A simple and efficient method to prepare a phosphorus modified g-C3N4 visible light photocatalyst
Abstract
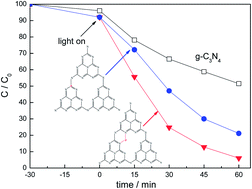
Phosphorus doped g-C3N4 was prepared by a simple method using dicyandiamide monomer and diammonium hydrogen phosphate as precursors. X-ray diffraction (XRD), UV-Vis spectroscopy, Fourier transform infrared spectra (FT-IR), photoluminescence (PL), X-ray photoelectron spectroscopy (XPS) and photocurrent measurement were used to characterize the prepared catalysts. The results indicated that the introduction of phosphorus inhibited the crystal growth of graphitic carbon nitride, decreased the band gap energy and increased the separation efficiency of photogenerated electrons and holes. The P doping site is influenced by the phosphorus source. Interstitial P doping is more effective in improving the photocatalytic activity of Rhodamine B (RhB) degradation compared with substitutional P doping. A possible mechanism was proposed.

 Please wait while we load your content...
Please wait while we load your content...