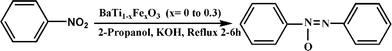A highly efficient iron doped BaTiO3 nanocatalyst for the catalytic reduction of nitrobenzene to azoxybenzene†
Ch. Srilakshmi*a,
H. Vijay Kumara,
K. Praveenab,
C. Shivakumaraa and
M. Muralidhar Nayakc
aSolid State and Structural Chemistry Unit (SSCU), Indian Institute of Science (IISc), Bangalore 560012, India. E-mail: ch.srilakshmi@sscu.iisc.ernet.in; Fax: +91 80 2360 1310
bMaterial Research Centre (MRC), Indian Institute of Science (IISc), Bangalore 560012, India
cInorganic and Physical Chemistry Division (IPC), Indian Institute of Science (IISc), Bangalore 560012, India
First published on 1st April 2014
Abstract
In the present study, we report an efficient and high yielding catalytic reduction of nitrobenzene in the presence of 2-propanol or molecular H2 over Fe doped BaTiO3 nanocatalysts. This method provides 100% conversion of nitrobenzene with 93% and 95% AOB yield with 2-propanol and molecular H2 under the described experimental conditions, thus leading to efficient synthesis of azoxybenzene from nitrobenzene. The optimum Fe content to obtain high azoxybenzene yields was 2.5% and 10% in the presence of 2-propanol and molecular H2 respectively.
Azoxy compounds are versatile organic compounds that have received considerable attention due to their physiological activity and their applications as liquid crystalline materials.1–4 Azoxybenzene has been used in dyes, analytical reagents, reducing agents, stabilizers, food additives, pigments, and polymerization inhibitors.5–8
The synthesis of azoxybenzene is carried out through the oxidation of aromatic amines and reduction of nitro compounds by using various stoichiometric reagents.9–23 Due to environmental concern associated with homogeneous routes using stoichiometric routes it is desired to develop an alternative, efficient, reusable solid phase catalyst for the direct conversion of nitrobenzene to azoxybenzene. To date, very few catalytic routes are reported on reduction of nitrobenzene to azoxybenzene.24,25 However, known catalysts for hydrogenation reactions are less active and selective. Thus, a synthetic method that can differentiate and catalyze a selective reduction of nitrobenzene using a simple, inexpensive catalyst would be valuable and have potential synthetic applications. Recently, Zhu et al.26 reported that gold nanoparticles supported on TiO2 nanocrystals painted H-titanate nanofibers could selectively reduce nitrobenzene to azoxybenzene under visible light irradiation. In the present study we report selective reduction of nitrobenzene in presence of either 2-propanol and molecular H2 over Fe doped BaTiO3 catalysts.
Although Perovskite type mixed oxides have been used previously for a variety of reactions including oxidation of CO27 and NH3,28 and NOx decomposition.29 Their application for catalytic reduction of nitrobenzene is still unexplored. Hence in the present manuscript we describe the development of solid phase, reusable and robust catalyst with high catalytic activity and selectivity for the selective reduction of nitrobenzene to azoxybenzene. The catalysts were thoroughly characterized using electron microscopy, XRD, FT-IR, (see, Fig. S2, ESI†) N2 physisorption methods (see, Table S1, ESI†).
The indexed PXRD patterns of BaTi1−xFexO3 (0.0 ≤ x ≤ 0.3) powders calcined at 850 °C for 4 h (Fig. S1, ESI†) showed BaTiO3 (BTO) phase (JCPDS no. 031-0174) with cubic symmetry and space group, Pm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m. XRD patterns of the Fe doped samples were matched with the XRD of the BTO sample and shows doping at B site. Trace amounts of BaCO3 was observed as impurity in the catalysts. The formation of BaCO3 phase might be due to the reaction between BaO and CO2 produced at the elevated temperature during calcinations.
m. XRD patterns of the Fe doped samples were matched with the XRD of the BTO sample and shows doping at B site. Trace amounts of BaCO3 was observed as impurity in the catalysts. The formation of BaCO3 phase might be due to the reaction between BaO and CO2 produced at the elevated temperature during calcinations.
Transmission electron microscopic images of the catalysts (Fig. 1) illustrates a narrow distribution of particle sizes, in the range of 40–50 nm for BaTiO3 (Fig. 1A) and in the range of 30–40 nm for Fe doped BaTiO3 catalysts (Fig. 1B–D). The particles are spherical in shape and are agglomerated. The particle size estimated from TEM reasonably agree (see, Table S2, ESI†) with those obtained from XRD (30 to 60 nm) using Scherrer equation. There is a decrease in the average particle size for Fe doped BTO catalysts with increase in Fe content in the catalysts, could be due to the particle growth hindrance caused by oxygen vacancies, lattice distortion as well as internal stress arising from the substitution of Ti with Fe as described by Wei et al.30 Fig. 1E shows a typical lattice image of a 50 nm nanocrystalline grain of BTO catalyst (Fig. 1A). Clear lattice fringes with d110 = 0.28 nm reveal that well-crystallized BTO nanoparticles are formed. The surrounding edges of the particle are very smooth and no surface steps were observed. Fig. 1F shows a surface profile HRTEM image of a 50 nm BTO grain (Fig. 1A). It is noticed that the surface facets are parallel to the (100) and (110) planes. Small islands of 3–4 atom layer thickness were frequently observed around the edge of the particle. The contrast variations across Fig. 1F are due to the thickness variations associated with the fine-scale surface facets and surface roughness.
 | ||
| Fig. 1 TEM images of (a) BTO (b) BTO-Fe-2.5 (c) BTO-Fe-5 (d) BTO-Fe-30 (e) lattice image of a 50 nm nanocrystalline grain of BTO (f) surface profile HRTEM image of 50 nm BTO grain. | ||
X-ray photoelectron spectra (XPS) spectra of the Fe 2p region (Fig. S3, ESI†) showed very less intense peaks for BTO-Fe-2.5 and BTO-Fe-5 indicating the high dispersion of Fe particles. BTO-Fe-10 catalyst showed Fe 2p3/2 broad peak at 713.9 eV and Fe 2p1/2 at 726.5 eV which was due to Fe3+. With further increase in the Fe content all the catalysts showed broad peaks for Fe 2p3/2 with maxima between 709.25 and 710.52 indicating the presence of both Fe2+/Fe3+in the catalysts.
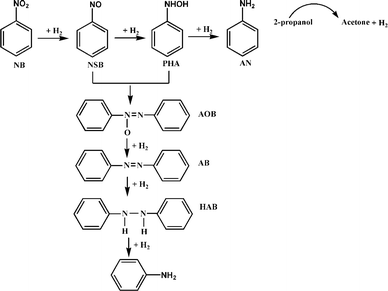
The detailed investigation on the optimization of the reaction conditions and discussion is provided in ESI (see, pages S6–S8†). The catalytic reduction performed on pure BTO catalyst in presence of 2-propanol (Table 1, entry 1) showed 99% conversion of nitrobenzene (NB) and 56.4% AOB yield. Whereas, the reaction conducted on the Fe doped catalysts showed 100% NB conversion on the catalysts with Fe content from 2.5 to 15% (BTO-Fe-2.5 to BTO-Fe-15). The decrease in conversion on BTO-Fe-20 and BTO-Fe-30 catalyst is due to presence of iron in its oxidized state as the reaction is conducted in the absence of H2. This is clearly understood as the reaction results obtained in H2 atmosphere showed 100% conversion on all the catalysts (see, Table 2, entries 2–7). The maximum yield of 93% AOB was obtained on BTO-Fe-2.5 catalyst and was higher than that of other catalysts indicating 2.5% Fe is the optimal amount to obtain maximum AOB yield. The reduction in AOB yield with increase in Fe content in the catalysts is due to formation of other reduction products like azobenzene and aniline via side reactions (see Scheme 1).
| Ent. | Catalyst | Yield (%) | Conversion (%) | Selectivity (%) AOB | ||
|---|---|---|---|---|---|---|
| AOB | AB | AN | ||||
| a Reaction conditions: nitrobenzene (20 mmol), KOH pellets (20 mmol), catalyst (150 mg) and 2-propanol (20 ml), refluxed for 2–6 h at 80 °C; AOB – azoxybenzene, AB – azobenzene, AN – aniline, conversion %= ((Ci − Cf)/Ci) × 100; (Ci & Cf) – initial and final amount of nitrobenzene (mol%), yield% = (%conversion of nitrobenzene × %selectivity of product)/100. | ||||||
| 1 | BTO | 56.4 | 21.8 | 20.8 | 99 | 57 |
| 2 | BTO-Fe-2.5 | 93 | 0 | 7 | 100 | 93 |
| 3 | BTO-Fe-5 | 82 | 0 | 18 | 100 | 82 |
| 4 | BTO-Fe-10 | 81 | 0 | 19 | 100 | 81 |
| 5 | BTO-Fe-15 | 80 | 0 | 20 | 100 | 80 |
| 6 | BTO-Fe-20 | 56 | 0.92 | 35 | 92 | 61 |
| 7 | BTO-Fe-30 | 59 | 1 | 28 | 88 | 67 |
| 8 | BaFeO3 | 38.8 | 8.61 | 31.7 | 79 | 49 |
| 9 | BaO–TiO2–Fe2O3 | 36.5 | 9.12 | 35.8 | 82 | 44.5 |
| Ent. | Catalyst | Yield (%) | Conversion (%) | Selectivity (%) AOB | ||
|---|---|---|---|---|---|---|
| AOB | AB | AN | ||||
| a Reaction conditions: nitrobenzene (20 mmol), KOH pellets (20 mmol), catalyst (150 mg) and 2-propanol (20 ml), refluxed for 2–6 h at 80 °C, %selectivity = (mole% of desired product/mole% of all products) × 100. | ||||||
| 1 | BTO | 64 | 0 | 26 | 90 | 71 |
| 2 | BTO-Fe-2.5 | 92 | 4 | 4 | 100 | 92 |
| 3 | BTO-Fe-5 | 93 | 0 | 7 | 100 | 93 |
| 4 | BTO-Fe-10 | 95 | 0 | 5 | 100 | 95 |
| 5 | BTO-Fe-15 | 86 | 0 | 14 | 100 | 86 |
| 6 | BTO-Fe-20 | 85 | 0 | 15 | 100 | 85 |
| 7 | BTO-Fe-30 | 87 | 0 | 13 | 100 | 87 |
| 8 | Fe(NO3)3·9H2O | 58 | 6 | 24 | 88 | 66 |
For comparison purpose, catalytic reduction of nitrobenzene was also studied on the respective mixed oxide and perovskite such as BaO–TiO2–Fe2O3 and BaFeO3 catalysts in the presence of 2-propanol. The conversion and yields obtained on these catalysts (see entries 8 and 9 in Table 1) are low compared to all the catalysts in the present study. This result clearly demonstrates the effect of Fe doping in the BaTiO3 matrix in the catalytic reduction of nitrobenzene.
Molecular H2 has been employed as the hydrogen source, and the catalytic reduction was performed at atmospheric pressure, (see Table 2). The conversion as well as yield indeed is improved in the presence of H2 atmosphere. 100% conversion was achieved on all the catalysts (see Table 2, entries 2–7). A maximum AOB yield of 95% was obtained on BTO-Fe-10 catalyst (see entry 4 in Table 2). The better results obtained in the presence of H2 atmosphere could be due to active metal i.e. iron remain in reduced state under reaction conditions.
For the sake of comparison the catalytic reduction also conducted with homogeneous Fe(NO3)3 (150 mg) in the presence of H2 and the results are presented in Table 2 (entry 8). The conversion of nitrobenzene and selectivity of AOB were found to be lesser compared to Fe supported on BaTiO3. This result clearly indicates the influence of BaTiO3 support for Fe for the conversion of nitrobenzene. Optimum content of Fe found to be 2.5 wt% on BaTiO3 for the conversion of nitrobenzene with 2-propanol. The optimum contents of Fe is found to be 10 wt% on BaTiO3 for the conversion of nitrobenzene using molecular hydrogen as reducing agent.
From the above results it can be understood that Fe doped catalysts exhibits similar or higher NB conversion, in particular, significantly higher yields towards AOB was achieved than on pure BTO catalyst.
The possible reaction pathways are outlined in Scheme 1. The two reaction routes possible in the catalytic reduction of nitrobenzene are direct route and condensation route. Direct route involves the formation of aniline as the major product. And the condensation route involves a condensation reaction between formed intermediates of nitrosobenzene (NSB) and phenyl hydroxylamine (PHA) leads to the formation of azoxybenzene. The formation of dimeric products of AOB and AB are mainly favoured by a basic reaction environment. Azoxybenzene is easily converted into azobenzene and hydrazobenzene (HAB) which can be subsequently hydrogenolysed to AN at atmospheric pressure. From the above results it can inferred that the Fe on BaTiO3 is promoting the reductive coupling between NSB and PHA to selectively form the AOB. Whereas, the pure BaTiO3, BaO–TiO2–Fe2O3, BaFeO3 and Fe(NO3)3·9H2O are less selective towards AOB formation.
Conclusions
In conclusion, we have developed a highly efficient catalytic system consisting of Fe doped BaTiO3, for the catalytic reduction of nitrobenzene to azoxybenzene using either 2-propanol or molecular H2 as hydrogen source and KOH as promoter. This is the first demonstration of the selective catalytic reduction of nitrobenzene to azoxybenzene over solid phase perovskite catalysts using both 2-propanol and molecular H2 as hydrogen source. This method provides 100% conversion with 93% AOB yield with 2-propanol and 100% conversion with 95% AOB yield with molecular H2 thus leading to efficient synthesis of azoxybenzene from nitrobenzene. These results indicate that these materials provide great promise towards the important goal of developing improved catalysts for selective catalytic reduction.Acknowledgements
CSL acknowledge the financial support from Department of Science and Technology (DST) through INSPIRE faculty program and KP acknowledges Union Grants Commission (UGC) for awarding Kothari fellowship.Notes and references
- T. Ikeda and O. Tsutumi, Science, 1995, 268, 1873 CAS
.
- J. P. Snyder, V. T. Bandurco, F. Darack and H. Olsen, J. Am. Chem. Soc., 1974, 96, 5158 CrossRef CAS
.
- D. Campbell, L. R. Dix and P. Rostron, Dyes Pigm., 1995, 29, 77 CrossRef CAS
.
- T. L. Gilchrist, in Comprehensive Organic Synthesis, ed. B. M. Trost and I. Fleming, Pergamon, Oxford, 1991, vol. 7, p. 735 Search PubMed
; D. H. Rosenblatt and E. P. Burrows, in The Chemistry of Amino, Nitroso and Nitro Compounds and Their Derivatives, ed. S. Patai, Wiley, Chichester, 1982, p. 1085 Search PubMed
.
- R. Egli, in Colour Chemistry: The Design and Synthesis of Organic Dyes and Pigments, ed. A. P. Peter and H. S. Freeman, Elsevier, London, 1991, ch. VII Search PubMed
.
- S. C. Catino and E. Farris, Concise Encyclopedia of Chemical Technology, Wiley, New York, 1985 Search PubMed
.
- K. Venkataraman, in The Chemistry of Synthetic Dyes, Academic Press, London, 1970, ch. VI Search PubMed
.
- E. Buncel, Acc. Chem. Res., 1975, 6, 132 CrossRef
.
- O. D. Wheeler and D. Gonzales, Tetrahedron, 1964, 20, 189 CrossRef CAS
.
- H. E. Baumgarten, A. Staklis and E. M. Miller, J. Org. Chem., 1965, 30, 1203 CrossRef CAS
.
-
(a) R. W. Murray, K. Iyanar, J. Chen and J. T. Wearing, Tetrahedron Lett., 1996, 37, 805 CrossRef CAS
; (b) G. Barak and Y. Sasson, J. Org. Chem., 1989, 54, 3484 CrossRef CAS
.
- R. W. Murray, R. Jeyaraman and L. Mohan, Tetrahedron Lett., 1986, 27, 2335 CrossRef CAS
.
- A. McKillop and J. A. Tarbin, Tetrahedron, 1987, 43, 1753 CrossRef CAS
.
- R. A. Sheldon and J. Dakka, Catal. Today, 1994, 19, 215 CrossRef CAS
.
- S. Tollari, M. Cuscela and F. Porta, J. Chem. Soc., Chem. Commun., 1993, 1510 RSC
.
- S. Tollari, D. Vergani, S. Banfi and F. Porta, J. Chem. Soc., Chem. Commun., 1993, 442 RSC
.
- S. Satoru, M. Miura and M. Nomura, J. Org. Chem., 1989, 54, 4700 CrossRef CAS
; H. Huang, D. Sommerfeld, B. C. Dunn, C. R. Lloyd and E. M. Eyring, J. Chem. Soc., Dalton Trans., 2001, 1301 RSC
.
- R. O. Hutchins, D. W. Lamson, L. Rua, C. Milewski and B. Maryanoff, J. Org. Chem., 1971, 36, 803 CrossRef CAS
.
- H. J. Shine and H. E. Mallory, J. Org. Chem., 1962, 27, 2390 CrossRef CAS
.
- S. A. Buckler, L. Doll, F. K. Lind and M. Epstein, J. Org. Chem., 1962, 27, 794 CrossRef CAS
.
- N. S. Kozlov and V. A. Soshin, Uch. Zap. Permsk. Gos. Pedagog. Inst., 1965, 32, 84 Search PubMed
; N. S. Kozlov and V. A. Soshin, Chem. Abstr, 1967, 66, 2290k Search PubMed
.
- E. F. Degering and E. F. Hitch, J. Am. Chem. Soc., 1951, 73, 1323 CrossRef
.
- H. E. Bigelow and A. Palmer, Org. Synth., 1943, 2, 57 Search PubMed
.
- D. K. Dutta, Synth. Commun., 2006, 36, 1903 CrossRef CAS
.
- B. Pal, T. Torimoto, K. Okazaki and B. Ohtani, Chem. Commun., 2007, 483 RSC
.
- Z. Zheng, J. Zhao, H. Liu, J. Liu, A. Bo and H. Zhu, ChemCatChem, 2013, 5, 2382 CrossRef CAS
.
- G. Kremenic, J. M. L. Nieto, J. M. D. Tascon and L. G. Tejuca, J. Chem. Soc., Faraday Trans. 1, 1985, 81, 939 RSC
.
- Y. Wu, T. Yu, B. Dou, C. Wang, X. Xie, Z. Yu, S. Fan, Z. Fan and L. Wang, J. Catal., 1989, 88, 120 Search PubMed
.
- L. G. Tejuca, J. L. G. Fierro and J. M. D. Tascon, Adv. Catal., 1989, 36, 237 CAS
.
- X. Wei, G. XU, Z. Ren, Y. Wang, G. Shen and G. Han, Mater. Lett., 2008, 62, 3666 CrossRef CAS PubMed
.
Footnote |
| † Electronic supplementary information (ESI) available: XRD, FTIR, N2 physisorption, XPS, optimization of reaction conditions results. See DOI: 10.1039/c4ra00595c |
| This journal is © The Royal Society of Chemistry 2014 |