Zirconium–Beta zeolite as a robust catalyst for the transformation of levulinic acid to γ-valerolactone via Meerwein–Ponndorf–Verley reduction†
Jie Wang,
Stephan Jaenicke and
Gaik-Khuan Chuah*
Department of Chemistry, National University of Singapore, 3 Science Drive 3, Kent Ridge, 117543, Singapore. E-mail: chmcgk@nus.edu.sg; Fax: +65 6779 1691; Tel: +65 6516 2839
First published on 28th February 2014
Abstract
Zr–Beta zeolite is a robust and active catalyst for the Meerwein–Ponndorf–Verley reduction of levulinic acid to γ-valerolactone, a versatile intermediate for bio-fuels and chemicals. In a batch reactor, γ-valerolactone was formed with a selectivity of >96%. In a continuous flow reactor, >99% yield of γ-valerolactone was obtained with a steady space-time-yield of 0.46 molGVLgZr−1 h−1 within 87 h, on a par with that of noble metal based catalysts. The high activity of this catalyst was attributed to the presence of Lewis acidic sites with moderate strength. Due to the relatively few basic sites, it is not poisoned by the acidic reactant. Its robustness in liquid and gas phase reactants coupled with good thermal stability makes Zr–Beta a green regenerable catalyst that can be used directly on levulinic acid without the need for derivatization.
1 Introduction
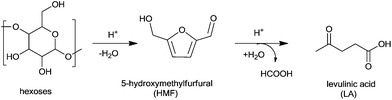
The threat of future shortage and ultimately of the depletion of fossil fuels has made the utilization of biomass especially attractive to researchers and manufacturers.1–5 In a bio-refinery, bio-fuels and value-added chemicals are produced from renewable bio-feedstocks.6–8 The US Department of Energy has identified several biomass-derived compounds as platform molecules on which to focus future research endeavors.9,10 Levulinic acid is one of these platform molecules; it can be obtained through hydrolysis/dehydration of hexoses such as glucose and fructose, or hexose-containing polymers like starch and cellulose (Scheme 1).11–15 The utilization of nonedible lignocelluloses is particularly attractive as it avoids any potential competition with food supplies.16–18The hydrogenation of levulinic acid gives γ-valerolactone (GVL), which is a sustainable liquid for energy and carbon-based chemicals.19–23 Recently, the group of Dumesic24 developed a route to convert γ-valerolactone into branched alkanes with molecular weights appropriate for liquid transportation fuels. Esterification of pentenoic acid derived from γ-valerolactone gives “valeric biofuels” which are potential substitutes for gasoline and diesel components.25 Useful chemicals such as 1,4-pentanediol or 2-methyltetrahydrofuran can be obtained by chemoselective hydrogenolysis of γ-valerolactone.13,26,27
As biomass derivatives have high oxygen content, an oxygen removal step is essential for the upgrading of biomass feedstock to biofuels and chemicals.10,28 This normally requires external H2 incurring drawbacks such as the need for pressure equipment, the loss of the petroleum-derived H2 in the form of water, and the use of noble metal-based homogeneous or heterogeneous catalysts.29–34 Instead of gaseous H2, the catalytic transfer hydrogenation (CTH) process offers an alternative approach by using hydrogen donors such as formic acid, formate salts, sec-alcohols, cyclohexene and hydrazine.35 The use of formic acid for reduction is especially attractive since an equimolar amount of formic acid is formed during the production of levulinic acid from carbohydrates.36–39 However, the reaction requires expensive noble metal catalysts36–38 or harsh conditions.39,40 Furthermore, the noble metals catalyze the decomposition of formic acid so that the reduction seems to proceed via in situ formed H2 rather than transfer hydrogenation, necessitating the use of a closed system.36–38
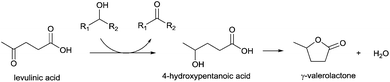
Instead of formic acid, the carbonyl group in levulinic acid can be reduced by secondary alcohols via the Meerwein–Ponndorf–Verley (MPV)41–43 reduction (Scheme 2). Wise and Williams44 showed that although the carbonyl group of levulinic acid is not chemically labile, the reaction is favored due to lactonisation of 4-hydroxypentanoic acid (4-HPA) to γ-valerolactone. However, little attention was paid to this strategy, until Chia and Dumesic45 recently screened different metal oxides for this reaction and found that zirconium oxide was the most active. In the flow sheet of the bio-refinery, the biomass is primarily transferred into 5-hydromethylfurfural (HMF) which further hydrolyses to levulinic acid.46 Following this, a biphasic system could be employed to extract the levulinic acid from the aqueous phase.47,48 If sec-alcohols are employed as the extraction reagent, they can form alternative hydrogen donors other than formic acid. The MPV reaction has the advantage that inexpensive non-noble metal catalysts can be used. Whilst the traditional MPV catalyst was aluminium alkoxide, a number of heterogeneous catalysts such as zeolites,49–52 mesoporous materials,53,54 metal oxides or hydroxides,55–62 hydrotalcites63,64 and K3PO4 (ref. 65) have been reported in recent times. The conditions for liquid phase MPV reduction are usually mild, taking place under ambient pressure at the boiling point of the secondary alcohol.
Our previous work has shown that zirconium-based catalysts, especially Zr–Beta zeolites, are highly active for the MPV reduction of substituted cyclohexanones and α,β-unsaturated aldehydes.52,66,67 This motivated us to investigate the use of Zr-Beta in the MPV reduction of levulinic acid to γ-valerolactone. By incorporating very small amounts of zirconium (Si/Zr ∼75–200) into the zeolite framework, isolated zirconium atoms with Lewis acidic property are formed. Computational studies using density functional theory show that the zirconium ions are located at specific crystallographic positions of the zeolite framework and play an important role in adsorption and activation of reactants.68 The ability of the catalyst to function under acidic conditions is important for the title reaction. Hydrous zirconia (ZrO(OH)n) and zirconia (ZrO2) have been compared. The activities were tested in a batch reactor as well as in a continuous flow reactor for potential industrial utilization.
2 Experimental
2.1 Catalyst preparation
2.2 Characterization
The surface area and porosity of the samples were determined by nitrogen adsorption (Micromeritics Tristar 3000). Prior to each measurement, the sample was thoroughly degassed under a nitrogen flow for 4 h. The degassing temperature was 100 °C for ZrO(OH)n calcined below 300 °C, and 300 °C for the other catalysts. The crystalline phase was determined by powder X-ray diffraction (Siemens D5005 equipped with Cu anode and variable slits). The diffractograms were measured at a step size of 0.02° and a dwell time of 1 s. The elemental composition was determined by inductively coupled plasma-atomic emission spectroscopy (ICP-AES) after dissolving the sample in HF. Infrared spectra of the samples in KBr were recorded in the range of 4000–400 cm−1 using a Bio-Rad Excalibur FT-IR spectrometer with a resolution of 4 cm−1. Thermogravimetric analyses were performed on a Dupont SDT 2960 apparatus to determine the water content in the hydrous zirconia. The sample was kept at 100 °C for 0.5 h to remove physically absorbed water, and then heated to 800 °C at 10 °C min−1. The degree of hydroxylation was calculated from the weight loss. The desorption of residual organics on used Zr-Beta as well as the catalyst with adsorbed levulinic acid or γ-valerolactone was measured by thermogravimetry combined with mass spectrometry (Mettler-Toledo TGA/DSC Stare with Pfeiffer Thermostar mass spectrometer). The acidic and basic nature of the samples was quantified by temperature programmed desorption of NH3 and CO2, respectively. The sample was pretreated at its calcined temperature for 2 h in a flow of helium (50 ml min−1). After cooling to 100 °C, NH3 or CO2 gas was introduced for 15 min. The sample was flushed with helium for another 2 h before heating at 10 °C min−1. The desorption of NH3 or CO2 was monitored by a quadrupole mass spectrometer (Balzers Prisma 200). Lewis and Brønsted acidity was investigated by FT-IR spectra of adsorbed pyridine. A self-supporting sample disc (10–20 mg) was pretreated in an evacuated (100 Pa) glass cell at 300 °C for 2 h. After cooling to room temperature, a background spectrum was recorded using a Bio-Rad Excalibur FT-IR spectrometer. The sample was exposed to pyridine for 15 min and re-evacuated at 100–300 °C for 1 h before measuring the IR spectra.2.3 Catalytic activity evaluation
3 Results and discussion
3.1 Materials
The X-ray diffractograms of Zr-Beta samples (Si/Zr 75–200) showed the characteristic peaks of the zeolite Beta phase (Fig. 1). The samples had high surface areas between 436–503 m2 g−1. The ZrAl-Beta samples showed similar textural properties as the Zr–Beta zeolites (Table 1). All the samples were largely microporous with total pore volumes of 0.26–0.29 cm3 g−1. The ICP analyses confirmed that both aluminium and zirconium were successfully incorporated into the samples, although the Si/Al and Si/Zr ratios in the zeolite were slightly higher than in the initial synthesis gel. | ||
| Fig. 1 X-ray diffraction patterns of (a) Zr-Beta-75 (b) Zr-Beta-100 (c) Zr-Beta-150 (d) Zr-Beta-200 and (e) ZrAl-Beta-100. | ||
The as-synthesized hydrous zirconia dried at 100 °C had a high surface area of 400 m2 g−1 (Table 2) and contained micropores as well as mesopores up to 12 nm (Fig. S1†). After calcination at 300 °C, the surface area was reduced to 271 m2 g−1. Although there was a reduction in the micropore volume, the pore size distribution of the mesopores remained similar to that of the as-synthesized sample. However, after calcination at 400 and 600 °C, the surface area decreased to 141 and 51 m2 g−1, respectively. The samples calcined above 400 °C contained bigger mesopores in the range of 8–20 nm. XRD showed that the hydrous zirconia samples were amorphous, but after calcination at 400 °C, crystalline zirconia with predominantly monoclinic phase was formed (Fig. S2†). The percentage of monoclinic phase increased from 77 to 83% as the calcination temperature was raised from 400 to 600 °C. The transformation to zirconia is accompanied by condensation of the hydroxyl groups in hydrous zirconia and the weight loss was monitored by thermogravimetry. Samples calcined at higher temperatures had smaller weight loss.
| Sample | Surf. area (m2 g−1) | Mean porea ϕ (nm) | Pore vol. (cm3 g−1) | Monoclinic phase (%) | Weight lossb (%) |
|---|---|---|---|---|---|
| a Pore size distributions were calculated from the desorption branch of the isotherms by the BJH method.b From TGA results in the range of 100 to 800 °C. | |||||
| ZrO(OH)n-100 | 400 | 5.3 | 0.56 | Amorp. | 11.8 |
| ZrO(OH)n-200 | 326 | 5.3 | 0.47 | Amorp. | 8.3 |
| ZrO(OH)n-300 | 271 | 5.5 | 0.42 | Amorp. | 7.1 |
| ZrO(OH)n-400 | 141 | 8.4 | 0.35 | 77 | 4.2 |
| ZrO(OH)n-500 | 84 | 12.5 | 0.32 | 81 | 3.8 |
| ZrO(OH)n-600 | 51 | 17.0 | 0.26 | 83 | 3.6 |
The amount of water, n, associated with the hydrous zirconia, ZrO2·nH2O, decreased from 0.92 in the as-synthesized sample to 0.30 after calcination at 400 °C. These results correlate well with that from IR spectroscopy (Fig. S3†). Hydrous zirconia has absorption bands at ca. 3400 cm−1 due to O–H stretching, ca. 1620 cm−1 due to the scissor bending mode of coordinated molecular water and ca. 1390 cm−1 due to atmospheric CO2 adsorbed on the sample forming a bicarbonate-like species.69,70 All these bands decreased in intensity as the sample was heated to higher temperatures.
The overall acidity and basicity of the samples was determined by TPD of NH3 and CO2, respectively (Table 3). For ZrO(OH)n-300, desorption of NH3 was observed from 120 to 300 °C with a maximum at 200 °C (Fig. 2). When hydrous zirconia had been calcined to 400 °C and higher, the NH3 desorption curve became bimodal with maxima at around 200 °C and 420 °C. The lower temperature peak below 300 °C was assigned to desorption from weak acid sites while NH3 desorption above this temperature was taken to be indicative of strong acidic sites. After calcination at 400 °C, the amorphous hydrous zirconia transformed to crystalline zirconia. The results show that the resulting oxide has weak as well as strong acid sites, with the latter being predominant. The density of both weak and strong acid sites decreased with higher calcination temperature, due to a loss in surface area. In contrast to hydrous zirconia, Zr-Beta-100 has acid sites of moderate strength. The NH3 desorption occurred between 220 to 390 °C, which is in the temperature range intermediate between the low and temperature peaks observed for hydrous zirconia. Furthermore, the density of acidic sites, 45 μmol g−1, was lower than that for the zirconia samples.
| Sample | Aciditya (μmol g−1) | L/B ratiob | Basicity (μmol g−1) | |
|---|---|---|---|---|
| Weak | Strong | |||
| a Integrated from the NH3 TPD peak areas with cutoff at ∼300 °C.b Lewis/Brønsted ratio from pyridine IR after heating at 100 °C for 1 h. | ||||
| ZrO(OH)n-300 | 137 | — | 6.7 | 132 |
| ZrO(OH)n-400 | 99 | 204 | 2.1 | 112 |
| ZrO(OH)n-500 | 25 | 146 | n.d | 53 |
| ZrO(OH)n-600 | 12 | 35 | n.d | 25 |
| Zr-Beta-100 | 45 (In total) | 8.4 | 3.8 | |
| ZrAl-Beta-25 | n.d. | 0.85 | n.d | |
 | ||
| Fig. 2 NH3 desorption profiles of (a) Zr-Beta-100 (b) ZrO(OH)n-300 (c) ZrO(OH)n-400 (d) ZrO(OH)n-500 and (e) ZrO(OH)n-600. | ||
The nature of the acid sites on the catalysts was determined by FT-IR measurements after adsorption of pyridine (Fig. S4† and Table 3). The bands at 1446 and 1540 cm−1 are assigned to pyridine adsorbed at Lewis and Brønsted acid sites, respectively.71 On ZrO(OH)n-300 and Zr-Beta-100, Lewis acid sites dominated, with Lewis/Brønsted ratio of 6.7 and 8.4, respectively. Only weak Brønsted acid sites were found for these two samples as the intensity of the 1540 cm−1 band decreased significantly after heating at 100 °C. On the other hand, ZrO(OH)n-400 and ZrAl-Beta-25 showed stronger Brønsted acid sites as the 1540 cm−1 band was still present even after heating to 200 °C. These samples had a higher density of Brønsted acid sites than Zr-Beta-100.
The presence of basic sites was probed by CO2 TPD. There was hardly any desorption of CO2 from Zr-Beta-100 showing that it has very few basic sites. In contrast, the amount of CO2 desorbed from hydrous zirconia/zirconia was in the range of 25 to 132 μmol g−1, reflecting the amphoteric nature of these samples. With increase of calcination temperature from 300 to 600 °C, the CO2 desorption was shifted to higher temperatures, indicating an increase in the basicity of the samples (Fig. S5†).
3.2 Effect of sec-alcohol and reaction temperature
The MPV reduction of levulinic acid was tested using a number of different secondary alcohols. The alcohols were used in excess, acting both as solvent and as hydrogen donor. Normally, 2-propanol is the best reducing agent.52,65 However, in the liquid phase reduction of levulinic acid with 2-propanol, only 5.6% conversion was obtained after 18 h despite using 200 mg of Zr-Beta-100 (Table 4). The low activity could be due to the aliphatic nature of levulinic acid as Zr-Beta had previously been shown to be a highly efficient catalyst for the MPV reduction of cyclohexanones and α,β-unsaturated aldehydes.52 The formation of γ-valerolactone involves the tandem reduction of levulinic acid to 4-hydroxypentanoic acid followed by cyclization and loss of one molecule of water. By using 2-propanol as the hydrogen donor and solvent, the reaction temperature is limited to 82 °C in open system which may be too low to remove the water formed. To increase the reaction temperature, aliphatic secondary alcohols with higher boiling points like 2-butanol and 2-pentanol were employed.| Alcohol | Temp. (°C) | Time (h) | LA conv. (%) | GVL sel. (%) | GVL yield (%) |
|---|---|---|---|---|---|
| a Reaction condition: 200 mg Zr-Beta-100, 1 mmol LA, 5 ml sec-alcohol.b 100 mg Zr-Beta-100 as the catalyst.c By-products from cyclohexanol were formed. | |||||
| 2-propanol | 82 | 18 | 5.6 | 72 | 4.0 |
| 2-butanol | 100 | 18 | 77 | 93 | 72 |
| 2-pentanol | 100 | 18 | 58 | 95 | 55 |
| 2-pentanol | 118 | 10 | 100 | 96 | 96 |
| 2-pentanol | 118 | 22b | 88 | 95 | 83 |
| Cyclohexanol | 150 | 6 | 100c | 82 | 82 |
The conversion increased significantly to 77% when using 2-butanol at 100 °C. Although the reducing capability of 2-pentanol is not as good as that of 2-butanol, giving only 58% conversion of levulinic acid at 100 °C, its higher boiling point of 118 °C enabled a faster MPV reduction in liquid phase. Hence, after 10 h, full conversion was achieved. For all these linear alcohols, the only by-product was due to esterification between levulinic acid and the corresponding alcohol. Cyclohexanol has an even higher boiling point, but by-products such as cyclohexene were formed when the reaction was conducted at 150 °C.
3.3 Effect of substrate concentration
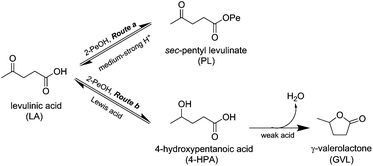
The sec-alcohol was used in excess (46 equiv.) and served both as the solvent and reducing agent. As levulinic acid has both carbonyl and carboxyl groups, two competing reactions, MPV reduction and esterification, can occur simultaneously (Scheme 3). An increase in the concentration of the limiting substrate accelerates both reactions. However, at higher concentrations of levulinic acid, the selectivity to γ-valerolactone was lower, showing that the esterification rate increased more than the MPV reduction. Use of 2 ml of 2-pentanol (7.2 wt% levulinic acid) resulted in only 75% selectivity to γ-valerolactone at full conversion whereas with 5 ml of 2-pentanol (2.9 wt% levulinic acid), 98% selectivity with 46% conversion was observed after 6 h (Table S1†). Even without adding any catalyst, the yield of the ester increased with higher levulinic acid concentration (Table S2†). Hence, a dilute system of 1 mmol (2.9 wt%) levulinic acid in 5 ml solvent was employed in the liquid phase to eliminate to reduce the rate of esterification and improve the selectivity to γ-valerolactone.3.4 Catalyst screening for liquid phase transformation
Comparing the Zr–Beta zeolites with different Si/Zr ratios, Zr-Beta-100 showed the best activity with 100% conversion and a very high selectivity of 96% to γ-valerolactone (Table 5). The only by-product was sec-pentyl levulinate due to esterification. Over Zr-Beta-150 and Zr-Beta-200, the conversion was lower, 71% and 27%, respectively. This may be attributed to the lower zirconium content in these samples. Despite their lower activity, the selectivity to γ-valerolactone was high, between 97–99%. On the other hand, Zr-Beta-75 contained more zirconium than Zr-Beta-100 but this catalyst was less active and selective, suggesting that an optimum number of isolated zirconium sites is essential for reaction. Based on the levulinic acid converted per g of Zr, the initial rates of the Zr-Beta catalysts with Si/Zr of 75–200 was between 16–31 mmolGVLgZr−1 h−1. The low initial rate indicates the difficulty to reduce the aliphatic carbonyl group in levulinic acid. In comparison, the initial rate for the MPV reduction of cyclohexanone and α,β-unsaturated aldehydes,52 is generally 20–30 times larger even at a lower temperature of 82 °C. To check for any leaching, the Zr-Beta-100 was hot filtered from the reaction medium after the conversion had reached 40%. No further increase in conversion was observed in the catalyst-free reaction mixture, showing that the MPV reduction of levulinic acid is heterogeneously catalyzed by Zr-Beta.| Catalyst | Time (h) | LA conv. (%) | GVL sel. (%) | GVL yield (%) | Initial ratec (mmolGVLgZr−1 h−1) |
|---|---|---|---|---|---|
| a Reaction conditions: 200 mg catalyst, 1 mmol LA, 5 ml 2-pentanol, 118 °C, reflux.b Reaction conditions: 100 mg catalyst, 2 mmol LA, 5 ml 2-butanol, 150 °C and autogenous pressure plus 5 bar He in autoclave.c Calculated based on the conversion after first 2 h. | |||||
| Zr-Beta-75 | 10 | 88 | 93 | 82 | 19 |
| Zr-Beta-100 | 10 | 100 | 96 | 96 | 30 |
| Zr-Beta-150 | 10 | 71 | 97 | 69 | 31 |
| Zr-Beta-200 | 10 | 27 | >99 | 27 | 16 |
| ZrAl-Beta-25 | 6 | 100 | 71 | 71 | — |
| ZrAl-Beta-100 | 6 | 91 | 79 | 72 | — |
| ZrO(OH)n-100 | 24 | 11 | 74 | 8.3 | 0.071 |
| ZrO(OH)n-200 | 24 | 13 | 67 | 8.8 | 0.075 |
| ZrO(OH)n-300 | 24 | 16 | 67 | 11 | 0.096 |
| ZrO(OH)n-400 | 24 | 37 | 62 | 23 | 0.22 |
| ZrO(OH)n-500 | 24 | 33 | 33 | 11 | 0.16 |
| ZrO(OH)n-600 | 24 | 12 | 45 | 5.4 | 0.076 |
| Zr-Beta-100 | 6 | 100b | 92 | 92 | — |
| ZrO(OH)n-200 | 16 | 68b | 56 | 38 | — |
| ZrO(OH)n-400 | 16 | 89b | 46 | 41 | — |
| ZrO(OH)n-600 | 16 | 45b | 36 | 16 | — |
The incorporation of Al into Zr-Beta led to Brønsted acidity which promoted the conversion of levulinic acid. Over ZrAl-Beta-25, the time for full conversion was reduced from 10 h to 6 h. However, the presence of strong Brønsted acid sites accelerated the esterification to sec-pentyl levulinate so that at 100% conversion, the selectivity to γ-valerolactone was only 71% (Scheme 3, Route a). Furthermore, the Brønsted acid sites catalyzed other side reactions of pentanol leading to hemiacetals, pentene and its isomers. Hence, the MPV reduction of the carbonyl group in levulinic acid is best catalyzed by a Lewis acid catalyst as the presence of Brønsted acid sites will reduce the overall selectivity to γ-valerolactone.
Compared to Zr-Beta zeolites, the ZrO(OH)n-T samples were less active and selective. After 24 h, the conversion was only between 11 to 37% (Table 5). The selectivity to γ-valerolactone decreased from 74 to 45% with increasing calcination temperature of the hydrous zirconia. The best catalyst among the hydrous zirconia was ZrO(OH)n-400 with 23% yield of γ-valerolactone. The dependence of the catalytic activity and selectivity on the calcination temperature may be attributed to its effect on the density of surface hydroxyl groups and the acidity of the samples. Surface hydroxyl groups are important in the MPV reaction as they react with the reducing alcohol to form the alkoxide intermediate.55 With increase of calcination temperature, the density of hydroxyl groups decreased as they condensed to form the oxide. On the other hand, NH3 TPD results show that stronger acid sites are formed that would favor the cyclization step. Hence a balance in the density of surface hydroxyls and acid strength is important for activity of the hydrous zirconia.
The activity of hydrous zirconia and Zr-Beta-100 were next tested under conditions similar to those reported by Chia and Dumesic.45 The authors used a closed system at a higher temperature of 150 °C with 2-butanol as the reducing alcohol (Table 5). It was found that a high γ-valerolactone yield of 92% could be achieved based on the zirconium oxide catalyst. However, an extremely dilute system (1 wt% levulinic acid) was necessary. When the concentration was increased to 5 wt%, the selectivity was only 42% at 51% conversion, giving a γ-valero-lactone yield of 22% These results are comparable with this work where 16% yield of γ-valerolactone was obtained over ZrO(OH)n-600 (Table 5). We observed that hydrous zirconia calcined at a lower temperature of 400 °C instead of 600 °C, ZrO(OH)n-400, was more active with 89% conversion and 46% yield of γ-valerolactone. Compared to hydrous zirconia, Zr-Beta-100 was able to maintain a very high γ-valerolactone selectivity of 92% at full conversion even at 5 wt% levulinic acid (Table 5).
The better activity of Zr–Beta zeolite for transforming levulinic acid to γ-valerolactone can be attributed to its Lewis acidity of moderate strength and its lack of basic sites which otherwise would react with the carboxylic acid functional group in the substrate. Chia and Dumesic45 found that levulinic acid was less readily reduced than levulinate esters over zirconia. They suggested that this was due to strong binding of levulinic acid to the basic sites on zirconia, leading to blockage and loss of activity. As Zr-Beta has very few basic sites, such inhibitory effects cannot occur. Furthermore, the microporous structure of the zeolite coupled with the absence of strong acid sites hinder the competing esterification reaction, so that a very high γ-valerolactone selectivity of 93–99% could be obtained.
The influence of the acid group in levulinic acid was investigated by using methyl levulinate as substrate. Over Zr-Beta-100, the conversion after 2 h was about twice that of levulinic acid (Fig. 3). The higher rate of reaction may be explained by the higher hydrophobicity of methyl levulinate which leads to improved adsorption at the surface of the hydrophobic Zr-Beta. The effect was more pronounced over hydrous zirconia where the 2 h-conversion of methyl levulinate was 4.5–11 times higher than levulinic acid. As with levulinic acid, the most active hydrous zirconia sample was ZrO(OH)n-400 with 100% conversion of methyl levulinate after only 6 h.
3.5 Continuous flow reactions over Zr–Beta zeolite
Zr-Beta-100 catalyst was also used in a continuous flow reactor to explore its stability, reusability and potential for industrial application. The feed was 5 wt% levulinic acid in 2-propanol. Initial tests showed that at a reaction temperature of 150 °C with a weight hourly space velocity (WHSV) of 0.16 h−1, full conversion was sustained for around 10 h (Fig. S6†). Due to the high boiling points of levulinic acid (245–246 °C) and γ-valerolactone (207–208 °C), it is highly probable that at this low temperature, these molecules remained significantly adsorbed at the surface of the catalyst. Thermogravimetric measurements of Zr-Beta-100 that had been used in the reaction for 34 h showed a weight loss of 9.7% after heating in air to 500 °C (Fig. S7†). The weight loss profile of the used Zr-Beta was similar to that of the catalyst adsorbed with levulinic acid with a steep weight loss between 200 to 350 °C (Fig. S8†). The hydrogen/carbon (H/C) ratio of the used Zr-Beta was 1.57, which is close to the H/C ratio of levulinic acid and γ-valerolactone rather than that of coke in zeolites (H/C around 1.25).72,73 The catalytic activity was recovered after calcination which also shows that the deactivation was mainly due to the adsorption of substrate and product molecules rather than by heavy coke formation.To promote the desorption of molecules, the reaction temperature was raised to 250 °C, which is higher than the boiling points, and the space velocity was also increased. At WHSV of 0.64 h−1, the γ-valerolactone yield remained at > 99% for as long as 87 h (Fig. 4). After this time, the conversion dropped and increasing amounts of propyl levulinate were formed. However, the activity was fully regained by recalcining the catalyst in air at 500 °C. Full conversion could again be maintained for prolonged periods of 72 and 78 h in the second and third run, respectively. The stability of the catalyst is of significant importance for industrial applications. The productivity of >99% γ-valerolactone yield under WHSV of 0.64 h−1 for the long period works out to 0.46 molGVLgZr−1 h−1. Pure γ-valerolactone could be obtained by simply distilling off the solvent 2-propanol for reuse in subsequent cycles. The isolated yield was about 93% and the product collected was in high purity as confirmed by 1H and 13C NMR (Fig. S9†).
 | ||
| Fig. 4 GVL yield as a function of time on stream for the MPV of levulinic acid over Zr-Beta-100 at 250 °C in a continuous flow reactor of WHSV = 0.64 h−1. Catalyst was regenerated at 110 h and 218 h. | ||
At higher space velocities of 2–4 h−1, the average rate of γ-valerolactone formation was around 0.68–0.71 molGVLgZr−1 h−1 (5.7 molGVLgZr−1 h−1 for the first 15 min with WHSV of 4.0 h−1) (Fig. S10†). The sustained productivity using Zr-Beta-100 compares very well with the reported values for metal-based catalysts in vapour phase where the productivity ranges from 0.09 to 0.36 molGVLgmetal−1 h−1 (Table 6). Due to its high activity for MPV reduction, Zr-Beta offers an alternative to γ-valerolactone formation from levulinic acid without the need for hydrogenation using precious metal catalysts. Furthermore, the reaction can be carried out without the need for high pressure of H2.
| Catalyst | T (°C) | P (bar) | H2 Sourcea | GVL yield (%) | Productivity (molGVLgmetal−1 h−1) | Stability (h) | Ref. |
|---|---|---|---|---|---|---|---|
| a 2-PrOH: 2-propanol; HCOOBu: butyl formate; 2-BuOH: 2-butanol. | |||||||
| Zr-Beta | 250 | 1 | 2-PrOH | >99 | 0.46 | 87 | — |
| Ru/C | 265 | 1 | H2 | 99 | 0.09 | 50 | 74 |
| RuSn/C | 220 | 35 | H2 | 93 | 0.36 | <100 | 75 |
| Pd/C + Ru/C | 170 | 35 | HCOOBu | 95 | 0.11 | 400 | 76 |
| Cu/SiO2 | 265 | 10 | H2 | >99 | 0.09 | 100 | 26 |
| ZrO2 | 150 | 20 | 2-BuOH | 20–40 | <0.001 | 150 | 45 |
4 Conclusions
Using sec-alcohol as the hydrogen source, biomass-derived levulinic acid was converted to γ-valerolactone via a tandem MPV reduction and cyclisation/lactonization. Zr–Beta zeolite (Si/Zr 100) is highly active and selective for the γ-valerolactone production, both in liquid phase batch reactions and in a gas phase continuous flow system. Quantitative conversion with >99% yield of γ-valerolactone was obtained with a steady generation rate of 0.46 molGVLgZr−1 h−1 during a single run in the flow reactor. The presence of Lewis acid sites with moderate strength and only relatively few basic sites are key factors for its good activity and lack of poisoning by the acidic substrate. Due to its high thermal stability, Zr-Beta could be easily recalcined and reused. In comparison, hydrous zirconia and zirconia were less active for the MPV reduction of levulinic acid with the most active hydrous zirconia catalyst being that obtained after calcination at 400 °C.Acknowledgements
Financial support for this work from the National University of Singapore grants R-143-000-476-112 and R-143-000-550-112 is gratefully acknowledged.Notes and references
- G. W. Huber, S. Iborra and A. Corma, Chem. Rev., 2006, 106, 4044–4098 CrossRef CAS PubMed.
- A. Corma, S. Iborra and A. Velty, Chem. Rev., 2007, 107, 2411–2502 CrossRef CAS PubMed.
- M. J. Climent, A. Corma and S. Iborra, Green Chem., 2011, 13, 520–540 RSC.
- J. A. Melero, J. Iglesias and A. Garcia, Energy Environ. Sci., 2012, 5, 7393–7420 CAS.
- P. Gallezot, Chem. Soc. Rev., 2012, 41, 1538–1558 RSC.
- B. Kamm, M. Kamm, P. R. Gruber and S. Kromus, in Biorefineries Industrial Processes and Products: Status Quo and Future Directions, ed. B.Kamm, P. R. Gruber and M. Kamm, Wiley-VCH, Weinheim, 2006, vol. 1, pp. 3–40 Search PubMed.
- J. H. Clark, V. Budarin, F. E. Deswarte, J. J. Hardy, F. M. Kerton, A. J. Hunt, R. Luque, D. J. Macquarrie, K. Milkowski and A. Rodriguez, Green Chem., 2006, 8, 853–860 RSC.
- J. H. Clark, F. E. I. Deswarte and T. J. Farmer, Biofuels, Bioprod. Biorefin., 2009, 3, 72–90 CrossRef CAS.
- J. J. Bozell and G. R. Petersen, Green Chem., 2010, 12, 539–554 RSC.
- J. C. Serrano-Ruiz, R. Luque and A. Sepúlveda-Escribano, Chem. Soc. Rev., 2011, 40, 5266–5281 RSC.
- S. W. Fitzpatrick, US Pat., 5608105, 1997.
- J. J. Bozell, L. Moens, D. Elliott, Y. Wang, G. Neuenscwander, S. Fitzpatrick, R. Bilski and J. Jarnefeld, Resour., Conserv. Recycl., 2000, 28, 227–239 CrossRef.
- H. Mehdi, V. Fábos, R. Tuba, A. Bodor, L. T. Mika and I. T. Horváth, Top. Catal., 2008, 48, 49–54 CrossRef CAS PubMed.
- O. O. James, S. Maity, L. A. Usman, K. O. Ajanaku, O. O. Ajani, T. O. Siyanbola, S. Sahu and R. Chaubey, Energy Environ. Sci., 2010, 3, 1833–1850 CAS.
- R. Weingarten, W. C. Conner and G. W. Huber, Energy Environ. Sci., 2012, 5, 7559–7574 CAS.
- R. Palkovits, Angew. Chem., Int. Ed., 2010, 49, 4336–4338 CrossRef CAS PubMed.
- D. W. Rackemann and W. O. S. Doherty, Biofuels, Bioprod. Biorefin., 2011, 5, 198–214 CrossRef CAS.
- S. Van de Vyver, J. Geboers, P. A. Jacobs and B. F. Sels, ChemCatChem, 2011, 3, 82–94 CrossRef CAS.
- I. T. Horváth, H. Mehdi, V. Fábos, L. Boda and L. T. Mika, Green Chem., 2008, 10, 238–242 RSC.
- J. C. Serrano-Ruiz and J. A. Dumesic, Energy Environ. Sci., 2011, 4, 83–99 CAS.
- W. R. H. Wright and R. Palkovits, ChemSusChem, 2012, 5, 1657–1667 CrossRef CAS PubMed.
- J. Zhang, S. Wu, B. Li and H. Zhang, ChemCatChem, 2012, 4, 1230–1237 CrossRef CAS.
- D. M. Alonso, S. G. Wettstein and J. A. Dumesic, Green Chem., 2013, 15, 584–595 RSC.
- J. Q. Bond, D. M. Alonso, D. Wang, R. M. West and J. A. Dumesic, Science, 2010, 327, 1110–1114 CrossRef CAS PubMed.
- J. P. Lange, R. Price, P. M. Ayoub, J. Louis, L. Petrus, L. Clarke and H. Gosselink, Angew. Chem., Int. Ed., 2010, 49, 4479–4483 CrossRef CAS PubMed.
- P. P. Upare, J. M. Lee, Y. K. Hwang, D. W. Hwang, J. H. Lee, S. B. Halligudi, J. S. Hwang and J. S. Chang, ChemSusChem, 2011, 4, 1749–1752 CrossRef CAS PubMed.
- X.-L. Du, Q.-Y. Bi, Y.-M. Liu, Y. Cao, H.-Y. He and K.-N. Fan, Green Chem., 2012, 14, 935–939 RSC.
- J. J. Bozell, Science, 2010, 329, 522–523 CrossRef CAS PubMed.
- R. A. Bourne, J. G. Stevens, J. Ke and M. Poliakoff, Chem. Commun., 2007, 4632–4634 RSC.
- K. Yan, C. Jarvis, T. Lafleur, Y. Qiao and X. Xie, RSC Adv., 2013, 3, 25865–25871 RSC.
- A. M. Hengne and C. V. Rode, Green Chem., 2012, 14, 1064–1072 RSC.
- A. M. R. Galletti, C. Antonetti, V. De Luise and M. Martinelli, Green Chem., 2012, 14, 688–694 RSC.
- S. G. Wettstein, J. Q. Bond, D. M. Alonso, H. N. Pham, A. K. Datye and J. A. Dumesic, Appl. Catal., B, 2012, 117, 321–329 CrossRef PubMed.
- W. Luo, U. Deka, A. M. Beale, E. R. van Eck, P. C. Bruijnincx and B. M. Weckhuysen, J. Catal., 2013, 301, 175–186 CrossRef CAS PubMed.
- R. A. W. Johnstone, A. H. Wilby and I. D. Entwistle, Chem. Rev., 1985, 85, 129–170 CrossRef CAS.
- L. Deng, J. Li, D. M. Lai, Y. Fu and Q. X. Guo, Angew. Chem., Int. Ed., 2009, 48, 6529–6532 CrossRef CAS PubMed.
- L. Deng, Y. Zhao, J. Li, Y. Fu, B. Liao and Q. X. Guo, ChemSusChem, 2010, 3, 1172–1175 CrossRef CAS PubMed.
- X. L. Du, L. He, S. Zhao, Y. M. Liu, Y. Cao, H. Y. He and K. N. Fan, Angew. Chem., Int. Ed., 2011, 50, 7815–7819 CrossRef CAS PubMed.
- Z. Shen, Y. L. Zhang, F. M. Jin, X. F. Zhou, A. Kishita and K. Tohji, Ind. Eng. Chem. Res., 2010, 49, 6255–6259 CrossRef CAS.
- D. Kopetzki and M. Antonietti, Green Chem., 2010, 12, 656–660 RSC.
- J. S. Cha, Org. Process Res. Dev., 2006, 10, 1032–1053 CrossRef CAS.
- G. K. Chuah, S. Jaenicke, Y. Z. Zhu and S. H. Liu, Curr. Org. Chem., 2006, 10, 1639–1654 CrossRef CAS.
- J. R. Ruiz and C. Jimenez-Sanchidrian, Curr. Org. Chem., 2007, 11, 1113–1125 CrossRef CAS.
- N. J. Wise and J. M. J. Williams, Tetrahedron Lett., 2007, 48, 3639–3641 CrossRef CAS PubMed.
- M. Chia and J. A. Dumesic, Chem. Commun., 2011, 47, 12233–12235 RSC.
- R.-J. van Putten, J. C. van der Waal, E. de Jong, C. B. Rasrendra, H. J. Heeres and J. G. de Vries, Chem. Rev., 2013, 113, 1499–1597 CrossRef CAS PubMed.
- Y. Román-Leshkov, J. N. Chheda and J. A. Dumesic, Science, 2006, 312, 1933–1937 CrossRef PubMed.
- E. Nikolla, Y. Roman-Leshkov, M. Moliner and M. E. Davis, ACS Catal., 2011, 1, 408–410 CrossRef CAS.
- E. Creyghton, S. Ganeshie, R. Downing and H. Van Bekkum, J. Mol. Catal. A: Chem., 1997, 115, 457–472 CrossRef CAS.
- A. Corma, M. E. Domine, L. Nemeth and S. Valencia, J. Am. Chem. Soc., 2002, 124, 3194–3195 CrossRef CAS PubMed.
- A. Corma, M. E. Domine and S. Valencia, J. Catal., 2003, 215, 294–304 CrossRef CAS.
- Y. Zhu, G. K. Chuah and S. Jaenicke, J. Catal., 2006, 241, 25–33 CrossRef CAS PubMed.
- P. P. Samuel, S. Shylesh and A. P. Singh, J. Mol. Catal. A: Chem., 2007, 266, 11–20 CrossRef CAS PubMed.
- A. Ramanathan, M. C. Castro Villalobos, C. Kwakernaak, S. Telalovic and U. Hanefeld, Chem.–Eur. J., 2008, 14, 961–972 CrossRef CAS PubMed.
- G. Chuah and S. Jaenicke, Appl. Catal., A, 1997, 163, 261–273 CrossRef CAS.
- P. S. Kumbhar, J. Sanchez-Valente, J. Lopez and F. Figueras, Chem. Commun., 1998, 535–536 RSC.
- S. Liu, S. Jaenicke and G. Chuah, J. Catal., 2002, 206, 321–330 CrossRef CAS.
- M. Kotani, T. Koike, K. Yamaguchi and N. Mizuno, Green Chem., 2006, 8, 735–741 RSC.
- F. J. Urbano, M. A. Aramendia, A. Marinas and J. M. Marinas, J. Catal., 2009, 268, 79–88 CrossRef CAS PubMed.
- J. Ramos, V. Díez, C. Ferretti, P. Torresi, C. Apesteguía and J. Di Cosimo, Catal. Today, 2011, 172, 41–47 CrossRef CAS PubMed.
- J. F. Miñambres, M. A. Aramendía, A. Marinas, J. M. Marinas and F. J. Urbano, J. Mol. Catal. A: Chem., 2011, 338, 121–129 Search PubMed.
- J. K. Bartley, C. Xu, R. Lloyd, D. I. Enache, D. W. Knight and G. J. Hutchings, Appl. Catal., B, 2012, 128, 31–38 CrossRef CAS PubMed.
- T. Jyothi, T. Raja and B. Rao, J. Mol. Catal. A: Chem., 2001, 168, 187–191 CrossRef CAS.
- J. R. Ruiz, C. Jiménez-Sanchidrián and J. M. Hidalgo, Catal. Commun., 2007, 8, 1036–1040 CrossRef CAS PubMed.
- R. Radhakrishnan, D. M. Do, S. Jaenicke, Y. Sasson and G. K. Chuah, ACS Catal., 2011, 1, 1631–1636 CrossRef.
- Y. Zhu, S. Jaenicke and G. Chuah, J. Catal., 2003, 218, 396–404 CrossRef CAS.
- Y. Nie, S. Jaenicke and G. K. Chuah, Chem.–Eur. J., 2009, 15, 1991–1999 CrossRef CAS PubMed.
- G. Yang, E. A. Pidko and E. J. M. Hensen, J. Phys. Chem. C, 2013, 117, 3976–3986 CAS.
- G. Guo and Y. Chen, J. Mater. Sci., 2004, 39, 4039–4043 CrossRef CAS.
- S. Axpuac, M. Aramendía, J. Hidalgo-Carrillo, A. Marinas, J. Marinas, V. Montes-Jiménez, F. Urbano and V. Borau, Catal. Today, 2012, 187, 183–190 CrossRef CAS PubMed.
- E. Parry, J. Catal., 1963, 2, 371–379 CrossRef CAS.
- D. M. Bibby, R. F. Howe and G. D. Mclellan, Appl. Catal., A, 1992, 93, 1–34 CrossRef CAS.
- J. Kim, M. Choi and R. Ryoo, J. Catal., 2010, 269, 219–228 CrossRef CAS PubMed.
- P. P. Upare, J.-M. Lee, D. W. Hwang, S. B. Halligudi, Y. K. Hwang and J.-S. Chang, J. Ind. Eng. Chem., 2011, 17, 287–292 CrossRef CAS PubMed.
- D. M. Alonso, S. G. Wettstein, J. Q. Bond, T. W. Root and J. A. Dumesic, ChemSusChem, 2011, 4, 1078–1081 CrossRef CAS PubMed.
- E. I. Gurbuz, D. M. Alonso, J. Q. Bond and J. A. Dumesic, ChemSusChem, 2011, 4, 357–361 CrossRef PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Fig. S1–S10, Tables S1 and S2. See DOI: 10.1039/c4ra01120a |
| This journal is © The Royal Society of Chemistry 2014 |