the lubrication mechanism of such a system and to discover how to shorten the running-in time and reduce material wear. We propose that this lubrication could be used in silicon nitride bearings after further improvements of some existing limitations, such as the brittleness of glass and the acidic environment of the lubricant.
2. Materials and methods
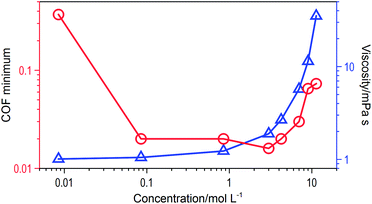
Lactic acid (85% w/w, a mixture of dextrorotatory and levorotatory molecules) was purchased from Sinopharm Chemical Reagent Beijing Co., Ltd. The lactic acid was then diluted with deionized water to a range of concentrations (from 0.009 M to 11.3 M). The pH value and viscosity of each solution is shown as Table 1.| Concentration (mol L−1) | pH value | Viscosity (mPa s) |
|---|---|---|
| 0.009 | 2.96 | 1.01 |
| 0.085 | 2.17 | 1.05 |
| 0.85 | 1.58 | 1.23 |
| 3 | 1.39 | 1.89 |
| 4.25 | 1.08 | 2.67 |
| 7 | 0.77 | 5.72 |
| 9 | 0.46 | 11.4 |
| 11.3 | <0 | 35.1 |
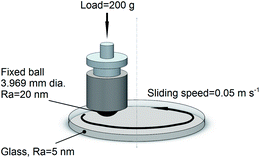
The friction force was measured constantly in ball-on-disc experiments at a sampling frequency of 1 kHz. The ball-on-disc device was installed on a UMT-3 tribometer (CETR). The load used in the experiments was 200 g, corresponding to a Hertzian contact pressure up to 400 MPa. The rotating speed was maintained at 120 rpm, corresponding to a linear speed of around 50 mm s−1. The rotating radius was 4 mm, and the disc acceleration was 12 rad s−2. The lubricant amount in each test was 30 μL, added as droplets onto the disc by pipette. The lubricant covered the entire area of the wear track. Each test was repeated 3 times.
The experimental set-up is sketched in Fig. 1. The upper ceramic ball was made of Si3N4, with a diameter of 5/32 inches (3.969 mm) and a mean surface roughness (Ra) less than 20 nm. The lower glass discs had a mean surface roughness of 5 nm. Before testing, the balls and discs were cleaned by ultrasonic washing in ethanol and deionized water, 5 min each.
To investigate the effect of surface wettability on the friction, the hydroxylation of the lower glass surfaces was enabled by immersing the samples in piranha solution for 5 h. The piranha solution was a mixture of 98% sulfuric acid (H2SO4) and hydrogen peroxide (H2O2) at a mixture ratio 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. In addition, the ContourGT-K 3D optical microscope (BRUKER) was used to measure the surface topography of the glass discs.
1. In addition, the ContourGT-K 3D optical microscope (BRUKER) was used to measure the surface topography of the glass discs.
3. Results and discussion
3.1 Lubrication of lactic acid on the glass surface
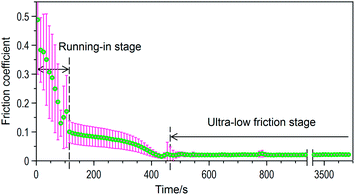
Lactic acid solution was introduced into the gap between the glass disc surface and the silicon nitride ball surface. Fig. 2 shows the typical evolution of friction coefficient vs. time, which was obtained utilizing 0.85 M lactic acid solution.As glass is a hard and brittle material, a typical running-in process is a friction reduction process accompanied by drastic fluctuations due to the acutely worn process, resulting in the large standard deviation observed in Fig. 2. For the sake of clarity, we define the running-in process as the initial procedure that enables the friction coefficient to fall to about 0.1. After the running-in period, the friction coefficient became low and steady, followed by the long-lasting ultra-low friction stage (more than 3600 s) with a friction coefficient of ca. 0.02 occurring within the next several minutes.
In addition, the tribology tests with other material combinations of the tribopairs were also performed, such as ceramic/ceramic, ceramic/silicon, and ceramic/DLC. The friction coefficient of about 0.08 to 0.15 was achieved using these material combinations. Further, we focused on ceramic/glass tribopairs, as the tests of the ceramic ball and glass disc obtained the best lubrication performance.
3.2 Influence on friction coefficient of lactic acid concentration
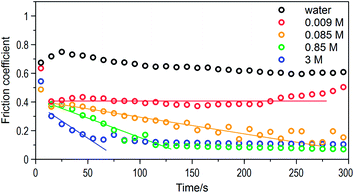
The results of friction coefficient varied with time using lactic acid solutions of different concentrations (0.009 M, 0.085 M, 0.85 M, 3 M) and pure water were recorded (Fig. 3). Predictably, pure water could not lubricate the ball-on-disc rubbing, with a friction coefficient in the range of 0.6 to 0.8, while the extremely dilute solution with a concentration of 0.009 M could effectively reduce the friction coefficient to about 0.4. Taking the 0.085 M lactic acid solution as lubricant, a pronounced running-in process appeared together with an obvious reduction of friction. The running-in period was found to be notably shortened by the increasing lactic acid concentration. Consequently, the friction coefficients after the running-in process for 0.085 M, 0.85 M and 3 M solutions were almost the same at 0.1. This indicated that the effect of concentration was only present during the running-in period rather than as the final friction coefficient.The wear scars of the ceramic balls and glass disks were observed by microscopy (Fig. 4). The wear scar area on the balls was roughly the same, although the shape of the wear scars changed with different lubricants. The contact pressure remained unchanged when the concentration of the lactic acid solution was less than 0.85 mol L−1. After lubrication by 0.85 mol L−1 lactic acid solution, the wear scar on the ball was elliptical, and the wear track on the disc was narrower and shallower than that of the other two samples. Even an addition of a small amount of lactic acid (0.009 mol L−1) to the deionized water reduced the wear of the disc, while the viscosity of the 0.009 M lactic acid solution was approximately equal to that of pure water. This indicated that the interaction between the glass surface and the lactic acid molecules reduced friction and wear in ball-on-disc sliding contact.
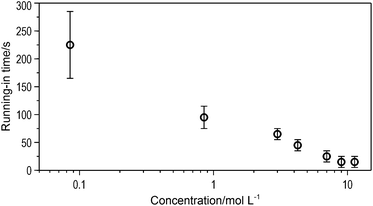
We also measured the friction coefficient of other solutions that were more concentrated than 3 M. The length of running-in time decreased with concentration, as shown in Fig. 5. The running-in process almost disappeared when the surfaces were lubricated by a highly concentrated solution. On the one hand, the higher concentration of the lubricant, the faster the lactic acid adsorption on the glass disc. Moreover, the highly concentrated solutions had high viscosities, making it easier for them to separate the two sliding surfaces.
Despite the fact that highly concentrated solutions also can reduce the friction coefficient, the lubricating ability did not increase linearly with concentration. The final stable friction coefficients of each test are listed in Fig. 6. The optimal lactic acid concentration ranged between 0.085 M and 3 M, within which the friction coefficient can go down to ca. 0.02. If the concentration continued to rise, the final stable friction coefficient did not decrease. On the contrary, the final stable friction coefficient would rise with the content of lactic acid in the solution. Considering the significantly increased viscosity, the highly concentrated solution would generate additional resistance, so that the final stable friction coefficient increased. When the viscosity became a major influencing factor, the lubrication mechanism had little relationship with the adsorption layer on the sliding surfaces.
Because it was easy to separate the two friction surfaces using a high viscosity lubricant, the separated surfaces were not effectively run in. This was another reason that the lubrication state could not enter into the ultra-low friction stage with highly concentrated solutions. Therefore, we focused on the dilute lactic acid solutions in the following experiments.
3.3 Influence on friction of surface hydroxyl groups
When a new glass surface is produced by cooling from a high temperature or by fracture, many unsaturated bonds or broken bonds will exist on the surface. In an acid solution, the broken bond will attract hydrogen ions to form surface hydroxyl groups that can change the surface energy of glass. To study the effect of surface hydroxyl groups on friction, we hydroxylated the glass discs by the piranha solution before the tribology tests.As shown in Fig. 7, the contact angles of before and after hydroxylated glass discs were ca. 56 and 20 degrees, respectively. The surface topography measured by 3D optical microscopy showed that the roughness of hydroxylated glass surface had hardly changed. After the hydroxylation pretreatment, the glass surface became more hydrophilic and had a large number of surface hydroxyl groups.
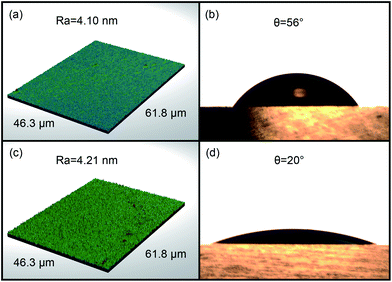 | ||
| Fig. 7 Surface topography of the glass discs and video images of a water drop on glass: (a and b), the original glass disc; (c and d), the hydroxylated glass disc. | ||
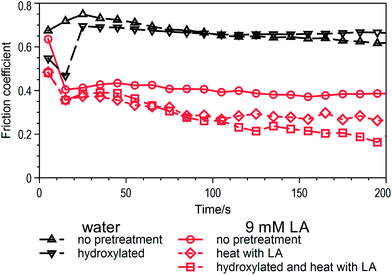
Fig. 8 shows the friction results of the original glass and the hydroxylated glass lubricated by lactic acid solutions of different concentrations. It is interesting to compare the results with lubrication by deionized water, especially in the first several seconds. When water alone was used as the lubricant on the hydroxylated glass disc, it performed as well as 0.009 M lactic acid in the first 20 s. Then, the friction coefficient rose, exactly like rubbing on the original glass discs because the hydroxylated glass surface had worn out. For the very dilute lactic acid solution, the friction coefficient can be maintained at about 0.4 M, which was 0.6 M when lubricated by deionized water. The friction coefficient would not rise when the hydroxylated surface had worn out because there were enough hydrogen ions in the solution to hydroxylate the new surface. This indicated that glass surface hydroxyl groups were conducive to the reduction of friction. However, there was no running-in process in the test that lubricated with 0.009 M lactic acid solution. Therefore, the surface hydroxyl groups were not the cause of the generation of a running-in process during friction.
The main factor causing a gradual decline in the friction coefficient should be the adsorption of lactic acid molecules on the glass surface. The existence of hydrogen ions in the solution or the surface hydroxyl groups was a necessary condition for adsorption. Glass surfaces can adsorb some organic compounds through the surface hydroxyl groups (![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) SiOH), some by physical adsorption and others by chemical adsorption by the hydroxyl reaction. To investigate whether lactic acid could adsorb on glass, another pretreatment was introduced. The glass discs were immersed in concentrated lactic acid and heated to 60 °C in a water bath for 1 h, and then rinsed with deionized water before the tribology tests. The diamonds and squares in Fig. 8 show the friction results with different glass discs lubricated by lactic acid solution in a concentration of 0.009 M. The lubrication on the discs that had been heated with lactic acid was better than those that had not been treated. This indicated that some lactic acid molecules had adsorbed onto the glass surface during the water bath pretreatment. That was equivalent to increasing the content of lactic acid during the friction test, so that a running-in process appeared no matter that the added lubricant was still very dilute. In the running-in period, lactic acid molecules continuously adsorbed on the glass surface because of intense friction and the local temperature rise. The best performance occurred when the glass disc had been first hydroxylated and then heated together with lactic acid in the water bath. The hydroxylated glass surface had more surface hydroxyl groups that made it possible to adsorb more lactic acid molecules.
SiOH), some by physical adsorption and others by chemical adsorption by the hydroxyl reaction. To investigate whether lactic acid could adsorb on glass, another pretreatment was introduced. The glass discs were immersed in concentrated lactic acid and heated to 60 °C in a water bath for 1 h, and then rinsed with deionized water before the tribology tests. The diamonds and squares in Fig. 8 show the friction results with different glass discs lubricated by lactic acid solution in a concentration of 0.009 M. The lubrication on the discs that had been heated with lactic acid was better than those that had not been treated. This indicated that some lactic acid molecules had adsorbed onto the glass surface during the water bath pretreatment. That was equivalent to increasing the content of lactic acid during the friction test, so that a running-in process appeared no matter that the added lubricant was still very dilute. In the running-in period, lactic acid molecules continuously adsorbed on the glass surface because of intense friction and the local temperature rise. The best performance occurred when the glass disc had been first hydroxylated and then heated together with lactic acid in the water bath. The hydroxylated glass surface had more surface hydroxyl groups that made it possible to adsorb more lactic acid molecules.
4. Conclusions
In summary, lactic acid solutions have an excellent lubricating ability on glass surfaces, with a coefficient down to 0.02 under a Hertzian contact pressure around 400 MPa. There was an obvious running-in process that appeared together with the reduction of friction when dilute solutions were used as lubricants. During the running-in process, the lactic acid molecules continuously adsorbed on the glass surface. This adsorption was carried out under acidic conditions because the hydrogen ions can add many hydroxyl groups, which are necessary for adsorption, onto the glass surface. The formation of the hygroscopically attached layer that separated the surfaces and held the water molecules significantly reduced the friction and wear of the silicon nitride ceramic/glass contact. Accordingly, there was a long time stable ultra-low friction state lubricated by lactic acid solution after the running-in period. The higher the concentration of lactic acid solution, the shorter the running-in time. However, the ultimate friction coefficient would increase with the viscosity if the concentration was too high.Acknowledgements
The work was financially supported by the National Key Basic Research Program of China (2013CB934200), the National Natural Science Foundation of China (51305225), and Research Fund of the Tsinghua Univeristy (20131089320).References
- D. Dowson, History of tribology, Longman, 1979 Search PubMed.
- J. P. Gong and Y. Osada, Prog. Polym. Sci., 2002, 27, 3–38 CrossRef CAS.
- M. Scherge, S. S. Gorb and S. N. Gorb, Biological Micro- and Nanaotribology, Springer, 2001 Search PubMed.
- T. Murakami, S. Yarimitsu, K. Nakashima, Y. Sawae and N. Sakai, Friction, 2013, 1, 150–162 CrossRef PubMed.
- J. Klein, Friction, 2013, 1, 1–23 CrossRef PubMed.
- Z. Liu, J. Yang, F. Grey, J. Z. Liu, Y. Liu, Y. Wang, Y. Yang, Y. Cheng and Q. Zheng, Phys. Rev. Lett., 2012, 108, 205503 CrossRef.
- Z. Jin and D. Dowson, Friction, 2013, 1, 100–113 CrossRef PubMed.
- A. G. Ogston and J. E. Stanier, J. Physiol., 1953, 119, 244–252 CAS.
- J. H. Bongaerts, J. J. Cooper-White and J. R. Stokes, Biomacromolecules, 2009, 10, 1287–1294 CrossRef CAS PubMed.
- D. P. Chang, N. I. Abu-Lail, F. Guilak, G. D. Jay and S. Zauscher, Langmuir, 2008, 24, 1183–1193 CrossRef CAS PubMed.
- D. A. Swann, R. B. Hendren, E. L. Radin and S. L. Sotman, Arthritis Rheum., 1981, 24, 22–30 CrossRef CAS.
- A. Oloyede, P. Gudimetla, R. Crawford and B. A. Hills, Clin Biomech, 2004, 19, 534–542 CrossRef CAS PubMed.
- I. M. Schwarz and B. A. Hills, Rheumatology, 1998, 37, 21–26 CrossRef CAS.
- M. Müller, S. Lee, H. A. Spikes and N. D. Spencer, Tribol. Lett., 2003, 15, 395–405 CrossRef.
- M. A. Faers, Adv. Colloid Interface Sci., 2003, 106, 23–54 CrossRef CAS.
- T. J. Hakala, P. Laaksonen, V. Saikko, T. Ahlroos, A. Helle, R. Mahlberg, H. Haehl, K. Jacobs, P. Kuosmanen, M. B. Linder and K. Holmberg, RSC Adv., 2012, 2, 9867–9872 RSC.
- U. Raviv, S. Giasson, N. Kampf, J. Gohy, R. Jérôme and J. Klein, Nature, 2003, 425, 163–165 CrossRef CAS PubMed.
- M. Chen, W. H. Briscoe, S. P. Armes and J. Klein, Science, 2009, 323, 1698–1701 CrossRef CAS PubMed.
- S. Arad, L. Rapoport, A. Moshkovich, D. van Moppes, M. Karpasas, R. Golan and Y. Golan, Langmuir, 2006, 22, 7313–7317 CrossRef CAS PubMed.
- J. Li, Y. Liu, J. Luo, P. Liu and C. Zhang, Langmuir, 2012, 28, 7797–7802 CrossRef CAS PubMed.
- J. Xu, J. B. Luo, S. H. Liu, G. X. Xie and L. Ma, Tribol.-Mater., Surf. Interfaces, 2008, 2, 72–76 CrossRef CAS PubMed.
- J. Li, C. Zhang, L. Sun, X. Lu and J. Luo, Langmuir, 2012, 28, 15816–15823 CrossRef CAS PubMed.
- J. Li, C. Zhang and J. Luo, Langmuir, 2013, 29, 5239–5245 CrossRef CAS PubMed.
- B. S. Kim, J. S. Hrkach and R. Langer, Biomaterials, 2000, 21, 259–265 CrossRef CAS.
- J. E. McKinney, J. M. Antonucci and N. W. Rupp, J. Dent. Res., 1988, 67, 831–835 CrossRef CAS PubMed.
- K. Yoshida, T. Horiuchi, M. Kano and M. Kumagai, Plasma Processes Polym., 2009, 6, S96–S101 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2014 |