Tetrahydropyranyl ether (THPE) formation in hydroxyl group protection and conversion to other useful functionalities
Brijesh Kumar
a,
Mushtaq A. Aga
a,
Abdul Rouf
a,
Bhahwal A. Shah
b and
Subhash C. Taneja
*a
aBio-organic Chemistry Division, Indian Institute of Integrative Medicine (CSIR), Canal Road, Jammu, India-180001. E-mail: drsctaneja@gmail.com
bNatural Product Microbe, Indian Institute of Integrative Medicine (CSIR), Canal Road, Jammu, India-180001
First published on 11th April 2014
Abstract
This short review highlights the various methods of formation of tetrahydropyranyl ethers (THPEs) as a method for the protection of simple alcohols as well as a diverse range of complex molecules using a variety of reagents and reaction conditions, i.e., acid catalyst, heterogeneous catalyst and neutral reagent mediated reactions. We also discuss their direct conversion to other useful functionalities.
1. Introduction
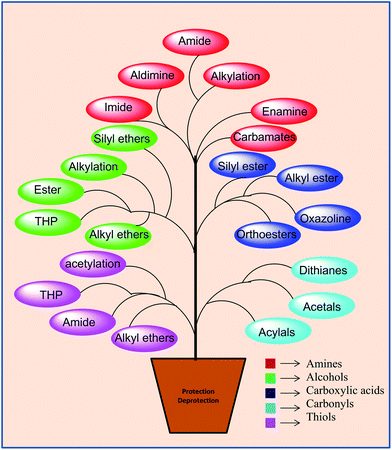
Protecting groups play an indispensable role in the synthesis of complex multifunctional molecules. Continuing efforts worldwide to develop ideal protection methodologies has led to the introduction of a number of protecting groups over the years; also, several books1,35 and reviews2 have appeared on the subject. The functional groups that have attracted the most attention are the amino, thio, carboxylic, carbonyl and hydroxyl groups (Fig. 1).The amino group is found in a number of biologically significant compounds such as peptides, nucleosides, and amino acids; likewise, thiol (–SH) and carboxyl groups also constitute an important part of various drug moieties interacting with receptors or antigens involved in the development of disease.
A tremendous amount of work has gone into developing suitable strategies towards the protection and deprotection of these functional groups. The most frequently used methods for amino group protection are N-alkylation using alkyl halides, amide or imide formation using acetic acid/acetyl chloride/acetic anhydride or phthaloyl anhydride,3 aldimine and enamine formation,4 while thiols are generally protected by acetylation or tetrahydropyranylation (THPRN),5–7 and the protection of carboxylic acids is normally facilitated by ester formation with alcohols,8,9 alkyl halides,10 chloroformates11 and dimethyl carbonate.12
However, in the protection of aldehydes and ketones, a relatively small repertoire of protecting groups has been employed and of these, acetals (O,O), thiocetals (S,S),13 oxathiolanes (O,S),14 1,1-diacetate nitrogenous derivatives (imines, enamines, oximes, hydrazones, semicarbazones)15 and O-methoxycarbonyl-cyanohydrin16 have proven to be the most useful.
2. Hydroxy group protection
Hydroxy compounds (for instance, alcohols, phenols, steroids, and sugars) have an immense significance in our life. Most of these compounds are used for many different scientific, medical and industrial applications. Many compounds with alcoholic and phenolic functionalities are associated with pharmacological activities, for example antitumor (podophyllotoxin, etoposide, taxol, vinca alkaloids, bleomycin, doxorubicin); antibiotic (amoxicillin, kanamycin, neomycin, erythromycin, tetracycline, gentamycin); anti-AIDs (crixivan, zidovudine); cardiac vascular system normalizing (digoxin, digitoxin, gitoxin, quinidine, propranolol, atenolol); anti-pyretic (paracetamol); anaesthetic (propofol); analgesic (morphine); acting on the central nervous system (L-dopa) and as vitamins (pyridoxine/vitamin B6, riboflavin/vitamin B2, ascorbic acid/vitamin C). Moreover, steroids (cholesterol, stigmasterol, oestrone, and testosterone) are also used as medicines; generally, they also bear one or more hydroxyl groups (Fig. 2). The protection of hydroxy groups is a key step in both the synthesis of various polyfunctional organic molecules and in further reactions of these compounds.17Though more than 150 hydroxy-protecting groups have been reported,18 the search for novel OH protective groups is still highly desirable, as molecular targets increase in their complexities and new fields such as supported-oligosaccharide synthesis emerge.19 This has led to the development of a variety of techniques, such as ester formation (acylation, tosylation) and ether formation (silyl ethers, allyl ethers, THP ethers and other alkoxyalkyl ethers), for their protection. Acylation (acetate, benzoate, pivaloate and levulinate formation), the most frequently used method of protection, is generally carried out using carboxylic acids or acyl chlorides or the corresponding anhydrides.
Esterification of carboxylic acids is potentiated by various catalysts such as montmorillonite,20 metal-exchanged as well as LaY zeolites,21 and phosphorus pentoxide supported on silica gel (SiO2/P2O5).22 For acylation with acyl chlorides or anhydrides, the use of bases such as triethylamine, pyridine or 4-(dimethylamino)-pyridine (DMAP) as a catalyst is essential. In the case of base-sensitive substrates, Lewis acids, such as p-toluene-sulfonic acid,23 zinc chloride,24 cobaltous chloride,25 distannoxane,26 scandium triflate27 and silica-supported fluoroboric acid (SiO2/HBF4) are used as catalysts.28 Protic and Lewis acids that are absorbed on different organic or inorganic polymeric materials can also be used; they are prepared by mixing the reagents and the support materials. Chakraborti et al. have reported that acetylation of structurally varied alcohols and phenols is catalyzed competently by numerous perchloric acids absorbed on silica gel (e.g., SiO2/HClO4).29
In the synthesis of various naturally occurring glycosides and other glycol conjugates, protection of the hydroxyl group is an essential step that is effected either by acetylation30 or by levulinate ester formation.31 In addition, pivaloyl esters are formed if high hydrolytic stability of the ester is required. Neutral alumina is used where solventless conditions are needed, and also with microwave irradiations.32 Phenols bearing electron-donating groups can be protected using methyl benzoate in the presence of iron(III) sulfate supported silica, which as such cannot be acetylated.33 Besides, basic alumina is also used as an efficient catalyst for the esterification of phenols even in the absence of solvent, using microwave irradiations for 4–5 min in the presence of pyridine.34
Tosylation is another frequently used method for hydroxyl group protection,35 generally carried out using p-toluenesulfonic acid, sulfonyl chloride and sulfonyl anhydrides in the presence of pyridine, tri-ethylamine and 1,4-diazabicyclo [2.2.2.] octane (DABCO). The assorted alcohols are protected by treating with p-toluenesulfonic acid in the presence of silica chloride.36 Primary hydroxy groups in polyhydroxy compounds can be selectively tosylated in the presence of TsOH in combination with Fe3+-exchanged montmorillonite clay K10.37 In case of symmetrical diols, selectivity of tosylation depends upon the concentration of TsOH in the reaction mixture.
The protection of a hydroxyl group can also be facilitated by forming ethers such as silyl ethers, allyl ethers, benzyl ethers, THP ethers and alkoxyalkyl ethers. The formation of a silyl ether is an excellent method for protection of alcohols and phenols; many silylating methods are known and among them, trimethylsilylation (TMS) and trimethylsilylethoxymethyl (SEM) are commonly used. Trimethylsilylation is generally carried out using hexamethyldisilazane (HMDS) in presence of kaolinitic clay,38 montmorillonite K10,39 envirocat EPZG40 and zirconium sulfophenyl phosphonate.41 The SEM ethers are formed using SEMCl in the presence of alumina-supported potassium fluoride (Al2O3/KF) and can be efficiently used in the protection of both electron-rich and electron-poor phenols;42 however, they cannot be used in the protection of alcohols.
Protection using benzyl ethers, diphenyl-methyl (DPM) ether, 9-fluorenyl ether and allyl ethers are the most frequently used methods in carbohydrate chemistry, since these ethers are very stable under both acidic and basic conditions. Benzyl ethers are formed using benzyl bromide in the presence of NaH,43 whereas DPM ethers are formed by reacting with diphenyl-methanol in the presence of Yb(OTf)3 or FeCl3.44 Although allyl ether formation is the most popular method, its utilization has some limitations because the double bond present in the allyl group makes it susceptible to various reactions, such as halogenations and catalytic hydrogenation.45 Allyl ethers are formed using allyl bromide in the presence of NaH or allyl trichloroacetimidate catalysed by trifloromethane sulfonic acid46 or allyl chloroformate followed by Pd-catalyzed decarboxylation.47 Besides, allyl ethers can be selectively cleaved by oxidation with DDQ.48 Alkoxyalkyl ether formation is another method of protection of hydroxyl groups. It is highly preferable because the alkoxyalkyl ethers are very stable and are resistant to strong bases, alkyllithiums, LAH and Grignard reagents.49 Methoxymethyl (MOM) ethers, methoxypropyl ethers and THP ethers are the most commonly prepared alkoxyalkyl ethers. MOM ethers are prepared by using chloromethyl methyl ether (MOMCl) or dimethoxymethane in the presence of catalysts such as envirocat EPZG,50 montmorillonite K10 (ref. 51) and zeolites52 (e.g., NaY zeolite, acid Y zeolite, ZSM-5, mordenite). As MOMCl is highly carcinogenic, dimemethoxymethane is preferred over it. However, deprotection of MOM ethers requires harsh acidic conditions because of the high stability of such ethers.
Tetrahydropyranyl ethers (THPEs) have found extensive application in organic synthesis as they can be easily synthesized from a variety of hydroxy-group-containing compounds by an acid catalyzed reaction using 3,4-dihydro-2H-pyran (DHP). Tetrahydropyranylation is one of the preferred methods in organic synthesis due to high stability of THP ethers in different reaction conditions, such as strongly acidic or basic pH and presence of oxidizing or reducing agents, in addition to the ease of deprotection (Table 1).
| Bases | LDA | NEt3, Py | t-BuOK | KOH | DCC | Pyridine |
|---|---|---|---|---|---|---|
| Nucleophiles | RLi | RMgX | RCuLi | Enolates | NH3, RNH2 | NaOCH3 |
| Electrophiles | RCOCl | RCHO | CH3I | NXS | CCl2 | Bu3SnH |
| Reduction | H2/Ni | H2/Rh | H2/Pd | Na/NH3 | LiAlH4 | NaBH4 |
| Oxidation | KMnO4 | OsO4 | CrO3/Py | RCOOOH | I2, Br2, Cl2 | MnO2/CH2Cl2 |
THPEs are stable to bases and the deprotection is done through acid hydrolysis. It is important to point out that the introduction of a THP ether onto a chiral molecule results in the formation of diastereoisomers due to an additional stereogenic centre present in the tetrahydropyran ring, which can make both the NMR interpretation and the handling of the reaction products somewhat troublesome.
3. THP as a protecting group
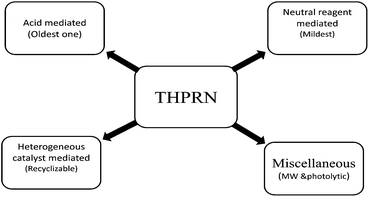
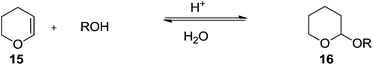
Tetrahydropyranyl ethers are prepared from dihydropyran (versatile vinyl ether) by reacting with alcohols under mild acid catalysis (p-toluenesulfonic acid, or more effectively, boron trifluoride etherate) (Scheme 1).The THP protection can be catalyzed by a wide range of catalysts (Scheme 2) and the methodology may be broadly divided into following categories:
3.1. Acid mediated
3.2. Neutral reagent mediated
3.3. Heterogeneous catalyst mediated
3.4. Miscellaneous
3.1. Acid mediated
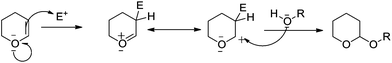
The acid-mediated reaction has a special relevance in numerous chemical reactions. There are several possible chemical compounds that can act as sources for the protons to be transferred in an acid catalysis system. Usually this is done to create a more likely electron abstraction from the double bond of DHP to produce an oxonium ion intermediate, which then abstracts the electron from the nucleophile to produce THP ethers.Usually, tetrahydropyranylation is carried out by acid-catalyzed addition of alcohols and phenols to 2,3-dihydro-2H-pyran (DHP) in an organic solvent at room temperature. Various methods for the formation of THP ethers in acidic conditions have been reported and used frequently for protecting hydroxy groups in multi-step organic synthesis. Most of these reported methods use acidic reagents in an aprotic solvent, such as CH2Cl2, THF, acetone or toluene. In 1934, R. Paul53 observed that 2-methoxytetrahydropyran was formed while adding methyl alcohol to dihydropyran with HCl. Later, Woods and Kramer54 modified the procedure developed by Paul and synthesized a number of acetals from 2,3-dihydropyran, and Schwalm et al.55 used PTSA for THPRN.
THPRN of alcohols has also been reported under solvent-free conditions using catalytic amounts of SnCl2–2H2O (ref. 56) and InCl3 immobilized in 1-butyl-3-methylimidazolium hexafluorophosphate (an ionic liquid) in excellent yields and under mild reaction conditions. The use of ionic liquid offers the advantage of compatibility with a wide range of functional and protecting groups such as THP, TBDMS, TBDPS, PMB, MOM ethers, acetonides, olefins and epoxides. Moreover, aluminum chloride hexahydrate as a catalyst also enables solvent-free THPRN of alcohols and phenols at moderate temperatures with the simple addition of methanol, regenerating the corresponding alcohols and phenols, rendering these protection and deprotection sequences as very efficient transformations at high substrate to catalyst ratios.57 Yadav et al.58 employed THPRN in monoprotection of diols using InCl3. One such method affected highly selective monoprotection of symmetrical diols using a catalytic amount of polystyrene-supported AlCl3.59 Nagaiah et al.60 used niobium(V) chloride to convert a diversity of alcohols and phenols into their corresponding THP-ethers in excellent yields. THPRN of alcohols and phenols with dihydropyran (DHP) has also been performed using a catalytic amount of ZrCl4 in CH2Cl2.61 Pachamuthu and Vankar62 employed CAN in CH3CN at room temperature to prepare the corresponding THP ethers of a variety of alcohols.
Majid et al.63 developed a simple, mild and efficient method of THPRN of alcohols using ferric perchlorate. The protection of hydroxy groups as tetrahydropyranyl ethers and carbonyl functionalities as oxathioacetals and thioacetals has also been achieved using a catalytic amount of silica-supported perchloric acid under solvent-free conditions.64 Bismuth triflate, a non-toxic catalyst, is insensitive to air and small amounts of moisture; it can also be efficiently employed in THPRN under solvent-free conditions.65 Additionally, the CeCl3·7H2O/NaI system under solvent-free conditions can also be employed in highly chemo-selective and environmentally benevolent THPRN of alcohols and phenols.66 This reaction bears a great advantage of being performed under extremely mild conditions.67 Majid and co-workers reported chemo-selective tetrahydropranylation of primary alcohols in the presence of secondary and tertiary alcohols and phenols,68 using tin(IV) porphyrin triflate as catalyst in THF. Babak et al.69 reported THRPN in the presence of a catalytic amount of lithium trifluoromethanesulfonate (LiOTf) and also using Brønsted acidic ionic liquids [BMIm][HSO4] or [BMIm][H2PO4].70 Sulfated zirconia (SO42−/ZrO2) has also been reported to catalyze tetrahydropranylation of alcohols and phenols under solvent-free reaction conditions and reusability of the catalyst.71 In(OTf)3 catalyzed THPRN formation of alcohols in dichloromethane is also described.72 Tetrahydropyranyl ethers can also be synthesized in a mild, chemoselective and convenient fashion, even in the presence of many acid-sensitive functional groups using acetyl chloride and dihydropyran.73
3.2. Neutral reagent mediated
Neutral reaction conditions essentially involve a reaction at room temperature, atmospheric pressure and almost neutral pH. Such reaction conditions generally come with the advantage of having no serious effects on other sensitive (acid/base) functionalities present in the reactants.In 2009, Taneja and co-workers74 used allyl tetrahydropyranyl ether (ATHPE) as a versatile THP-protecting reagent. In combination with NBS/I2, the O-allyl group can be easily substituted by hydroxyls (including tertiary OH) or thiols in presence of other reactive groups such as halogens, nitro, acetonide, and alkenes under mild reaction conditions (near neutral pH and in ambient temperature).
Kotke and co-workers75 reported tetrahydropyranylation of sterically hindered and acid-sensitive substrates in the presence of N,N′-bis[3,5-bis(trifluoromethyl)phenyl]thiourea catalyst. Bartoli et al.76 reported a highly chemoselective method for the protection of free hydroxy compounds with DHP using CeCl3·7H2O/NaI as a catalyst under solvent-free conditions. Molecular iodine has also been employed as a highly efficient catalyst for tetrahydropyranylation using 3,4-dihydro-2H-pyran in DCM at room temperature. It was generated in situ from Fe(NO3)3·9H2O/NaI.77 Sampath et al.78 also used molecular iodine for THP protection. Bernady et al.79 reported non-acidic condition for THP protection using PPh3 and DEAD in THF; this method is also effective for phenols. Olah et al.80 reported synthesis of tetrahydropyranyl ethers with dihydropyran in the presence of Me3SiI under mild, neutral conditions and short reaction times. Bismuth(III) nitrate pentahydrate [Bi(NO3)3·5H2O]81 has also been found to be an effective catalyst for THPRN of alcohols and phenols in the presence of a large number of other protecting groups such as isopropylidene, benzylidene and thioacetal.
Tetrahydropyranylation of primary alcohols has also been selectively carried out in the presence of secondary and tertiary alcohols and phenols using PdCl2(CH3CN)2 as a catalyst in tetrahydrofuran (THF), while other protection groups such as p-toluenesulfonyl, tert-butyldiphenylsilyl, benzyloxycarbonyl, allyl, benzyl, and benzoyl remained intact under these conditions.82 Miyashita et al.83 used PPTS for THPRN under very mild reaction conditions. CuSO4·5H2O has also been reported to bring about smooth conversions of various alcohols and phenols into their corresponding THP ethers under mild reaction conditions.84 Moreover, the preparation and catalytic application of N,N-dibromo-N,N-1,2-dethanediylbis(benzene sulfonamide) for THPRN of different alcohols and phenols has also been reported.85 Catalytic behavior of water-insoluble cesium and rubidium tungstosilicates has also been studied in THPRN of phenols.86 Cesium salts were found to be more active than rubidium salts. THPRN has also been reported using 3,4-dihydro-2H-pyran in the presence of various other catalysts such as anhydrous calcium chloride,87 LiBr,88 dicyanoketene ethylene acetal,89 lithium perchlorate in diethyl ether (LPDE),90 and (TBA)2S2O8.91
3.3. Heterogeneous catalyst mediated
The ion-exchange resin Dowex 50WX4-100 efficiently catalyzed the protection of a variety of alcohols with DHP in dichloromethane at ambient conditions.92 Silica-gel-supported aluminium chloride as a heterogeneous Lewis acid catalyst has proven to be a simple, effective, highly chemoselective and reusable catalyst for the preparation of 2-tetrahydropyranyl ethers of alcohols and phenols.93 Shimizu et al.94 used sulfonic acid group-functionalized silica as a highly effective and reusable catalyst for THPRN of alcohols. Solid silica-based sulfonic acid catalysts have also been employed in the conversion of alcohols and phenols into corresponding THP ether. The catalyst shows high thermal stability (up to 300 °C) and can be recovered and reused for at least eight reaction cycles without loss of reactivity.95 Vanadyl(IV) acetate has also been utilized in the synthesis of THPE of a variety of alcohols, thiols and phenols under mild conditions, with excellent yields at a faster rate in a heterogeneous medium.96 Molybdophosphoric and tungstophosphoric acids supported on silica–alumina, obtained by means of sol–gel method, catalyzed the phenol THPRN in environmentally benign reaction conditions.97Some inexpensive and readily available naturally occurring clays, frequently utilized as competent and versatile catalysts for organic reactions (e.g., K10 clay,98 Spanish sepiolite clay99 or natural kaolinitic clay100), have also been utilized for the THPRN of hydroxy compounds. Likewise, envirocat (an EPZG exhibiting both Brønsted and Lewis acid characteristics)101 and acid zeolites (e.g., Y zeolite with silica–alumina ratio of 4.86)102 have also been utilized for the highly efficient THPRN of alcohols and phenols in short reaction times. Moreover, the protection of phenols and alcohols could be performed under solventless conditions by using zeolites with different SARs (5.9 and 13.9, respectively).103 Corma et al.104 utilized the zeolitic material (ITQ-2) as catalyst to protect alcohols and phenols, including naphthols and steroids. Mesoporous (H-MCM-41) molecular sieves (SAR 51.8) represent another zeolite-type material utilized for the reaction with bulky molecules.105 Sulfuric acid adsorbed on silica gel has also been very efficiently exploited for the preparation of THP ethers of alcohols.106 Steroids, cinnamic and propargylic alcohols were quantitatively converted into the corresponding THP ethers using this reagent system (SiO2/H2SO4).107 The same reaction could be performed selectively using silica chloride, prepared by treating silica with thionyl chloride.108 Additionally, silica-supported Lewis acid SiO2/TaCl5, affected THPRN at a very low concentration and short reaction time.109 This protocol is highly useful in the case of protection of benzyloxy and acid-labile sugar substrates containing acetal groups. Ranu et al. used alumina-supported zinc chloride for THPRN of alcohols through a simple solvent-free reaction.110
Zirconia, in its pure as well as its modified form, has also been employed in microwave-accelerated THPRN of alcohols and phenols. Moreover, THP ethers of allylic and acetylenic alcohols were formed without isomerization of double and triple bonds, as well as bulky substrates, such as cholesterol and naphthols, in high yields and short reaction times. Additionally, treatment of a variety of alcohols and phenols with DHP in the presence of a catalytic amount of sulfated zirconia (ZrO2/SO4) (a popular solid super-acid catalyst that exhibits the highest acid strength) gave corresponding THP ethers in high yields.111 The procedure had also been efficiently applied to highly acid-sensitive alcohols such as allyl and propargyl alcohols.112 This solid catalyst has also been utilized for the THPRN of hydroquinone protected as the benzyl ether.113 Similarly, tetrahydropranylation was also reported with α-Zr(O3PCH3)1.2(O3PC6H4SO3H)0.8, even in presence of C–C double and triple bonds, with the yields not being affected by the steric hindrance of reagents.114 ZrO2-pillared clay (Zr-PILC) has been used for the selective mono THPRN of symmetrical diols and simple alcohols with good selectivity and conversion under solvent-free conditions as a mild recyclable solid Lewis acid catalyst both by heating and microwave irradiation.115
Campelo et al.116 employed AlPO4 as a solid acid catalyst using an excess of DHP for the protection of alcohols and phenols in short reaction times and without the formation of the troublesome oligomeric pyrenes. Sulfated charcoal in combination with a 3 Å molecular sieve has been used in the protection of a variety of alcohols and phenols as THP ethers.117 Recently, tetrahydropyranylation was performed using catalyst supported on organic polymers: e.g., Hon et al.118 used acetonyltriphenylphosphonium bromide (ATPB) supported on polystyrene. Olah et al.119 reported the use of Nafion for the THPRN of primary and secondary alcohols. Zeolite H-beta, as a recyclable acid catalyst, has been suggested as a useful alternative to known methods for the production of THP ethers under mild reaction conditions.120 Small pore size zeolite viz., modified zeolite-type adsorbent E4A, has been found to be a simple, recyclable and environmentally friendly catalyst for THPRN of various alcohols and phenols.121
3.4. Miscellaneous
THP ethers of alcohols have also been prepared by photolysis of DHP, using 1,5-dichloro-9,10-anthraquinone as catalysts under visible light. The reaction could be conducted under ambient fluorescent lighting or with sunlight, as well as in a Rayonet reactor.122Microwave-assisted organic synthesis became an increasingly popular technique in academic and industrial research due to advantages such as especially shorter reaction times and rapid optimization of chemical reactions. In addition, iodine-catalyzed THPRN under microwave irradiation has also been achieved for selective protection of one hydroxyl group in an n-symmetrical diol.123
4. Deprotection of tetrahydropyranyl ethers
The selective removal of a protecting group is of equal importance and significance as is its introduction in an organic synthesis. Acetals and ketals are generally deprotected by reducing them to either ethers or hydrocarbons under a variety of reducing conditions, e.g., trialkylsilanes in the presence of Bronsted or Lewis acids. THP ethers are mixed acetals, whereas for the deprotection of the THP ethers a transacetalization methodology is preferred. Owing to the great impact of THP ethers on the protection of hydroxyl groups, the development of its deprotection methods has also received considerable attention. Liu and Wong124 achieved the deprotection of THP ethers by selectfluor catalysis in an efficient fashion.Bismuth triflate has been found to bring about deprotection of THP ethers of a range of alcohols and phenols under solvent-free conditions.125 Williams et al.126 developed a simple and efficient method for the deprotection of THPE in a facile manner using Al(OTf)3 in the presence of methanol. Also, Tajbakhsh and co-workers127 reported a chemoselective and competent method for the deprotection of THP ethers with a H2O2/Mn(III) Schiff-base complex. Hiromichi et al.128 carried out the reaction of tetrahydropyranyl (THP) ethers with triethylsilyl trifluoromethanesulfonate (TESOTf)-2,4,6-collidine to chemoselectively afford alcohol and 4-triethylsiloxybutanal in good yields. The weakly basic reaction conditions facilitate deprotection without affecting acid-labile protecting groups. Maulide et al.129 achieved chemoselective, catalytic deprotection of tetrahydropyranyl (THP) ethers in the presence of enol triflates by the action of cerium(IV) ammonium nitrate (CAN). Ti(III) chloride was found to be a mild and effective catalyst for the deprotection of tetrahydropyranyl ethers of alcohols and phenols not affecting allyl ether, benzyl ether, tert-butyldiphenylsilyl (TBDPS) ether, p-toluenesulfonate ester and isomerizable double bonds.130 Narender et al.131 reported oxidative deprotection of tetrahydropyranyl ethers with N-bromosuccinimide using (−)-cyclodextrin in water. Pore et al.132 employed silica-sulfuric acid as a reusable solid acid catalyst for the deprotection of tetrahydropyranyl ethers. Various THP ethers can be deprotected to the parent alcoholic or phenolic compounds in CH2Cl2/MeOH (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) by employing bromodimethylsulfonium bromide as catalyst.133 THP ethers of alcohols in absolute MeOH have also been converted to the corresponding alcohols using a catalytic amount of decaborane.134 Mohammadpoor and co-workers135 reported that the treatment of tetrahydropyranyl (THP) ethers with Bi(III) salts like BiCl3, Bi(TFA)3 and Bi(OTf)3 in MeOH provided a simple and efficient process for the conversion of ethers into corresponding alcohols. Cupric chloride dihydrate in MeOH has also been used in the deprotection of tetrahydropyranyl ethers to the corresponding alcohols.136 Tomoko Mineno137 reported indium triflate-mediated deprotection of tetrahydropyran ethers in aqueous MeOH.
2) by employing bromodimethylsulfonium bromide as catalyst.133 THP ethers of alcohols in absolute MeOH have also been converted to the corresponding alcohols using a catalytic amount of decaborane.134 Mohammadpoor and co-workers135 reported that the treatment of tetrahydropyranyl (THP) ethers with Bi(III) salts like BiCl3, Bi(TFA)3 and Bi(OTf)3 in MeOH provided a simple and efficient process for the conversion of ethers into corresponding alcohols. Cupric chloride dihydrate in MeOH has also been used in the deprotection of tetrahydropyranyl ethers to the corresponding alcohols.136 Tomoko Mineno137 reported indium triflate-mediated deprotection of tetrahydropyran ethers in aqueous MeOH.
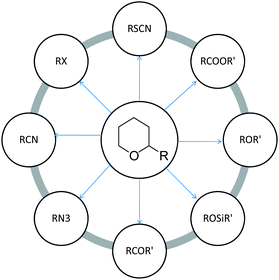
5. Direct conversion of THP ethers in different functionalities
THP ethers have the immense advantage of being easily convertible to corresponding functionalities such as halides, sulphides, esters, cyanides, alkyl ethers, silyl ethers, isothiocyanate and carbonyl compounds using a variety of methods.The interconversion of THPEs into acetate is a useful transformation in organic synthesis. Das et al.138 developed an efficient and direct method for the conversion of THPEs into the corresponding acetates using acetic anhydride in the presence of Amberlyst-15 as a catalyst. Rafiee and co-workers139 converted THPEs derived from primary alcohols into the corresponding acetates and formates by the action of EtOAc, HOAc, acetic anhydride and ethyl formate in the presence of K5CoW12O40·3H2O as catalyst. Tetrahydropyranyl ethers derived from secondary alcohols and phenols could be transformed into the corresponding acetates using acetic anhydride, However K5CoW12O40·3H2O was ineffective for esterification with EtOAc, HOAc, and ethyl formate. Movassagh et al.140 reported conversion of tetrahydropyranyl ethers into their corresponding esters with acid chlorides in the presence of montmorillonite K-10. A green chemical method for the direct conversion of alcohol tetrahydropyranyl ethers into the corresponding acetates has been reported with various substituted acetyl chlorides and sodium iodide in high yields.141 Bi(III) salts such as BiCl3, Bi(TFA)3 and Bi(OTf)3 were found to be efficient catalysts for the transformation of THPEs to their corresponding acetates and formates with acetic acid and ethyl formate.142 Ranu et al.143,144 developed a highly selective conversion of THPEs to acetates by indium tri-iodide. Chandrasekhar et al.145 used TiCl4/Ac2O and Bakos et al.146 used acid chlorides and a catalytic amount of ZnCl2 for the conversion of THPE to acetates.
Direct conversion of alcohol silyl ethers to diphenylmethyl (DPM) ethers can be easily performed by reaction with diphenylmethyl formate in the presence of a catalytic amount of trimethylsilyl trifluoromethanesulfonate.147 The reaction of THP ethers with Ac2O, Bi(NO3)3 and a catalytic amount of DABCO under microwave irradiation led to the corresponding acetates.148
Direct transformation of tetrahydropyranyl ethers into the corresponding halides is an attractive method.149 Sonnet et al.150 developed a protocol for the conversion of the alcohol tetrahydropyranyl ether into a bromide, chloride, methyl ether, nitrile or trifluoroacetate. Various reagents systems, viz., 4-aminophenyldiphenylphosphinite,151 SiO2–Cl/NaI, NaI (or LiBr) BF3–Et2O (or ClSiMe3)152 and CBr4–Ph3P.153 have been widely used for this transformation. A THPE using pyridinium chlorochromate and TFA has been employed for their direct conversion to aldehydes.154
PPh3/DDQ[n-Bu4N]OCN has been used as a reagent system for the conversion of THPEs to the corresponding alkyl isocyanates.155 Nasser et al.156 exploited 4-aminophenyl diphenylphosphinite for the conversion of THPEs to their corresponding thiocyanates or isothiocyanates in the presence of Br2 and NH4SCN. A combination of triphenylphosphine and 2,3-dichloro-5,6-dicyanobenzoquinone has also been provided for the conversion of THPEs to their corresponding thiocyanates.157 In situ generated Ph3P(SCN)2 has also been used for the conversion of THPEs; viz., primary and secondary alkyls and also benzylic to their corresponding thiocyanates.158
Direct conversion of THPEs into the corresponding benzyl ethers can be achieved in one pot with Et3SiH and PhCHO in the presence of a catalytic amount of TMSOTf.159 Oriyama et al.160 developed a reagent system of trialkylsilyl trifluoromethanesulfonate and NEt3 to readily convert THP ethers into corresponding trialkylsilyl ethers, e.g. Ph(CH2)3OSiR3, in good yields. Direct conversion of THP ethers into tert-butyldimethylsilyl ethers has also been facilitated with CF3SO3SiMe2CMe3 and Me2S.161
Akhlaghinia et al.162 reported triphenylphosphine/2,3-dichloro-5,6-dicyanobenzoquinone/tetrabutylammonium azide as an efficient reagent system for conversion of THP ethers to corresponding alkyl azides. For the conversion of tetrahydropyranyl ethers to their corresponding alkyl cyanides, triphenylphosphine/2,3-dichloro-5,6-dicyanobenzoquinone/n-Bu4NCN system has been employed.163 THP ethers of primary fatty alcohols can be converted to the corresponding fatty acids using Jones reagents.164
Fig. 3 summarizes the types of one-step transformation of tetrahydropyranyl ethers into various useful functionalities.
6. Conclusions
This review focussed on hydroxyl group protection via tetrahydropyranylation and deprotection of the resulting THPE, and direct conversion of THPE into various useful functionalities.Acknowledgements
The authors (BK, MAA and AR) thank UGC and CSIR, New Delhi, for the award of senior research fellowships.Notes and references
- T. W. Greene and P. G. M. Wuts, Protective Groups in Organic Synthesis, John Wiley, New York, 3rd edn, 1999 Search PubMed.
- (a) H. Kunz and H. Waldmann, Protecting Groups in Comprehensive Organic Synthesis, ed. B. M. Trost and I. Fleming, Pergamon Press, Oxford, U.K., 1991, p. 631 Search PubMed; (b) M. Schelhaas and H. Waldmann, Angew. Chem., Int. Ed. Engl., 1996, 35, 2056 CrossRef CAS; (c) K. Jarowicki and P. J. Kocieński, J. Chem. Soc., Perkin Trans. 1, 2001, 2109 RSC; (d) A. C. Spivey and R. Srikaran, Annu. Rep. Prog. Chem., Sect. B: Org. Chem., 2001, 97, 41 RSC.
- S. Chandrasekhar, M. Takhi and G. Uma, Tetrahedron Lett., 1997, 38, 8089 CrossRef CAS.
- R. S. Varma, R. Dahiya and S. Kumar, Tetrahedron Lett., 1997, 38, 2039 CrossRef CAS.
- A.-X. Li, T.-S. Li and T.-H. Ding, J. Chem. Soc., Chem. Commun., 1997, 1389 RSC.
- T.-S. Li and A.-X. Li, J. Chem. Soc., Perkin Trans. 1, 1998, 1913 RSC.
- R. Kumareswaran, K. Pachamuthu and Y. D. Vankar, Synlett, 2000, 1652 CAS.
- B. Manohar, V. R. Reddy and B. M. Reddy, Synth. Commun., 1998, 28, 3183–3187 CrossRef CAS.
- H. Yang, B. Li and Y. Fang, Synth. Commun., 1994, 24, 3269–3275 CrossRef CAS.
- J. C. Lee and Y. Choi, Synth. Commun., 1998, 28, 2021 CrossRef CAS.
- P. Gros, P. Le Perchec, P. Gauthier and J. P. Senet, Synth. Commun., 1993, 23, 1835 CrossRef CAS.
- S. R. Kirumakki, N. Nagaraju, K. V. V. S. B. S. R. Murthy and S. Narayanan, Appl. Catal., A, 2002, 226, 175 CrossRef CAS.
- H. K. Patney, Synth. Commun., 1993, 23, 1523 CrossRef CAS.
- (a) B. Pério and J. Hamelin, Green Chem., 2000, 2, 252 RSC; (b) D. E. Ponde, V. H. Deshpande, V. J. Bulbule, A. Sudalai and A. S. Gajare, J. Org. Chem., 1998, 63, 1058 CrossRef.
- (a) R. S. Varma and R. Dahiya, Synlett, 1997, 1245 CrossRef CAS PubMed; (b) A. R. Kiasat, F. Kazemi and K. Nourbakhsh, Phosphorus, Sulfur Silicon Relat. Elem., 2004, 179, 569 CrossRef CAS.
- D. Berthiaume and D. Poirier, Tetrahedron, 2000, 56, 5995 CrossRef CAS.
- (a) A. Studer, S. Hadida, R. Ferritto, S.-Y. Kim, P. Jegar, P. Wipf and D. P. Curran, Science, 1997, 275, 826 CrossRef; (b) B. M. Trost and D. L. Van Vrauken, Chem. Rev., 1996, 96, 422 CrossRef PubMed; (c) V. Amarnath and A. D. Broom, Chem. Rev., 1977, 77, 217 CrossRef.
- P. J. Kocienski, Protecting Groups, 4th edn, Thieme, Stuttgart, 2005 Search PubMed.
- R. Palmacci, M. C. Hewitt and P. H. Seeberger, Angew. Chem., Int. Ed., 2001, 40, 4433 CrossRef.
- B. M. Choudary, V. Bhaskar, M. L. Kantam, K. K. Rao and K. V. Raghavan, Green Chem., 2000, 2, 67 RSC.
- N. Narender, P. Srinivasu, S. J. Kulkarni and K. V. Raghavan, Synth. Commun., 2000, 30, 1887 CrossRef CAS.
- H. Eshghi, M. Rafei and M. H. Karimi, Synth. Commun., 2001, 31, 771 CrossRef CAS PubMed.
- A. C. Cope and E. C. Herrick, Org. Synth., 1963, 4, 304 Search PubMed.
- R. H. Baker and F. G. Bordwell, Org. Synth., 1955, 3, 141 Search PubMed.
- J. Iqbal and R. R. Srivastava, J. Org. Chem., 1992, 57, 2001 CrossRef CAS.
- A. Orita, K. Sakamoto and Y. Hamada, Tetrahedron, 1999, 55, 2899 CrossRef CAS.
- K. Ishihara, M. Kubota, H. Kurihara and H. Yamamoto, J. Am. Chem. Soc., 1995, 117, 4413 CrossRef CAS.
- A. K. Chakraborti and R. Gulhane, Tetrahedron Lett., 2003, 44, 3521 CrossRef CAS.
- A. K. Chakraborti and R. Gulhane, Chem. Commun., 2003, 1896 RSC.
- P. M. Bhaskar and D. Loganathan, Tetrahedron Lett., 1998, 39, 2215 CrossRef CAS.
- N. Barroca and J. C. Jacquinet, Carbohydr. Res., 2000, 329, 679 CrossRef.
- R. S. Varma, M. Varma and A. K. Chatterjee, J. Chem. Soc., Perkin Trans. 1, 1993, 999 RSC.
- T. Nishiguchi and H. Taya, J. Chem. Soc., Perkin Trans. 1, 1990, 172 RSC.
- S. Paul, P. Nanda, R. Gupta and A. Loupy, Tetrahedron Lett., 2002, 43, 4261 CrossRef CAS.
- P. Kocienski, Protecting Groups, Thieme, Stuttgart, Germany, 1994 Search PubMed.
- B. Das, V. S. Reddy and M. R. Reddy, Tetrahedron Lett., 2004, 45, 6717 CrossRef CAS PubMed.
- B. M. Choudary, N. S. Chowdari and M. L. Kantam, Tetrahedron, 2000, 56, 7291 CrossRef CAS.
- T. T. Upadhyay, T. Daniel, A. Sudalai, T. Ravindranathan and K. R. Sabu, Synth. Commun., 1996, 26, 4539 CrossRef.
- Z.-H. Zhang, T.-S. Li, F. Yang and C.-G. Fu, Synth. Commun., 1998, 28, 3105 CrossRef CAS.
- B. P. Bandgar and P. P. Wadgaonkar, Synth. Commun., 1997, 27, 2069 CrossRef CAS.
- M. Curini, F. Epifano, M. C. Marcotullio, O. Rosati and U. Costantino, Synth. Commun., 1999, 29, 541 CrossRef CAS.
- B. E. Blass, C. L. Harris and D. E. Portlock, Tetrahedron Lett., 2001, 42, 1611 CrossRef CAS.
- P. J. Kociensky, Protecting Groups, Georg Thieme, Stuttgart, 2004 Search PubMed.
- G. V. M. Sharma, T. Rajendra Prasad and A. K. Mahalingam, Tetrahedron Lett., 2001, 42, 759 CrossRef CAS.
- R. Gigg and R. Conant, Carbohydr. Res., 1982, 100, c5–c9 CrossRef CAS.
- H.-P. Wessel, T. Iversen and D. R. Bundle, J. Chem. Soc., Perkin Trans. 1, 1985, 2250 Search PubMed.
- F. Guibe and Y. Saint M'Leux, Tetrahedron Lett., 1981, 26, 5048 Search PubMed.
- J. S. Yadav, S. Chandrasekhar, G. Sumithra and R. Kache, Tetrahedron Lett., 1996, 37, 6606 Search PubMed.
- J. P. Yardley and H. Fletcher, Synthesis, 1976, 244 CrossRef CAS PubMed.
- B. P. Bandgar, C. T. Hajare and P. P. Wadgaonkar, J. Chem. Res., Synop., 1995, 90 Search PubMed.
- M. L. Kantam and P. L. Santhi, Indian J. Chem., Sect. B: Org. Chem. Incl. Med. Chem., 1996, 35, 260 Search PubMed.
- P. Kumar, S. V. N. Raju, R. S. Reddy and B. Pandey, Tetrahedron Lett., 1994, 35, 1289 CrossRef CAS.
- R. Paul, Bull. Soc. Chim., 1934, 5, 973 Search PubMed.
- G. Forrest Woods and D. N. Kramer, J. Am. Chem. Soc., 1947, 69, 2246 CrossRef.
- R. Schwalm, H. Binder and D. Funhoff, J. Appl. Polym. Sci., 2000, 78, 208 CrossRef CAS.
- D. Gogoi, N. Baruah and G. Bez, Synth. Commun., 2007, 37, 597 CrossRef.
- V. N. Vasudevan and R. S. Varma, Tetrahedron Lett., 2002, 43, 1146 Search PubMed.
- J. S. Yadav, B. S. V. Reddy and D. Gnaneshwar, New J. Chem., 2003, 27, 204 Search PubMed.
- B. Tamami and B. K. Parvanak, Tetrahedron Lett., 2004, 45, 718 Search PubMed.
- K. Nagaiah, B. V. S. Reddy, D. Sreenu and A. V. Narsaiah, ARKIVOC, 2005, 3, 199 Search PubMed.
- N. Rezai, F. A. Meybodi and P. Saleh, Synth. Commun., 2000, 30, 1799 CrossRef CAS.
- K. Pachamuthu and Y. D. Vankar, J. Org. Chem., 2001, 66, 7513 CrossRef.
- M. M. Heravi, F. K. Behbahani, H. A. Oskooie and S. R. Hekmat, Tetrahedron Lett., 2005, 46, 2545 Search PubMed.
- A. T. Khan, T. Parvin and L. H. Choudhury, Synthesis, 2006, 2502 Search PubMed.
- J. R. Stephens, P. L. Butler, C. H. Clow, M. C. Oswald, R. C. Smith and R. S. Mohan, Eur. J. Org. Chem., 2003, 3827 CrossRef CAS.
- R. Sanz, A. Martinez, G. J. M. Alvarez and F. Rodriquez, Eur. J. Org. Chem., 2006, 1383 CrossRef CAS.
- B. Giuseppe, G. Riccardo, G. Arianna, M. Enrico, M. Massimo, M. Paolo, P. Melissa and S. Letizia, Eur. J. Org. Chem., 2006, 6, 1482 Search PubMed.
- M. Majid, T. Shahram, M. Valiollah, M.-B. Iraj and G. Shadab, Inorg. Chim. Acta, 2010, 363, 1528 Search PubMed.
- K. Babak and M. Jafar, Tetrahedron Lett., 2002, 43, 5355 Search PubMed.
- Z. Duan, Y. Gu and Y. Deng, Synth. Commun., 2005, 35, 1945 Search PubMed.
- B. M. Reddy, P. M. Sreekanth and P. Lakshmanan, J. Mol. Catal. A: Chem., 2005, 237, 100 CrossRef PubMed.
- T. Mineno, Tetrahedron Lett., 2002, 43, 7978 CrossRef.
- C.-E. Yeom, Y. J. Shin and B. M. Kim, Bull. Korean Chem. Soc., 2007, 28, 103 CrossRef CAS.
- B. Kumar, M. A. Aga, D. Mukherjee, S. S. Chimni and S. C. Taneja, Tetrahedron Lett., 2009, 50, 6236 CrossRef CAS PubMed.
- M. Kotke and P. R. Schreiner, Synthesis, 2007, 790 Search PubMed.
- G. Bartoli, R. Giovannini, A. Giuliani, E. Marcantoni, M. Massaccesi, P. Melchiorre, M. Paoletti and L. Sambri, Eur. J. Org. Chem., 2006, 1482 Search PubMed.
- R. Amin, R. Sadegh and K. Ardeshir, Monatsh. Chem., 2009, 140, 667 Search PubMed.
- H. M. S. Kumar, B. V. S. Reddy, E. J. Reddy and J. S. Yadav, Chem. Lett., 1999, 857 CrossRef CAS.
- K. F. Bernady, M. B. Floyed, J. F. Poletro and M. J. Weiss, J. Org. Chem., 1979, 44, 1438 CrossRef CAS.
- G. A. Olah, A. Husain, B. P. Singh, P. Donald and B. Katherine, Synthesis, 1985, 704 Search PubMed.
- A. T. Khan, S. Ghosh and L. H. Choudhury, Eur. J. Org. Chem., 2005, 4896 Search PubMed.
- Y.-G. Wang, X.-X. Wu and Z.-Y. Jiang, Tetrahedron Lett., 2004, 45, 2976 Search PubMed.
- M. Miyashita, L. A. Yoshikoshi and P. A. Griecol, J. Org. Chem., 1977, 42, 3772 CrossRef CAS.
- A. T. Khan, L. H. Choudhury and S. Ghosh, Tetrahedron Lett., 2004, 45, 7894 Search PubMed.
- A. Khazaei, A. Rostami and M. Mahboubifar, Catal. Commun., 2007, 8, 388 Search PubMed.
- G. P. Romanelli, J. C. Autino, M. N. Blanco and L. R. Pizzio, Appl. Catal., A, 2005, 295, 215 CrossRef PubMed.
- B. P. Bandgar, V. S. Sadavarte, L. S. Uppalla and S. V. Patil, Monatsh. Chem., 2003, 134, 428 CrossRef.
- M. A. Reddy, L. R. Reddy, N. Bhanumathi and R. K. Rama, Synth. Commun., 2000, 30, 4328 Search PubMed.
- M. Tsuyoshi and M. Yukio, Synth. Commun., 1995, 25, 1987 Search PubMed.
- B. B. Sobhana and B. K. Kuppuswamy, Tetrahedron Lett., 1998, 39, 9288 Search PubMed.
- C. H. Chul, J. J. Chul, C. Kyu II and K. Y. Hae, Heteroat. Chem., 1995, 6, 338 Search PubMed.
- P. S. Poon, A. K. Banerjee, L. Bedoya, M. S. Laya, E. V. Cabrera and K. M. Albornoz, Synth. Commun., 2009, 39, 3377 CrossRef.
- K. P. Borujeni, Synth. Commun., 2006, 36, 2710 CrossRef.
- K. Shimizu, E. Hayashi, T. Hatamachi, T. Kodamab and Y. Itayama, Tetrahedron Lett., 2004, 45, 5138 Search PubMed.
- B. Karimi and M. Khalkhali, J. Mol. Catal. A: Chem., 2005, 232, 117 CrossRef PubMed.
- B. M. Choudary, V. Neeraja and M. L. Kantam, J. Mol. Catal. A: Chem., 2001, 175, 172 CrossRef.
- G. Romanelli, P. Vázquez, L. Pizzio, N. Quaranta, J. Autino, M. Blanco and C. Cáceres, Appl. Catal., A, 2004, 261, 170 CrossRef PubMed.
- S. Hoyer, P. Laszlo, M. Orlović and E. Polla, Synthesis, 1986, 655 CrossRef CAS.
- J. M. Campelo, A. Garcia, F. Lafont, D. Luna and J. M. Marinas, Synth. Commun., 1994, 24, 1345 CrossRef CAS.
- A. Cornelis and P. Laszlo, Synlett, 1994, 155 CrossRef CAS.
- B. P. Bandgar, S. R. Jagtap, B. B. Aghade and P. P. Wadgaonkar, Synth. Commun., 1995, 25, 2211 CrossRef CAS.
- P. Kumar, C. U. Dinesh, R. S. Reddy and B. Pandey, Synthesis, 1993, 1069 CrossRef CAS.
- R. Ballini, F. Bigi, S. Carloni, R. Maggi and G. Sartori, Tetrahedron Lett., 1997, 38, 4169 CrossRef CAS.
- I. Rodriguez, M. J. Climent, S. Iborra, V. Fornés and A. Corma, J. Catal., 2000, 192, 441 CrossRef CAS.
- K. R. Kloetstra and H. Van Bekkum, J. Chem. Res., Synop., 1995, 26 CAS.
- M. M. Heravi, D. Ajami and M. Ghassemzadeh, Synth. Commun., 1999, 29, 1013 CrossRef CAS.
- F. Chávez and R. Godinez, Synth. Commun., 1992, 22, 159 CrossRef.
- N. Ravindranath, C. Ramesh and B. Das, Synlett, 2001, 1777 CrossRef CAS PubMed.
- S. Chandrasekhar, M. Takhi, Y. R. Reddy, S. Mohapatra, C. R. Rao and K. V. Reddy, Tetrahedron, 1997, 53, 14997 CrossRef CAS.
- B. C. Ranu and M. Saha, J. Org. Chem., 1994, 59, 8269 CrossRef CAS.
- T. Riemer, D. Spielbauer, M. Hunger, G. A. Mekhemer and H. Knozinger, Chem. Commun., 1994, 1181 RSC.
- A. Sarkar, O. S. Yemul, B. P. Bandgar, N. B. Gaikwad and P. P. Wadgaonkar, Org. Prep. Proced. Int., 1996, 28, 613 CrossRef.
- B. M. Reddy and P. M. Sreekanth, Synth. Commun., 2002, 32, 3561 CrossRef CAS PubMed.
- M. Curini, F. Epifano, M. C. Marcotullio and O. Rosati, Tetrahedron Lett., 1998, 39, 8159 CrossRef CAS.
- V. Singh, V. Sapehiyia and G. L. Kad, Catal. Commun., 2004, 5, 468 CrossRef PubMed.
- J. M. Campelo, A. Garcia, F. Lafont, D. Luna and J. M. Marinas, Synth. Commun., 1992, 22, 2335 CrossRef CAS.
- H. K. Patney, Synth. Commun., 1991, 21, 2329 CrossRef CAS.
- Y.-S. Hon, C.-F. Lee, R.-J. Chen and P.-H. Szu, Tetrahedron, 2001, 57, 5991 CrossRef CAS.
- G. A. Olah, A. Husain and B. P. Singh, Synthesis, 1983, 892 CrossRef CAS.
- J. E. Choi and K.-Y. Ko, Bull. Korean Chem. Soc., 2001, 1178 Search PubMed.
- A. Hegedus, I. Vıgh and Z. Hell, Synth. Commun., 2004, 34, 4152 CrossRef PubMed.
- R. P. Oates and P. B. Jones, J. Org. Chem., 2008, 73, 4745 CrossRef PubMed.
- N. Deka and J. C. Sarma, J. Org. Chem., 2001, 66, 1947 CrossRef CAS PubMed.
- J. Liu and C.-H. Wong, Tetrahedron Lett., 2002, 43, 4037 CrossRef CAS.
- J. R. Stephens, P. L. Butler, C. H. Clow, M. C. Oswald, R. C. Smith and R. S. Mohan, Eur. J. Org. Chem., 2003, 3831 Search PubMed.
- D. Williams, G. Bradley, S. B. Simelane, M. Lawton and H. H. Kinfe, Tetrahedron, 2010, 66, 4576 Search PubMed.
- T. Mahmood, H. Rahman, G. Hamid and R. Raheleh, Lett. Org. Chem., 2008, 5, 312 Search PubMed.
- F. Hiromichi, O. Takashi, O. Takuya, S. Yoshinari, K. Ozora, O. Kazuhisa and K. Yasuyuki, Adv. Synth. Catal., 2007, 349, 646 Search PubMed.
- M. Nuno and E. M. Istvan, Synlett, 2005, 14, 2198 Search PubMed.
- A. Nayak and S. Kumar, Synthesis, 2005, 1, 74 Search PubMed.
- M. Narender, M. S. Reddy and K. R. Rao, Synthesis, 2004, 11, 1743 Search PubMed.
- D. M. Pore, U. V. Desai, R. B. Mane and P. P. Wadgaonkar, Synth. Commun., 2004, 34, 2142 CrossRef PubMed.
- A. T. Khan, E. Mondal, B. M. Borah and S. Ghosh, Eur. J. Org. Chem., 2003, 21, 4117 Search PubMed.
- J. Y. Joo, P. E. Soo, L. J. Hee and Y. C. Min, Bull. Korean Chem. Soc., 2002, 23, 792 Search PubMed.
- M. B. Iraj, K. Bahram and K. Sadegh, Synth. Commun., 2002, 32, 1637 Search PubMed.
- D. K. Joju, U. T. Bhalerao and V. B. Rao, Indian J. Chem., Sect. B: Org. Chem. Incl. Med. Chem., 2000, 39, 862 Search PubMed.
- M. Tomoko, Tetrahedron Lett., 2002, 43, 7978 Search PubMed.
- B. Das, M. R. Reddy, K. R. Reddy, R. Ramu and P. Thirupathi, J. Mol. Catal. A: Chem., 2006, 248, 188 Search PubMed.
- E. Rafiee, S. H. Tangestaninejad, M. H. Habibi, I. Mohammadpoor-Baltork and V. Mirkhani, Russ. J. Org. Chem., 2005, 41, 395 CrossRef PubMed.
- B. Movassagh and K. Atrak, Synth. Commun., 2003, 33, 3912 Search PubMed.
- T. Oriyama, K. Kobayashi, T. Suzuki and M. Oda, Green Chem., 2002, 4, 31 RSC.
- I. Mohammadpoor-Baltork and A. R. Khosropour, Synth. Commun., 2002, 32, 2439 Search PubMed.
- B. C. Ranu and A. Hajra, J. Chem. Soc., Perkin Trans. 1, 2001, 18, 2265 Search PubMed.
- B. C. Ranu and A. Hajra, J. Chem. Soc., Perkin Trans. 1, 2001, 4, 357 Search PubMed.
- S. Chandrasekhar, T. Ramachandar, M. V. Reddy and T. Mohamed, J. Org. Chem., 2000, 65, 4731 CrossRef.
- (a) T. Bakos and I. Vincze, Synth. Commun., 1989, 19, 528 CrossRef; (b) S. Kim and W. J. Lee, Synth. Commun., 1986, 16, 665 Search PubMed.
- T. Suzuki, K. Kobayashi, K. Noda and T. Oriyama, Synth. Commun., 2001, 31, 2766 CrossRef PubMed.
- K. Asadolah, M. M. Heravi and R. Hekmatshoar, Russ. J. Org. Chem., 2009, 45, 1111 CrossRef.
- (a) S. Kim and J. H. Park, J. Org. Chem., 1988, 53, 3111 CrossRef CAS; (b) A. Tanaka and T. Oritani, Tetrahedron Lett., 1997, 38, 7223 CrossRef CAS; (c) Y. C. Hwang and F. W. Fowler, J. Org. Chem., 1985, 50, 2719 CrossRef CAS; (d) G. W. Brown, in The Chemistry of the Hydroxyl Group, ed. S. Patai, Interscience, London, 1971, Part 1, p. 592 Search PubMed; (e) S. Wolfe, J. C. Godfrey, C. T. Holdrege and Y. G. Perron, Can. J. Chem., 1968, 46, 2549 CrossRef CAS.
- P. E. Sonnet, Synth. Commun., 1976, 6, 26 CrossRef.
- F. Habib, G. Mohammad and I. Nasser, Can. J. Chem., 2006, 84, 1012 Search PubMed.
- Y. D. Vankar and K. Shah, Tetrahedron Lett., 1991, 32, 1084 CrossRef.
- A. Wagner, M. P. Heitz and C. Mioskowski, Tetrahedron Lett., 1989, 30, 558 Search PubMed.
- P. E. Sonnet, Org. Prep. Proced. Int., 1978, 10, 94 CrossRef.
- B. Akhlaghinia and S. Samiei, Phosphorus, Sulfur Silicon Relat. Elem., 2009, 184, 2529 Search PubMed.
- I. Nasser, F. Habib and G. Mohammad, Phosphorus, Sulfur Silicon Relat. Elem., 2009, 184, 2019 Search PubMed.
- I. Nasser, F. Habib and N. Najmeh, Tetrahedron, 2006, 62, 5501 Search PubMed.
- I. Nasser, F. Habib and S. H. Reza, Phosphorus, Sulfur Silicon Relat. Elem., 2005, 180, 2096 Search PubMed.
- T. Suzuki, K. Ohashi and T. Oriyama, Synthesis, 1999, 9, 1563 Search PubMed.
- T. Oriyama, K. Yatabe, S. Sugawara, Y. Machiguchi and G. Koga, Synlett, 1996, 6, 525 Search PubMed.
- S. Kim and I. S. Kee, Tetrahedron Lett., 1990, 31, 2900 Search PubMed.
- B. Akhlaghinia, Phosphorus, Sulfur Silicon Relat. Elem., 2005, 180, 1604 CrossRef.
- B. Akhlaghinia, Phosphorus, Sulfur Silicon Relat. Elem., 2004, 179, 1786 Search PubMed.
- R. Gruiec, N. Noiret and H. Patin, J. Am. Oil Chem. Soc., 1995, 72, 1085 CrossRef.
| This journal is © The Royal Society of Chemistry 2014 |