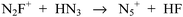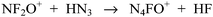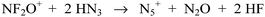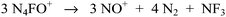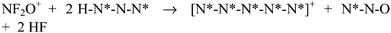4-Oxo- or 1-oxo-N7O+? A computational and experimental study†
Tao
Yu
*ac,
Ying-Zhe
Liu
a,
Ralf
Haiges
b,
Karl O.
Christe
*b,
Wei-Peng
Lai
a and
Bo
Wu
*c
aComputer-Aided Energetic Materials Design Group, Xi'an Modern Chemistry Research Institute, Xi'an 710065, P. R. China. E-mail: fischer@wo.cn; Tel: +86-029-88294020
bLoker Research Institute, University of Southern California, Los Angeles, CA 90089-1661, USA. E-mail: kchriste@usc.edu; Tel: +1-213-7403552
cMultiscale Computational Materials Facility, School of Materials Science and Engineering, Fuzhou University, Fuzhou 350108, P. R. China. E-mail: wubo@fzu.edu.cn; Tel: +86-0591-38725008
First published on 16th June 2014
Abstract
In a previous paper [Inorg. Chem., 2010, 49, 1245], we studied the reaction of F2NO+ with an excess of HN3 which led to the quantitative formation of N5+ and N2O. Based on 15N-labeling experiments and theoretical calculations, the formation of a 4-oxo-N7O+ intermediate with a decomposition energy barrier of about 40 kcal mol−1 was proposed. Since this relatively high barrier disagreed with our failure to experimentally observe this cation, a thorough theoretical study of the isomerization, dissociation and formation pathways of N4FO+ and N7O+ was carried out at the B3LYP and G3B3 levels at 240 K. It was found that the self-decomposition of 4-oxo-N7O+ to NO+ and N2 has a considerably lower barrier of only 19.6 kcal mol−1 and, therefore, would be more likely than a self-decomposition to N5+ and N2O. Additional calculations also showed that alternate reaction pathways between the stable and well-characterized z-N4FO+ intermediate product and HN3 involving 7- or 9-membered cyclic transition states, can lead to the observed N5+ and N2O products with the observed 15N distribution and barriers as low as 20.7 kcal mol−1. The transition states for these reactions contain a 1-oxo-N7O+ component which can decompose without a barrier to N5+ and N2O. These alternative pathways involving an unstable 1-oxo-N7O+ cation are in better agreement with experiment than the one involving 4-oxo-N7O+. The correctness of this re-interpretation was experimentally verified by a 15N-labeling experiment between α- and γ-15N-labeled HN3 and unlabeled N4FO+ which resulted exclusively in unlabeled N2O and α- and γ-15N-labeled N5+. Therefore, we conclude that in the reaction of NF2O+ with excess HN3 the experimental and theoretical evidence supports only the formation of an unstable 1-oxo-N7O+ cation, and that for the preparation of the symmetric 4-oxo-N7O+ cation different synthetic approaches will be required.
Introduction
Polynitrogen compounds are of great interest as energetic materials.1–3 In 1999, N5+ was synthesized by the reaction of HN3 with N2F+ at −78 °C in dry HF (Scheme 1).2Application of the same approach to the reaction of the closely related NF2O+ cation with HN3 resulted in the formation of the stable z-N4FO+ and e-N4FO+ cations, when a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio of the starting materials was used (Scheme 2).3
1 molar ratio of the starting materials was used (Scheme 2).3
However, when an excess of HN3 was used, the expected N7O+ cation (Scheme 3) could not be experimentally observed even at low temperatures.4
Surprisingly, N5+ and N2O were observed in quantitative yield as the only reaction products (Scheme 4).
Based on theoretical calculations and a 15N-labeling experiment, using unlabeled NF2O+ and α- and γ-15N-labeled HN3, which resulted in terminally labeled N2O and N5+ labeled equally in all five positions, the formation of a 4-oxo-N7O+ intermediate was proposed.4 However, this interpretation presented a major problem. The predicted barrier for the decomposition of 4-oxo-N7O+ to N5+ and N2O had a high value of ∼42 kcal mol−1.4 This high barrier should have allowed its experimental observation. This discrepancy prompted the Chinese group to carry out a very intensive computational study which suggested a more likely alternate reaction path with a much lower activation energy barrier of only 20.7 kcal mol−1, while also replicating the observed 15N-labeled products. The correctness of the alternate reaction scheme was experimentally verified by the American group by a second 15N-labeling experiment. The detailed results of these combined studies are presented in this paper, showing that 1-oxo-N7O+ is much less stable to decomposition to N5+ and N2O than the 4-oxo isomer and is the likely unobserved unstable intermediate in the reaction of NF2O+ with an excess of HN3.
Results and discussion
A. Selection of computational method
In this section, we first test the applicability of the B3LYP, MP2 and CCSD(T) methods by comparing the optimized structures of small molecules and cations (1 to 6) which are related to the reaction of N7O+, with corresponding known experimental structures,5–9 and by calculating their enthalpies of formation. The most important structural parameters of 1 to 6 optimized with the B3LYP, MP2 and CCSD(T) methods, are shown in Fig. 1. Compared with the experimental values, the B3LYP method slightly overestimates bond lengths of 1 to 6, but the deviations are within 0.01 Å (except for the N–O bond in 1), while the deviations of the bond angles are within 2°. Thus, the B3LYP method can provide satisfactory geometries for the systems of interest. The CCSD(T) method performs excellently when compared to the experimental geometries in gas phase. The deviations of bond lengths of 2, 3, 4 and 6 are less than 0.004 Å, and 0.2° on bond angles of 2. However, the CCSD(T)/aug-cc-pVQZ calculations are costly. The performance of MP2 is not as good as expected, especially for the short N–N bonds in 2, 4, 5 and 6. | ||
| Fig. 1 Computed (at B3LYP/aug-cc-pVDZ, MP2/aug-cc-pVQZ, and CCSD(T)/aug-cc-pVQZ levels) and experimental geometries of F2NO+ (1), HN3 (2), HF (3), N2O (4), N5+ (5) and N2F+ (6). The bond lengths in angstroms and bond angles in degrees. The experimental geometries: F2NO+,5 crystal structure of F2NO+AsF6−; HN3,6 microwave spectroscopy; HF,7 microwave spectroscopy; N2O,7 infrared and Raman spectroscopy; N5+,8 crystal structure of N5+Sb2F11−; and N2F+,9 millimeter-wave spectroscopy. | ||
The computed enthalpies of formation of 1 to 6 using the B3LYP, MP2, CCSD(T) and G3B3 methods and experimental enthalpies of formation of 1 to 4 and 6 are presented in Table 1. Among the four computational methods, the computed values of the composite G3B3 method for 1 to 4 and 6 are the closest to experiment, and the deviations are within 1 kcal mol−1. The deviation between G3B3 and CCSD(T) of complete basis set (353.3 kcal mol−1 was predicted by the best available calculation10) is about 2 kcal mol−1. Compared with experimental (1 to 4) or G3B3 (5 and 6) values, the absolute deviations of B3LYP, MP2 and CCSD(T) are 1.2 to 3.4, 4.2 to 26.4 and 0.4 to 10.7 kcal mol−1, respectively. Evidently, the G3B3 method can evaluate precise energies, and the B3LYP calculations achieve acceptable accuracy at moderate cost. MP2 fails to predict the precise enthalpies of formation for polynitrogen system and provides a negative enthalpy of formation for 4. The results of Pople's 6-311++G** and Petersson's CBS-QB3 are also presented in Fig. S1 and Table S1 of ESI.† There are little difference between B3LYP/aug-cc-pVDZ and B3LYP/6-311++G** geometries. The enthalpies of formation of B3LYP/aug-cc-pVDZ and G3B3 are a little better than those of B3LYP/6-311++G** and CBS-QB3, respectively.
| Species | B3LYP | MP2 | CCSD(T) | G3B3 | Expt. |
|---|---|---|---|---|---|
| a The enthalpies of formation were calculated from the atomic energies in the gas phase. The ionization energy of the nitrogen atom was used to calculate the enthalpies of formation of the cations. b The experimental enthalpies of formation, the atomic energies, the ionization energy of the nitrogen atom, and the appearance energies of NF3O and cis-N2F2 are taken from ref. 7. | |||||
| NF2O+ (1) | 234.6 | 212.8 | 237.6 | 233.4 | 232.4 |
| HN3 (2) | 68.0 | 56.9 | 77.3 | 70.0 | 70.3 |
| HF (3) | −61.8 | −69.4 | −64.8 | −65.2 | −65.1 |
| N2O (4) | 21.7 | −0.6 | 26.3 | 19.4 | 19.6 |
| N5+ (5) | 348.4 | 324.8 | 361.9 | 351.2 | — |
| N2F+ (6) | 293.5 | 274.8 | 295.5 | 290.6 | 283.4 ± 4.6 |
On the basis of the above results, we will use in the following discussions B3LYP to optimize all of the geometries, and B3LYP and G3B3 to explore the reaction pathways and to calculate the free energy activation barriers. Unless otherwise specified, G3B3 energies are mainly used in the text. Numbering schemes for the compounds and cations are given in Fig. S2 of the ESI.† The notations for the transition states and complexes are TS and C, respectively, followed by the numbers of compounds and cations.
B. Isomerization and dissociation of N4OF+
In this investigation, five isomers (7 to 11) of N4FO+ were optimized. The isomerization and dissociation pathways of these isomers are shown in Fig. 2. The isomers with the two lowest free energies are 7 (e) and 8 (z), and their free energies are almost the same. The isomerization barrier from 7 to 8 is 18.0 kcal mol−1, and the reverse barrier is equal to the forward one. Consequently, there is no thermodynamically dominant geometry for 7 and 8. The dissociation of 9 to C4/6, the ring opening of 9 to 7, dissociation of 10 to C4/6, and transformation of 11 to 7 cross barriers of less than 15 kcal mol−1, which implies that 9, 10 and 11 dissociate or transform easily. 11 can also decompose to NF, NO+ and N2 with a 3.1 kcal mol−1 barrier (see Fig. S3†). Hence, 7 and 8 are the two primary isomers of N4FO+.The dissociation of 7 to C4/6 has two pathways: one is concerted, and the other one is a stepwise pathway via the intermediate 9. The concerted pathway viaTS7C4/6 has a very high barrier of 69.1 kcal mol−1. Because the dissociation barrier of 9 to C4/6, i.e., the second step of the dissociation of 7 to C4/6, is only 7.1 kcal mol−1, the limiting step is the first step, i.e., the cyclization of 7 to 9viaTS7/9 with a high barrier of 36.4 kcal mol−1. The dissociation of 8 to C4/6 also has two pathways: F can approach either β-N (TS8C4/6b) or γ-N (TS8C4/6a). The dissociation barrier for 8 to C4/6viaTS8C4/6a is 38.8 kcal mol−1, and viaTS8C4/6b it is 44.0 kcal mol−1. Therefore the dissociation of z- and e-N4FO+ to N2F+ and N2O will not proceed easily.
Furthermore, decomposition of N4FO+ to N2F+ and N2O is not supported by the experimentally observed decomposition of α- and γ-15N-labeled N4FO+ to unlabeled NO+, labeled N2 and NF3 (Scheme 5).
However, we found the following more likely pathway. The e-N4FO+ cation (7) can go through the transition state (TS7CI) involving the breakage of the α-N–β-N bond and an F shift from N(O) to α-N, with a dissociation barrier of only 21.1 kcal mol−1. The z-N4FO+ cation (8) has a similar pathway viaTS8CI, but the barrier is somewhat higher (39.6 kcal mol−1). ONNF+ in CI can decompose to NO+ and N2 with a 27.2 kcal mol−1 barrier (see Fig. S4†). The barrier from 8 to NO+ and FN3 (CII) is 46.9 kcal mol−1 and is considerably larger. Therefore, the most favorable decomposition pathways of 7 and 8 are 7 → TS7CI → CI and 8 → TS7/8 → 7 → TS7CI → CI, respectively, with maximum barriers of 21.1 kcal mol−1.
Due to a potential catalysis by HF (3), the dissociation of z-N4FO+ (8) in the presence of 3 was also studied. However, as shown in Fig. S5,† the participation of HF increases the dissociation barriers to more than 60 kcal mol−1. Thus, these pathways can be ruled out.
C. Formation of N4OF+
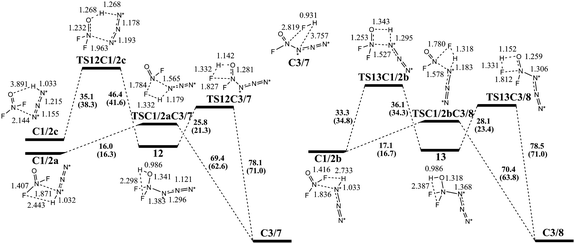
The reactions of NF2O+ (1) with HN3 (2) in the absence of HF leading to the formation of e-N4FO+ (7) and z-N4FO+ (8) are shown in Fig. 3. During the reaction of 1 with 2, the different interactions between two molecules can involve three complexes (C1/2a, C1/2b and C1/2c) as reaction entrances, among which C1/2a ultimately forms C3/7 along a concerted pathway with a barrier of 16.0 kcal mol−1, and C1/2c ultimately forms C3/7 along a stepwise pathway with one intermediate and two transition states with a maximum barrier of 35.1 kcal mol−1, while C1/2b ultimately forms C3/8 along either a concerted pathway with a barrier of 17.1 kcal mol−1 or a stepwise pathway with one intermediate and two transition states with a maximum barrier of 33.3 kcal mol−1. Obviously, from a thermodynamic point of view, the preferred formation pathways of 7 and 8 are the concerted ones from C1/2a and C1/2b, respectively. It should be noted that in the transition states along the concerted pathways the hydrogen atom is still bound to nitrogen and bridges to an F atom, while in the first step of the stepwise pathways, the H atom attaches itself to the O atom and starts to depart from the α-N atom, resulting in a higher barrier.Since the reaction of NF2O+ with HN3 was carried out in HF solution, it was important to study the reactions of 1 with 2 in the presence of HF (Fig. 4). As can be seen, HF significantly lowers the barriers for the formation of 7 and 8, and catalyzes the reaction of 1 with 2. Since HF is one of the reaction products, this reaction is auto-catalytic, similar to our previous report on the auto-catalysis of polyesterification.11 However, the presence of HF does not change the barrier ordering of these pathways, and the preferred formation pathways of 7 and 8 are still the concerted ones from C1/2/3a and C1/2/3b, respectively. The corresponding barriers are 12.6 and 14.7 kcal mol−1, respectively.
Comparing the barriers of the concerted reaction pathways viaC1/2a and C1/2b, it is found that, independent of the absence or presence of HF, the barriers for the formation of 7 and 8 are always very similar, and no low-barrier pathways for the formation of 9, 10 and 11 from 1 and 2 as reactants were discovered. Therefore the reaction of 1 with 2 produces both 7 and 8.
D. Formation of N5+via N2F+ and HN3
The direct formation mechanism of N5+ (5) from N2F+ (6) and HN3 (2) has been reported.12 Our calculations indicated that the barrier from C2/6 to C3/5 is 10.2 kcal mol−1 (Fig. 5). We have also investigated the reaction of 2 with 6 in the presence of HF (3). It was found that a transition state with a six-membered ring is formed (TSC2/3/6C3/3/5 in Fig. 6) along the pathway, and the corresponding barrier is only 6.6 kcal mol−1. Hence, this is the preferred pathway to the formation of 5 from 2 and 6, and HF takes part in the reaction as a catalyst. The formation of 5via2 and 6 is not the limiting pathway.E. Isomerization and self-dissociation of N7O+
In our calculations, six isomers (14 to 19) of N7O+ were found. Their isomerization and dissociation pathways are shown in Fig. 6. The 14, 15 and 16 isomers are the three possible 4-oxo-isomers, i.e., the oxygen is attached to the central nitrogen atom, and the two azido ligands point either in the same direction as the oxygen (15) (syn–syn), in the opposite direction (16) (anti–anti), or one in the same direction and the other one in the opposite direction (14) (syn–anti). The (anti–anti) isomer 16 lies 14.6 kcal mol−1 above the (syn–anti) isomer 14 and its isomerization barrier to 14 is only 1.0 kcal mol−1. Hence 16 is highly unstable and was not further considered. The (syn–syn) isomer 15 is the lowest in energy and lies 1.9 kcal mol−1 below the (syn–anti) isomer 14. The isomerization barrier for the conversion of (anti–syn) 14 to (syn–syn) 15 is also low and amounts only to 11.3 kcal mol−1. Isomers 17 and 18 are cyclic isomers and lie 24.2 and 23.0 kcal mol−1, respectively, above the (syn–syn) isomer 15. Compound 19 is the 1-oxo-isomer and lies 18.3 kcal mol−1 above 15. Thus, (syn–syn) 15 is the dominant isomer of N7O+.The dissociation of (anti–syn) N7O+14 to N2O (4) and N5+ (5) can proceed along three different pathways: (1) the formation of a new N–N bond between the α and γ positions viaTS14C4/5a with a five-membered ring and a barrier of 39.5 kcal mol−1; (2) a dissociation pathway involving the TS14C4/5b transition state with a four-membered ring and a high barrier of 59.5 kcal mol−1; (3) a stepwise pathway involving an intermediate (18) with a five-membered ring crossing a lower barrier of 23.1 kcal mol−1. Similarly, the dissociation of (syn–syn) N7O+15 to N2O (4) and N5+ (5) can proceed along two different pathways: (1) a dissociation pathway involving only one transition state with a four-membered ring, TS15C4/5, with a very high barrier of 63.4 kcal mol−1; (2) a stepwise pathway involving an intermediate (17) with a five-membered ring crossing a lower barrier of 25.4 kcal mol−1. These conclusions are consistent with those from the previous investigation.4 Based on these barriers and whether starting from 14 or 15, the most favorable dissociations are the stepwise pathways with ring-like intermediates. These intermediates, 17 and 18, are isomers of N7O+. They are located in a shallow potential well and their isomerization or dissociation barriers are only several kcal mol−1. However, they tend to isomerize rather than dissociate because the barriers of isomerization are lower than those of dissociation by about 1 kcal mol−1. Although from a thermodynamic point of view these stepwise pathways having barriers as low as 23.1 kcal mol−1 might be worthy of consideration, they must be rejected because they do not result in the experimentally observed 15N labels.
Another important result from our calculations is the finding that the barrier for the conversion of the 1-oxo-N7O+ isomer 19 to N2O 4 and N5+5 (not denoted in Fig. 6) is only 0.2 kcal mol−1 (0 K without zero-point vibrational energy) at the B3LYP level. Therefore, 19 is able to dissociate very easily into 4 and 5, while reproducing the previously reported 15N labels. Although 19 cannot be directly prepared from NF2O+ and excess HN3, it is present as a component of the important TSC2/3/8C3/3/4/5a transition state in the reaction of N4FO+ with HN3 in the presence of HF and thus provides the best explanation for all of our experimental observations, including the additional 15N-labeling experiments which will be described below.
There are also three possible pathways for the decomposition of N7O+ to N3+ or NO+: (1) (anti–syn) N7O+14 decomposes to N3+ with evolution of N2 and NO2viaTS14CIII. It undergoes the break of the α-N–β-N bond on one side and the break of the α-N–N(O) bond on the other side with a barrier of 40.4 kcal mol−1; (2) (syn–syn) N7O+15 decomposes to N3+viaTS14CIII with a barrier of 40.7 kcal mol−1; (3) the pathway of dissociation to N5O+ goes through a transition state (TS15CIV) involving the break of a α-N–β-N bond and an azide group shifts from N(O) to α-N with a dissociation barrier of 19.6 kcal mol−1. N5O+ in CIV readily decomposes to NO+ with evolution of N2 with a barrier of only 5.8 kcal mol−1 (Fig. S6†). Therefore, the optimum pathways of decomposition of 14 and 15 are 14 → TS14/15 → 15 → TS15CIV → CIV and 15 → TS15CIV → CIV, respectively. Again, the barrier of 19.6 kcal mol−1 would be reasonably low, but these pathways cannot explain the experimentally observed reaction of NF2O+ with excess HN3 which produced equally labeled N5+ and terminally labeled N2O.4
F. Dissociation of N7O+ with excess HN3
Due to the presence of excess HN3 in the reaction system, the dissociation of N7O+ in the presence of HN3 was also investigated. The corresponding dissociation pathways are displayed in Fig. 7. The presence of HN3 lowers the dissociation barrier of 14 to C4/5 along the concerted pathway, with the H atom attaching to the α-N of 14 and departing from the α-N of HN3, but the barrier of 44.9 kcal mol−1 is still high. The barrier order parallels again the electronegativity order. At the same time, in the presence of HN3, the anti–syn isomerization of N7O+ was found via an intermediate C5/20, but for both isomerization directions, from 14 to 15 or from 15 to 14, the barriers are more than 30 kcal mol−1. Thus the presence of HN3 does not facilitate the isomerization, compared to the reaction in its absence (see Fig. 6). It should be mentioned that the processes give rise to the formation of N5+. In addition, the dissociation of N5OH+20 was investigated. It undergoes two transition states with one intermediate in between (Fig. S7†). Both barriers are not more than 8 kcal mol−1 and are lower than the barriers of the reaction of 20 with 5 back to 14 and 15. Thus, the intermediate 20 rather dissociates than transforms to 14 or 15, and the dissociation barriers of 14 and 15 with excess HN3 are 30.6 and 32.5 kcal mol−1, respectively.The stepwise N7O+ decomposition mechanisms involve attachment of the azide groups to N(O) in the first step and decomposition through TS14C4/5a (Fig. 6) in the second step. The intermediates 22 and 23 (Fig. 7) are novel with ten nitrogen atoms. The performance of these stepwise pathways is still poor, because they cross limiting barriers of more than 50 kcal mol−1.
Because of the catalytic effect of HF, the dissociation of N7O+ in the presence of HN3 and HF was also studied. The corresponding dissociation pathways are presented in Fig. 8. There is no significant change in the presence of HF, compared to the presence of HN3 only. The lowest dissociation barriers of 14 and 15 with excess HN3 in the presence of HF are 33.6 and 33.4 kcal mol−1, respectively.
All the pathways in Fig. 7 and 8 are in accord with the experimental 15N labels.4 Moreover, we tried to calculate the barriers of reactions between 15 and HF. However, barriers in excess of 60 kcal mol−1 must be overcome. In summary, the barriers of dissociation of 14 and 15 in the presence of HN3 are higher than 30 kcal mol−1, and the reactions are not catalyzed.
G. Formation of N7O+ and N5+via N4OF+ and HN3
The reaction pathways between e-N4FO+ (7) or z-N4FO+ (8) and HN3 (2) in the absence of HF (3) are presented in Fig. 9. As already mentioned, the barriers involving the proton transfer from 2 to the oxygen of NF2O+ in the stepwise mechanisms for the formation of N4FO+, are much higher than those involving concerted mechanisms. Consequently, they do not easily lead to the formation of 1-oxo-N4FO+ (10). The formation of 10 undergoes three transition states and two intermediates (25 and 28), and the limiting pathway, the proton transfer to O(N), crosses a barrier of 49.2 kcal mol−1. The mechanisms of proton transfer to O(N) is complex. Firstly, 7 or 8 and 2 form complexes C2/7 or C2/8via a long-range Coulombic force with no activation energy barrier. C2/7 can form 5 along two different pathways viaTSC2/7C5/24a and TSC2/7C5/24b, and crosses barriers of 28.0 and 63.7 kcal mol−1, respectively. 24 is highly unstable, and its dissociation barrier is only 3.0 kcal mol−1 (Fig. S8†). The γ-N of 7 prefers to attach to the α-N of 2 rather than attach to the γ-N of 2. Another pathway for the formation of 5 starts from 25 through TS14C4/5a, and crosses a barrier of 47.5 kcal mol−1. Alternatively, C2/7 can concertedly form 14 and 15viaTSC2/7C3/14 and TSC2/7C3/15, and crosses the barriers of 27.9 and 29.9 kcal mol−1, respectively. Consequently in the reaction of 2 with 7, there is no dominant product formed between 5, 14 and 15.C2/8 can form 5 along three different pathways, viaTSC2/8C3/4/5a, TSC2/8C3/4/5b and TSC2/8C3/4/5c, crossing barriers of 24.4, 35.1 and 51.2 kcal mol−1, respectively. The γ-N of 8 also prefers to attach to the α-N of 2, and α-N of 2 is more reactive than γ-N of 2. It is logical that the reactions involving γ-N of 2 aren't considered for the dissociation of N7O+ in the presence of excess HN3. Alternatively, C2/8 can form 14 and 16viaTSC2/8C3/14 and TSC2/8C3/16, and crosses barriers of 29.8 and 31.9 kcal mol−1, respectively. Consequently, in the reaction of 2 with 8 the dominant product out of 5, 14 and 16 is probably 5. Except for the pathway viaTSC2/8C3/4/5b, all the pathways agree with the observed 15N labels.
HF acts as an auto-catalyst in these reaction systems. Thus, the reactions of 7 or 8 with HN3 in the presence of HF were investigated and are shown in Fig. 10. Generally, the reaction barriers are lower in the presence of HF than in its absence, however, the presence of HF does not affect the order of the barriers. During the reactions of 7 with 2 in the presence of HF, the formation barriers of 14 and 15 are 21.7 and 20.7 kcal mol−1, respectively, while the lower formation barrier of 5 is 29.1 kcal mol−1. Consequently, in the reaction of 7 with 2 in the presence of HF the dominant product out of 5, 14 and 15 is probably 15. During the reactions of 8 with HN3 in the presence of HF, the formation barriers of 14 and 16 are 21.6 and 26.7 kcal mol−1, respectively, while the lower formation barrier of 5 is 20.7 kcal mol−1. Therefore, in the reaction of 8 with HN3 in the presence of HF the dominant product out of 5, 13 and 15 is probably 5.
In conclusion, in the absence of HF, the reaction of z-N4FO+ (8) and HN3 (2) viaTSC2/8C3/4/5a has the lowest barrier (24.4 kcal mol−1), and, in the presence of HF, the production of N5+ (5) viaTSC2/3/8C3/3/4/5a has the lowest barrier at the B3LYP level. Although at the G3B3 level the barrier for this reaction is the same (both = 20.7 kcal mol−1) as the production of 4-oxo-N7O+ (15) viaTSC2/3/7C3/3/15, the consumption of 8 might contribute to the transformation of 7 to 8. Compared to the barriers of the reactions of NF2O+ with HN3, the reactions of e-N4FO+ and z-N4FO+ with HN3 must cross higher barriers, which may be one of reasons why the subsequent reactions need excess HN3.
H. Summary of the computational study
In this investigation, the reaction of F2NO+ with HN3 and the subsequent reactions, together with the isomerization and dissociation of N4FO+ and N7O+, have been studied using B3LYP and G3B3 methods. Whether in the presence or absence of HF, the reaction of F2NO+ with HN3 can produce N4FO+ whose primary isomers are e-N4FO+ (7) and z-N4FO+ (8). The decomposition of e-N4FO+ and z-N4FO+ to ONNF+ (and subsequently NO+) crosses a limiting barrier of 21.1 kcal mol−1. In accord with the previous study,4 the self-decomposition barrier of 4-oxo-N7O+viaTS14C4/5a is 39.5 kcal mol−1. Based on the computational results, the formation of N2O and N5+ following the 15N labels can be divided into four cases (Table 2). (1) N4FO+ dissociates directly into N2O and N2F+, and then N5+ is formed by the reaction of N2F+ with HN3; (2) 4-oxo-N7O+ dissociates into N2O and N5+ itself; (3) 4-oxo-N7O+ dissociates into N2O and N5+, catalyzed by HN3; and (4) the reaction between N4FO+ and HN3 produces N2O and N5+. Based on the data in Table 2, the first case has the highest barrier, 38.8 kcal mol−1, and is not reasonable. As a matter of fact, its barrier is higher than that of the decomposition to NO+. In the second case, the barrier, 39.5 kcal mol−1, is higher than that of 19.6 kcal mol−1 of the decomposition to N2 and 1-oxo-N5O+. Therefore, if syn–syn-4-oxo-N7O+ (15) (the lowest energy 4-oxo-N7O+ isomer) is formed, it would decompose to NO+ with evolution of N2. Consequently, the formation of N2O and N5+ does not imply that 4-oxo-N7O+ is the necessary intermediate. In the third case, the barrier of 30.6 kcal mol−1 is lower than that of 39.5 kcal mol−1 for the uncatalyzed self-decomposition to N5+, but still higher than that of 19.6 kcal mol−1 for the self-decomposition to NO+. In the fourth case, z-N4FO+ and HN3 produce N2O and N5+ with a barrier of 24.4 kcal mol−1 in the absence of HF and 20.7 kcal mol−1 in its presence, while the 15N labels of the products agree with experiment.4 Based on the analysis of the four cases, 4-oxo-N7O+ has not been formed as an intermediate in the reaction of NF2O+ with excess HN3, and N2O and N5+ are probably formed in the 4th case, i.e., by the HF catalyzed reaction of N4FO+ with HN3. The latter reaction involves with TSC2/3/8C3/3/4/5a a cyclic transition state containing a 1-oxo-N7O+ cation which can decompose to N5+ and N2O with a barrier of only 0.2 kcal mol−1 and results in the experimentally observed 15N labels.4 The adjacently longest N–N distance of TSC2/3/8C3/3/4/5a is the N of α-position of 2 approaching to N of γ-position of 8. The distance computed at B3LYP level is 1.455 Å compared with 1.449 Å for experimental7 (electron diffraction and microwave spectroscopy) N–N single bond length of hydrazine. TSC2/3/8C3/3/4/5a involves a 1-oxo-N7O+ component as 19, and it should decompose to N2O and N5+ with few energy barrier. The comparison between geometries of 19 and TSC2/3/8C3/3/4/5a is shown in Fig. 11. The solvent effects were also estimated, see Table 2. The result of the 4th case as the optimum pathway is not changed. The correctness of this conclusion was experimentally confirmed by the reaction of unlabeled N4FO+ and α- and γ-labeled HN3 which produced unlabeled N2O and α- and γ-15N-labeled N5+ (see below).| Case | Pathway | Barrier | Solvent effect | |
|---|---|---|---|---|
| B3LYP | G3B3 | |||
| a There are three key parameters, static dielectric constant, dynamic dielectric constant, and solvent radius in this model. The polynomial fitting parameters of static dielectric constant on HF are a = 0.50352 × 103, b = −0.19297 × 101 and c = 0.14372 × 10−2. Thus the static dielectric constant of HF at 240 K was set to 123.17. The dynamic dielectric constant is equal to the square of refraction index. The refraction index of HF at 298 K is 1.1574, and the approximate correction factor of temperature is 0.00045. Thus the dynamic dielectric constant of HF at 240 K was set to 1.280. The solvent radius of HF, recommended by tight calculation of molecular volume using the Monte-Carlo integration, was set to 2.41 angstrom. All the data of chemical and physical properties are taken from ref. 7. b From Fig. 2. c From Fig. 6. d From Fig. 7. e From Fig. 10. | ||||
| 1st | 8 → TS8C4/6ab | 36.8 | 38.8 | −0.2 |
| 2nd | 14 → TS14C4/5ac | 36.9 | 39.5 | −3.1 |
| 3rd | C2/14 → TSC2/14C5/20d | 25.8 | 30.6 | 0.2 |
| 4th | C2/3/8 → TSC2/3/8C3/3/4/5ae | 16.0 | 20.7 | −2.9 |
 | ||
| Fig. 11 Computed geometries of 19 and TSC2/3/8C3/3/4/5a at B3LYP/aug-cc-pVDZ level. The bond lengths in angstroms and all the systems possess one positive charge. | ||
I. 15N labeling experiment between unlabeled N4FO+ and α- and γ-labeled HN3
In the original report on 4-oxo-N7O+, the following 15N labeling experiment was carried out using unlabeled NF2O+ and an excess of 50% α- and γ-labeled HN3, with the 15N label being equally distributed over all five N atoms of N5+ and the terminal N of N2O (Scheme 6).The formation of a 4-oxo-N7O+ cation intermediate was proposed4 to explain the experimentally observed labels. However, the predicted barrier for the self-decomposition of 4-oxo-N7O+ to N5+ and N2O was 42.0 kcal mol−1 at the B3LYP/6-311G(2df) level of theory. This high barrier was in stark contrast to our inability to experimentally observe 4-oxo-N7O+, even at low temperatures, by NMR spectroscopy. Based on the extensive computational study described above, an alternate pathway involving the reaction of N4FO+ with HN3 was proposed yielding the correct 15N distribution while at the same time crossing a barrier of only 20.7 kcal mol−1. Furthermore, the predicted cyclic transition state involves a 1-oxo-N7O+ component which can decompose with a barrier of only 0.2 kcal mol−1 to N5+ and N2O. The correctness of the proposed alternate pathway was experimentally tested by the following 15N label experiment between unlabeled N4FO+ and 50% α- and γ-labeled HN3. If 4-oxo-N7O+would be the intermediate (pathways B and C), the products should be N2O with the terminal N being labeled and N5+ with all labels. On the other hand, if the alternate pathway A involving a cyclic 1-oxo-N7O+ intermediate is correct, the 15N should be found exclusively in the central and terminal positions of N5+ and none in N2O (Scheme 7).
The results from this labeling experiment are shown in Fig. 12. It is clearly seen that only pathway A agrees with the observed 15N-labels.
 | ||
| Fig. 12 14N- and 15N-NMR spectra of the products from the reaction of unlabeled N4FO+ and 50% α- and γ-labeled HN3. | ||
The 15N labels are exclusively in the α- and γ-positions of N5+ and none in N2O. In addition to the labeled N5+, strong 15N signals are observed for the protonated form of the α- and γ-labeled HN3 starting material, H2N3+, due to the well-known3,13 equilibrium between HN3 and HF which was used as a solvent for recording the spectra.
Conclusions
The results from both the computational study and the experimental labeling experiment demonstrate that 4-oxo-N7O+ has not been formed in the reaction of NF2O+ with excess HN3. Instead, a competing pathway involving a cyclic transition state TSC2/3/8C3/3/4/5a, containing a 1-oxo-N7O+ cation which can decompose to N5+ and N2O with a barrier of only 0.2 kcal mol−1, produces the observed N5+ and N2O with the correct labels. Although syn–syn-4-oxo-N7O+ is the minimum energy isomer of N7O+ and has reasonably high self-decomposition barriers of 19.6 kcal mol−1 to NO+ and N2, and of 39.5 kcal mol−1 to N5+ and N2O, it is not formed in the reaction of NF2O+ with excess HN3, and different synthetic approaches must be utilized for its synthesis. The correctness of our conclusions was confirmed by two 15N labeling experiments of unlabeled NF2O+ and N4FO+ with α- and γ-labeled HN3 which produced terminally labeled N2O and N5+ with labels in all 5 positions, and unlabeled N2O and α- and γ-labeled N5+, respectively. Our study shows that 1-oxo-N7O+ is much less stable than syn–syn-4-oxo-N7O+, and its instability is responsible for the difficulties to experimentally observe N7O+.Computational methods
The geometry optimization calculations were performed for the reactants, intermediates, transition states and products using DFT (density functional theory) B3LYP method (the exchange–correlation potential is constructed from Becke's three parameter formula for exchange (B3)14 along with the Lee–Yang–Parr parameter for correlation (LYP)15). Then, harmonic frequency analysis calculations were used to verify that each species is a minima or saddle point. To confirm the right connection of each transition state to its forward and backward minima, intrinsic reaction coordinate (IRC)16,17 calculations were carried out. The refined energies of each species was also calculated at the composite G3B3 (ref. 18 and 19) level. For the small molecules and cations, the CCSD(T)20–24 (the single and double excitation coupled cluster theory including triple excitation non-iteratively) and MP2 (ref. 25–29) (the second order perturbation theory) methods combined with the frozen-core option, were also used. Dunning's correlation-consistent basis sets30 aug-cc-pVXZ (X-zeta basis, X = D, T, Q, 5, 6) were used in this study. The moderate basis set aug-cc-pVDZ was combined with B3LYP, and the aug-cc-pVQZ one with MP2 and CCSD(T). The Polarizable Continuum Model31 (PCM) of integral equation form32,33 (IEF) was applied to solvent effect calculations on single point energies based on optimized geometries of important pathways at B3LYP/aug-cc-pVDZ level. The calculation of standard enthalpies of formation involved the design of chemical reaction and calculating their enthalpies of reaction. We employed the reaction that the target compound is formed by its constituent atoms in the gas phases. The temperature for computing free energies was 240 K, in accord with the experiment. The free energy activation barriers were derived from the differences between transition states and complexes of reactants or products. All calculations were carried out using the Gaussian 09 program package.34Experimental section
Caution! neat HN3 is highly explosive and should, whenever possible, be handled only in solution. N4FO+- and N5+-salts react violently with water or organic materials. Anhydrous HF can cause severe burns, and skin contact must be avoided.A. Materials and apparatus
All reactions were carried out in Teflon-FEP ampules that were closed by stainless steel valves. Volatile materials were handled in a stainless steel/Teflon-FEP vacuum line.35 All reaction vessels and the vacuum line were passivated with ClF3 prior to use. Nonvolatile materials were handled in the dry argon atmosphere of a glove box. The 14N and 15N NMR spectra were recorded at room temperature unlocked in anhydrous HF as solvent on a Bruker AMX-500 NMR instrument using a 5 mm broadband probe. Neat CH3NO2 (δ = 0 ppm) was used as an external standard.The starting materials, N4FO+SbF6−3 and HN38 were prepared by literature methods. HF was dried by storage over BiF5.36
B. Reaction of N4FO+SbF6− with HN3
In a typical experiment, N4FO+SbF6− (0.3 mmol) was added to a thin-walled 4 mm o.d. Teflon-FEP ampule, which was closed by a stainless steel valve. On the vacuum line, anhydrous HF (270 mg) was condensed in at −196 °C and the N4FO+SbF6− was dissolved in the HF at room temperature. The ampule was cooled back to −196 °C, and a mixture of HN3 (0.4 mmol) and HF (750 mg) was condensed in. The FEP ampule was heat-sealed and slowly warmed to ambient temperature. It was then inserted into a standard 5 mm o.d. glass NMR tube and transferred to the probe of the NMR spectrometer.Acknowledgements
We acknowledge helpful discussions with Dr Hai-Bo Chang (Tsinghua University) and Dr Xiao-Fang Chen (Dalian Institute of Chemical Physics, CAS). The graphical abstract was designed and produced with the help of Mr Shuo Zhang who graduated from Xi'an Academy of Fine Arts. The work at XAMCRI and FZU was partially supported by the National Science Foundation of China (nos. 21303133 and 51171046). The work at USC was financially supported by the Office of Naval Research and the Defense Threat Reduction Agency.Notes and references
- K. O. Christe, Propellants, Explos., Pyrotech., 2007, 32, 194 CrossRef CAS.
- K. O. Christe, W. W. Wilson, J. A. Sheehy and J. A. Boatz, Angew. Chem., Int. Ed., 1999, 38, 2004 CrossRef CAS.
- W. W. Wilson, R. Haiges, J. A. Boatz and K. O. Christe, Angew. Chem., Int. Ed., 2007, 46, 3023 CrossRef CAS PubMed.
- K. O. Christe, R. Haiges, W. W. Wilson and J. A. Boatz, Inorg. Chem., 2010, 49, 1245 CrossRef CAS PubMed.
- A. Vij, X. Zhang and K. O. Christe, Inorg. Chem., 2001, 40, 416 CrossRef CAS.
- B. P. Winnewisser, J. Mol. Spectrosc., 1980, 82, 220 CrossRef CAS.
- (a) D. R. Lide, CRC handbook of chemistry and physics, CRC Press, Boca Raton, FL, 84th edn, 2003–2004 Search PubMed; (b) NIST Chemistry WebBook.
- A. Vij, W. W. Wilson, V. Vij, F. S. Tham, J. A. Sheehy and K. O. Christe, J. Am. Chem. Soc., 2001, 123, 6308 CrossRef CAS PubMed.
- P. Botschwina, P. Sebald, M. Bogey, C. Demuynck and J.-L. Destombes, J. Mol. Spectrosc., 1992, 153, 255 CrossRef CAS.
- D. A. Dixon, D. Feller, K. O. Christe, W. W. Wilson, A. Vij, V. Vij, H. D. B. Jenkins, R. M. Olson and M. S. Gordon, J. Am. Chem. Soc., 2004, 126, 834 CrossRef CAS PubMed.
- T. Yu, H.-B. Chang, W.-P. Lai and X.-F. Chen, Polym. Chem., 2011, 2, 89230 Search PubMed.
- W.-G. Xu, G.-L. Li, L.-J. Wang, S. Li and Q.-S. Li, Chem. Phys. Lett., 1999, 314, 300 CrossRef CAS.
- K. O. Christe, W. W. Wilson, D. A. Dixon, S. I. Khan, R. Bau, T. Metzenthin and R. Lu, J. Am. Chem. Soc., 1993, 115, 1836 CrossRef CAS.
- A. D. Becke, J. Chem. Phys., 1993, 98, 5648 CrossRef CAS PubMed.
- C. Lee, W. Yang and R. G. Parr, Phys. Rev. B: Condens. Matter Mater. Phys., 1988, 37, 785 CrossRef CAS.
- C. Gonzalez and H. B. Schlegel, J. Chem. Phys., 1989, 90, 2154 CrossRef CAS PubMed.
- C. Gonzalez and H. B. Schlegel, J. Chem. Phys., 1991, 95, 5853 CrossRef CAS PubMed.
- A. G. Baboul, L. A. Curtiss, P. C. Redfern and K. Raghavachari, J. Chem. Phys., 1999, 110, 7650 CrossRef CAS PubMed.
- L. A. Curtiss, K. Raghavachari, P. C. Redfern, V. Rassolov and J. A. Pople, J. Chem. Phys., 1998, 109, 7764 CrossRef CAS PubMed.
- J. Cizek, Adv. Chem. Phys., 1969, 14, 35 CrossRef CAS.
- G. D. Purvis III and R. J. Bartlett, J. Chem. Phys., 1982, 76, 1910 CrossRef PubMed.
- G. E. Scuseria, C. L. Janssen and H. F. Schaefer III, J. Chem. Phys., 1988, 89, 7382 CrossRef CAS PubMed.
- G. E. Scuseria and H. F. Schaefer III, J. Chem. Phys., 1989, 90, 3700 CrossRef CAS PubMed.
- J. A. Pople, M. Head-Gordon and K. Raghavachari, J. Chem. Phys., 1987, 87, 5968 CrossRef CAS PubMed.
- M. Head-Gordon, J. A. Pople and M. J. Frisch, Chem. Phys. Lett., 1988, 153, 503 CrossRef CAS.
- S. Saebø and J. Almlöf, Chem. Phys. Lett., 1989, 154, 83 CrossRef.
- M. J. Frisch, M. Head-Gordon and J. A. Pople, Chem. Phys. Lett., 1990, 166, 275 CrossRef CAS.
- M. J. Frisch, M. Head-Gordon and J. A. Pople, Chem. Phys. Lett., 1990, 166, 281 CrossRef CAS.
- M. Head-Gordon and T. Head-Gordon, Chem. Phys. Lett., 1994, 220, 122 CrossRef CAS.
- T. H. Dunning Jr, J. Chem. Phys., 1989, 90, 1007 CrossRef PubMed.
- J. Tomasi, B. Mennucci and R. Cammi, Chem. Rev., 2005, 105, 2999 CrossRef CAS PubMed.
- J. Tomasi, B. Mennucci and E. Cancès, J. Mol. Struct. THEOCHEM, 1999, 464, 211 CrossRef CAS.
- G. Scalmani and M. J. Frisch, J. Chem. Phys., 2010, 132, 114110 CrossRef PubMed.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery, Jr, J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, , K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, O. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski and D. J. Fox, Gaussian 09, Revision B. 01, Gaussian, Inc., Wallingford CT, 2009 Search PubMed.
- K. O. Christe, W. W. Wilson, C. J. Schack and R. D. Wilson, Inorg. Synth., 1986, 24, 39 CrossRef CAS.
- K. O. Christe, W. W. Wilson and C. J. Schack, J. Fluorine Chem., 1978, 11, 71 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Optimized structures of 1 to 6 at the B3LYP/6-311++G** level, the computed enthalpies of formation in gas phase at 298.15 K for 1 to 6 at the B3LYP/6-311++G** and CBS-QB3 levels, compounds and cations mapping list for their serial numbers and supplementary reaction pathways. See DOI: 10.1039/c4ra02140a |
| This journal is © The Royal Society of Chemistry 2014 |