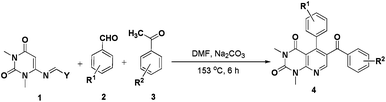
Exploration of uracils: pot and step economic production of pyridine core containing templates by multicomponent aza-Diels–Alder reaction†
Manas M. Sarmah and
Dipak Prajapati*
Medicinal Chemistry Division, CSIR-North-East Institute of Science and Technology, Jorhat, Assam 785006, India. E-mail: dr_dprajapati2003@yahoo.co.uk; Fax: +91 376 2370011
First published on 13th May 2014
Abstract
An efficient pot and step economic protocol for synthesis of pyrido[2,3-d]pyrimidine derivatives from a multicomponent aza-Diels–Alder reaction of uracil analogues, aromatic aldehydes and acetophenones in the presence of Na2CO3 in DMF was developed. The key step of the reaction is in situ generation of the reactive dienophile from aldehyde and acetophenone and their subsequent reaction with diene to give the desired product.
Introduction
Multicomponent reactions (MCRs) are one pot synthetic operations where concurrent combination of more than two common and convenient reactants furnishes a single product containing significant portions of all reacting counterparts. Spectacular results have been obtained by synthetic organic chemists for construction of complex and structurally diverse molecules by using MCRs with advantages such as high degree of synthetic efficiency, molecular diversity and step-economy.1 The concept of aza-Diels–Alder reaction strategy is successfully applied in numerous multicomponent reactions for construction of electrifying molecules.2Over the years, pyridine containing molecules has attracted much attention due to its extensive availability throughout nature. The field of pyridine and its derivatives is a promising area of research as this structural motif appears in a large number of pharmaceutical agents and natural products.3 Very recently, Saikia et al. developed a convenient method to synthesize pyrido[2,3-d]pyrimidines which were found to possess remarkable activities against microorganisms such as B. subtilis, S. aureus, K. pneumonia and E. coli.4a PD180970 (a novel pyrido[2,3-d]pyrimidine derivative), was successfully applied to inhibit Bcr-Abl and induce apoptosis in Bcr-Abl expressing leukemic cells.4b Pyrido[2,3-d]pyrimidine is also known to act as potent inhibitor of dihydrofolate reductase (DHFR);4c,d a target site in most of the parasitic diseases. In 2012, Škedelj's group showed that 6-arylpyrido[2,3-d]pyrimidines had notable action as ATP-competitive inhibitor of bacterial D-alanine: D-alanine ligase (Ddl).4e These discoveries have stimulated considerable interest in the synthesis of pyridine containing carbocycles through new and efficient routes.
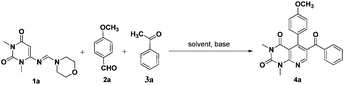
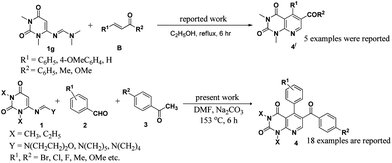
Uracil or pyrimidine-2,4-dione is one of the four essential components in the nucleic acid of RNA. A wide range of works on uracil are reported by synthetic chemists5 as well as biologists.6 General methods for the synthesis of uracil derivatives involve the annulation reactions starting from suitably substituted uracils and related systems.7 We have recently synthesized a series of iminoquinazolinedione derivative from vinyl uracils without using any solvent or catalyst.7f We also developed efficient methodologies for syntheses of some complex tetrahydroquinazolinedione and dihydropyrido[2,3-d]pyrimidine derivatives7g and, method for the formation of a library of dihydropyrido[4,3-d]pyrimidines was also well-documented by applying vinyl uracils in a microwave-assisted multicomponent reaction.7h,i 6-(Morpholinomethyleneamino)-1,3-dimethyluracil 1a is a reactive diene for [4 + 2]-cycloaddition reactions and a diverse array of potential products can be obtained by its synthetic manipulation. The molecule can simply be synthesized within two hours from N,N-dimethyl-6-aminouracil, morpholine and triethyl orthoformate under reflux conditions and subsequent recrystallization of the crude solid from ethanol.8 In continuation to our studies on uracil molecules,7f–n we describe in this paper an efficient three-component aza-Diels–Alder procedure for the synthesis of various pyrido[2,3-d]pyrimidine derivatives from the equimolar reaction mixture of 6-(morpholinomethyleneamino)-1,3-dimethyluracil, aromatic aldehydes and acetophenones in DMF (Scheme 1). To the best of our knowledge, only one report was available in the literature for the synthesis of uracil derivatives using 6-(morpholinomethyleneamino)-1,3-dimethyluracil as one of the starting reactants7i and thus we believe, our work will be able to draw considerable attention of organic chemists to explore the diene properties of 6-(morpholinomethyleneamino)-1,3-dimethyluracil.
Results and discussion
Our initial effort was to develop an appropriate solvent system and reaction condition to perform the proposed reaction. The model reaction of 6-(morpholinomethyleneamino)-1,3-dimethyluracil (1a, 1 mmol), p-anisaldehyde (2a, 1 mmol) and acetophenone (3a, 1 mmol) was investigated under different solvent systems, and the results obtained are indicated in Table 1. The reaction did not occur in dry condition and the starting materials were recovered quantitatively. It was observed that all solvent systems showed ‘no progress with no base’ in either reflux or stirring conditions (entries 2–11, Table 1). We were excited to notice that presence of catalytic amount of an organic base (e.g., morpholine, piperidine and pyrrolidine) could initiate the reaction but only in solvents with high boiling points, particularly in DMF. However, the percentage of conversion into product was very less (entries 12–16, Table 1). We then changed our methodology and planned to carry out a series of reactions at the boiling point of all solvents with moderately stronger bases. It was observed that good to better results were obtained when the reaction was carried out in presence of catalytic amount of inorganic bases (entries 19–24, Table 1). Because of easy handling and availability we preferred Na2CO3 (ref. 9) over other bases and the model reaction was screened in presence of Na2CO3 at various concentrations. We found that more than 80% of the product, 6-benzoyl-5-(4-methoxyphenyl)-1,3-dimethylpyrido[2,3-d]pyrimidine-2,4-dione 4a could be obtained within 6 hours, when the reaction mixture with Na2CO3 (10 mol%) was vigorously stirred in DMF at its boiling point. The yield of the product was unaffected by a further increase in the loading of the catalyst but a lower yield of the product was obtained from the reaction with 20 mol% of Na2CO3. By considering the side effects associated with the use of dimethyl sulfoxide (DMSO) for example, sedation, headache, nausea, dizziness, burning eyes, constipation etc. DMF was considered as better solvent for the cycloaddition. We have also taken aim to reduce the time of the reaction for formation of the product by application of microwaves but very poor conversion into product was observed under microwave conditions. Using the optimized reaction conditions, the feasibility of the reaction scheme was studied in detail by varying the uracils, aldehydes and acetophenones and the results obtained are summarized in Fig. 1.| Entry | Solvent | Base | Yieldd (%) |
|---|---|---|---|
| a Reaction conditions: a mixture of 6-(morpholinomethyleneamino)-1,3-dimethyluracil (1a, 1 mmol), p-anisaldehyde (2a, 1 mmol), acetophenone (3a, 1 mmol) wasb refluxed/stirred for 10 h without base;c vigorously stirred in presence of a base at the boiling point points of solvents till the completion of reaction.d Isolated yield. | |||
| 1 | Neat | — | NRb |
| 2 | Water | — | NRb |
| 3 | Chloroform | — | NRb |
| 4 | DCM | — | NRb |
| 5 | DCE | — | NRb |
| 6 | MeOH | — | NRb |
| 7 | EtOH | — | NRb |
| 8 | Acetonitrile | — | NRb |
| 9 | Toluene | — | NRb |
| 10 | DMF | — | NRb |
| 11 | DMSO | — | NRb |
| 12 | EtOH | Morpholine | NR |
| 13 | Acetonitrile | Piperidine | NR |
| 14 | Water | Piperidine | NR |
| 15 | DMF | Morpholine | Trace |
| 16 | DMSO | Pyrrolidine | Trace |
| 17 | o-Xylene | Na2CO3, 10 mol% | Trace |
| 18 | DMF | Na2CO3, 5 mol% | 40c |
| 19 | DMF | Na2CO3, 10 mol% | 85c |
| 20 | DMF | Na2CO3, 15 mol% | 85c |
| 21 | DMF | Na2CO3, 20 mol% | 70c |
| 22 | DMSO | Na2CO3, 10 mol% | 72c |
| 23 | DMF | K2CO3, 10 mol% | 70c |
| 24 | DMF | Cs2CO3, 10 mol% | 70c |
| 25 | DMF | Et3N | 12c |
During our generalization studies we were satisfied to find that the reaction was effective with aldehydes and acetophenones bearing electron-withdrawing and, -donating substituents on the aromatic ring. In all cases, regiospecific formation of the aromatic pyrido[2,3-d]pyrimidine templates were observed in excellent yields together with trace amounts of corresponding α,β-unsaturated ketones arising from aromatic aldehydes and acetophenones as byproduct which were removed during column chromatography. It can be mentioned here that yields of the products were better with aldehydes and acetophenones bearing electron withdrawing groups on both and/or on either sides of the aromatic rings. A lower yield of product was observed when heteroaromatic and conjugated aromatic aldehydes were employed (relative to the reaction with 6-(morpholinomethyleneamino)-1,3-dimethyluracil, benzaldehyde and acetophenone). It was observed that 6-(morpholinomethyleneamino)-1,3-dimethyluracil 1a was more reactive than 6-(piperidinylmethyleneamino)-1,3-dimethyluracil 1b, 6-(pyrrolidinylmethyleneamino)-1,3-dimethyluracil 1c, and vinyl uracils prepared from N,N-diethyl-6-aminouracil (1d–1f); leading to better yields of pyrido[2,3-d]pyrimidines. However, the reaction failed when aliphatic aldehydes and ketones were employed and only the condensation product between the aldehyde and ketone was formed. An attempt to perform the reaction with aliphatic aldehydes and ketones at high temperatures led to the decomposition of the starting materials. All of the products obtained were characterized by different methods such as, IR and NMR spectroscopy, mass spectrometry and elemental analysis.
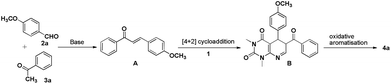
A plausible mechanism for the formation of the pyrido[2,3-d]pyrimidine is shown in Scheme 2. It is believed that the base promotes the formation of α,β-unsaturated ketone A between p-anisaldehyde 2a and acetophenone 3a which then undergoes a cycloaddition reaction with the diene system of uracil 1, followed by elimination of the amine moiety to generate dihydropyrido[2,3-d]pyrimidine derivative B. Oxidative aromatisation of B under the reaction conditions then leads to the formation of pyrido[2,3-d]pyrimidines 4a.
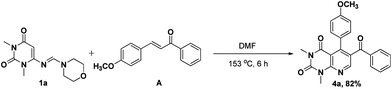
To verify our proposed mechanism, a two component reaction was carried out between a pre-formed unsaturated ketone A and 6-(morpholinomethyleneamino)-1,3-dimethyluracil 1a under the same reaction conditions (Scheme 3). As expected, the derivative 4a was obtained in comparable yield (82%). We further confirmed our mechanistic postulate by monitoring the model reaction at different time intervals (by thin layer chromatography) and observed that an intense spot appeared with Rf value 0.75 (ethyl acetate–hexane 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) within less than two hours. After two hours we stopped the reaction and isolated the compound responsible for the spot whose NMR spectra corresponded to A. These consequences showed that the experimental results were highly consistent with the proposed mechanism. Earlier we reported a two-component reaction of uracil amidine 1g with α,β-unsaturated compound B in ethanol to generate very limited numbers of pyrido[2,3-d]pyrimidines 4′in 6 h.10 But it is noteworthy that the multicomponent reaction with 10 mol% Na2CO3 in alcoholic medium was reluctant to undergo transformation despite long reaction time (Fig. 2). The step and pot economy principle (pre-functionalization of α,β-unsaturated ketone was not required) together with good yields associated with our new methodology make the approach more economical and useful over previous scheme to synthesize pyrido[2,3-d]pyrimidines.
2) within less than two hours. After two hours we stopped the reaction and isolated the compound responsible for the spot whose NMR spectra corresponded to A. These consequences showed that the experimental results were highly consistent with the proposed mechanism. Earlier we reported a two-component reaction of uracil amidine 1g with α,β-unsaturated compound B in ethanol to generate very limited numbers of pyrido[2,3-d]pyrimidines 4′in 6 h.10 But it is noteworthy that the multicomponent reaction with 10 mol% Na2CO3 in alcoholic medium was reluctant to undergo transformation despite long reaction time (Fig. 2). The step and pot economy principle (pre-functionalization of α,β-unsaturated ketone was not required) together with good yields associated with our new methodology make the approach more economical and useful over previous scheme to synthesize pyrido[2,3-d]pyrimidines.
Conclusions
In summary, we have demonstrated an aza-Diels–Alder reaction which efficiently leads to the synthesis of a diverse range of pyrido[2,3-d]pyrimidine derivatives. A wide variety of substituted aromatic aldehydes, cinnamaldehyde, heteroaromatic aldehydes, and substituted acetophenones were shown to undergo the reaction with different uracil molecules to give exclusive amount of desired products. Overall, our developed methodology requires simple and easily available starting materials and we consider that this reaction will create interest among chemists to investigate the diene behavior of some uracil derivatives.Acknowledgements
We are grateful to CSIR, New Delhi for financial support to this work. We also thank the Director, CSIR-NEIST, Jorhat for his keen interest and constant encouragement.Notes and references
- For reviews on MCRs, see: Multicomponent Reactions, ed. J. Zhu and H. Bienayme, Wiley-VCH, Weinheim, Germany, 2005 Search PubMed.
- (a) A. Islas-Jácome, A. Gutiérrez-Carrillo, M. A. García-Garibay and E. González-Zamora, Synlett, 2014, 403–406 Search PubMed; (b) Z. Chen, B. Wang, Z. Wang, G. Zhu and J. Sun, Angew. Chem., Int. Ed., 2013, 52, 2027–2031 CrossRef CAS PubMed; (c) A. Islas-Jácome, L. E. Cárdenas-Galindo, A. V. Jerezano, J. Tamariz, E. González-Zamora and R. Gámez-Montaño, Synlett, 2012, 2951–2956 Search PubMed; (d) Y.-H. He, W. Hu and Z. Guan, J. Org. Chem., 2012, 77, 200–207 CrossRef CAS PubMed; (e) L. He, M. Bekkaye, P. Retailleau and G. Masson, Org. Lett., 2012, 14, 3158–3161 CrossRef CAS PubMed; (f) A. Islas-Jácome, E. González-Zamora and R. Gámez-Montaño, Tetrahedron Lett., 2011, 52, 5245–5248 CrossRef PubMed; (g) D. Borkin, E. Morzhina, S. Datta, A. Rudnitskaya, A. Sood, M. Török and B. Török, Org. Biomol. Chem., 2011, 9, 1394–1401 RSC; (h) R. Burai, C. Ramesh, M. Shorty, R. Curpan, C. Bologa, L. A. Sklar, T. Oprea, E. R. Prossnitz and J. B. Arterburn, Org. Biomol. Chem., 2010, 8, 2252–2259 RSC; (i) H. Sundén, I. Ibrahem, L. Eriksson and A. Córdova, Angew. Chem., Int. Ed., 2005, 44, 4877–4880 CrossRef PubMed; (j) R. Lavilla, M. C. Bernabeu, I. Carranco and J. L. Díaz, Org. Lett., 2003, 5, 717–720 CrossRef CAS PubMed.
- H. J. Roth and A. Kleeman, Pharmaceutical chemistry, Wiley, New York, 1988, vol. 1: drug synthesis Search PubMed.
- (a) L. Saikia, B. Das, P. Bharali and A. J. Thakur, Tetrahedron Lett., 2014, 55, 1796–1801 CrossRef CAS PubMed; (b) P. L. Rosée, Cancer Res., 2002, 62, 7149–7153 Search PubMed; (c) A. Gangjee, A. Vasudevan, S. F. Queener and R. L. Kisliuk, J. Med. Chem., 1995, 38, 1778–1785 CrossRef CAS; (d) A. Gangjee, A. Vasudevan, S. F. Queener and R. L. Kisliuk, J. Med. Chem., 1996, 39, 1438–1446 CrossRef CAS PubMed; (e) V. Škedelj, E. Arsovska, T. Tomašić, A. Kroflič, V. Hodnik, M. Hrast, M. Bešter-Rogač, G. Anderluh, S. Gobec, J. Bostock, I. Chopra, A. J. O'Neill, C. Randall and A. Zega, PLoS One, 2012, 7, 39922 Search PubMed.
- (a) M. Szostak, B. Sautier and D. J. Procter, Org. Lett., 2014, 16, 452–455 CrossRef CAS PubMed; (b) O. Talhi, D. C. G. A. Pinto and A. M. S. Silva, Synlett, 2013, 24, 1147–1149 CrossRef CAS PubMed; (c) T. Miyazawa, K. Umezaki, N. Tarashima, K. Furukawa, T. Ooi and N. Minakawa, Chem. Commun., 2013, 49, 7851–7853 RSC; (d) D. W. Cho, C. W. Lee, J. G. Park, S. W. Oh, N. K. Sung, H. J. Park, K. M. Kim, P. S. Mariano and U. C. Yoon, Photochem. Photobiol. Sci., 2011, 10, 1169–1180 RSC; (e) H. Ito, K. Yumura and K. Saigo, Org. Lett., 2010, 12, 3386–3389 CrossRef CAS PubMed.
- (a) S. Das, A. J. Thakur, T. Medhi and B. Das, RSC Adv., 2013, 3, 3407–3413 RSC; (b) H. Miyakoshi, S. Miyahara, T. Yokogawa, K. T. Chong, J. Taguchi, K. Endoh, W. Yano, T. Wakasa, H. Ueno, Y. Takao, M. Nomura, S. Shuto, H. Nagasawa and M. Fukuoka, J. Med. Chem., 2012, 55, 2960–2969 CrossRef CAS PubMed; (c) A. Okamoto, Org. Biomol. Chem., 2009, 7, 21–26 RSC; (d) J. A. Valderrama and D. Vásquez, Tetrahedron Lett., 2008, 49, 703–706 CrossRef CAS PubMed; (e) C. Zhi, Z. Long, A. Manikowski, N. C. Brown, P. M. Tarantino Jr, K. Holm, E. J. Dix, G. E. Wright, K. A. Foster, M. M. Butler, W. A. LaMarr, D. J. Skow, I. Motorina, S. Lamothe and R. Storer, J. Med. Chem., 2005, 48, 7063–7074 CrossRef CAS PubMed; (f) R. Kumar, N. Sharma, M. Nath, H. A. Saffran and D. L. J. Tyrrell, J. Med. Chem., 2001, 44, 4225–4229 CrossRef CAS PubMed.
- (a) P. S. Naidu and P. J. Bhuyan, RSC Adv., 2014, 4, 9942–9945 RSC; (b) N. Tolstoluzhsky, P. Nikolaienko, N. Gorobets, E. V. Van der Eycken and N. Kolos, Eur. J. Org. Chem., 2013, 5364–5369 CrossRef CAS; (c) S. Paul, G. Pal and A. R. Das, RSC Adv., 2013, 3, 8637–8644 RSC; (d) S. Samai, G. C. Nandi, S. Chowdhury and M. S. Singh, Tetrahedron, 2011, 67, 5935–5941 CrossRef CAS PubMed; (e) K. C. Majumdar, S. Ponra and D. Ghosh, Synthesis, 2011, 1132–1136 CrossRef CAS PubMed; (f) M. M. Sarmah, D. Bhuyan and D. Prajapati, Synlett, 2013, 24, 1667–1670 CrossRef CAS PubMed; (g) M. M. Sarmah, D. Prajapati and W. Hu, Synlett, 2013, 24, 0471–0474 CrossRef CAS PubMed; (h) M. M. Sarmah, R. Sarma, D. Prajapati and W. Hu, Tetrahedron Lett., 2013, 54, 267–271 CrossRef CAS PubMed; (i) R. Sarma, M. M. Sarmah and D. Prajapati, J. Org. Chem., 2012, 77, 2018–2023 CrossRef CAS PubMed; (j) D. Prajapati, K. J. Borah and M. Gohain, Synlett, 2007, 595–598 CrossRef CAS PubMed; (k) D. Prajapati, M. Gohain and A. J. Thakur, Bioorg. Med. Chem. Lett., 2006, 16, 3537–3540 CrossRef CAS PubMed; (l) D. Prajapati and A. J. Thakur, Tetrahedron Lett., 2005, 46, 1433–1436 CrossRef CAS PubMed; (m) M. Gohain, D. Prajapati and B. J. Gogoi, Synlett, 2004, 1179–1182 CAS; (n) A. J. Thakur, P. Saikia, D. Prajapati and J. S. Sandhu, Synlett, 2001, 1299–1301 CrossRef CAS PubMed.
- Y. N. Tkachenko, E. B. Tsupak and A. F. Pozharskii, Chem. Heterocycl. Compd., 2000, 36, 307–310 CrossRef CAS.
- For examples of reactions in presence of Na2CO3, see: (a) H.-J. Eom, D.-W. Lee, Y.-K. Hong, S.-H. Chung, M.-g. Seo and K.-Y. Lee, Appl. Catal., A, 2014, 472, 152–159 CrossRef CAS PubMed; (b) P. Borah, P. S. Naidu, S. Majumder and P. J. Bhuyan, RSC Adv., 2013, 3, 20450–20455 RSC; (c) L. Donati, S. Michel, F. Tillequin and F.-H. Porée, Org. Lett., 2010, 12, 156–158 CrossRef CAS PubMed; (d) L. Wang, C. Tan, X. Liu and X. Feng, Synlett, 2008, 2075–2077 CAS; (e) C. Lu and J.-M. Lin, Catal. Today, 2004, 90, 343–347 CrossRef CAS PubMed.
- P. Saikia, A. J. Thakur, D. Prajapati and J. S. Sandhu, Indian J. Chem., Sect. B: Org. Chem. Incl. Med. Chem., 2002, 41, 804–807 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra02273d |
| This journal is © The Royal Society of Chemistry 2014 |