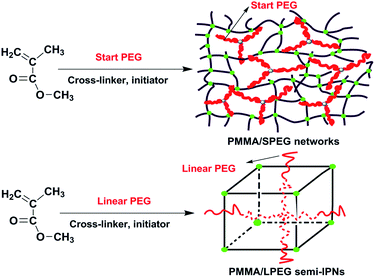
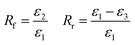Shape memory behavior and mechanism of poly(methyl methacrylate) polymer networks in the presence of star poly(ethylene glycol)
Xingjian Liab,
Tuo Liuab,
Yaru Wangab,
Yi Pan*a,
Zhaohui Zhenga,
Xiaobin Ding*a and
Yuxing Penga
aChengdu Institute of Organic Chemistry, Chinese Academy of Sciences, Chengdu, 610041, China. E-mail: xbding@cioc.ac.cn; Fax: +86 28 85233426; Tel: +86 28 5233426
bUniversity of Chinese Academy of Sciences, Beijing, 100081, China
First published on 11th April 2014
Abstract
The shape memory poly(methyl methacrylate)/star poly(ethylene glycol) (PMMA/SPEG) networks and poly(methyl methacrylate)/linear poly(ethylene glycol) semi-interpenetrating networks (PMMA/LPEG semi-IPNs) were synthesized. The influence of two structural parameters for two network systems, the crosslinking density of the PMMA network and molecular weight of PEG, on the macroscopic properties of the polymer networks such as thermal properties, mechanical properties, dynamic mechanical properties and shape memory properties has been studied. The results showed that all samples for PMMA/SPEG networks had uniform deformation properties with recovery ratios above 98%. By contrast, PMMA/LPEG semi-IPNs showed poor recovery ratios. For two network systems, whereas the shape recovery speed decreased with increasing crosslinking density and molecular weight of PEG, the shape fixity ratio increased. The shape fixity ratio and shape recovery speed for PMMA/SPEG networks is highly influenced by the two structural parameters of the networks, which have a negligible effect on its shape recovery ratio. It is proven that the embedding of the star-shaped structures greatly improved the shape recovery ratio and recovery speed of the PMMA/SPEG networks with excellent mechanical properties compared to PMMA/LPEG semi-IPNs, which was caused by the star-shaped structures in the PMMA/SPEG networks according to the tube model theory proposed by Edwards and the analysis of the creep properties of the materials.
1. Introduction
Thermo-responsive shape memory polymers (SMPs) are those which have the capability of changing their shapes from a temporary shape to a permanent shape upon application of an external thermal stimulus.1,2 They have attracted great attention of scientists and engineers in terms of their intrinsic advantages such as highly tunable properties, larger attainable strain, lower density, easier processing and lower cost,3–5 which provide great potential for applications in biomedicine,6,7 intelligent textiles,8 aerospace engineering,9 self-healing materials,10 micro-optical or fluidics devices11,12 and emerging 4D printing.13In principle, thermally induced SMPs consist of netpoints and switches. The netpoints determine the permanent shape of polymer network and can be of a chemical or physical nature. The switches are responsible for controlling the shape fixity and recovery.4,8 Generally, the material properties are mainly determined by their intrinsic structure. Consequently, SMPs with new macromolecular structures can be designed and synthesized based on their structural principles.14,15 And then their relationship between the structure and shape memory properties can be investigated so as to improve their relatively small recovery stresses, long response time, low achievable number of cycles and poor mechanical properties, which are some of the major drawbacks and the issues that remain challenging in the field of shape memory materials.16
A great deal of effort has been devoted to optimize the properties of SMPs through the reinforcement and molecular structure design of SMPs. First of all, incorporating small amounts of micro/nano-sized reinforcing fillers into SMPs significantly improved the mechanical performance and shape memory capabilities, especially, shape recovery stress levels.17–20 For instance, Miaudet et al. achieved the 150 MPa recovery stress in CNT/polyvinyl alcohol fiber, which is two orders of magnitude higher than that of neat SMPs.21 Recently, a novel family of organic–inorganic shape memory hybrids possessing high elastic modulus and excellent shape memory properties was developed by Agarwal et al.22 The hybrids called nanoscale ionic materials consist of SiO2 core and polyethylene glycol corona. The rubbery modulus of the material is of order 100 MPa at room temperature at a moderate particle volume fraction of 0.16. In recent ground breaking work, Leng et al. took an in-depth look at the synergistic effect between micro/nano-sized carbon fibers and various conductive materials by changing the composite structure, significantly improving the shape recovery performance.23–26 On the other hand, the molecular structure design of SMPs has also gradually attracted wide attention of researchers. For instance, our group first reported the SMPs with semi-interpenetrating networks (semi-IPNs) structure based on the hydrogen-bonding interaction between poly[(methyl methacrylate)-co-(N-vinyl-2-pyrrolidone)] and linear poly(ethylene glycol) (PMMA-co-VP/LPEG).27 The polymers quickly returned to the initial shape in 45 s with a shape recovery ratio of above 99%. And on this basis, our group for the first time realized the quintuple shape memory effect in PMMA/LPEG semi-IPNs containing broadened glass transition and crystalline segments.28 The SMPs with AB-polymer networks structure was first reported by Lendlein's group using oligo-(ε-caprolactone) dimethacrylate as crosslinker and n-butyl acrylate as comonomer, which reached uniform deformation properties with recovery ratios above 99%.29 Later, various types of AB–polymer networks showing shape-memory properties were reported.30–34
It is well-known that star polymers show unique properties in comparison with their linear counterparts due to their well-defined branched structures, large number of functional groups available in a small volume, controlled functionality and composition.35 Consequently, the star-shaped structures applied in SMPs have also attracted attention of researchers. Such as the star-shaped hybrid SMPs were representatively reported by Song etc. using a well-defined star-branched macromer constructed from a polyhedral oligomeric silsesquioxane (POSS) nanoparticle core functionalized with eight polyester arms.36 The star-shaped POSS-polycaprolactone shape memory polyurethanes were prepared by Mya etc.37,38 Recently, our group in a communication also reported PMMA SMP networks with the star-shaped structures in the presence of star-shaped PEG, which exhibited an extraordinary combination of rapid and full shape recovery with excellent mechanical properties when compared to PMMA/LPEG semi-IPNs by the synergistic effect of star polymer and polymer network.39 A “void” hierarchical structural model was proposed for PMMA/SPEG networks to elucidate the improvement of mechanical and shape memory properties. As the cross-linked PMMA network has a high Young's modulus but is quite brittle on its own, we have assumed that the dramatically enhanced mechanical strength is due to an effective relaxation of stress by SPEG filled in the “void” which dissipates the fracture energy and prevents crack development. However, higher crosslinker concentration or larger scale filler should cause a tighter network structure, which would affect the size of “void”, and substantially influenced the synergistic effect of star polymer and polymer network. And the detailed structure–property relationships for such systems have not been attempted, while the network properties are expected to play an important role in controlling the mechanical and shape memory performance and need to be addressed for the successful development of SMPs with the star-shaped structures.
The purpose of the present investigation is to study the shape memory behavior and mechanism of poly(methyl methacrylate) polymer networks in the presence of star poly(ethylene glycol). In the article, the effect of varying levels of chemical crosslinking and varying scale levels of filler via varying SPEG molecular weights on essential aspects of shape memory behavior and thermomechanical behavior was investigated. In order to highlight the effect of SPEG on the mechanical properties, thermomechanical behavior and shape memory performance of PMMA/SPEG networks, PMMA/LPEG semi-IPNs were also synthesized and investigated in a similar process. Schematic diagram for preparation and architecture of PMMA/SPEG networks and PMMA/LPEG semi-IPNs is shown in Scheme 1. The shape memory behavior of PMMA/SPEG networks differing from PMMA/LPEG semi-IPNs was explained according to the tube model proposed by Edwards, which was demonstrated by the analysis of creep properties of the materials.
2. Experimental section
2.1. Materials
Methyl methacrylate (MMA) and 2,2′-azoisobutyronitrile (AIBN) were of analytical grade and were obtained from the Chengdu Reagent Factory. Ethylene glycol dimethacrylate (EGDMA) (Aldrich) was used as a crosslinker without further purification. MMA was distilled under reduced pressure before use. AIBN, used as a radical initiator, was recrystallized from methanol solution. LPEG purchased from local commercial company with weight-average molecular weights of 1000, 2000, and 5000 was dried by heating at 70 °C for 7 h under a vacuum. SPEG was purchased from Polymer Source Inc. The weight-average molecular weights of the arm chain were 1000, 2000, and 5000 respectively. SPEG was dried by heating at 70 °C for 7 h under a vacuum before use.2.2. The preparation of PMMA/SPEG networks
The PMMA/SPEG networks were prepared by radical polymerization and crosslinking of 62 wt% MMA in the presence of 0.5 wt% AIBN based on MMA weight as an initiator, 0.5–5 wt% EGDMA based on MMA weight as a crosslinker and 38 wt% SPEG. Nitrogen was bubbled through the reaction mixture for 15 min to remove any oxygen from the mixture, and the mixture was then injected into the space between two glass plates separated by silicone rubber spacers (1 mm thick). Polymerization was carried out at 55 °C for 24 h. All specimens were annealed and then dried under a vacuum at room temperature for 10 d to remove any unreacted monomer.According to the molecular weight of SPEG and the amount of crosslinker, the PMMA/SPEG networks are divided into two series, namely the crosslinker concentration series (A1–A4) and SPEG series (A5–A7), as shown in Table 1.
| SPEG | PEG | C (wt%) | Crosslinker concentration (wt%) | |||
|---|---|---|---|---|---|---|
| Notation | Mw | Notation | Mw | |||
| (1) Crosslinker concentration of network series | ||||||
| A1 | 6000 | B1 | 2000 | 38 | 0.5 | |
| A2 | 6000 | B2 | 2000 | 38 | 2 | |
| A3 | 6000 | B3 | 2000 | 38 | 3.5 | |
| A4 | 6000 | B4 | 2000 | 38 | 5 | |
| (2) Molecular weight of SPEG and LPEG series | ||||||
| A5 | 3000 | B5 | 1000 | 38 | 0.5 | |
| A6 | 6000 | B6 | 2000 | 38 | 0.5 | |
| A7 | 15000 | B7 | 5000 | 38 | 0.5 | |
2.3. The preparation of PMMA/LPEG semi-IPNs
The PMMA/LPEG semi-IPNs were prepared by the same process in the presence of LPEG based on 2.2. Section. According to the molecular weight of LPEG and the amount of crosslinker, the PMMA/LPEG semi-IPNs is divided into two series, namely the crosslinker concentration series (B1–B4) and LPEG series (B5–B7), as shown in Table 1.2.3. Characterization
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ curves.
δ curves.Bending test which examined the shape recovery time is as follows:39,40 a straight strip (2 × 12 × 60 mm) of the specimen was folded at 76 °C, then cooled to room temperature to maintain the deformation. The deformation sample was then heated again and the shape recovery time was recorded.
3. Results and discussion
3.1. Thermal properties
The thermal properties of the SMPs comprising star-shaped structures and semi-IPNs were determined by DSC with the second heating thermograms, as shown in Fig. 1, 2 and Table 2. As representatively shown in Fig. 1, PMMA/SPEG networks (A) and PMMA/LPEG semi-IPNs (B) have the same PEG weight content ranging while the crosslinker concentration and the molecular weight of PEG is varying. Star-shaped polymers were more compact than linear homologous because of their higher segment densities, which would lead to a much greater phase separation, i.e. PEG crystal and amorphous part of PEG remains dissolved in the PMMA network,41 in A specimen with further increment of crosslinker concentration supported by DSC results (Fig. 1). A samples revealed a much smaller crystalline phase transition than that of B samples at low cross-linker concentration, respectively, and the PEG chains of SPEG favored to crystallize at high crosslinker concentration, while PEG crystal in B samples keep much more slowly increasing as increment of crosslinker concentration. It can be observed that A samples showed higher melting enthalpy (ΔHm) than that of B samples, when the crosslinking density is more than 3 wt%. This is ascribed to higher segment densities of SPEG than LPEG. SPEG tend to gather together in the PMMA network, resulting in a higher degree of crystallinity and ΔHm. | ||
| Fig. 1 Heat of fusion of PMMA/SPEG networks (A, solid circles) and PMMA/LPEG semi-IPNs (B, solid squares) as a function of the crosslinker concentration in the PMMA networks, respectively. | ||
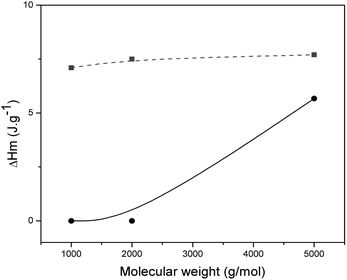 | ||
| Fig. 2 Heat of fusion of PMMA/SPEG networks (A, solid circles) and PMMA/LPEG semi-IPNs (B, solid squares) as a function of the molecular weight in the PMMA networks, respectively. | ||
| Sample | Tm (°C) | ΔHm (J g−1) | Crystallinity (%) |
|---|---|---|---|
| A1 | — | — | — |
| A2 | 46 | 5.8 | 3.1 |
| A3 | 47 | 24.9 | 13.2 |
| A4 | 49 | 28.1 | 14.9 |
| A5 | — | — | — |
| A6 | — | — | — |
| A7 | 52 | 5.7 | 2.9 |
| B1 | 50 | 7.1 | 3.8 |
| B2 | 51 | 14.4 | 7.6 |
| B3 | 52 | 16.5 | 8.7 |
| B4 | 52 | 20.7 | 10.9 |
| B5 | 50 | 7.1 | 3.7 |
| B6 | 51 | 7.5 | 3.9 |
| B7 | 53 | 7.6 | 4.0 |
Since the crystallinity of PEG is directly related to its molecular weight, the influence of molecular weight of PEG on the crystallinity of the two PMMA networks was investigated while maintaining the same crosslink density (0.5 wt%). As seen from Fig. 2, with increasing of the molecular weight of PEG, the ΔHm of PMMA/LPEG semi-IPNs (B5–B7) showed a little change, while the ΔHm of PMMA/SPEG networks (A5–A7) increased rapidly, but lower than the corresponding ΔHm of PMMA/LPEG semi-IPNs. It is chiefly because the star-shaped structures disturbed the crystallization of PEG, leading to a lower degree of the crystallinity in PMMA/SPEG networks under the same PEG weight content and crosslinking density.
3.2. Mechanical properties
As representatively shown in Fig. 3, PMMA/SPEG networks (A) and PMMA/LPEG semi-IPNs (B) have the same crosslinker concentration ranging from 0.5 wt% to 5 wt% based on MMA weight, respectively. The elongation at break of both series decreased greatly with the increment of crosslinker concentration, respectively. However, a much more sharply decrease was observed for PMMA/SPEG networks when compared to PMMA/LPEG semi-IPNs. In particular, the elongation at break of A4 sample with 5 wt% crosslinker concentration was only 8%. The higher elongation at break of PMMA/SPEG networks at low crosslinker concentration compared to the counterparts may be ascribed to the movable-star PEG in the “void”, which effectively absorbed the crack energy either by viscous dissipation or by large deformation of the PEG chains under deformation, preventing the crack growth to a macroscopic level.42 Comparing with linear counterparts, however, the increasing crosslinker concentration tended to format a tighter network resulting in a much more difficult movement for the star branching point under stretching, which substantially sharply decreased the deformability of the PMMA/SPEG networks.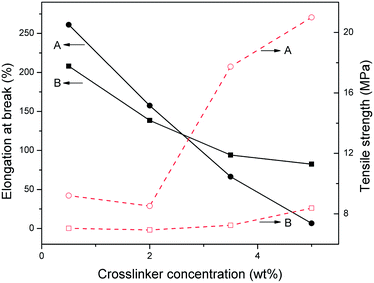 | ||
| Fig. 3 Mechanical properties of PMMA/SPEG networks (A) and PMMA/LPEG semi-IPNs (B) obtained at 25 °C as a function of the crosslinker concentration in the PMMA networks, respectively. | ||
Fig. 3 also shows the relationship between tensile strength and crosslinker concentration of both series. The tensile strength in A specimen decreased firstly and then increased sharply to 21 MPa by increasing crosslinker concentration, while the values in B specimen keep increasing slowly to 8.3 MPa at the studied crosslinker concentration range, respectively. The higher tensile strength of PMMA/SPEG networks at low cross-linker concentration resulted from the effective relaxation of locally applied stress and dissipation of the crack energy through diffusive fluctuation of the movable-star PEG in the “void”,42 as well as the additional crosslinker of star branching point in the network to enhance the tensile strength. These crosslinkers are not fixed in the network, which can move with the sample's macroscopic deformation, and subsequently improved the elongation at break discussed above. Whereas, the “void” tended to be more compact for the tighter network by increasing the crosslinker concentration, which would restrict the movable range for the star PEG and have a bad effect on the improvement of tensile strength, that's the reason that the tensile strength of A2 sample is slight lower than that of A1 sample. On the other hand, with increasing crosslinking density, the degree of SPEG crystallinity in PMMA/SPEG networks increased obviously as showed in analysis of thermal properties. The presence of crystals can play the role of physical crosslinking points. Thus it is also responsible for the fact that the tensile strength of PMMA/SPEG networks increased sharply while reducing the elongation at break. However, for PMMA/LPEG semi-IPNs, the crosslinking density had no significant effect on the crystallinity of the material, resulting in a small change in the tensile strength.
The effect of PEG molecular weight in the PMMA networks on mechanical properties is shown in Fig. 4. It is remarkable that PMMA/SPEG networks exhibited more excellent tensile strength and elongation at break than those of PMMA/LPEG semi-IPNs as the increasing of PEG molecular weight. The excellent performance of PMMA/SPEG networks is attributed to the effective relaxation of locally applied stress and dissipation of the crack energy provided by diffusive fluctuation of the movable-star PEG in the “void” at low cross-linker concentration.
 | ||
| Fig. 4 Mechanical properties of PMMA/SPEG networks (A) and PMMA/LPEG semi-IPNs (B) obtained at 25 °C as a function of the molecular weight in the PMMA networks, respectively. | ||
3.3. Dynamic mechanical properties
Storage modulus (E′)-temperature and loss angle (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ)-temperature curves in Fig. 5 obtained by DMA displayed how the thermomechanics of both series can be altered by controlling the amount of the crosslinking monomer. Tg and initial storage modulus (E0) at 0 °C of both series increased with an increase in crosslink density. More precise thermo-mechanical values containing E0 and Tg of the eight networks can be seen in Fig. 5. When increasing the crosslink density, the spacing between the linear chains built from MMA and the crosslinking monomer diminishes. The chains are linked closer together with a higher degree of steric hindrance and lower chain mobility. Thus, with an increase in crosslink density, the decrease in the chain spacing allows for less conformational motion, and resulting in an increase in both E0 and Tg.
δ)-temperature curves in Fig. 5 obtained by DMA displayed how the thermomechanics of both series can be altered by controlling the amount of the crosslinking monomer. Tg and initial storage modulus (E0) at 0 °C of both series increased with an increase in crosslink density. More precise thermo-mechanical values containing E0 and Tg of the eight networks can be seen in Fig. 5. When increasing the crosslink density, the spacing between the linear chains built from MMA and the crosslinking monomer diminishes. The chains are linked closer together with a higher degree of steric hindrance and lower chain mobility. Thus, with an increase in crosslink density, the decrease in the chain spacing allows for less conformational motion, and resulting in an increase in both E0 and Tg.
 | ||
Fig. 5 Storage modulus (E′)–temperature and loss angle (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ)–temperature curves of PMMA/SPEG networks (A1–A4) and PMMA/LPEG semi-IPNs (B1–B4). δ)–temperature curves of PMMA/SPEG networks (A1–A4) and PMMA/LPEG semi-IPNs (B1–B4). | ||
It's worth noting that E0 value of the PMMA/SPEG networks exhibits considerable variation as crosslinker concentration ranging from 0.5 wt% to 5 wt%. The E0 value of A4 specimen is more than twice as much as that of A1 specimen. When the cross-linker concentration goes over 3.5 wt%, E0 value PMMA/LPEG semi-IPNs is approximately constant. The same was of course true in Tg value. The nature of the phenomenon lies in the structural differences because SPEG in PMMA/SPEG networks have many branch points in comparison with PMMA/LPEG semi-IPNs given that both series have similar polymer chain composition. Therefore, as an increase in crosslinking density, the free volume of the polymer network is reduced, and then the branch points of SPEG are limited significantly by mesh in PMMA network. Thus, these characteristics make it difficult for the free arms in SPEG to move compared to LPEG. So, the branch points of SPEG are primarily responsible for the more considerable variation of E0 and Tg value for the PMMA/SPEG networks.
Storage modulus (E′)–temperature and loss angle (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ)–temperature curves of both series with the increment of PEG molecular weight displayed in Fig. 6. As shown in Fig. 6, both series exhibit similar thermodynamic behavior. E0 and Tg value of A samples is all less than the corresponding B samples, but the change in these parameters is small. With increasing molecular weight of SPEG and LPEG, E0 and Tg of PMMA/SPEG networks and PMMA/LPEG semi-IPNs increased. It should be briefly noted that increasing the molecular weight of the PEG may lead to more severe microphase separation of the PEG unit in PMMA network as demonstrated analysis of thermal properties, which enhanced crystallization of MMA network. Hence, the role of physical cross-linking points provided by more crystals results in dispersion strengthening effect, which further impedes the movement of the polymer chain segments and contributes to a higher value both E0 and Tg.
δ)–temperature curves of both series with the increment of PEG molecular weight displayed in Fig. 6. As shown in Fig. 6, both series exhibit similar thermodynamic behavior. E0 and Tg value of A samples is all less than the corresponding B samples, but the change in these parameters is small. With increasing molecular weight of SPEG and LPEG, E0 and Tg of PMMA/SPEG networks and PMMA/LPEG semi-IPNs increased. It should be briefly noted that increasing the molecular weight of the PEG may lead to more severe microphase separation of the PEG unit in PMMA network as demonstrated analysis of thermal properties, which enhanced crystallization of MMA network. Hence, the role of physical cross-linking points provided by more crystals results in dispersion strengthening effect, which further impedes the movement of the polymer chain segments and contributes to a higher value both E0 and Tg.
 | ||
Fig. 6 Storage modulus (E′)–temperature and loss angle (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ)–temperature curves of PMMA/SPEG networks (A5–A7) and PMMA/LPEG semi-IPNs (B5–B7). δ)–temperature curves of PMMA/SPEG networks (A5–A7) and PMMA/LPEG semi-IPNs (B5–B7). | ||
3.4. Shape memory properties
Uniaxial tensile test is then employed for evaluating shape memory properties of these polymer materials. In general, the increasing crosslinker concentration tends to decrease the sample's deformability due to the formation of a tighter network, which may result in a reduction in shape recovery performance for both series.43 However, as shown in Fig. 7, PMMA/SPEG networks showed much better shape recovery (more than 98%) than that of semi-IPNs (75% to 93%) at the study crosslinker concentration range, respectively, which was opposite to the above consideration, i.e. the resultant entropic force after deformation would be minimized because of the movable branch points in the fixed network can maximize the configurational entropy of the whole networks during the heating and deformation process. This phenomenon was also observed in SMP coupon samples and can simply be explained based on Gibbs free energy.44 Shape memory is driven by a favorable increase in entropy, thus lowering the free energy of the system.45 When PMMA/SPEG networks are deformed at near its Tg, a significant amount of mechanical force is needed for the deformation because the chains tethering with the spital-confined movable branch point can effectively absorb the external force energy, and the energy is stored in enthalpic internal energy wells during the subsequent cooling process. In this case, shape recovery is driven by both a favorable increase in entropy and decrease in enthalpy, which results in a better and fast shape recovery discussed later. | ||
| Fig. 7 Shape recovery ratio and shape fixity ratio of PMMA/SPEG composites (A1–A4) and PMMA/LPEG semi-IPNs (B1–B4). | ||
Furthermore, the shape recovery time of both network systems increased by the increment of crosslinker concentration as shown in Fig. 8. This can be explained by the restorative forces due to crosslinking. If the shape memory restorative forces are high enough, the polymer will take advantage of the increased chain mobility and visco-elastically return to its original shape. Since crosslinking is directly related to a polymer's rubbery modulus, which governs the restorative stresses during shape recovery, an increase in crosslinking will provide the decreased chain mobility and may enable large shape recovery time.45 In both network systems, the PEG crystal is expected to serve the purpose of switching segment, and the concentration of switching segment plays an important role in determining the shape memory properties of SMP systems.43 The increasing crosslinker concentration resulted in an improvement in the shape fixity for both series because the accumulating crystalline PEG phase helped retain the shape, while PMMA/SPEG networks show bad shape fixity at lower crosslinker concentration, which may be ascribed to the larger relaxation of residual energy caused by deformation and subsequent cooling process above-mentioned compared with semi-IPNs at low crosslinker concentration, which would cause a large shrinkage upon removing the grip from the sample and indicate a poor shape fixity.40 However, the effect of increasing PEG crystals for retaining the temporary shape become dominating at higher crosslinker concentration, which was also supported by DSC results, and A4 sample exhibited a much better shape fixity among all samples.
When the molecular weight of the PEG increased gradually, similar trends of shape memory properties containing shape recovery ratio, shape fixity ratio and recovery time are observable in both network systems. As can be seen in Fig. 9 and 10, the shape recovery ratio and shape fixity ratio of both systems increase, while their recovery time prolongs gradually with increasement of PEG molecular weight. However, PMMA/SPEG networks shape recovery ratio and shape recovery speed are significantly higher than the corresponding PMMA/LPEG semi-IPNs. Therein, the shape recovery ratio of the PMMA/SPEG networks remains stable at 98% level showing excellent shape recovery properties. The enhanced crystallization of SPEG is expected to be chiefly responsible for the excellent shape memory properties of the SMP systems.
 | ||
| Fig. 9 Shape recovery ratio and shape fixity ratio of PMMA/SPEG composites (A5–A7) and PMMA/LPEG semi-IPNs (B5–B7). | ||
3.5. Molecular mechanism of shape memory effect
In order to vividly describe the shape memory behavior of both network systems, an impressive picture sequence demonstrates the macroscopic effect of shape memory for A5 and B5 samples in Fig. 11. The shape memory properties evaluated by bending deformation test of A5 and B5 samples differ from those by uniaxial tensile test caused by different test methods, test conditions and sample size.46,47 A5 sample in PMMA/SPEG networks exhibited excellent shape memory recovery performance and fast response speed, compared with B5 sample in the PMMA/LPEG semi-IPNs. A5 sample achieved full shape recovery in a matter of 51 s. The B5 sample quickly returned to the initial shape in 128 s with a shape recovery ratio of above 98%. Both of two systems, having the same molecular mechanism of shape memory effect, consist of switch units and net-points. The chemically crosslinked networks as net-points determine the permanent shape. The thermal reversible phase transition, that is glass transition, as switch units are responsible for the reversible controlling the shape fixity and recovery upon a specific and predetermined external stimulus.4The mechanism by which the reason that PMMA/SPEG networks can show the excellent shape memory performance compared to PMMA/LPEG semi-IPNs can be explained is revealed on the basis of the tube model theory proposed by Edwards.48 Just as linear chains, the chain loses memory of its present configuration by diffusing out of the surrounding “tube” into a new one. However, in a star polymer (SPEG), the branch point prevents reptation, and star arms explore new configurations by retreating along their tubes and poking out again in a new direction. Retraction is entropically unlikely, so arm retraction is thermally activated.49 Meanwhile, when the star polymers move along one of the “tube” direction under the effect of outside force, it inevitably drag the remaining arms to move, which produce a significant decrease in entropy value for the star polymer relative to linear polymer. On the other hand, the shape memory capacity of polymers lies in the entropy-driven tendency for polymer chains to adopt a randomly coiled configuration. The intrinsic mechanism for shape memory behavior of polymers is the freezing and activation of the long-range motion of polymer chain segments below and above Ttrans, respectively.36 From above consideration, star architecture can more effectively achieve complete freezing of chain segment motion and thus prevent chain recoiling below Ttrans to facilitate temporary shape fixation and full activation of chain recoiling above Ttrans to facilitate shape recovery, which substantially improve shape recovery ratio and shape fixity ratio of PMMA/SPEG networks.
Furthermore, since branching points hinder star polymers from crawling, reptation motion of the polymer chains along the surrounding tube in star polymers takes longer time than that in linear chain based on the tube model. Hence, the relaxation stress of star PEG in PMMA/SPEG networks elapses much more slowly, which enables subsequently much more internal stress in PMMA/SPEG networks to be stored during the deformation process than PMMA/LPEG semi-IPNs. Consequently, PMMA/SPEG networks exhibit more outstanding shape recovery ratio and shape recovery speed. On the other hand, well distribution of the star-shaped structures in the crosslinking PMMA networks could effectively reduce the heterogeneity of the polymer networks, which is partly conducive to improve the recovery speed of the PMMA/SPEG network.50 Because there is a certain influence rule of the relaxation time and the shape recovery behavior based on thermodynamic constitutive frameworks,51 the following creep properties of both network systems were analyzed to confirm the theoretical analysis above according to stress relaxation behavior.
3.6. Creep properties
The effect of the crosslink density and molecular weight of PEG on stress relaxation behavior of both network systems was assessed by creep properties, and the results are shown in Fig. 12 and 13, respectively. The creep compliance of both network systems decreased with increasing of crosslink density and molecular weight of PEG. This is attributed to the fact that the presence of a growing number of chemical crosslinks and physical cross-linking points provided by PEG crystals lowers chain mobility of both network systems, respectively. It is desirable that the creep to be a minimum for an effective fixing of a shape memory polymer.43,52 It's evident that the changing trends of the shape fixity ratio of both network systems follow the conclusion.Moreover, it is clear that all the PMMA/SPEG networks show much higher creep compared to PMMA/PEG semi-IPNs except for A4 and A7 sample. This may be because SPEG filled in the large “voids” in PMMA networks needs to complete larger deformation of arm chains to dissipate localized internal stress relaxation than LPEG, leading to larger creep. However, more meaningful than the above is the creep compliance of all the PMMA/LPEG semi-IPNs tend quickly towards stability while the creep compliance of all the PMMA/SPEG networks gradually increases over time. This arises from the large molecular volume and impediment of branch points for SPEG, which brings about longer time of reptation motion in SPEG than that of LPEG. Thus, stress relaxation of PMMA/SPEG networks takes longer time than that of PMMA/LPEG semi-IPNs. So, PMMA/SPEG networks show relatively larger creep compliance and longer stress relaxation time, which indicates poor shape fixity and a large and rapid recovery compared to PMMA/LPEG semi-IPNs. The analysis results of creep properties illuminate that the above-mentioned theoretical analysis that the presence of star-shaped structure will help improve the shape memory performance based on the tube model proposed by Edwards is quite reasonable.
4. Conclusion
PMMA/SPEG networks and PMMA/LPEG semi-IPNs were synthesized and investigated. Both structural parameters, the crosslinking density of the PMMA network and molecular weight of PEG, strongly influenced the macroscopic properties of the both networks. When compared with PMMA/LPEG semi-IPNs, PMMA/SPEG networks exhibit excellent shape memory recovery and fast response speed, which was caused by the star-shaped structures in PMMA/SPEG networks according to the tube model theory proposed by Edwards. The study is expected to provide new ideas and theories for the design of shape memory polymer materials having new structures, and to broaden the application area of shape memory polymer materials.Acknowledgements
The authors would like to thank the National Natural Science Foundation of China (Grant no. 51173185 and 51303179) for the financial support of this research.References
- A. Lendlein and S. Kelch, Angew. Chem., Int. Ed., 2002, 41, 2034–2057 CrossRef CAS.
- S. Mondal, Mini-Rev. Org. Chem., 2009, 6, 114–119 CrossRef CAS.
- C. Liu, H. Qin and P. T. Mather, J. Mater. Chem., 2007, 17, 1543–1558 RSC.
- J. L. Hu, Y. Zhu, H. H. Huang and J. Lu, Prog. Polym. Sci., 2012, 37, 1720–1763 CrossRef CAS PubMed.
- T. Xie, Polymer, 2011, 52, 4985–5000 CrossRef CAS PubMed.
- W. Small IV, P. Singhal, T. S. Wilson and D. J. Maitland, J. Mater. Chem., 2010, 20, 3356–3366 RSC.
- W. M. Huang, C. L. Song, Y. Q. Fu, C. C. Wang, Y. Zhao, H. Purnawali, H. B. Lu, C. Tang, Z. Ding and J. L. Zhang, Adv. Drug Delivery Rev., 2013, 65, 515–535 CrossRef CAS PubMed.
- J. L. Hu and S. J. Chen, J. Mater. Chem., 2010, 20, 3346–3355 RSC.
- X. Lan, Y. Liu, H. Lv, X. Wang, J. Leng and S. Du, Smart Mater. Struct., 2009, 18, 024002 CrossRef.
- X. F. Luo and P. T. Mather, ACS Macro Lett., 2013, 2, 152–156 CrossRef CAS.
- H. X. Xu, C. J. Yu, S. D. Wang, V. Malyarchuk, T. Xie and J. A. Rogers, Adv. Funct. Mater., 2013, 23, 3299–3306 CrossRef CAS.
- A. Balasubramanian, R. Morhard and C. J. Bettinger, Adv. Funct. Mater., 2013, 23, 4832–4839 CAS.
- Q. Ge, H. J. Qi and M. L. Dunn, Appl. Phys. Lett., 2013, 103, 131901 CrossRef PubMed.
- X. L. Wu, W. M. Huang, Y. Zhao, Z. Ding, C. Tang and J. L. Zhang, Polymers, 2013, 5, 1169–1202 CrossRef CAS PubMed.
- X. F. Luo and P. T. Mather, Curr. Opin. Chem. Eng., 2013, 2, 103–111 CrossRef PubMed.
- I. A. Rousseau, Polym. Eng. Sci., 2008, 48, 2075–2089 CAS.
- Y. J. Liu, H. Y. Du, L. W. Liu and J. S. Leng, Smart Mater. Struct., 2014, 23, 023001 CrossRef.
- H. Meng and G. Q. Li, Polymer, 2013, 54, 2199–2221 CrossRef CAS PubMed.
- S. A. Madbouly and A. Lendlein, Adv. Polym. Sci., 2010, 226, 41–95 CrossRef CAS.
- Q. H. Meng and J. L. Hu, Composites, Part A, 2009, 40, 1661–1672 CrossRef PubMed.
- P. Miaudet, A. Derré, M. Maugey, C. Zakri, P. M. Piccione, R. Inoubli and P. Poulin, Science, 2007, 318, 1294–1296 CrossRef CAS PubMed.
- P. Agarwal, M. Chopra and L. A. Archer, Angew. Chem., Int. Ed., 2011, 50, 8670–8673 CrossRef CAS PubMed.
- J. S. Leng, H. B. Lu, Y. J. Liu and S. Y. Du, Appl. Phys. Lett., 2007, 91, 144105 CrossRef PubMed.
- H. B. Lu, K. Yu, S. H. Sun, Y. J. Liu and J. S. Leng, Polym. Int., 2010, 59, 766–771 CAS.
- H. B. Lu, W. L. Yin, W. M. Huang and J. S. Leng, RSC Adv., 2013, 3, 21484–21488 RSC.
- H. B. Lu, W. M. Huang and J. S. Leng, Composites, Part B, 2014, 62, 1–4 CrossRef PubMed.
- G. Q. Liu, C. L. Guan, H. S. Xia, F. Q. Guo, X. B. Ding and Y. X. Peng, Macromol. Rapid Commun., 2006, 27, 1100–1104 CrossRef CAS.
- J. Li, T. Liu, Y. Pan, S. Xia, Z. Z. Zheng, X. B. Ding and Y. X. Peng, J. Mater. Chem., 2011, 21, 12213–12217 RSC.
- A. Lendlein, A. M. Schmidt and R. Langer, Proc. Natl. Acad. Sci. U. S. A., 2001, 98, 842–847 CAS.
- M. Behl, I. Bellin, S. Kelch, W. Wagermaier and A. Lendlein, Adv. Funct. Mater., 2009, 19, 102–108 CrossRef CAS.
- A. T. Neffe, B. D. Hanh, S. Steuer and A. Lendlein, Adv. Mater., 2009, 21, 3394–3398 CrossRef CAS PubMed.
- J. Li, T. Liu, Y. Pan, S. Xia, Z. Z. Zheng, X. B. Ding and Y. X. Peng, Macromol. Chem. Phys., 2012, 213, 2246–2252 CrossRef CAS.
- C. M. Yakacki, R. Shandas, C. Lanning, B. Rech, A. Eckstein and K. Gall, Biomaterials, 2007, 28, 2255–2263 CrossRef CAS PubMed.
- C. M. Yakacki, R. Shandas, D. Safranski, A. M. Ortega, K. Sassaman and K. Gall, Adv. Funct. Mater., 2008, 18, 2428–2435 CrossRef CAS PubMed.
- N. Hadjichristidis, J. Polym. Sci., Part A: Polym. Chem., 1999, 37, 857–871 CrossRef CAS.
- J. W. Xu and J. Song, Proc. Natl. Acad. Sci. U. S. A., 2010, 107, 77652–77657 Search PubMed.
- K. Y. Mya, H. B. Gose, T. Pretsch, M. Bothe and C. He, J. Mater. Chem., 2011, 21, 4827–4836 RSC.
- M. Bothe, K. Y. Mya, E. M. J. Lin, C. C. Yeo, X. H. Lu, C. B. He and T. Pretsch, Soft Matter, 2012, 8, 965–972 RSC.
- T. Liu, J. Li, Y. Pan, Z. Z. Zheng, X. B. Ding and Y. X. Peng, Soft Matter, 2011, 7, 1641–1643 RSC.
- C. C. Tsai, C. C. Chang, C. S. Yu, S. A. Dai, T. M. Wu, W. C. Su, C. N. Chen, F. M. C. Chen and R. J. Jeng, J. Mater. Chem., 2009, 19, 8484–8494 RSC.
- G. Q. Liu, X. B. Ding, Y. P. Cao, Z. H. Zheng and Y. X. Peng, Macromol. Rapid Commun., 2005, 26, 649–652 CrossRef CAS.
- Y. H. Na, T. Kurokawa, Y. Katsuyama, H. Tsukeshiba, J. P. Gong, Y. Osada, S. Okabe, T. Karino and M. Shibayama, Macromolecules, 2004, 37, 5370–5374 CrossRef CAS.
- D. Ratna and J. Karger-Kocsis, Polymer, 2011, 52, 1063–1070 CrossRef CAS PubMed.
- Y. P. Liu, K. Gall, M. L. Dunn and P. Mccluskey, Smart. Mater. Struct., 2003, 12, 947 CrossRef CAS.
- C. M. Yakacki, R. Shandas, C. Lanning, B. Rech, A. Eckstein and K. Gall, Biomaterials, 2007, 28, 2255–2263 CrossRef CAS PubMed.
- K. Yu, Q. Ge and H. J. Qi, Nat. Commun., 2014, 5, 1–9 Search PubMed.
- F. Castro, K. K. Westbrook, K. N. Long, R. Shandas and H. J. Qi, Mech. Time-Depend. Mater., 2010, 14, 219–241 CrossRef.
- M. Doi and S. F. Edwards, The theory of polymer dynamics, Clarendon, Oxford, 1986 Search PubMed.
- S. T. Milner and T. C. B. Mcleish, Macromolecules, 1997, 30, 2159–2166 CrossRef CAS.
- Q. Ge, K. Yu, Y. Ding and H. J. Qi, Soft Matter, 2012, 8, 11098–11105 RSC.
- H. B. Lu and W. M. Huang, Smart Mater. Struct., 2013, 22, 105021 CrossRef.
- P. Thorsten, Polymers, 2010, 2, 120–158 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2014 |