Redox-active gold nanoclusters immobilized ZnO nanorod electrodes for electrochemical sensing applications†
Abstract
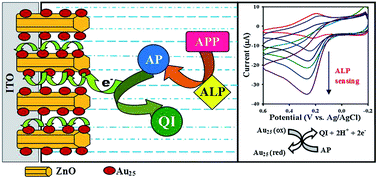
Well-defined composite electrodes were fabricated by combining redox-active Au25 nanoclusters with highly oriented ZnO nanorods. The structure and electrocatalytic activity of the composite electrodes were examined for the development of an amperometric sensor for alkaline phosphatase.

 Please wait while we load your content...
Please wait while we load your content...