Selective fluorescence assay of aluminum and cyanide ions using chemosensor containing naphthol†
Soojin Kim‡
a,
Jin Young Noh‡b,
Sol Ji Parka,
Yu Jeong Nab,
In Hong Hwangb,
Jisook Minc,
Cheal Kim*b and
Jinheung Kim*a
aDepartment of Chemistry and Nano Science, Global Top 5 Research Program, Ewha Womans University, Seoul 120-750, Korea. E-mail: jinheung@ewha.ac.kr; Fax: +82-2-3277-3419; Tel: +82-2-3277-4453
bDepartment of Fine Chemistry, Seoul National University of Science and Technology, Seoul, Korea. E-mail: chealkim@snust.ac.kr
cNational Forensic Service, Seoul, 158-707, Korea
First published on 7th April 2014
Abstract
The selective assay of aluminum and cyanide ions is reported using fluorescence enhancement and quenching of a phenol–naphthol based chemosensor (PNI) in aqueous and nonaqueous solvents, respectively. PNI gave no significant fluorescence in water. The binding properties of PNI with metal ions were investigated by UV-vis, fluorescence, and electrospray ionization mass spectrometry in a Bis–Tris buffer solution. The addition of aluminum ions switches on the fluorescence of the sensor PNI in water, comparable to relatively very low fluorescence changes in the presence of various other metal ions. The complex stability constant (Ka) for the stoichiometric 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complexation of PNI with aluminium ions was obtained by fluorimetric titrations and NMR experiments. However, upon treatment with cyanide ions, the fluorescence of PNI was selectively turned off and the yellow solution of PNI turned to red in methanol. Other comparable anions, such as F−, Cl−, Br−, I−, CH3COO−, and H2PO4−, afforded no apparent fluorescence quenching. The interaction of PNI with cyanide ions was studied by NMR experiments.
1 complexation of PNI with aluminium ions was obtained by fluorimetric titrations and NMR experiments. However, upon treatment with cyanide ions, the fluorescence of PNI was selectively turned off and the yellow solution of PNI turned to red in methanol. Other comparable anions, such as F−, Cl−, Br−, I−, CH3COO−, and H2PO4−, afforded no apparent fluorescence quenching. The interaction of PNI with cyanide ions was studied by NMR experiments.
Introduction
Fluorescent probes for selective detection of various biologically and environmentally relevant small anions and metal cations have attracted increasing interest due to the lasting impact of these ions' toxic effects in biological systems. Due to the widespread use of aluminum in food and cooking related goods, the possibility of human exposure to aluminum has increased. Long-term intake of excess aluminum ions is possible to spread throughout all tissues in humans and animals, and eventually accumulates in the bone. The iron binding proteins in the human body are the major carrier of Al3+ in plasma. Aluminum ions can enter the brain and reach the placenta and fetus by such in vivo carriers. Aluminum ions can stay quite a while in various organs and tissues before they are excreted through the urine. In addition, aluminum ions may be one of possible factors of Alzheimer's disease reported to result from the accumulation of oxidative damage induced by metal ions and may result in bad influence to the central nervous system in humans.1–4 The average daily human intake of aluminum ions recommended by WHO is about 3–10 mg.5,6 In the study of chemosensors for aluminum ion, Schiff base complexes showed various applications, such as analytical, biological, and clinical fields. Recently, Schiff base-type chemosensors have also been reported as a major class for sensing aluminum ions.7–12Cyanide ions are extremely toxic to mammals and lead to death above a certain level. WHO recommends the cyanide concentration in drinking water to be kept lower than ca. 2 μM.13 Chemicals containing cyanide ions are widely used in electroplating, gold mining, and polymer production.14–16 Recently, some chemosensors specific for cyanide have been reported based on fatal toxicity of cyanide in biological systems and environment.17–30 More examples of selective chemosensors for cyanide are needed to develop fast and accurate detection of cyanide in various situations.
In this study, we report the anion and metal cation-binding properties of a new imine probe bearing phenol and naphthol moieties which is the π-conjugated Schiff base receptor. This imine probe exhibited fluorescence enhancement with high selectivity upon binding to aluminum ions in aqueous solution, and fluorescence quenching with high selectivity to cyanide anions in methanol. The in vivo application of the probe as an intracellular sensor of Al3+ is also reported by confocal fluorescence microscopy.
Results and discussion
Fluorometric assay of aluminum ion
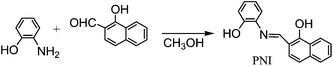
The chemosensor, ortho-phenol-ortho-naphtholmethylimine (PNI) was prepared by the reaction of 1-hydroxy-2-naphthaldehyde and 2-aminophenol with 61% yield (Scheme 1 and Experimental section). The binding properties of the PNI receptor was examined towards various metal ions, such as Ag+, Na+, K+, Ca2+, Mg2+, Ni2+, Cd2+, Mn2+, Co2+, Cu2+, Zn2+, Hg2+, Pb2+, Al3+, Sc3+, Cr3+, Fe3+, Ga3+, and In3+ (34 equiv. each) in 50 mM Bis–Tris buffer (pH = 7.0). The fluorescence spectral response in the presence of each metal ion was shown in Fig. 1. Relative to no fluorescence of PNI in aqueous solution, significant spectral changes were observed in the case of Al3+. No significant changes observed with other metal ions demonstrate the high selectivity of PNI to other metal ions. PNI is even highly selective for Al3+ over Ga3+ and In3+. | ||
| Fig. 1 Emission spectra of PNI (10 μM) in the presence of various metal ions (340 μM) using Eλ = 450 nm in 50 mM Tris buffer (pH = 7.0). | ||
The turn-on fluorescence response of PNI was observed for Al3+ with emission bands at 490 and 515 nm. Upon addition of 34 equiv. Al3+, the fluorescence intensity of PNI increased by 100-fold. The fluorescence spectra of PNI progressively changed upon addition of an incremental amount of Al3+ (Fig. 2a). The fluorometric titration curve showed a steady and smooth increase with increasing Al3+ concentration, which demonstrated an efficient fluorescence response (Fig. 2b). The binding affinity of PNI towards Al3+ was quantified based on fluorescence titration experiments, affording the association constant (Ka) of 2.5 × 103 M−1 (ESI, Fig. S1†). Upon binding of Al3+, the aromatic rings of PNI no more rotate around the imine group, and two aromatic rings could be on the same plane to make a good conjugated system through the imine. This conjugation probably caused the fluorescence enhancement. In the case of other metal ions which can bind to PNI in a similar manner, the fluorescence would be quenched by each metal coordination.
 | ||
| Fig. 2 (a) Changes in emission intensity of 10 μM PNI upon the addition of Al3+ (0–0.6 mM), Eλ = 450 nm in 50 mM Tris buffer. (b) Binding isotherm monitored by the fluorescence increase at 515 nm. | ||
The absorption spectra of PNI also changed upon addition of Al3+ ions (Fig. 3). The bands at 285, 455, and 473 nm decreased and no characteristic new bands appeared.
 | ||
| Fig. 3 Changes in the UV-vis spectrum of PNI (10 μM) as a function of the Al3+ concentration in 50 mM Tris buffer. | ||
Fig. 4 illustrate the fluorescence response of PNI to Al3+ in the presence of other competing metal ions, respectively. Except for Cu2+, Cr3+, and Fe3+, backgrounds of most metal ions do not interfere with the detection of Al3+ by PNI in Tris buffer (pH = 7.0). Chemosensors reporting fluorescence studies with Al3+ in the presence of competing metal ions showed inhibition by Ag+, Cu2+, Cr3+, and Fe3+,11,12 but PNI was not significantly inhibited by the other metal ions. In order to understand the interference by Cu2+, Cr3+, and Fe3+, absorption spectra were obtained using the mixtures of Al3+ and other metal ions. In the presence of Cu2+, Cr3+, or Fe3+, the absorption spectrum of PNI and Al3+ were different from that obtained without such interfering metal ions. Based on the spectral changes, the inference derived from the displacement of Al3+.
 | ||
| Fig. 4 Aluminum ion (34 μM) response for PNI (10 μM) in the absence and presence of competing metal ions (34 μM) in 50 mM Tris buffer. L stands for PNI. | ||
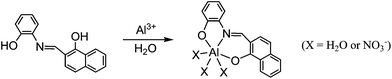
A Job's plot as well as binding analysis using the Benesi–Hildebrand plot established that a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complex of PNI (Scheme 2 and Fig. S2†). Al3+ could be detected down to 0.13 μM based on the 3α/slope when 5 μM PNI was employed. The 1
1 complex of PNI (Scheme 2 and Fig. S2†). Al3+ could be detected down to 0.13 μM based on the 3α/slope when 5 μM PNI was employed. The 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complex was supported by the electrospray mass spectrum of PNI and Al3+ (Fig. S3, ESI†). The ESI-MS peak at m/z = 412.07 (calculated = 412.04) was attributed to {[PNI + Al3+ + 2NO3−]}+.
1 complex was supported by the electrospray mass spectrum of PNI and Al3+ (Fig. S3, ESI†). The ESI-MS peak at m/z = 412.07 (calculated = 412.04) was attributed to {[PNI + Al3+ + 2NO3−]}+.
Colorimetric and fluorometric assay of cyanide ion
The anion binding affinity of PNI was evaluated by monitoring their UV-vis and steady-state emission properties as a function of anion concentration in methanol. The changes in the UV-vis spectrum of PNI were observed as a function of CN− concentration (Fig. 5a). The bands at 286, 320, 457, and 480 nm decreased and a new band at 300 nm appeared. Two clear isosbestic points observed at 368 and 400 nm indicated a clean conversion throughout the titration. A photograph of the color changes induced upon anion treatment to methanol solutions of PNI is shown in Fig. 5b. An obvious color change from yellow to red was observed only upon treatment with cyanide. Previously, some organic dyes have been reported as colorimetric sensors for cyanide ions, in which all probes were covalently bound with cyanide upon addition.22,23,29The fluorescence spectrum of free PNI showed an emission band at 518 nm. As shown in Fig. 6, only cyanide ions caused significant fluorescence changes of PNI compared to other anions examined. The fluorescence maximum was shifted to 540 nm upon treatment with cyanide. The fluorescence responses of PNI were unperturbed by F−, Cl−, Br−, I−, CH3COO−, and H2PO4− at 100-fold excess. The titration experiment showed a progressive intensity decrease at 518 nm (Fig. 7).
 | ||
| Fig. 6 Fluorescence spectral changes of 50 μM PNI obtained at 518 nm upon the addition of 100 equiv. of various anions in methanol. All fluorescence spectra were acquired with excitation at 450 nm. | ||
 | ||
| Fig. 7 (a) Fluorescence titrations of 50 μM PNI with tetraethylammonium cyanide (0 to 500 equiv.) in CH3OH. (b) Ratio of fluorescence intensities at 518 nm as a function of cyanide concentration. | ||
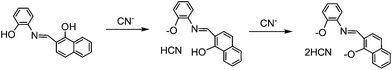
Further insights into the nature of PNI and cyanide interactions were studied by 1H-NMR titration experiments. Addition of cyanide to PNI did not show any significant changes in the NMR spectra (Fig. S4†). The well resolved resonance signals of aromatic protons of PNI became a little broad and shifted upon addition of cyanide. Interestingly, the imine proton signal also appeared no evident shift in the absence and presence of cyanide. Usually, the neighboring protons to cyanide bonding covalently to probes were shifted significantly in NMR spectra.18,21,31 Therefore, the fluorescence behavior of PNI towards cyanide can be explained as a deprotonation process of phenol and naphthol (Scheme 3). Such a fluorescence quenching of a probe upon treatment of cyanide was also proposed due to deprotonation.25
Live cell imaging
We examined the bioimaging application of PNI for mapping aluminum ions in living cells. HeLa cells were first exposed to 0 and 1 μM Al(NO3)3 for 4 h and then incubated with the chemosensor (10 μM) for 20 min. Low background fluorescence was observed in the cells (Fig. 8a) that had not been exposed to Al(NO3)3. The background fluorescence derived from cellular metal ions, such as Na+, Ca2+, Mg2+, Zn2+, etc., which also afforded low fluorescence with PNI (Fig. 8b). Weak but still discernible fluorescence was observed in the cells previously exposed Al(NO3)3 at 1 μM, compared to the unexposed cells. These results indicate that the fluorescence intensities in the exposed cells depended on the Al(NO3)3 concentrations. The PNI probe afforded strong fluorescence in the presence of intracellular Al3+, thus demonstrating its suitability for determining the exposure level of cells to aluminum ions.Conclusions
We have described a new fluorescent Schiff-base chemosensor PNI for sensing aluminum and cyanide ions. PNI exhibited high selectivity for Al3+ in aqueous solution over competing relevant metal ions and a large turn-on response for detecting aluminum ions. In addition, the treatment with anions to the methanol solution leads to different photophysical behaviors. PNI showed the responses specific for cyanide ions, resulting in readily distinguished by color. Furthermore, PNI is capable of mapping aluminum levels in live cells, which might be exploited as specific and effective sensors for intracellular aluminum ions.Experimental section
Materials and instrumentation
All the solvents and reagents (analytical grade and spectroscopic grade) were obtained from Sigma-Aldrich and used as received. Water was purified with a MilliQ purification system. The metal ion solutions were prepared with metal nitrate salts in methanol. 1H and 13C-NMR spectra were recorded on a Varian 400 spectrometer (Palo Alto, CA, USA). Chemical shifts (δ) are reported in ppm, relative to tetramethylsilane Si(CH3)4. Absorption spectra were recorded at 25 °C using a Perkin-Elmer model Lambda 2S UV/Vis spectrometer (Waltham, MA, USA). Emission spectra were recorded on a Perkin-Elmer LS45 fluorescence spectrometer. Electrospray ionization mass spectra (ESI-MS) were collected on a Thermo Finnigan (San Jose, CA, USA) LCQTM Advantage MAX quadrupole ion trap instrument, by infusing samples directly into the source at 25 μL min−1 with a syringe pump. The spray voltage was set at 4.7 kV and the capillary temperature at 70 °C. Elemental analysis for carbon, nitrogen, and hydrogen was carried out by using a Flash EA 1112 elemental analyzer (thermo) in Organic Chemistry Research Center of Sogang University, Korea. To confirm the accuracy and reliability of the analytical procedure, the concentrations of various metal ions were checked by inductively coupled plasma (ICP) spectroscopic analysis.Synthesis of ortho-phenyljulolidineimine (PNI)
A solution of 2-aminophenol (0.12 g, 1.1 mmol) in methanol was added to a solution containing 1-hydroxy-2-naphthaldehyde (0.18 g, 1 mmol) in methanol. The reaction mixture was stirred for 5 h at room temperature until an orange precipitate appeared. The resulting precipitate was filtered and washed 2 times with ice methanol. The yield: 0.13 g (61%). 1H NMR (DMSO-d6, 400 MHz): 14.61 (d, J = 12 Hz, 1H), 10. 42 (s, 1H), 8.86 (d, J = 12 Hz, 1H), 8.32 (d, J = 8 Hz, 1H), 7.70 (d, J = 8 Hz, 1H), 7.65 (m, 2H), 7.42 (t, J = 8 Hz, 1H), 7.21 (d, J = 8 Hz, 1H), 7.09 (t, J = 8 Hz, 1H), 7.03 (d, J = 8 Hz, 1H), 6.94 (t, J = 8 Hz, 1H), 6.81 (d, J = 8 Hz, 1H). 13C NMR (DMSO-d6, 400 MHz): d 171.08, 163.64, 146.84, 136.69, 129.98, 128.54, 128.38, 128.27, 128.00, 127.81, 127.49, 125.25, 124.42, 123.63, 121.67, 120.44, 118.92, 115.61, 110.57 ppm. Anal. calcd for C17H13NO2 (263.09): C, 77.55; H, 4.98; N, 5.32. Found: C, 77.51; H, 5.01; N, 5.28%.UV-vis measurements of aluminum ion
Receptor PNI was dissolved in methanol and PNI were diluted with Bis–Tris buffer to make the final concentration of 10 μM. Al(NO3)3·9H2O was dissolved in Bis–Tris buffer. A certain amount of the Al3+ solution was transferred to each receptor PNI solution. After shaking the vials for a few minutes, UV-vis spectra were taken at room temperature.Fluorescence measurements of aluminum ion
Receptor PNI was dissolved in methanol and PNI was diluted in Bis–Tris buffer to make the final concentration of 10 μM. Al(NO3)3 was dissolved in Bis–Tris buffer. The Al3+ solution was transferred to each receptor PNI solution prepared above. After shaking the vials for a few minutes, fluorescence spectra were taken at room temperature.Competition of aluminum ion to other metal ions
M(NO3)x were dissolved in Bis–Tris buffer, respectively. Each metal solution (10 mM) was added into each PNI solution (10 μM). Then, Al3+ solution was added into the mixed solution of PNI containing a competing metal ion.Job plot measurement of aluminum ion
Al(NO3)3 dissolved in Bis–Tris buffer was added to each PNI solution diluted in Bis–Tris buffer. Each vial had a total volume of 3 mL. After shaking the vials for a few minutes, fluorescence spectra were taken at room temperature.UV-vis and fluorescence measurements of cyanide
Tetraethylammonium cyanide in methanol was transferred to each PNI solution (50 μM). After shaking the vials for a few minutes, UV-vis and fluorescence spectra were taken at room temperature.Competition with other anions
Each anion solution (tetraethylammonium salts) was added into 3 mL of each PNI solution (50 μM). Then, CN− solution was added into the mixture of each competing anion and PNI.Fluorescent imaging of intracellular Al3+ in cells
Human HeLa cell line were cultured in DMEM (Dulbecco's Modified Eagle Medium) which were supplemented with 100 units per ml penicillin, 100 mg ml−1 streptomycin, and 10% heat-inactivated fetal bovine serum at 37 °C in a humidified incubator. Cells were seeded on an 18 × 18 mm cover glass (Marienfeld, Lauda-Koenigshofen, Germany) at density 2 × 105 cells in culture media. HeLa cells were incubated with 1 μM of Al(NO3)3 in culture media for 4 hour at 37 °C. After washing with PBS three times to remove the remaining Al(NO3)3, cells were then incubated with 100 μM PNI for 20 min at RT. The treated cells were washed with PBS and mounted onto a glass slide with ClearMount™ aqueous mounting medium (Invitrogen). The fluorescent images of the mounted HeLa cells were obtained by using a confocal laser scanning microscope (CLSM LSM510, Carl Zeiss) with a 480 nm excitation and LP 520 nm emission filters at various magnifications (200× to 400×).Acknowledgements
This work was supported by the National Research Foundation of Korea Grant funded by the Korea Government (NRF-2013R1A2A2A03015101, 2012001725, 2012008875), and the National R&D program of the Ministry of Education, Science and Technology (MEST), Korea: Development of Molecular Sensing Technology (Code number 2012-009832).References
- C. S. Cronan, W. J. Walker and P. R. Bloom, Nature, 1986, 324, 140–143 CrossRef CAS.
- G. D. Fasman, Coord. Chem. Rev., 1996, 149, 125–165 CrossRef CAS.
- P. Nayak, Environ. Res., 2002, 89, 101–115 CrossRef CAS.
- J. R. Walton, J. Inorg. Biochem., 2007, 101, 1275–1284 CrossRef CAS PubMed.
- B. Valeur and I. Leray, Coord. Chem. Rev., 2000, 205, 3–40 CrossRef CAS.
- J. Barcelo and C. Poschenrieder, Environ. Exp. Bot., 2002, 48, 75–92 CrossRef CAS.
- J. Lee, H. Kim, S. Kim, J. Y. Noh, E. J. Song, C. Kim and J. Kim, Dyes Pigm., 2013, 96, 590–594 CrossRef CAS PubMed.
- S. Kim, J. Y. Noh, K. Y. Kim, J. H. Kim, H. K. Kang, S.-W. Nam, S. H. Kim, S. Park, C. Kim and J. Kim, Inorg. Chem., 2012, 51, 3597–3602 CrossRef CAS PubMed.
- D. Maity and T. Govindaraju, Eur. J. Inorg. Chem., 2011, 36, 5479–5485 CrossRef.
- S. Sen, T. Mukherjee, B. Chattopadhyay, A. Moirangthem, A. Basu, J. Marek and P. Chattopadhyay, Analyst, 2012, 137, 3975–3981 RSC.
- J. Y. Noh, S. Kim, I. H. Hwang, G. Y. Lee, J. Kang, S. H. Kim, J. Min, S. Park, C. Kim and J. Kim, Dyes Pigm., 2013, 99, 1016–1021 CrossRef CAS PubMed.
- H. M. Park, B. N. Oh, J. H. Kim, W. Qiong, I. H. Hwang, K.-D. Jung, C. Kim and J. Kim, Tetrahedron Lett., 2011, 52, 5581–5584 CrossRef CAS PubMed.
- W. H. Organization, Guidelines for Drinking-Water Quality, 1996 Search PubMed.
- W. Gerhartz, S. Yamamoto, F. T. Campbell, R. Pfefferkorn and J. F. Rounsaville, Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, New York, 1999 Search PubMed.
- S. I. Baskin and T. G. Brewer, Medical Aspects of Chemical and Biological Warfare, TMM Publications, Washington, 1997 Search PubMed.
- H. Hachiya, S. Ito, Y. Fushinuki, T. Masadome, Y. Asano and T. Imato, Talanta, 1999, 48, 997–1004 CrossRef CAS.
- (a) S.-Y. Chung, S.-W. Nam, J. Lim, S. Park and J. Yoon, Chem. Commun., 2009, 2866–2868 RSC; (b) Z. Xu, X. Chen, H. N. Kim and J. Yoon, Chem. Soc. Rev., 2010, 39, 127–137 RSC.
- K.-S. Lee, H.-J. Kim, G.-H. Kim, I. Shin and J.-I. Hong, Org. Lett., 2008, 10, 49–51 CrossRef CAS PubMed.
- F. Garcia, J. M. Garcia, B. Garcia-Acosta, R. Martinez-Manez, F. Sanceno and J. Soto, Chem. Commun., 2005, 2790–2792 RSC.
- J. V. Ros-Lis, M. D. Marcos, R. Martinez-Manez, K. Rurack and J. Soto, Angew. Chem., Int. Ed., 2005, 44, 4405–4407 CrossRef CAS PubMed.
- C.-L. Chen, Y.-H. Chen, C.-Y. Chen and S.-S. Sun, Org. Lett., 2006, 8, 5053–5056 CrossRef CAS PubMed.
- M. Jamkratoke, V. Ruangpornvisuti, G. Tumcharen, T. Tuntulani and B. Tomapatanaget, J. Org. Chem., 2009, 74, 3919–3922 CrossRef CAS PubMed.
- T. Agou, M. Sekine, J. Kobayashi and T. Kawashima, Chem.–Eur. J., 2009, 15, 5056–5062 CrossRef CAS PubMed.
- J. L. Sessler and D.-G. Cho, Org. Lett., 2008, 10, 73–75 CrossRef CAS PubMed.
- Y. M. Chung, B. Raman, D.-S. Kim and K. H. Ahn, Chem. Commun., 2006, 186–188 RSC.
- R. Badugu, J. R. Lakowicz and C. D. Geddes, J. Am. Chem. Soc., 2005, 127, 3635–3641 CrossRef CAS PubMed.
- T. W. Hudnall and F. P. Gabba, J. Am. Chem. Soc., 2007, 129, 11978–11986 CrossRef CAS PubMed.
- P. Anzenbacher, Jr, D. S. Tyson, K. Juriskova and F. N. Castellano, J. Am. Chem. Soc., 2002, 124, 6232–6233 CrossRef PubMed.
- Z. Ekmekci, M. D. Yilmaz and E. U. Akkaya, Org. Lett., 2008, 10, 461–464 CrossRef CAS PubMed.
- (a) J. H. Lee, A. R. Jeong, I.-S. Shin, H.-J. Kim and J.-I. Hong, Org. Lett., 2010, 12, 764–767 CrossRef CAS PubMed; (b) S.-Y. Na, J.-Y. Kim and H.-J. Kim, Sens. Actuators, B, 2013, 188, 1043–1047 CrossRef CAS PubMed; (c) K.-H. Hong and H.-J. Kim, Supramol. Chem., 2013, 25, 24–27 CrossRef CAS.
- J. Ren, W. Zhu and H. Tian, Talanta, 2008, 75, 760–764 CrossRef CAS PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available: ESI-MS, Job's plot, and additional NMR. See DOI: 10.1039/c4ra01245c |
| ‡ Both authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2014 |