Efficient and regioselective synthesis of bicyclic pyrrolidones or bicyclic pyridones by cyclocondensation of heterocyclic ketene aminals with nitro-phenylpropiolate†
Juan Fan‡
a,
Qin-Yi Yang‡a,
Guo-Jin Hea,
Xiao-Guang Xiea,
Hong-You Zhua,
Yi Jin*ac and
Jun Lin*ab
aKey Laboratory of Medicinal Chemistry for Natural Resource (Yunnan University), Ministry Education, School of Chemical Science and Technology, Yunnan University, Kunming, 650091, P. R. China. E-mail: jinyi@ynu.edu.cn; linjun@ynu.edu.cn; Fax: +86 871 65033215
bState Key Laboratory of Elemento-organic Chemistry, Nankai University, Tianjin, 300071, P. R. China
cAdvanced Analysis and Measurement Center, Yunnan University, Kunming, 650091, P. R. China
First published on 23rd June 2014
Abstract
Heterocyclic ketene aminals (HKAs) underwent nucleophilic addition to the α, β-unsaturated C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C of nitro-phenylpropiolate, obtaining a series of novel bicyclic pyrrolidones or bicyclic pyridones, whereas the opposite regioselectivity was observed for direct addition of HKAs to the β-C-position or α-C-position of the unsaturated ester; furthermore, this unprecedented nitro-substituted position regulated reactivity was validated by means of Density Functional Theory (DFT) calculations.
C of nitro-phenylpropiolate, obtaining a series of novel bicyclic pyrrolidones or bicyclic pyridones, whereas the opposite regioselectivity was observed for direct addition of HKAs to the β-C-position or α-C-position of the unsaturated ester; furthermore, this unprecedented nitro-substituted position regulated reactivity was validated by means of Density Functional Theory (DFT) calculations.
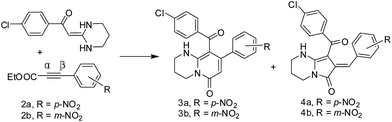
Nucleophilic addition to α, β-unsaturated carbonyl (such as acetylenic esters or alkyl propiolates) compounds has been extensively studied because it is one of the most useful methods in organic synthesis.1 The normal Michael addition predominantly consists of conjugate addition of nucleophiles to the β-C-position of the unsaturated ester. An efficient method of addition to the α-C-position of the unsaturated ester in the presence of a phosphine catalyst (such as triphenyl phosphine) has also been reported in the literatures.2 This two-step phosphine catalysed mechanism was initially inspired by the work of B. M. Trost et al.3 The first step in the process involves a Michael β-C-addition of PPh3 to alkynoate, generating an active phosphonium intermediate after proton exchange, which undergoes nucleophilic α-C-addition of the enolate followed by a H+ transfer and elimination of PPh3 to generate the product. Maury J. and co-workers reported radical addition with phtalimidomethyl iodide mediated by dialkylzinc can also favor regioselective addition of the radical intermediate to the α-C-position of the unsaturated ester.4 The α-C-position and β-C-position addition of organoboronic acids by palladium catalysis to alkyl propiolates has also been reported by Chang Ho Oh and co-workers,5 but they only obtained a mixture of the α-C-adduct and β-C-adduct products. Under catalyst-free and metal-free conditions, effective control of regioselectivity (α-addition vs. β-addition) is essential to maximize the synthetic utility; however, it is rather difficult to accomplish. Heterocyclic ketene aminals (HKAs) are versatile bifunctional nucleophiles for the synthesis of a wide variety of fused heterocyclic compounds; many five and six membered fused heterocycles have been reported.6 Here we report the first example of regioselective cyclocondensation of heterocyclic ketene aminals via α-C-addition or β-C-addition of a p-nitro-phenylpropiolate or m-nitro-phenylpropiolate, respectively.
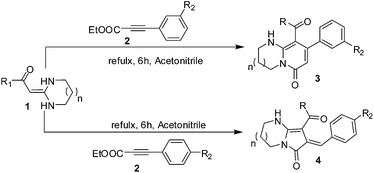
The regioselective cyclocondensation of a series of HKAs with p-nitrophenyl propiolate or m-nitrophenyl propiolate were examined (Table 1). The reaction of 1-(4-chlorophenyl)-2-(tetrahydropyrimidin-2(1H)-ylidene)-ethanone 1a with ethyl 3-(p-nitrophenyl)propiolate 2a or ethyl 3-(m-nitrophenyl)-propiolate 2b was first examined in a variety of solvents (such as 1,4-dioxane, THF, ethyl acetate and acetonitrile) in the presence of alkali catalysts (such as Et3N, Cs2CO3, potassium tert-butoxide and piperidine). The optimal yield and regioselective cyclocondensation of bicyclic pyrrolidone 3a or bicyclic pyridone 3b was obtained (entry 10) when a mixture of 1a and 2b (1.0 equiv.) in acetonitrile free of alkali catalyst was heated at reflux for 6 h. The reaction did not take place in 1,4-dioxane, and gave very low yields in THF and ethyl acetate whether it contained an alkali catalyst or was catalyst-free; however, in acetonitrile it afforded the highest product yield of 84% either with an alkali catalyst or catalyst-free in several hours. Interestingly, the reaction yielded only one α-C-adduct or β-C-adduct in acetonitrile. The cyclocondensation of 1a with ethyl 3-(p-nitrophenyl)propiolate 2a in acetonitrile afforded only the α-C-adduct 4a (bicyclic pyrrolidone) with good yield, and the β-C-adduct 3b (bicyclic pyridone) was prepared from 1a with ethyl 3-(m-nitrophenyl)propiolate (2b) under the same conditions.
| Entry | 2 | Solvent | Catalyst | t (°C) | Time (h) | Yield |
|---|---|---|---|---|---|---|
| 1 | 2b | Dioxane | — | Reflux | 24 | NR |
| 2 | 2b | Dioxane | Et3N | Reflux | 24 | NR |
| 3 | 2b | Dioxane | Cs2CO3 | Reflux | 24 | NR |
| 4 | 2b | Dioxane | (CH3)3COK | Reflux | 24 | NR |
| 5 | 2b | Dioxane | Piperidine | Reflux | 24 | NR |
| 6 | 2b | EtOAc | — | Reflux | 24 | <5% |
| 7 | 2b | EtOAc | Et3N | Reflux | 24 | <5% |
| 8 | 2b | THF | — | Reflux | 24 | (3b) 13% |
| 9 | 2b | THF | Et3N | Reflux | 24 | (3b) 10% |
| 10 | 2b | CH3CN | — | Reflux | 6 | (3b) 84% |
| 11 | 2b | CH3CN | Et3N | Reflux | 6 | (3b) 82% |
| 12 | 2b | CH3CN | Cs2CO3 | Reflux | 6 | (3b) 78% |
| 13 | 2b | CH3CN | (CH3)3COK | Reflux | 6 | (3b) 64% |
| 14 | 2a | CH3CN | — | Reflux | 6 | (4a) 79% |
| 15 | 2a | CH3CN | Et3N | Reflux | 6 | (4a) 80% |
| 16 | 2a | Dioxane | — | Reflux | 6 | NR |
To study the preparative scope of the new methodology, a series of HKAs (1c–q) were systematically varied (Table 2). A nitro-substituted position led to exclusively regioselective generation of bicyclic pyrrolidones or bicyclic pyridone products, and excellent yields were achieved within a few hours.
| Entry | 1 | n | 2 | R2 | Yield |
|---|---|---|---|---|---|
| 1 | (1c) R1 = 4-FC6H4 | 1 | 2b | NO2 | (3c) 91% |
| 2 | (1d) R1 = C6H5 | 1 | 2b | NO2 | (3d) 82% |
| 3 | (1e) R1 = 4-CH3C6H4 | 1 | 2b | NO2 | (3e) 84% |
| 4 | (1f) R1 = 4-CH3OC6H4 | 1 | 2b | NO2 | (3f) 85% |
| 5 | (1g) R1 = 2-ClC6H4 | 1 | 2b | NO2 | (3g) 88% |
| 6 | (1h) R1 = 4-FC6H4 | 2 | 2b | NO2 | (3h) 75% |
| 7 | (1i) R1 = 4-ClC6H4 | 2 | 2b | NO2 | (3i) 74% |
| 8 | (1j) R1 = C6H5 | 2 | 2b | NO2 | (3j) 70% |
| 9 | (1k) R1 = 4-CH3C6H4 | 2 | 2b | NO2 | (3k) 71% |
| 10 | (1l) R1 = 4-CH3OC6H4 | 2 | 2b | NO2 | (3l) 72% |
| 11 | (1c) R1 = 4-FC6H4 | 1 | 2a | NO2 | (4c) 85% |
| 12 | (1e) R1 = 4-CH3C6H4 | 1 | 2a | NO2 | (4e) 82% |
| 13 | (1h) R1 = 4-FC6H4 | 2 | 2a | NO2 | (4h) 74% |
| 14 | (1i) R1 = 4-ClC6H4 | 2 | 2a | NO2 | (4i) 73% |
| 15 | (1k) R1 = 4-CH3C6H4 | 2 | 2a | NO2 | (4k) 70% |
| 16 | (1l) R1 = 4-CH3OC6H4 | 2 | 2a | NO2 | (4l) 72% |
| 17 | (1m) R1 = 4-FC6H4 | 0 | 2a | NO2 | (4m) 75% |
| 18 | (1n) R1 = 4-FC6H4 | 0 | 2a | NO2 | (4n) 75% |
| 19 | (1o) R1 = C6H5 | 0 | 2a | NO2 | (4o) 70% |
| 20 | (1p) R1 = 4-CH3C6H4 | 0 | 2a | NO2 | (4p) 71% |
| 21 | (1q) R1 = 4-CH3OC6H4 | 0 | 2a | NO2 | (4q) 73% |
| 22 | (1q) R1 = 4-CH3OC6H4 | 0 | 2c | CN | (3r) 89% |
| 23 | (1q) R1 = 4-CH3OC6H4 | 0 | 2d | F | (3s) 87% |
From Table 2, the ring size of the amino group and the substituted phenyl group of HKA had a slight influence on the reactivity and product yield. Electron-withdrawing groups (such as, F and Cl) usually gave better yields (Table 2, entries 1 and 5). This may be due to the electron-withdrawing groups increasing the polarization of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C leading to increased electron density on the α-carbon of the carbonyl group, making it a better nucleophile. In addition, six-member HKAs gave better yields than five-member or seven-member HKAs (Table 2, entries 11 to 21). We believe that the six-member HKAs were easier to react with nitro-phenylpropiolate. It is encouraging to note that different ethyl 3-(EWG-phenyl) propionate (such as EWG is 3-CN or 3-F, Table 2, entries 22 to 23) can also react with HKA 1q to get correspondent bicyclic pyridones 3r or 3s, respectively.
C leading to increased electron density on the α-carbon of the carbonyl group, making it a better nucleophile. In addition, six-member HKAs gave better yields than five-member or seven-member HKAs (Table 2, entries 11 to 21). We believe that the six-member HKAs were easier to react with nitro-phenylpropiolate. It is encouraging to note that different ethyl 3-(EWG-phenyl) propionate (such as EWG is 3-CN or 3-F, Table 2, entries 22 to 23) can also react with HKA 1q to get correspondent bicyclic pyridones 3r or 3s, respectively.
To verify the structures of the bicyclic pyrrolidones and bicyclic pyridones derivatives, 3c and 4i were selected as representative compounds and characterized by X-ray crystallography (ESI† and Fig. 1).
 | ||
| Fig. 1 X-ray crystal structures of 3c and 4i;7 ellipsoids are draw at 30% probability level. | ||
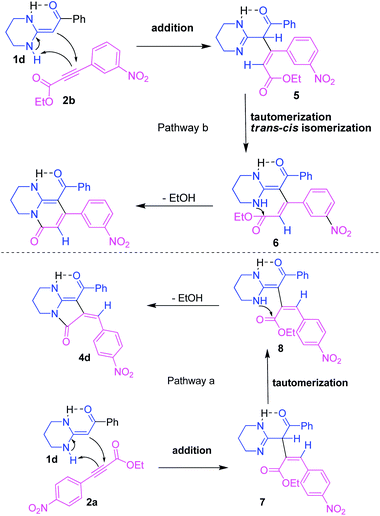
In our proposed mechanism (Scheme 1), the α-C of the ketene N,N-acetal 1d added to the β-C of m-nitro-phenylpropiolate 2b to afford 5. The intermediate 5 was followed by imine–enamine tautomerization and trans–cis isomerization to produce 6. Subsequently, the NH attacked the intramolecular carbonyl group and elimination of EtOH resulted in the target bicyclic pyridone 3d. In contrast, the α-C of the ketene N,N-acetal 1d added to the α-C of p-nitro-phenylpropiolate 2a to afford 7. The intermediate 7 was followed by imine–enamine tautomerization to produce 8. Subsequently, it reacted in the same way to obtain the target bicyclic pyrrolidone 4d.
In order to propose a rationale for these observations, theoretical calculations were performed to further rationalize the unprecedented regioselective cyclocondensation of HKAs with nitro-phenylpropiolate. First, due to the importance of orbital factors in such reactions, the energies of the frontier orbitals of all reactants in the different solvents were calculated at the B3LYP/6-31++G(d,p)8 level of theory (vibrational frequencies were calculated at the same level of theory to ensure that the obtained geometries were minimal). Natural bond orbital (NBO) calculations were used to visualize the molecular orbitals with the Gaussian 03 program package.9
The energy diagram shown in Table 3 enables qualitative comments on how orbital interactions influence reactivity in different solvents. The energy gaps between the HOMO (1d) and the LUMO of 2a and 2b are 3.231 eV and 3.480 eV in 1,4-dioxane, respectively, and 2.944 eV and 3.182 eV in THF. All the interactions are therefore weak and provide little driving force for the addition of 1d to 2a or 2b. In comparison, the energy gaps between the HOMO (1d) of the HKA and the LUMO of 2a and 2b are 2.266 eV and 2.500 eV in acetonitrile.
| THF | Acetonitrile | Dioxane | |
|---|---|---|---|
| LUMO (2a) | −2.303 | −3.011 | −2.986 |
| LUMO (2b) | −2.065 | −2.777 | −2.737 |
| HOMO (1d) | −5.247 | −5.277 | −6.217 |
| Δinter (2a–1d) | 2.944 | 2.266 | 3.231 |
| Δinter (2b–1d) | 3.182 | 2.500 | 3.480 |
This means that the HOMO–LUMO interaction should govern the reactivity and accelerate the addition of 1d to 2a or 2b in acetonitrile.
In addition, full geometry optimizations of the transition states of 1d added to 2a or 2b and their corresponding minima were performed. The transition state structures TS-7 and TS-7′ (Fig. 2) for HKA 1d added to the α-C or β-C of p-nitro-phenylpropiolate 2a through pathway b (Scheme 1), respectively, depict the H atom of the HKA transferring to the C atom of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C in the shortest distance, similar to double bond addition reaction.10 But the relative free energies (Fig. 3) of activation (ΔG) clearly indicate a preference for the attack by HKA 1d on the α-C of p-nitro-phenylpropiolate 2a for both TS (ΔGTS-7 = 56.2 kJ mol−1, ΔGTS-7′ = 28.5 kJ mol−1), affording the thermodynamically more stable product, hence confirming the experimentally observed regioselectivity of the HKA's attack on the α-C of p-nitro-phenylpropiolate 2a. Using the same method of calculation, the relative free energies of activation indicate a preference for the attack by HKA 1d on the β-C of m-nitro-phenylpropiolate 2b, also confirming the experimentally observed regioselectivity of the HKA's attack on the β-C of the m-nitro-phenylpropiolate 2b (see ESI†). Furthermore, NPA charges indicate that the α-C (NPAcharge = 0.139) of p-nitro-phenylpropiolate 2a is considerably more positive than β-C (NPAcharge = 0.036) at the B3LYP/6-31++G(d,p) level of theory, also consistent with the experimentally observed preference.
C in the shortest distance, similar to double bond addition reaction.10 But the relative free energies (Fig. 3) of activation (ΔG) clearly indicate a preference for the attack by HKA 1d on the α-C of p-nitro-phenylpropiolate 2a for both TS (ΔGTS-7 = 56.2 kJ mol−1, ΔGTS-7′ = 28.5 kJ mol−1), affording the thermodynamically more stable product, hence confirming the experimentally observed regioselectivity of the HKA's attack on the α-C of p-nitro-phenylpropiolate 2a. Using the same method of calculation, the relative free energies of activation indicate a preference for the attack by HKA 1d on the β-C of m-nitro-phenylpropiolate 2b, also confirming the experimentally observed regioselectivity of the HKA's attack on the β-C of the m-nitro-phenylpropiolate 2b (see ESI†). Furthermore, NPA charges indicate that the α-C (NPAcharge = 0.139) of p-nitro-phenylpropiolate 2a is considerably more positive than β-C (NPAcharge = 0.036) at the B3LYP/6-31++G(d,p) level of theory, also consistent with the experimentally observed preference.
 | ||
| Fig. 2 Optimized transition state structure. TS-7: 1d added to α-C of 2a; TS-7′: 1d added to β-C of 2a. | ||
 | ||
| Fig. 3 Free energy profile for HKAs 1d attack on α-C of p-nitro-phenylpropiolate 2a (red line); and HKAs 1d attack on β-C of p-nitro-phenylpropiolate 2a (black line). | ||
Conclusions
In conclusion, the efficient and regioselective synthesis of bicyclic pyrrolidones or bicyclic pyridones by cyclocon-densation of heterocyclic ketene aminals with nitro-phenylpropiolate has been described for the first time. The nitro-substituted position of phenylpropiolate can regulate the ketene N,N-acetals 1a–q added to the α-C or β-C of nitro-phenylpropiolate (2a and 2b). The observed regioselectivity was rationalized by means of orbital factors and DFT calculations, and the results described show again how the reactivity of HKA compounds can be tuned depending upon the synthetic goals. Future work will focus on analyzing more of the differences between the substituted group on phenylpropiolate responsible for the differences in regioselectivity for different HKAs' attacks on the phenylpropiolate.Acknowledgements
The authors gratefully acknowledge the Program for Changjiang Scholars and Innovative Research Team in University (IRT13095), the National Natural Science Foundation of China (nos. U1202221, 21262042, 21262043, 20902079 and 81160384), and thank the High Performance Computing Center at Yunnan University for use of the high performance computing platform.Notes and references
- (a) M. Hatano, T. Horibe and K. Ishihara, Angew. Chem., Int. Ed., 2013, 52, 4549–4553 CrossRef CAS PubMed; (b) S. Kobayashi, P. Xu, T. Endo, M. Ueno and T. Kitanosono, Angew. Chem., Int. Ed., 2012, 51, 12763–12766 CrossRef CAS PubMed; (c) M. Fernández, U. Uria, J. L. Vicario, E. Reyes and L. Carrillo, J. Am. Chem. Soc., 2012, 134, 11872–11875 CrossRef PubMed; (d) T. Nishimura, Y. Takiguchi and T. Hayashi, J. Am. Chem. Soc., 2012, 134, 9086–9089 CrossRef CAS PubMed.
- (a) M. Hanédanian, O. Loreau, M. Sawicki and F. Taran, Tetrahedron, 2005, 61, 2287–2294 CrossRef; (b) H. Liu and H. Jiang, Tetrahedron, 2008, 64, 2120–2125 CrossRef CAS; (c) H. Tavakol, Asian J. Chem., 2009, 1, 93–100 Search PubMed; (d) L. Meng, B. Hu, Q. Wu, M. Liang and S. Xue, Chem. Commun., 2009, 6089–6091 RSC; (e) M. Hanedanian, O. Loreau, F. Taran and C. Mioskowski, Tetrahedron Lett., 2004, 45, 7035–7038 CrossRef CAS; (f) B. M. Trost and G. R. Dake, J. Am. Chem. Soc., 1997, 119, 7595–7596 CrossRef CAS; (g) L. Meng, K. Tang, H. Liu, J. Xiao and S. Xue, Synlett, 2010, 1833–1836 CAS; (h) D. Tejedor, A. Santos-Exposito, G. Mendez-Abt, C. Ruiz-Perez and F. Garcia-Tellado, Synlett, 2009, 1223–1226 CrossRef CAS; (i) B. Mohtat, S. Jabbar, A. Ghasemi and I. Yavari, J. Chem. Res., 2008, 601–603 CrossRef CAS; (j) Y. Matsuya, K. Hayashi and H. Nemoto, Chem.–Eur. J., 2005, 11, 5408–5418 CrossRef CAS PubMed.
- B. M. Trost and G. R. Dake, J. Am. Chem. Soc., 1997, 119, 7595–7596 CrossRef CAS.
- J. Maury, D. Mouysset, L. Feray, S. R. A. Marque, D. Siri and M. P. Bertrand, Chem.–Eur. J., 2012, 18, 3241–3247 CrossRef CAS PubMed.
- C. H. Oh, H. H. Jung, K. S. Kim and N. Kim, Angew. Chem., Int. Ed., 2003, 42, 805–808 CrossRef CAS PubMed.
- (a) S. Yan, Y. Dong, Q. Peng, Y. Fan, J. Zhang and J. Lin, RSC Adv., 2013, 3, 5563–5569 CAS; (b) F. Yu, R. Huang, H. Ni, J. Fan, S. Yan and J. Lin, Green Chem., 2013, 15, 453–462 RSC; (c) F. Yu, S. Yan, L. Hu, Y. Wang and J. Lin, Org. Lett., 2011, 13, 4782–4785 CrossRef CAS PubMed; (d) S. Yan, Y. Chen, L. Liu, N. He and J. Lin, Green Chem., 2010, 12, 2043–2052 RSC.
- M. N. Burnett and C. K. Johnson, ORTEP-III: Oak Ridge Thermal Ellipsoid Plot Program for Crystal Structure Illustrations, Oak Ridge National Laboratory Report ORNL-6895 Search PubMed.
- A. D. Becke, J. Chem. Phys., 1993, 98, 5648 CrossRef CAS.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, J. A. Montgomery Jr, T. Vreven, K. N. Kudin, J. C. Burant, J. M. Millam, S. S. Iyengar, J. Tomasi, V. Barone, B. Mennucci, M. Cossi, G. Scalmani, N. Rega, G. A. Petersson, H. Nakatsuji, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, M. Klene, X. Li, J. E. Knox, H. P. Hratchian, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, P. Y. Ayala, K. Morokuma, G. A. Voth, P. Salvador, J. J. Dannenberg, V. G. Zakrzewski, S. Dapprich, A. D. Daniels, M. C. Strain, O. Farkas, D. K. Malick, A. D. Rabuck, K. Raghavachari, J. B. Foresman, J. V. Ortiz, Q. Cui, A. G. Baboul, S. Clifford, J. Cioslowski, B. B. Stefanov, G. Liu, A. Liashenko, P. Piskorz, I. Komaromi, R. L. Martin, D. J. Fox, T. Keith, M. A. Al-Laham, C. Y. Peng, A. Nanayakkara, M. Challacombe, P. M. W. Gill, B. Johnson, W. Chen, M. W. Wong, C. Gonzalez and J. A. Pople, GAUSSIAN 03 (Revision D.01), Gaussian, Inc., Wallingford, CT, 2003 Search PubMed.
- C. V. De Amicis, P. R. Graupner, J. A. Erickson, J. W. Paschal, H. A. Kirst, L. C. Creemer and P. E. Fanwick, J. Org. Chem., 2001, 66, 8431–8435 CrossRef CAS PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available: Full experimental procedures and data. CCDC 978904 (3c) and 979050 (4i). For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c4ra01966k |
| ‡ These authors contributed equally to this paper. |
| This journal is © The Royal Society of Chemistry 2014 |