Three novel d7/d10 metal complexes with N-heterocyclic ligand of 2,6-bis(3-pyrazolyl)pyridine: synthesis, structure, surface photovoltage spectroscopy and photocatalytic activity†
Ya Nan Houa,
Zhi Nan Wanga,
Feng Ying Bai*b,
Qing Lin Guana,
Xuan Wanga,
Rui Zhanga and
Yong Heng Xing*a
aCollege of Chemistry and Chemical Engineering, Liaoning Normal University, Huanghe Road 850#, Dalian City, 116029, P. R. China. E-mail: yhxing2000@yahoo.com; Fax: +86-0411-82156987; Tel: +86-0411-82156987
bCollege of Life Science, Liaoning Normal University, Dalian 11602, P. R. China. E-mail: baifengying2000@163.com; Fax: +86-0411-82156987; Tel: +86-0411-82156987
First published on 4th April 2014
Abstract
By introducing dicarboxylate acid ligands with flexible chains of different lengths as a second organic ligand, three complexes [Co2(μ2-ox)(H2L)2(HCOO)2] (1), [Co2(μ4-ad)0.5(HL)2]·(OH)·(H2O) (2), [Zn2(μ4-sub)0.5(HL)2]·(OH)·(H2O) (3) (H2L = 2,6-di-(5-phenyl-1H-pyrazol-3-yl)pyridine, ox = oxalic acid, ad = adipic acid, sub = suberic acid) were synthesized by a hydrothermal method. All complexes were characterized by elemental analysis, IR and UV-vis spectroscopy and single-crystal X-ray diffraction. Structural analysis reveals that in complex 1, the Co(II) atom was six-coordinated to form a distorted octahedron, while in complexes 2 and 3, the Co(II) and Zn(II) atoms were five-coordinated to form disordered tetragonal coordination geometries. Acting as a bridged ligand, the flexible dicarboxylate acid ligand in complex 1 coordinated with two metal atoms, while in complexes 2 and 3 it coordinated with four metal atoms. The surface photovoltage spectroscopy and photocatalytic activities of complexes 1, 2 and 3 were also investigated in detail.
Introduction
Pyrazole derivatives are an important family of organic photochromic compounds and have drawn considerable attention of chemical researchers especially in the field of metal–organic complex synthesis over the past few decades.1 Among these derivatives, pyridine-based tridentate compounds are very interesting polydentate N-heterocyclic ligands for coordination chemistry (Fig. 1).2 Therefore, it is necessary to conduct detailed studies to extend the knowledge of relevant structural types and establish proper synthetic strategies to design architectures with desirable physical properties.3 The conclusion that pyrazole derivatives could be regarded as a polydentate heterocyclic ligand is derived from the following considerations: (i) the three N atoms of the pyridine ring and two adjacent pyrazole rings could coordinate with metal atoms more easily and form stable tridentate complexes. Moreover, the remaining two N atoms of the two pyrazole rings could coordinate with other metal atoms to form binuclear, tetranuclear, and some more complicated complexes,4 for example, [{Co(H2L)2}(H2L)4][PF6]2 (H2L = 2,6-bis(5-phenylpyrazol-3-yl)-pyridine),5 [Fe6(bpp)4(μ3-O)2(μ-OMe)3(μ-OH)Cl2] (H2bpp = 2,6-bis(pyrazol-3-yl)pyridine),6 and [Cd2(μ-Tab)2(Tab)2(bdmppy)]2(PF6)8·H2O (TabH = 4-(trimethylammonio)benzenethiol, bdmppy = 2,6-bis(3,5-dimethyl-1H-pyrazol-1-yl)pyridine).3e (ii) There is a tautomeric equilibrium of the bis-pyrazole compound in solution indicating that the H atom of pyrazolyl NH can transfer to the adjacent N atoms.7 Hence, the ligand might lose one H atom on the pyrazole ring, two H atoms of two pyrazole rings or lose no H atom during the formation of the complex to keep the oxidation state balanced. (iii) They are commercially available or straightforward to synthesize. (iv) Particularly, the H atoms on 3-, 4- and 5- positions of the pyrazoyl ring are easily to be substituted. Furthermore, if these positions are substituted by phenyl rings or pyridine rings, it is easy to form a bulky conjugating system. Complexes based on these kinds of ligands, which possess bulky conjugating systems, present good properties of photoluminescence and photoelectric effect on the surface. By the ideas mentioned above, recently, we have successfully synthesized a series of complexes with pyridine-based pyrazole derivatives as ligands, 2,6-di-(5-phenyl-1H-pyrazol-3-yl)pyridine and 2,6-di-(5-methyl-1H-pyrazol-3-yl)pyridine (Fig. 2).3a,8 These two kinds of ligands commonly exhibit two kinds of coordination modes: quadridentate coordination mode (μ4-η1–η1–η1–η1) and trident chelate coordination mode.2b,9 | ||
| Fig. 2 Structure of 2,6-di-(5-phenyl-1H-pyrazol-3-yl)pyridine and 2,6-di-(5-methyl-1H-pyrazol-3-yl)pyridine. | ||
On the other hand, dicarboxylic acid ligands have been also widely used as ligands in metal coordination chemistry because of the following interesting features: (i) the two carboxylate groups are capable of adopting bidentate, monodentate or tridentate linking modes with the metal atoms; (ii) the possibility of obtaining mono- or di-anionic forms and (iii) the possibility to form secondary building blocks.10 To our knowledge, although number of metal coordination polymers with the rigid (R) and flexible (F) dicarboxylate acid ligands were reported,11 N-heterocyclic-transition metal complexes with flexible (F) dicarboxylate acid ligands of different lengths as an auxiliary ligand have been rarely reported.
Complexes based on these pyridine-based pyrazole derivatives have been applied in many fields such as the catalytic transfer hydrogenation of ketones,12 homogeneous catalysis,13 and biological activity.14 In particular, the ligands synthesized by us possess a bulky conjugating system, composed of one pyridine ring and two pyrazole rings, which made them widely researched in the field of photochromic materials. However, study about the photoelectric effect on the surface and photocatalytic activity of this kind of complex has been rarely reported.15 In this paper, we synthesized three transition metal complexes, i.e., [Co2(μ2-C2O4)(H2L)2(HCOO)2] (1), [Co2(μ4-ad)0.5(HL)2]·(OH)·(H2O) (2), and [Zn2(μ4-su)0.5(HL)2]·(OH)·(H2O) (3), with H2L ligands and performed studies of the structure, IR spectra, UV-vis spectra, and photoluminescent properties. The surface photovoltage spectra and photocatalytic activity of the three complexes were also investigated in detail.
Experimental section
General considerations
H2L was synthesized according to a modified previously reported method (Scheme S1†).16 A 30% aqueous solution of hydrogen peroxide was used as the primary oxidant. All other chemicals purchased were of reagent grade or better and used without further purification. IR spectra were recorded on a JASCO FT/IR-480 PLUS Fourier transform spectrometer with pressed KBr pellets in the range 200–4000 cm−1. The elemental analyses for C, H, and N were carried out on a Perkin Elmer 240C automatic analyzer. The luminescence spectra were reported on a JASCO F-6500 spectrofluorimeter (solid). UV-vis absorption spectra diffuse reflection was recorded with a JASCO V–570-UV/VIS/NIR spectrophotometer in the 200–2500 nm range. Surface photovoltage spectroscopy (SPS) and field-induced surface photovoltage spectroscopy (FISPS) measurements were conducted on a sample in a sandwich cell (ITO/sample/ITO) with the light source-monochromator-lock-in detection technique.17 Standard p-type silicon flakes were used to adjust the comparative phase, and a xenon lamp was used as an illuminator to supply radiation in the range of 300–800 nm.Synthesis
X-ray crystallographic determination
Suitable single crystals of four compounds were mounted on glass fibers for X-ray diffraction measurements. Reflection data were collected at room temperature on a Bruker AXS SMART APEX II CCD diffractometer with graphite monochromatized Mo Kα radiation (λ = 0.71073 Å). All the measured independent reflections (I > 2σ(I)) were used in the structural analyses, and semi-empirical absorption corrections were applied using the SADABS program.18 Crystal structures were solved by the direct method. All non-hydrogen atoms were refined anisotropically. Hydrogen atoms on carbon and nitrogen were fixed at calculated positions and refined by using a riding model. The hydrogen atoms of coordinated water and water molecules in the lattice were found in difference Fourier maps. All calculations were performed using the SHELX–97 program.19 For 3, C47–C54 of the suberic acid ligand were disordered over two positions with the site occupancy factors of 0.50/0.50. Crystal data and details of the data collection and the structure refinement are given in Table 1. The selected bond lengths and angles around metal atom of complexes 1–3 are listed in Table S1.†| Complex | 1 | 2 | 3 |
|---|---|---|---|
| a R = Σ||Fo| − |Fc||/Σ|Fo|, wR2 = [Σ(w(Fo2 − Fc2)2)/Σ(w(Fo2)2)]1/2; [Fo > 4σ(Fo)].b Based on all data. | |||
| Formula | C50H36N10O8Co2 | C49H39N10O4Co2 | C50H41N10O4Zn2 |
| M (g mol−1) | 1022.75 | 949.76 | 976.67 |
| Crystal system | Monoclinic | Triclinic | Triclinic |
| Space group | Cc | P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) |
P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) |
| a (Å) | 32.762(7) | 11.396(2) | 10.941(2) |
| b (Å) | 10.724(2) | 15.014(3) | 12.616(3) |
| c (Å) | 13.937(3) | 15.094(3) | 17.363(4) |
| α (°) | 90 | 72.47(3) | 105.61(3) |
| β (°) | 114.43(3) | 84.22(3) | 102.34(3) |
| γ (°) | 90 | 81.61(3) | 90.47(3) |
| V (Å3) | 4457.9(15) | 2431.8(8) | 2249.5(8) |
| Z | 4 | 2 | 2 |
| Dcalc (g cm−3) | 1.524 | 1.297 | 1.442 |
| F(000) | 2096 | 978 | 1006 |
| μ (Mo-Kα)/mm−1 | 0.814 | 0.735 | 1.124 |
| θ (°) | 3.21–27.48 | 3.09–27.48 | 3.25–25.35 |
| Reflections collected | 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 916 916 |
23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 324 324 |
18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 402 402 |
| Independent reflections (I > 2σ) | 9063(3844) | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 823(6272) 823(6272) |
8180(4139) |
| Parameters | 631 | 585 | 633 |
| Δ(ρ) (e Å−3) | 0.319 and −0.326 | 1.294 and −0.924 | 1.639 and −0.561 |
| Goodness of fit | 0.985 | 1.055 | 0.956 |
| Ra | 0.0698(0.1862) | 0.0863 (0.1403) | 0.0891(0.1674) |
| wR2b | 0.1162(0.1502) | 0.2504 (0.2919) | 0.2363(0.2902) |
Results and discussion
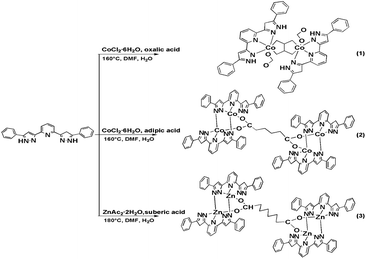
Synthesis
The three complexes were synthesized by the reaction of transition metal salts (CoCl2·6H2O and ZnAc2·2H2O), H2L ligand and dicarboxylate acid ligands of different lengths (oxalic acid, adipic acid and suberic acid) with the molar ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 under hydrothermal conduction (Scheme 1). During the process of these experiments, it was found that a mixture of H2O and DMF is better for crystal growth than that of H2O and EtOH. The crystals of the corresponding complexes were successfully obtained when we adjusted the pH value of the solution to 5 or 6; hence, the pH value of the solution is a very sensitive reaction parameter. It is worth mentioning that the formyl anion in the structure of complex 1 may be produced by DMF hydrolysis in the reaction course. This may be due to the fact that DMF can undergo hydrolysis in a weak acid (pH = 5).20
1 under hydrothermal conduction (Scheme 1). During the process of these experiments, it was found that a mixture of H2O and DMF is better for crystal growth than that of H2O and EtOH. The crystals of the corresponding complexes were successfully obtained when we adjusted the pH value of the solution to 5 or 6; hence, the pH value of the solution is a very sensitive reaction parameter. It is worth mentioning that the formyl anion in the structure of complex 1 may be produced by DMF hydrolysis in the reaction course. This may be due to the fact that DMF can undergo hydrolysis in a weak acid (pH = 5).20
Moreover, powder X-ray diffraction (PXRD) for the complexes 1, 2 and 3 was used to confirm the phase purity of the bulk samples. As shown in Fig. S1–S3,† all the peaks presented in the measured patterns closely match with those in the simulated patterns generated from single crystal diffraction data.
Structural analysis of complexes 1–3
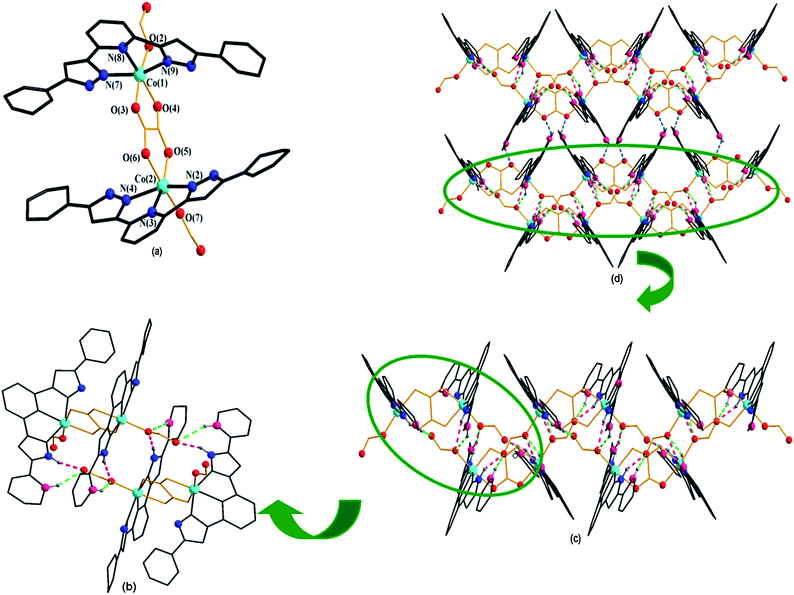
In the structure of the complex 1, the H2L ligand is coordinated with a Co atom in the tridentate coordination mode in a μ1–η1–η1–η1 chelating fashion, i.e., two N atoms from two pyrazolyl rings and one N atom from one pyridine ring. However, the oxalate ligand exhibits a μ2–η1–η1–η1–η1 bridging coordination mode, and the HCOO− group exhibits a terminal coordination mode. The adjacent Co atoms were connected by a bridging oxalate ligand to form a binuclear [Co-2,6-di(5-phenyl-1H-pyrazol-3-yl)pyridine]-oxalate-[Co-2,6-di(5-phenyl-1H-pyrazol-3-yl)pyridine] complex. Pyrazolyl rings from the two sides are distorted to the least-squares plane (central pyridyl ring) with the dihedral angle varying from 1.25(1.15)° to 13.89(1.16)°.
There are three kinds of hydrogen bonds in complex 1: (I) C–H⋯O type between the carbon atom (donor) from H2L and oxygen (acceptor) from a HCOO− group; (II) N–H⋯O type between the nitrogen atom (donor) from H2L and oxygen (acceptor) from HCOO−; and (III) C–H⋯O type between the carbon atom (donor) from H2L and oxygen (acceptor) from oxalate group. By hydrogen bonding of (I) and (II), adjacent molecular units were connected (Fig. 3b) to form an infinite supramolecular chain (Fig. 3c) like a zigzag. Furthermore, two adjacent chains were linked via hydrogen bonds (III) to form a 2D supramolecular network (Fig. 3d). The detailed data of the hydrogen bonds are listed in Table 2.
| D-H⋯A | d (D–H) Å | d (H⋯A) Å | d (D⋯A) Å | ∠DHA (°) |
|---|---|---|---|---|
| a Symmetry transformation used to generate equivalent atoms: #1: x, 1 − y, 0.5 + z; #2: x, 1 − y, −0.5 + z; #3: x, −y, 0.5 + z; #4: x, −y, −0.5 + z; #5: 1 − x, 1 − y, 1 − z. | ||||
| Complex 1 | ||||
| N1–H1A⋯O2#1 | 0.8600 | 1.8700 | 2.7227 | 170.00 |
| N5–H5A⋯O8#2 | 0.8600 | 1.9300 | 2.7466 | 157.00 |
| N6–H6A⋯O7#2 | 0.8600 | 2.0200 | 2.8638 | 165.00 |
| N10–H10A⋯O1#1 | 0.8600 | 1.9200 | 2.7140 | 152.00 |
| C11–H11A⋯O3#3 | 0.9300 | 2.3600 | 3.2299 | 155.00 |
| C23–H23⋯O8#2 | 0.9300 | 2.2500 | 3.1418 | 160.00 |
| C34–H34⋯O6#4 | 0.9300 | 2.3600 | 3.1891 | 148.00 |
| Complex 2 | ||||
| N1–H1A⋯O1W | 0.8600 | 1.9800 | 2.778 | 154.00 |
| N6–H6A⋯O3W#5 | 0.8600 | 1.9400 | 2.771 | 162.00 |
| Complex 3 | ||||
| N6–H6A⋯O1W | 0.8600 | 2.5200 | 3.120 | 127.00 |
| N6–H6A⋯O2W | 0.8600 | 1.8800 | 2.73 | 166.00 |
![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) . Two Co(II) centers, two H2L ligands, a half bridging adipic acid, one lattice water molecule and one lattice OH− group exist in the asymmetric unit of 2 (Fig. 4a). Co1 was five-coordinated by four nitrogen atoms (N2, N3, N4, N10) from two H2L ligands, in which N2, N3 and N4 belong to one H2L ligand while N10 comes from the pyrazole ring of another H2L ligand and one oxygen atom (O2) from the adipic acid ligand to generate a disordered tetragonal pyramid coordination geometry. The coordination environment of Co2 was similar to that of Co1. Co1 and Co2 were connected by four nitrogen atoms (N4, N5, N9 and N10) from two pyrazolyl rings of two H2L ligands and one carboxylate group of adipic acid to form a mixed three-bridging binuclear unit, which further stabilizes the structure of complex 2. In the molecular structure of complex 2 (Fig. 4b), H2L ligand adopted the μ2–ηN1–ηN1–ηN1–ηN1 coordination mode and adipic acid adopted the μ4–ηO1–ηO1–ηO1–ηO1 coordination mode. The contact distance between Co1 and Co2 in the binuclear unit is 3.6197 Å, and the contact distance between Co1 and Co1, which is linked by the adipic acid ligand is 12.4437 Å, while that of Co2 and Co2 is 10.9699 Å.
. Two Co(II) centers, two H2L ligands, a half bridging adipic acid, one lattice water molecule and one lattice OH− group exist in the asymmetric unit of 2 (Fig. 4a). Co1 was five-coordinated by four nitrogen atoms (N2, N3, N4, N10) from two H2L ligands, in which N2, N3 and N4 belong to one H2L ligand while N10 comes from the pyrazole ring of another H2L ligand and one oxygen atom (O2) from the adipic acid ligand to generate a disordered tetragonal pyramid coordination geometry. The coordination environment of Co2 was similar to that of Co1. Co1 and Co2 were connected by four nitrogen atoms (N4, N5, N9 and N10) from two pyrazolyl rings of two H2L ligands and one carboxylate group of adipic acid to form a mixed three-bridging binuclear unit, which further stabilizes the structure of complex 2. In the molecular structure of complex 2 (Fig. 4b), H2L ligand adopted the μ2–ηN1–ηN1–ηN1–ηN1 coordination mode and adipic acid adopted the μ4–ηO1–ηO1–ηO1–ηO1 coordination mode. The contact distance between Co1 and Co2 in the binuclear unit is 3.6197 Å, and the contact distance between Co1 and Co1, which is linked by the adipic acid ligand is 12.4437 Å, while that of Co2 and Co2 is 10.9699 Å.
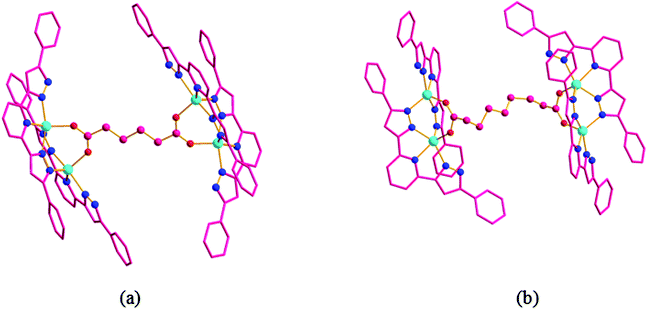 | ||
| Fig. 4 (a) The molecular structure of complex 2; (b) the molecular structure of complex 3 (the lattice water molecules and OH− group were all deleted). | ||
The single-crystal X-ray analysis reveals that complex 3 is also crystallized in the triclinic system with P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) space group, which is the same as complex 2. The structure of complex 3 is similar to that of the complex 2, and the only difference is that central metal of Co atoms and longer dicarboxylate ligand (adipic acid) were replaced by a Zn atom and suberic acid ligand, respectively. The asymmetric unit of 3 (Fig. 4c) is completed by two Zn atoms, two HL2 ligands, a half suberic acid ligand, one lattice formic acid molecule and one lattice water molecule. The coordination environments of zinc atoms are shown in Fig. 4c. Zn(II) is five-coordinated by four N atoms (N2, N3, N4, N10 for Zn1; N5, N7, N8, N9 for Zn2) from two (HL2)− ligands with Zn–N bond lengths in the range of 1.983–2.210 Å and one oxygen atom (O1 for Zn1; O2 for Zn2) from one suberic acid ligand with Zn–O bond lengths of 1.988 and 2.002 Å, to form a distorted tetrahedral geometry, which is best described by the structural parameter τ = 0.308 for Zn1 and 0.255 for Zn2.21 The angles of O–Zn–N and N–Zn–N are in the range of 94.72(11)°–124.23(11)° and 73.97(11)°–148.80(11)°, respectively. Pyrazolyl rings from two sides are distorted to the least-squares plane (central pyridyl ring) with a dihedral angle varying from 0.81(0.61)° to 3.85(0.38)°. In the molecular structure of the complex 3 (Fig. 4d), the flexible suberic acid ligand adopted the μ4-bridging coordination mode in which the carboxylate group of each side adopted the μ2–η1–η1 coordination mode to link four Zn atoms and formed a tetra-nuclear Zn–HL2–sub complex. The adjacent Zn atoms were connected by three different bridging groups from two (HL2)− ligands and one carboxylate group of sub2− ligand with Zn1⋯Zn2 contact distance of 3.68 Å, to form a building unit of Zn2(HL2)2. The Zn2(HL2)2 building unit is further linked by the flexible sub2− ligand with Zn1⋯Zn1 contact distance of 12.02 Å and Zn2⋯Zn2 contact distance of 12.432 Å.
space group, which is the same as complex 2. The structure of complex 3 is similar to that of the complex 2, and the only difference is that central metal of Co atoms and longer dicarboxylate ligand (adipic acid) were replaced by a Zn atom and suberic acid ligand, respectively. The asymmetric unit of 3 (Fig. 4c) is completed by two Zn atoms, two HL2 ligands, a half suberic acid ligand, one lattice formic acid molecule and one lattice water molecule. The coordination environments of zinc atoms are shown in Fig. 4c. Zn(II) is five-coordinated by four N atoms (N2, N3, N4, N10 for Zn1; N5, N7, N8, N9 for Zn2) from two (HL2)− ligands with Zn–N bond lengths in the range of 1.983–2.210 Å and one oxygen atom (O1 for Zn1; O2 for Zn2) from one suberic acid ligand with Zn–O bond lengths of 1.988 and 2.002 Å, to form a distorted tetrahedral geometry, which is best described by the structural parameter τ = 0.308 for Zn1 and 0.255 for Zn2.21 The angles of O–Zn–N and N–Zn–N are in the range of 94.72(11)°–124.23(11)° and 73.97(11)°–148.80(11)°, respectively. Pyrazolyl rings from two sides are distorted to the least-squares plane (central pyridyl ring) with a dihedral angle varying from 0.81(0.61)° to 3.85(0.38)°. In the molecular structure of the complex 3 (Fig. 4d), the flexible suberic acid ligand adopted the μ4-bridging coordination mode in which the carboxylate group of each side adopted the μ2–η1–η1 coordination mode to link four Zn atoms and formed a tetra-nuclear Zn–HL2–sub complex. The adjacent Zn atoms were connected by three different bridging groups from two (HL2)− ligands and one carboxylate group of sub2− ligand with Zn1⋯Zn2 contact distance of 3.68 Å, to form a building unit of Zn2(HL2)2. The Zn2(HL2)2 building unit is further linked by the flexible sub2− ligand with Zn1⋯Zn1 contact distance of 12.02 Å and Zn2⋯Zn2 contact distance of 12.432 Å.
The order of the corresponding bond lengths of M–N are M–Npyridine < M–Npyrazolyl for the complexes 1–3. In addition, pyrazolyl rings from two sides are distorted to the least-squares plane (central pyridine ring) with the different dihedral angles in the range of 0.81(0.61) to 13.89(1.16)°. This may be led by the dicarboxylate acid ligands of different lengths and their various coordination modes.
On the other hand, the comparison of the dicarboxylic acid ligands on the influence of the structures from the complexes 1–3 found that the coordination modes of the dicarboxylic ligands are also different. For ox2− ligand in the complex 1, ox2− ligand presents a μ2–η1–η1–η1–η1 bridging coordination mode linking two Co atoms, whereas the carboxylate group of the ad2− ligand in the complex 2 adopts a μ4–η1–η1–η1–η1 bridging coordination mode to link four Co atoms. The coordination fashion of the suberic acid ligand in the complex 3 is the same as that of the ad2− ligand. The bond length of the M–Ocarboxylate in complex 1 is longer than those of complexes 2 and 3, and the bond length of M–Ocarboxylate of the complex 2 is slightly longer than that of the complex 3. This phenomenon could be also attributed to the different coordination modes of the dicarboxylate acid ligands of different ligands in the three complexes.
IR spectra
In the IR spectrum of complex 1 (Fig. S4†), a broad absorption band appearing at 3439 cm−1 indicates the stretching vibrations of the N–H on the pyrazolyl rings and pyridine rings. A weak peak at 3137 cm−1 can be assigned to the stretching vibrations of the Ar-H on the phenyl rings. A weak peak appears at 3061 cm−1 because of the C–H stretching vibrations of the pyridine/pyrazolyl rings. The bands at 1598 cm−1 and 1310 cm−1 are attributed to the symmetrical stretching vibration and asymmetrical stretching vibration of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds, respectively. Absorptions at 1644 and 1448 cm−1 are the characteristics of stretching vibrations of C
O bonds, respectively. Absorptions at 1644 and 1448 cm−1 are the characteristics of stretching vibrations of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C or C
C or C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N of the phenyl and pyridine rings. The characteristic bands (C–C, C–N) of pyrazolyl and pyridine rings appear at 808 cm−1, 768 cm−1 and 1017 cm−1. IR spectra of complexes 2 and 3 (Fig. S5 and S6†) are similar to that of 1. The detailed attributions of IR (cm−1) for complexes 1–3 are listed in Table S2.†
N of the phenyl and pyridine rings. The characteristic bands (C–C, C–N) of pyrazolyl and pyridine rings appear at 808 cm−1, 768 cm−1 and 1017 cm−1. IR spectra of complexes 2 and 3 (Fig. S5 and S6†) are similar to that of 1. The detailed attributions of IR (cm−1) for complexes 1–3 are listed in Table S2.†
UV-vis spectra
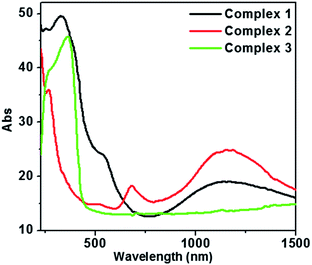
The electronic absorption spectra of complexes 1–3 are shown in Fig. 5. Bands at 254 nm for 1, 268 nm for 2, and 266 nm for 3 are attributed to the π–π* transition of the ligands. It is found that the π–π* transition was slightly blue-shifted as compared to the π–π* transition of H2L. Bands at 330 nm and 420 nm for 1, 340 nm for 2, and 368 nm and 418 nm for 3 are due to the LLCT (inter-ligand charge transfer transition). Broad peaks at 544 nm and 1158 nm for 1 and 506 nm 684 nm and 1156 nm for 2 should be caused by the d–d* transition of Co(II).22 This reveals that the absorption band for 2 is more slightly red-shifted than that of 1, and this phenomenon might be caused by the different coordination modes of the ligands (H2L, oxalic acid and adipic acid).Photoluminescence
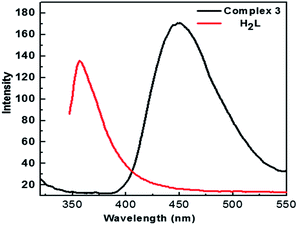
Photoluminescence examination of the Zn-containing compound 3, with a typical d10 transition-metal configuration, was examined at room temperature in the solid state. As shown in Fig. 6, a broad luminescence band at 415 nm was detected in the emission spectrum when it was excited at 365 nm. However, the free H2L ligand shows a strong emission band at 357 nm with an excitation of 337 nm. Accordingly, the emission peak of 3 should be attributed to the metal-to-ligand charge-transfer (MLCT) transition.23Photovoltage surface spectra
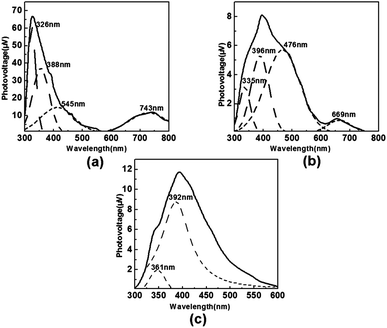
Surface photovoltage (SPV) is the most ordinary and the most sensitive method to study the photo-electric properties. The character of photovoltaic response of an organic semiconductor, inorganic semiconductor, or organic/inorganic semiconductor and the transition or diffusion of electrons on the surface of a solid sample could be detected via SPV,24 and it could be performed by the surface photovoltage spectroscopy (SPS). Surface photovoltage spectroscopy (SPS) is an effective technique to investigate the surface charge behavior of solid samples. SPS is a well-established contactless technique for the characterization of semiconductor materials, which relies on analyzing illumination-induced changes in the surface voltage. The studies had begun in the late 1940s, and after several decades of research, SPS had become a highly sensitive tool to detect the change of surface electronic behaviors for reasons of celerity, facility and without breakage.24,25 It is significant for research on the electronic transition of surfaces and interfaces because it not only relates to the electronic transition under light-inducing, but also reflects the separation and diversion of photo-generated charge. At present, SPV has been employed in the studies of charge transfer in photo-stimulated surface interactions, dye sensitization processes, photo-catalysis electronics, and charge transitions between different phases, surface electronic behaviors and photo-electric conversation. By studying the SPS of the sample, we can not only know the electron transition behavior on surface but also make a judgement of the type of semiconductor sample. In SPS, the detected signal is equivalent to the change in the surface potential barrier on illumination (δVs). The value is calculated by the equation: δVs = Vs − V0s, in which Vs and V0s are the surface potential barriers before and after illumination, respectively. Positive response of SPV (δVs > 0) means that the sample is characterized as a p-type semiconductor, whereas the negative one means the sample is an n-type semiconductor.17By analysis of the UV-vis spectra of the three complexes, we could regard the three compounds as an extended semiconductor. In this paper, we could combine energy-band theory with crystal-field theory to explain the signals of SPS. The central metal atoms of the three compounds coordinated with O atoms or N atoms directly, and we can take the multiple 2s 2p orbitals of O atoms and N atoms as the valence band and 4s 4p empty orbitals of Zn2+ or Co2+ ions as the conduction band. The empty d orbitals of central metal ions could be regarded as the impurity, which is in the region of the valence bond and conduction band.26 The SPS of complexes 1, 2 and 3 are shown in Fig. 7a–c, and some overlapping of signals was fixed after treatment using the Origin7.0 program. In complex 1, the peak at 326 nm was attributed to LLCT and the peak at 388 nm was attributed to LMCT of O → Co and N → Co. The peak at 545 nm and broad band at 743 nm might be caused by d–d* transition. Being similar with complex 1, there were also four peaks in the range of 300–800 nm of complex 2. The peak at 335 nm was attributed to LLCT, and a high peak of 396 nm might be caused by LMCT O → Co and N → Co. The d–d* transition caused the responses at 476 nm and 669 nm. However, in complex 3, only two peaks presented in SPS, and they were caused by LLCT (361 nm) and LMCT (392 nm) of O → Zn and N → Zn, respectively.
It is worth saying that every response of the signals moved to red-shift more or less when compared with the corresponding signals in the UV-vis spectra, and this phenomenon might be caused by the loss of energy in the process of energetic transition. Secondly, in complex 1, the peak of LLCT is twice as high as that of LMCT, while the opposite phenomenon appeared in complexes 2 and 3. By careful analysis and comparison of structures of the three compounds, the reason for this phenomenon should be attributed to the different coordinated types of the three compounds. In complex 1, the central metal atom coordinated with three N atoms from one H2L ligand.
However, in complexes 2 and 3, the central atoms coordinated not only with three N atoms of one H2L ligand but also another N atom from one pyrazole ring of another H2L ligand to form a circular system of (metal atom)–ligand–(metal atom)–ligand, which made it more effective for the electron transition between coordinated ligands and central metal atoms. Furthermore, for the three complexes and their absorption over the light range from 300 nm to 600 nm, this high utilization ratio made them behave as extraordinary potential photoexcitable materials.
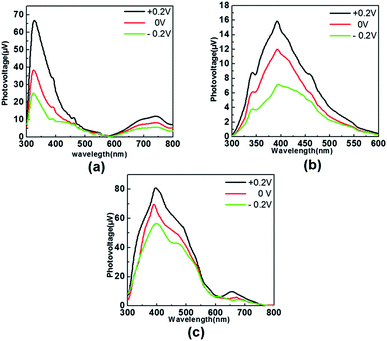
Field-induced surface photovoltage spectroscopy (FISPS) is a means to judge the type of the semiconductor and it can be measured by applying an external electric field to the sample with a transparent electrode.27 The intensity of the photovoltaic response signals was related to the efficiency of the separation of photoexcited electron–hole pairs and a positive electric field is beneficial to increase the efficiency whereas a negative one has the opposite effect. For a p-type semiconductor, when a positive electric field is applied on the semiconductor surface, the SPV response increases since the external field is consistent with the built-in field. On the contrary, when a negative electric field is applied, the SPV response is weakened. In contrast to p-type semiconductors, the SPV response intensity of n-type semiconductors increases as a negative field is applied and reduces as a positive electric field is applied.17 The FISPS results of complexes 1, 2 and 3 are shown in Fig. 8a–c. The intensities of the peaks all increased when the positive electronic (+0.2 V) field was applied, whereas they were reduced when the negative electronic field (−0.2 V) was applied. Hence, they behave as p-type semiconductors.
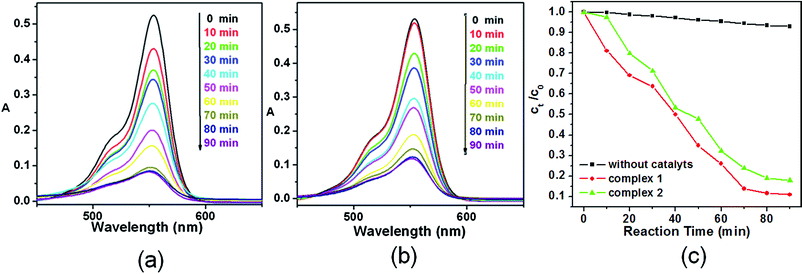
Photocatalytic activity
The photocatalytic activities of complexes 1–3 were studied in detail. Rhodamine B (RhB) was selected as a model dye to evaluate the photocatalytic activities of degeneration during purification in the wastewater. A 18 W Hg lamp was used as the UV light source. The distance between the reaction vessel and the light source was 15 cm. During the process of the decomposition reaction, a UV251 spectrophotometer was used to monitor the reaction under the specific wavelength. To rule out the possibility that the photocatalytic activities of 1 and 2 arise from molecular or oligomeric species formed through dissolution of the solid samples in the photocatalytic reaction systems, controlled experiments were conducted. We filtered the reaction suspensions after 10 h of irradiation to remove the solid catalyst, and fresh RhB was added into the respective filtrates for catalysis testing.Under the condition of the same temperature, intensity of the UV irradiation, concentration of the RhB solution and molar quantity of the catalysts, complexes 1–3 exhibited quite different photocatalytic activities under the UV irradiation. As shown in Fig. 9, approximately 85% and 80% RhB was degenerated in the absence of complexes 1 and 2, respectively, and the RhB solutions turned nearly colorless after about 70 min, indicating that the degeneration process was almost completed. However, nearly no degeneration was observed until 100 min when complex 3 was used as the catalyst. To our knowledge, the photocatalytic effect of MOFs was mainly influenced by factors such as the central metals, the extent of the conjugation, the coordinated environment, the structures of the complexes and the steric hindrance around the active metal centers.28 Although the detailed mechanism of the catalytic processes still needs to be understood by performing further experiments, we assumed that the cause of this phenomenon is the different central metals in this study. During the photocatalytic process of complexes 1 and 2, the highest occupied molecular orbital (HOMO) mainly comes from O and/or N 2p bonding orbitals (valence band) and the lowest unoccupied molecular orbital (LUMO) is from empty Co orbitals (conduction band).28e After irradiation by UV light, one electron in the HOMO was excited to the LUMO. The one-electron-shorted HOMO then extracted one electron from a H2O molecule and lead to the formation of ˙OH active species, which could then complete the process of photocatalytic degeneration. As to the different photocatalytic activities between complexes 1 and 2, these might be caused by the different coordinated environments and structures of complexes 1 and 2.
Conclusions
In this article, we synthesized three complexes using the 2,6-bis(3-pyrazolyl)pyridine as the main ligand and dicarboxylate acid ligands with flexible chains of different lengths as auxiliary ligands. All of the three complexes were characterized by single crystal X-ray diffraction analysis, elemental analysis and IR spectroscopy. The study of surface photovoltage spectra revealed that the three complexes all possessed good responses when the irradiation ranged from 300 nm to 800 nm. These good responses made complexes 1–3 potential p-type semiconductors. As to the investigation of photocatalytic activity, the experimental results revealed that complexes 1 and 2 exhibit good photocatalytic properties, while nearly no degeneration occurred when complex 3 was used as the catalyst. The different photocatalytic activities of complexes 1–3 were mainly caused by the metal centers Co(II) and Zn(II).Acknowledgements
We are very thankful to the Program for the National Natural Science Foundation of China (Grant no. 21371086).Notes and references
- (a) A. P. Sadimenko and S. S. Basson, Coord. Chem. Rev., 1996, 147, 247 CrossRef CAS; (b) R. Mukherjee, Coord. Chem. Rev., 2000, 203, 151 CrossRef CAS; (c) J. P. Zhang and S. Kitagawa, J. Am. Chem. Soc., 2008, 130, 907 CrossRef CAS PubMed; (d) M. A. Halcrow, Dalton Trans., 2009, 2059 RSC.
- (a) B. Schneider, S. Demeshko, S. Dechert and F. Meyer, Angew. Chem., Int. Ed., 2010, 49, 9274 CrossRef CAS PubMed; (b) T. D. Roberts, F. Tuna, T. L. Malkin, C. A. Kilnera and M. A. Halcrow, Chem. Sci., 2012, 3, 349 RSC; (c) J. I. van der Vlugt, S. Demeshko, S. Dechert and F. Meyer, Inorg. Chem., 2008, 47, 1576 CrossRef CAS PubMed.
- (a) Y. B. Zhou and W. Z. Chen, Dalton Trans., 2007, 5123 RSC; (b) S. M. Hawxwell and L. Brammer, CrystEngComm, 2006, 8, 473 RSC; (c) L. Wang, Q. Yang, H. Chen and R. X. Li, Inorg. Chem. Comm, 2011, 14, 1884 CrossRef CAS PubMed; (d) Y. B. Zhou, W. Z. Chen and D. Q. Wang, Dalton Trans., 2008, 1444 RSC; (e) A. X. Zheng, H. F. Wang, C. N. Lu, Z. G. Ren, H. X. Li and J. P. Lang, Dalton Trans., 2012, 41, 558 RSC; (f) B. Zhao, H. M. Shu, H. M. Hu, T. Qin and X. L. Chen, J. Coord. Chem., 2009, 62, 1025 CrossRef CAS; (g) L. T. Ghoochany, S. Farsadpour, Y. Sun and W. R. Thiel, Eur. J. Inorg. Chem., 2011, 23, 3431 CrossRef.
- (a) K. P. Rao, A. Thirumurugan and C. N. R. Rao, Chem.–Eur. J., 2007, 13, 3193 CrossRef CAS PubMed; (b) K. S. Banu, S. Mondal, A. Guha, S. Das, T. Chattopadhyay, E. Suresh, E. Zangrando and D. Das, Polyhedron, 2011, 30, 163 CrossRef CAS PubMed; (c) M. A. Halcrow, Coord. Chem. Rev., 2005, 249, 2880 CrossRef CAS PubMed.
- T. R. Scicluna, B. H. Fraser, N. T. Gorham, J. G. MacLellan and M. Massi, CrystEngComm, 2012, 12, 3422 RSC.
- D. Plaul, E. T. Spielberg and W. Plass, Z. Anorg. Allg. Chem., 2010, 636, 1268 CrossRef CAS.
- B. Zhao, H. M. Shu, H. M. Hu, T. Qin and X. L. Chen, J. Coord. Chem., 2009, 62, 1025 CrossRef CAS.
- (a) D. Rehder, G. Santoni, G. M. Licini, C. Schulzke and B. Meier, Coord. Chem. Rev., 2003, 237, 53 CrossRef CAS; (b) M. Časný and D. Rehder, Dalton Trans., 2004, 839 RSC; (c) D. Rehder, Inorg. Chem. Commun., 2003, 6, 604 CrossRef CAS; (d) M. T. Cocco, V. Onnis, G. Ponticelli, B. Meier, D. Rehder, E. Garribba and G. Micera, J. Inorg. Biochem., 2007, 101, 19 CrossRef CAS PubMed; (e) P. Friedländer, Ber. Dtsch. Chem. Ges., 1909, 42, 765 CrossRef.
- (a) S. Hu, Y. F. Zhao, H. M. Hu and L. Liu, Acta Crystallogr., 2011, 67, 945 Search PubMed; (b) L. J. Wan, C. S. Zhang, Y. H. Xing, Z. Li, N. Xing, L. Y. Wan and H. Shan, Inorg. Chem., 2012, 51(12), 6517 CrossRef CAS PubMed.
- C. N. R. Rao, S. Natarajan and R. Vaidhyanathan, Angew. Chem., Int. Ed., 2004, 43, 1466 CrossRef CAS PubMed.
- (a) K. L. Hou, F. Y. Bai, Y. H. Xing, J. L. Wang and Z. Shi, CrystEngComm, 2011, 13, 3884 RSC; (b) Z. Wang, Y. H. Xing, C. G. Wang, L. X. Sun, J. Zhang, M. F. Ge and S. Y. Niu, CrystEngComm, 2010, 12, 762 RSC; (c) C. G. Wang, Y. H. Xing, Z. P. Li, J. Li, X. Q. Zeng, M. F. Ge and S. Y. Niu, Cryst. Growth Des., 2009, 9(3), 1525 CrossRef CAS.
- (a) G. Zassinovich, G. Mestroni and S. Gladiali, Chem. Rev., 1992, 91, 1051 CrossRef; (b) S. Gladiali and G. Mestroni, in Transition Metals for Organic Synthesis: Building Blocks and Fine Chemicals, ed. M. Beller and C. Bolm, Wiley-VCH, Weinheim, Germany, 1999, p. 97 Search PubMed.
- S. Günnaz, N. Özdemir, S. Dayan, O. Dayan and B. Cetinkaya, Organometallics, 2011, 30, 4165 CrossRef.
- P. Wang, C. H. Leung, D. Ma, S. C. Yan and C. M. Che, Chem.–Eur. J., 2010, 16, 6900 CrossRef CAS PubMed.
- (a) Y. X. Chi, S. Y. Niu, J. Jin, R. Wang and Y. Li, Dalton Trans., 2009, 37, 7653 RSC; (b) Y. X. Chi, S. Y. Niu, Z. L. Wang and J. Jin, Eur. J. Inorg. Chem., 2008, 2336–2343 CrossRef CAS; (c) L. P. Sun, S. Y. Niu, J. Jin, G. D. Yang and L. Ye, Eur. J. Inorg. Chem., 2007, 3845 CrossRef CAS; (d) L. P. Sun, S. Y. Niu, J. Jin, G. D. Yang and L. Ye, Eur. J. Inorg. Chem., 2006, 5130 CrossRef CAS.
- (a) Y. Zhou, W. Chen and D. Wang, J. Chem. Soc., Dalton Trans., 2008, 1444 RSC; (b) D. Plaul, E. T. Spielberg and W. Z. Plass, Z. Anorg. Allg. Chem., 2010, 636, 1268 CrossRef CAS; (c) L. T. Ghoochany, S. Farsadpour, Y. Sun and W. R. Thiel, Eur. J. Inorg. Chem., 2011, 3431 CrossRef; (d) P. Wang, C. H. Leung, D. L. Ma, S. C. Yan and C. M. Che, Chem.–Eur. J., 2010, 16, 6900 CrossRef CAS PubMed; (e) Y. Luo, Z. Chen, R. Tang and L. Xiao, Chem. React. Eng. Technol., 2006, 22, 549 CAS; (f) Y. Luo, L. Xiao, Z. Chen, W. Huang and X. Tang, Chemical Research and Application, 2008, 20, 299 CAS.
- L. P. Sun, S. Y. Niu, J. Jin, G. D. Yang and L. Ye, Eur. J. Inorg. Chem., 2006, 5130 CrossRef CAS.
- G. M. Sheldrick, SADABS, Program for Empirical Absorption Correction for Area Detector Data, University of Göttingen, Göttingen, Germany, 1996 Search PubMed.
- G. M. Sheldrick, SHELXS 97, Program for Crystal Structure Refinement, University of Göttingen, Göttingen, Germany, 1997 Search PubMed.
- J. Marrot, K. Barthelet, C. Simonnet and D. Riou, C. R. Chim., 2005, 8, 971 CrossRef CAS PubMed.
- A. W. Addison and T. N. Rao, J. Chem. Soc., Dalton Trans., 1984, 1349 RSC.
- Z. P. Li, Y. H. Xing, Y. Z. Cao, X. Q. Zeng, M. F. Ge and S. Y. Niu, Polyhedron, 2009, 28, 865 CrossRef CAS PubMed.
- (a) S. L. Li, Y. Q. Lan, J. F. Ma, Y. M. Fu, J. Yang, G. J. Ping, J. Liu and Z. M. Su, Cryst. Growth Des., 2008, 8, 1610 CrossRef CAS; (b) Z. Su, J. Xu, J. Fan, D. J. Liu, Q. Chu, M. S. Chen, S. S. Chen, G. X. Liu, X. F. Wang and W. Y. Sun, Cryst. Growth Des., 2009, 9, 2801 CrossRef CAS; (c) X. L. Wang, C. Qin, E. B. Wang, Y. G. Li, N. Hao, C. W. Hu and L. Xu, Inorg. Chem., 2004, 43, 1850 CrossRef CAS PubMed; (d) X. Y. Cao, J. Zhang, Z. J. Li, J. K. Chen and Y. G. Yao, CrystEngComm, 2007, 9, 806 RSC.
- L. Kronik and Y. Shapira, Surf. Sci. Rep., 1999, 37, 1 CrossRef CAS.
- (a) W. H. Brattain, Phys. Rev., 1947, 72, 345 Search PubMed; (b) W. H. Brattain and J. Bardeen, Bell Syst. Tech. J., 1953, 32, 1 CrossRef; (c) C. G. B. Garrett and W. H. Brattain, Phys. Rev., 1955, 99, 376 CrossRef; (d) L. Kronik and Y. Shapira, Surf. Interface Anal., 2001, 31, 954 CrossRef CAS.
- W. Chen, H. M. Yuan, J. Y. Wang, Z. Y. Liu, J. J. Xu, M. Yang and J. S. Chen, J. Am. Chem. Soc., 2003, 125, 9266 CrossRef CAS PubMed.
- (a) J. Zhang, D. J. Wang, T. S. Shi, B. H. Wang, J. Z. Sun and T. J. Li, Thin Solid Films, 1996, 284/285, 596–599 CrossRef; (b) H. F. Mao, H. J. Tian, Q. F. Hou and H. J. Xu, Thin Solid Films, 1997, 300, 208 CrossRef.
- (a) B. Liu, Z.-T. Yu, J. Yang, H. Wu, Y.-Y. Liu and J.-F. Ma, Inorg. Chem., 2011, 50, 8967 CrossRef CAS PubMed; (b) H. Lin and P. A. Maggard, Inorg. Chem., 2008, 47, 8044 CrossRef CAS PubMed; (c) W.-Q. Kan, B. Liu, J. Yang, Y.-Y. Liu and J.-F. Ma, Cryst. Growth Des., 2012, 12, 2288 CrossRef CAS; (d) Q. Wu, W. L. Chen, D. Liu, C. Liang, Y. G. Li, S. W. Lin and E. Wang, Dalton Trans., 2011, 56 RSC; (e) L.-L. Wen, F. Wang, J. Feng, K.-L. Lv, C.-G. Wang and D.-F. Li, Cryst. Growth Des., 2009, 9, 3581 CrossRef CAS; (f) K. L. Lv and Y. M. Xu, J. Phys. Chem. B, 2006, 110, 6204 CrossRef CAS PubMed; (g) L.-L. Wen, F. Wang, J. Feng, K.-L. Lv, C.-G. Wang and D.-F. Li, Cryst. Growth Des., 2009, 9, 3581 CrossRef CAS; (h) M. Q. Hu and Y. M. Xu, Chemosphere, 2004, 54, 431 CrossRef CAS; (i) K. L. Lv and Y. M. Xu, J. Phys. Chem. B, 2006, 110, 6204 CAS.
Footnote |
| † Electronic supplementary information (ESI) available: The synthesis methods of H2L ligands, IR spectra, PXRD. CCDC 928253–928255. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c4ra02015d |
| This journal is © The Royal Society of Chemistry 2014 |