Synthesis of (−)-epibatidine†
Wen-Hua Chiou* and
Yu-Min Chiang
Department of Chemistry, National Chung Hsing University, Taichung, Taiwan 402, Republic of China. E-mail: wchiou@dragon.nchu.edu.tw; Fax: +886-4-22862547; Tel: +886-4-22840411-420
First published on 10th February 2014
Abstract
An asymmetric synthesis to the dendrobatid alkaloid (−)-epibatidine has been described, featuring chiral resolution of both optically pure 7-azabicyclo[2.2.1]heptanecarboxylic acid, and subsequent transformations to (−)-epibatidine. The methodology provides a flexible access to various substituted chiral epibatidine analogues.
In 1992, Daly and coworkers reported a dendrobatid alkaloid, epibatidine (1), isolated in trace amounts from the skin of the Ecuadoran poison frog Epipedobates tricolor.1,2 Epibatidine (1) has been found to be 200–400 times more potent than morphine as an analgesic, and appeared to act via a non-opioid mechanism since its effects are not blocked by the opiate receptor antagonist naloxone. In addition, epibatidine (1) is an extremely potent agonist of the nicotinic acetylcholine receptor.3 Due to its pharmacological activity, epibatidine (1) has attracted much attention from synthetic chemists, resulting in abundant approaches. However, its high toxicity also prevents therapeutic applications, and has prompted a search for safer analogues such as epiboxidine (2) (Fig. 1).4
Various strategies have been developed for efficient syntheses of the molecule, which have been reviewed by Trudell5 and Olivo.6 One of these approaches to synthesize the unique 7-azabicyclo[2.2.1]heptane framework in epibatidine is the cycloaddition reaction. N-Protected pyrroles could undergo Diels–Alder reactions with substituted acetylene or ethylene derivatives. Transannular SN2 displacement also provides a practical route to synthesize epibatidine.7 A well-established arrangement in a 1,4-disubstituted cyclohexyl ring system could trigger an SN2 reaction to give the 7-azabicyclo[2.2.1]heptane structure. Direct coupling of the 7-azabicyclo[2.2.1]heptan-2-one or its derivatives with the aromatic ring is an effective strategy for syntheses of epibatidine and analogues. Due to the diversity of the approach, many elegant approaches to synthesize 7-azabicyclo[2.2.1]heptane derivatives have been reported,8 including Aza-Prins-Pinacol rearrangement8e and Favorskii rearrangement of tropinone.9a,b As a part of the project devoted to asymmetric syntheses of alkaloids and derivatives for pharmaceutical purposes, here we describe a different approach to synthesize (−)-epibatidine (1). This strategy takes advantage of readily available carboxylic acid 3, and features a practical preparation of enantiopure acid 3 and construction of the 2-chloropyridine moiety from the carboxylic acid end.
Our approach commences with the preparation of racemic 7-azabicyclo[2.2.1]heptanecarboxylic acid, using Fevig' conditions,9c a modification based on Bai's procedure.9a,b We envision both optically active 7-azabicyclo[2.2.1]heptane carboxylic acids 3 are available through covalent bond modification with a chiral compound to two separable diastereomers, followed by removal of the chiral auxiliary. Subsequent functionality transformations of the carboxylic acid end effect the formation of the 2-chloropyridine moiety in epibatidine. Such an approach does not only synthesize the 2-chloropyridine moiety, but also allow diversity by construction of various bioisosteric rings or other modification. For example, epiboxidine (2) can be achieved by an acetoxime addition–cyclization protocol.8g With racemic acid 3 in hand, transformation of (±)-3 to separable diastereomers has been carried out by treating of acid 3 with SOCl2 to the resulting acid chloride, followed by reaction with various chiral auxiliaries.
We have utilized L-menthol and L-borenol as the chiral auxiliary, but both have failed to give separable diastereomeric adducts. Fortunately, after racemic acid 3 has been coupled with (4S)-benzyl-2-oxazolidinone, readily available from L-phenylalanine, two diastereomeric adducts were easily separated by column chromatography, to yield the less polar product 4a in 44% yield and the more polar product 4b in 41% yield, respectively (Scheme 1). In addition, recrystallization of oxazolidinone 4a within ethyl acetate and hexane provided a crystal for an X-ray analysis, which confirmed the absolute configuration as (1R,2S,4S)-7-azabicyclo[2.2.1]heptane moiety (Fig. 2).10 The results also disclosed the absolute configuration of the other diastereomer, amide 4b, as a (1S,2R,4R)-7-azabicyclo[2.2.1]heptane moiety. Subsequent basic hydrolysis conditions using H2O2 in THF for the less polar oxazolidinone derivative 4a proceeded successfully to yield optically pure (−)-acid 3 in 98% yield, while the more polar one 4b gave optically pure (+)-acid 3 in 93% yield (Scheme 1). Chiral HPLC analyses display the optical purity of (−)-acid 3 is more than 99% ee, (tR: 18.4 min for (+)-3, 26.3 min for (−)-3, see ESI†) (Fig. 3).
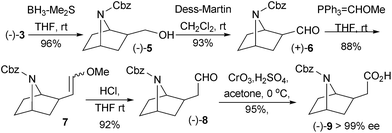
The next efforts were involved with the construction of the 5-substituted-2-chloropyridine moiety (Schemes 2 and 3). With optically pure acid (−)-3 in hand, we converted acid 3 to its homoaldehyde derivative by one-carbon homologation via an enol ether intermediate.11 Thus, reduction of acid (−)-3 with BH3·Me2S in THF produced alcohol (−)-5 in 96% yield with 99% ee. Oxidation of alcohol (−)-5 with Dess–Martin periodinane in CH2Cl2 afforded aldehyde (+)-6 in 93% yield. Direct treatment of oxazolidinone 4a with BH3·Me2S in THF, followed by Dess–Martin oxidation also afforded aldehyde (+)-6 in 86% yield over two steps. Treatment aldehyde (+)-6 with methoxymethylene yield, prepared by mixing of methoxymethyltriphenylphosphonium chloride with NaHMDS, yielded methyl vinyl ether 7 in 88% yield. About 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 ratio of the trans isomer to cis isomer was observed in 1H-NMR spectra, i.e. a doublet at 6.27 ppm with coupling constant 12.8 Hz implied the trans isomer while a doublet at 5.70 ppm with coupling constant 6.0 Hz implied the cis isomer. Hydrolysis of vinyl ether 7 in 1 N HCl in THF furnished homoaldehyde (−)-8 in 92% yield. To confirm the chiral integrity, homoaldehyde (−)-8 was oxidized by Jones reagent to homoacid (−)-9. Chiral HPLC analysis of homoacid (−)-9 confirmed the chiral integrity arrived intact during the processes mentioned above (>99% ee, Scheme 2).
2 ratio of the trans isomer to cis isomer was observed in 1H-NMR spectra, i.e. a doublet at 6.27 ppm with coupling constant 12.8 Hz implied the trans isomer while a doublet at 5.70 ppm with coupling constant 6.0 Hz implied the cis isomer. Hydrolysis of vinyl ether 7 in 1 N HCl in THF furnished homoaldehyde (−)-8 in 92% yield. To confirm the chiral integrity, homoaldehyde (−)-8 was oxidized by Jones reagent to homoacid (−)-9. Chiral HPLC analysis of homoacid (−)-9 confirmed the chiral integrity arrived intact during the processes mentioned above (>99% ee, Scheme 2).
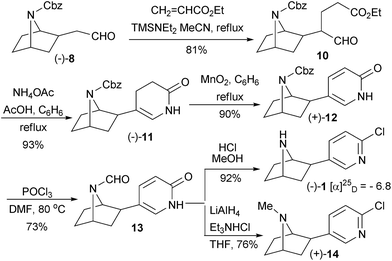
Homoaldehyde (−)-8 was treated with pyrrolidine in the presence of K2CO3 to the corresponding enamine, followed by reaction with ethyl acrylate, and then hydrolysis in an acidic media to furnish glutarate semialdehyde 10 in 55% yield. The yield was improved to 81% yield by Hagiwara's protocol,12 using TMSNEt2 and ethyl acrylate in refluxing acetonitrile. Glutarate semialdehyde 10 underwent a double condensation process with NH4OAc in refluxing benzene to yield dihydropyridone (−)-11 in 93% yield with 99% ee. Oxidation with 9 equivalents of MnO2 in refluxing benzene produced 5-substituted 2-pyridone (+)-12 in 90% yield. Slow addition of MnO2 during a long period was crucial for the yield in that addition at once brought about a yield decrease in this reaction. Treatment of 2-pyridone (+)-12 with POCl3 and in DMF did not only convert the 2-pyridone group to the 2-chloropyridine group, but also replaced the Cbz group by a formyl group as formamide 13 in 73% isolated yield. Since two sets of peaks with almost equal intensity have been observed in 13C-NMR, the product appeared to be a mixture of E/Z isomers with equal amount due to the restrict rotation of the formamide bonding. Exposure of formamide 13 in 5% HCl in MeOH afforded final product epibatidine (−)-1 in 92% yield with >99% ee. The specific rotation value of (−)-1, [α]25D − 6.8 (c: 1.04 CHCl3) was consistent with the reported value, [α]25D − 6.5 (c: 1.00, CHCl3).13 Treatment of formamide 13 with alane complex, prepared by mixing LiAlH4 and Et3N·HCl,14 produced N-methyl epibatidine analogue (+)-14 in 76% yield with 99% ee (Scheme 3).
In conclusion, we have described an efficient preparation of both enantiomers of 7-azabicyclo[2.2.1]heptane carboxylic acid 3, and a feasible strategy to synthesize a 5-substituted 2-chloropyridine structure from a cycloalkanecarboxylic acid, demonstrated as the synthesis of (−)-epibatidine 1. The methodology provides flexible access to various substituted alkaloids bearing a 7-azabicyclo[2.2.1]heptane moiety, which may benefit the development of more potent and safer analgesics. Subsequent extension of this methodology towards other natural products of interest is currently underway.
Acknowledgements
The authors thank the National Science Council, Taiwan (NSC101-2113-M-005-009-MY3) and the Nanotechnological Center of National Chung Hsing University for support of this research.Notes and references
- T. F. Spande, H. M. Garraffo, M. W. Edwards, H. J. C. Yeh, L. Pannell and J. W. Daly, J. Am. Chem. Soc., 1992, 114, 3475 CrossRef CAS
.
- J. W. Daly, H. M. Garraffo, T. F. Spande, M. W. Decker, J. P. Sullivan and M. Williams, Nat. Prod. Rep., 2000, 17, 131 RSC
.
- B. Badio and J. W. Daly, Mol. Pharmacol., 1994, 45, 563 CAS
.
- B. Badio, H. M. Garraffo, C. V. Plummer, W. L. Padgett and J. W. Daly, Eur. J. Pharmacol., 1997, 321, 189 CrossRef CAS
.
- Z. Chen and M. L. Trudell, Chem. Rev., 1996, 96, 1179 CrossRef CAS PubMed
.
- H. F. Olivo and M. S. Hemenway, Org. Prep. Proced. Int., 2002, 34, 1 CrossRef CAS
.
- For the synthesis published before 2002, please see ref. 5 and 6. Published after 2002, please see:
(a) Y. Hoashi, T. Yabuta and Y. Takemoto, Tetrahedron Lett., 2004, 45, 9185 CrossRef CAS PubMed
; (b) V. K. Aggarwal and B. Olofsson, Angew. Chem., Int. Ed., 2005, 44, 5516–5519 CrossRef CAS PubMed
; (c) C.-L. K. Lee and T.-P. Loh, Org. Lett., 2005, 7, 2965 CrossRef CAS PubMed
; (d) Y. Hoashi, T. Yabuta, P. Yuan, H. Miyabe and Y. Takemoto, Tetrahedron, 2006, 62, 365 CrossRef CAS PubMed
; (e) D. R. Boyd, N. D. Sharma, M. Kaik, P. B. A. McIntyre, P. J. Stevenson and C. C. R. Allen, Org. Biomol. Chem., 2012, 10, 2774 RSC
; (f) X. Huang, H. Shi, J. Ren, G. Liu, Y. Tang and B. Zeng, Chin. J. Chem., 2012, 30, 1305 CrossRef CAS
; (g) K. L. Jensen, C. F. Weise, G. Dickmeiss, F. Morana, R. L. Davis and K. A. Jorgensen, Chem.–Eur. J., 2012, 18, 11913 CrossRef CAS PubMed
.
- For the reports published before 2002, please see ref. 5 and 6. Published after 2002, please see:
(a) T. Tachihara, H. Watanabe and T. Kitahara, Heterocycles, 2002, 57, 781 CrossRef CAS
; (b) C. Che, G. Petit, F. Kotzyba-Hibert, S. Bertrand, D. Bertrand, T. Grutter and M. Goeldner, Bioorg. Med. Chem. Lett., 2003, 13, 1001 CrossRef CAS
; (c) A. J. Moreno-Vargas and P. Vogel, Tetrahedron: Asymmetry, 2003, 14, 3173 CrossRef CAS PubMed
; (d) M.-Y. Chang and H.-P. Chen, Heterocycles, 2005, 65, 1705 CrossRef CAS
; (e) A. Armstrong and S. E. Shanahan, Org. Lett., 2005, 7, 1335 CrossRef CAS PubMed
; (f) H. Kimura, T. Fujiwara, T. Katoh, K. Nishide, T. Kajimoto and M. Node, Chem. Pharm. Bull., 2006, 54, 399 CrossRef CAS
; (g) A. Armstrong, Y. Bhonoah and S. E. Shanahan, J. Org. Chem., 2007, 72, 8019 CrossRef CAS PubMed
; (h) E. Gomez-Sanchez, E. Soriano and J. Marco-Contelles, J. Org. Chem., 2007, 72, 8656 CrossRef CAS PubMed
; (i) E. Gomez-Sanchez, E. Soriano and J. Marco-Contelles, J. Org. Chem., 2008, 73, 6784 CrossRef CAS PubMed
; (j) A. Armstrong, Y. Bhonoah and A. J. P. White, J. Org. Chem., 2009, 74, 5041 CrossRef CAS PubMed
; (k) J. Bexrud and M. Lautens, Org. Lett., 2010, 12, 3160 CrossRef CAS PubMed
.
-
(a) D. Bai, R. Xu, G. Chu and X. Zhu, J. Org. Chem., 1996, 61, 4600 CrossRef CAS PubMed
; (b) R. Xu, G. Chu and D. Bai, Tetrahedron Lett., 1996, 37, 1463 CrossRef CAS
; (c) T. E. Fevig, T. E. Anjeh, J. P. Lawson, D. P. Walker and D. G. Wishka, U. S. Patent 0173063 A1, Pfizer Inc., 2006
.
- ESI.†.
- Although there are many reported valuable homologation methods, the present route has been taken for the reliable and safety reason.
- H. Hagiwara, N. Komatsubara, H. Ono, T. Okabe, T. Hoshi, T. Suzuki, M. Ando and M. Kato, J. Chem. Soc., Perkin Trans. 1, 2001, 316 RSC
.
- S. R. Fletcher, R. Baker, M. S. Chambers, R. H. Herbert, S. C. Hobbs, S. R. Thomas, H. M. Verrier, A. P. Watt and R. G. Ball, J. Org. Chem., 1994, 59, 1771 CrossRef CAS
.
- W. Chiou, G.-H. Lin, C.-C. Hsu, S. J. Chaterpaul and I. Ojima, Org. Lett., 2009, 11, 2659 CrossRef CAS PubMed
.
Footnote |
| † Electronic supplementary information (ESI) available: Experimental procedure, all 1H, 13C NMR spectra and assignment for all compounds, and HPLC chromatograms of 1, 3, 5, 9, 11, 12 and 14, and crystallographic data of 4a. CCDC reference number 954104. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c4ra00770k |
| This journal is © The Royal Society of Chemistry 2014 |