Phase transformation (cubic to rhombohedral): the effect on the NO2 sensing performance of Zn-doped flower-like In2O3 structures†
Pei Li,
Huiqing Fan*,
Yu Cai,
Mengmeng Xu,
Changbai Long,
Mengmeng Li,
Shenhui Lei and
Xinwei Zou
State Key Laboratory of Solidification Processing, School of Materials Science and Engineering, Northwestern Polytechnical University, Xi'an, China. E-mail: lipei8143706@163.com; Tel: +86-29-88494463
First published on 7th February 2014
Abstract
Cubic In2O3 (bcc-In2O3) was transformed into a mixture of bcc-In2O3 and rhombohedral In2O3 (rh-In2O3) by Zn doping. The Zn-doped flower-like In2O3 structures consisted of many thin sheets with a length of 0.4–1 μm, and cubes with a length of 200 nm, while the size of the microflowers was 1–3.5 μm. The Zn doping concentration significantly affected the phase transformation and the overall morphology of In2O3. Furthermore, the analysis of N2 adsorption–desorption measurements showed that the Zn-doped flower-like In2O3 structures (sample S5) adsorbed the largest amount of N2 and had the biggest surface area (46.41 m2 g−1), which contributed to an improvement in gas sensing performance. Finally, sensors based on the mixture of bcc- and rh-In2O3 structures exhibited a much higher response to NO2 than the pure bcc-In2O3 (sample S1), and the Zn-doped flower-like In2O3 structures (sample S5) exhibited the highest response of 27.4 ± 2.5 for 5 ppm NO2. Thus, the gas sensing performance of In2O3 was enhanced significantly by the phase transformation.
Introduction
In2O3 is a polymorph, consisting of the stable cubic phase (body-centered cubic, bcc-In2O3) and the metastable rhombohedral phase (denoted as rh-In2O3).1–3 Of the two phases, rh-In2O3 is regarded as the high-pressure polymorphic counterpart of cubic In2O3, which is a metastable phase at atmospheric pressure.4,5 As an important semiconductor with a wide band gap (2.93 ± 0.15 eV for bcc-In2O3 and 3.02 ± 0.15 eV for rh-In2O3),6 In2O3 has been applied in solar cells,7 UV lasers,8 detectors,9 sensors,10 etc. In recent years, the gas-sensing properties of In2O3 materials for trimethylamine (N(CH3)3),11 NH3,12 C2H5OH,13 HCHO,14 acetone,15 H2,16 CO,17 O3,18 H2S,19 Cl2,20 and NO221 gases have been investigated.Rhombohedral In2O3 has previously been obtained under extreme conditions, such as high pressure and high temperature,5,22 and rhombohedral In2O3 exhibits more stable conductivity than its cubic counterpart.23 The rh-In2O3 phase can be transformed into the bcc-In2O3 phase under certain physical and chemical conditions, if the change in the crystal structure can reduce the free energy of the system, which may affect the morphology and the gas sensing properties of In2O3. A great number of morphologies and various synthesis methods for bcc-In2O3 have been developed,24–30 but much less research has been done into the preparation of rh-In2O3.10,22,31 Moreover, studies of the phase-controlled synthesis of bcc-In2O3 and rh-In2O3 in one system are even rarer. Doping can not only improve the gas sensing performance of In2O3 materials, but also affects the crystallization of In2O3. Also, it may cause the phase transformation of In2O3. This transformation is an extremely complex issue. It is well known that many factors, such as reagents, pressure, temperature, chemical reaction time and the rate of temperature increase, have large effects on the final In2O3 products. Considering these factors, the experimental conditions would seriously affect the growth of the crystal and the phase transformation. Although the doping of Zn in In2O3 has been investigated in some previous studies,32,33 reports on the phase transformation of In2O3 are rare.
Herein, we report a facile hydrothermal route for the phase transformation of In2O3 structures (the pure bcc-In2O3 was transformed into a mixture of bcc- and rh-In2O3) with Zn doping. The morphologies of the Zn-doped flower-like In2O3 structures consisted of microflowers with a size of 1–3.5 μm, and cubes with a length of 200 nm. The obtained materials were analysed by X-ray diffraction (XRD), field emission scanning electron microscopy (FESEM), high resolution transmission electron microscopy (HRTEM), inductively coupled plasma mass spectrometry (ICP-MS), photoluminescence (PL) spectra, thermogravimetric (TG) analysis, and N2 adsorption–desorption. The gas sensing properties of the materials were also investigated. The introduction of a small quantity of Zn to the reaction system was found to play an important role in the phase transformation of In2O3, which consequently affected the gas sensing properties of NO2.
Experimental
Materials and synthesis
The chemical reagents indium nitrate hydrate (In(NO3)3·4.5H2O), zinc nitrate hydrate (Zn(NO3)2·6H2O), glycerol and ethylene glycol were obtained from Sinopharm Chemical Reagent Co., Ltd, Shanghai, China, and were of analytical grade and used without further purification. 1.5 mmol of In(NO3)3·4.5H2O, 5.0 mL glycerol, 10.0 mL ethylene glycol and different amounts of Zn(NO3)2·6H2O were added to distilled water under constant strong stirring. After stirring for 30 min, the solutions were transferred into 40 mL Teflon-lined stainless steel autoclaves. The autoclaves were heated at 140 °C for 12 h, and then cooled down to room temperature naturally. The white precipitates were separated by centrifugation, washed with distilled water and anhydrous ethanol several times, and dried at 80 °C for 4 h. Finally, Zn-doped flower-like In2O3 structures were obtained after thermal treatment at 500 °C for 2 h. We refer to these samples as S1, S2, S3, S4, S5 and S6, representing pure In2O3, and the samples with In![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Zn molar ratios of 15
Zn molar ratios of 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 12
1, 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 9
1, 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 7
1, 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, and 5
1, and 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, respectively.
1, respectively.
Characterization
The phase structure and purity of the as-synthesized products were examined using XRD (X'pert PRO MPD, Philips, Eindhoven, The Netherlands) with Cu Kα radiation (λ = 1.5406 Å) at 40 kV and 30 mA over the 2θ range 10–70°. The morphology of the obtained samples was investigated using FESEM (JSM-6701F, JEOL, Tokyo, Japan), and HRTEM (Tecnai F30G2, FEI, Hillsboro, OR, USA) operated at 300 kV accelerating voltage. Energy-dispersive X-ray (EDX) spectroscopy was also performed by using a microanalysis detector (X-MaxN 80 T, Oxford Instruments, Oxfordshire, UK). An ICP-MS spectrometer (7500A, Agilent, Santa Clara, CA, USA) was used for simultaneous multi-element detection of Zn. The analytical parameters of the ICP-MS instrument were a Babington nebulizer, a double pass quartz spray chamber, a 10 MHz frequency RF generator, 1220 W output power, 20 L min−1 Ar flow rate, 0.9 L min−1 auxiliary gas flow rate, 1–1.2 L min−1 nebuliser gas flow rate, 400 μL min−1 sample uptake rate, 0.1 s integration time, and 206.200 nm wavelength. Brunauer–Emmett–Teller (BET) surface area characterization of the In2O3 structures was obtained by full analysis of N2 adsorption–desorption tests (V-Sorb 2800P, Gold APP, Beijing, China). PL measurements were carried out at room temperature on a cold copper finger in a closed-cycle cryostat with grease at a 264 nm excitation wavelength (F-4600, Hitachi, Tokyo, Japan). TG (Q600-SDT, TA Instruments, New Castle, PA, USA) measurements were performed at a heating rate of 10 °C min−1 under a flow of nitrogen gas. AC impedance spectroscopy was measured with a precision impedance analyzer (4294A, Agilent, Santa Clara, CA, USA).Gas sensor fabrication and measurements
The basic fabrication process was as follows:34 the as-obtained In2O3 products were slightly ground with α-terpineol in an agate mortar to form a gas sensing paste. The paste was coated on an alumina tube with Au electrodes and platinum wires, dried under IR light for several minutes in air, and then sintered at 500 °C for 2 h. A Ni–Cr alloy crossing alumina tube was used as a heating resistor, which ensured both substrate heating and temperature control. In order to improve their stability, the gas sensors were aged at 300 °C for 240 h in air.The gas sensing properties were tested using a gas response instrument (HW-30A, Hanwei Ltd, Zhengzhou, China). The gas sensing properties of the In2O3 structures were tested in a glass test chamber, and the volume of the test chamber was 15 L. In the measuring electric circuit of the gas sensor, a load resistor was connected in series with a gas sensor. The circuit voltage Vc was 5 V, and the output voltage Vout was the terminal voltage of the load resistor RL. The working temperature of the sensor was adjusted by varying the heating voltage Vh. When a given amount of tested gas was injected into the chamber, the resistance of the sensor changed. As a result, the output voltage changed. The gas response S is defined as follows: S = Rg/Ra, where Rg and Ra are the resistance values measured in an oxidizing atmosphere and air, respectively. For each sample, three sensors were made by the same fabrication process, and each sensor was tested three times in the gas sensing testing process. The gas response values given in the text are the average values.
Results and discussion
Crystalline structure and morphology
Fig. 1a–f show the SEM images of the In precursor before calcination. According to Fig. 1a, the pure precursor consisted of cubes with a length of 100–500 nm. With the introduction of Zn, the morphology of the precursor changed to a mixture of cubes and microflowers, and the size of precursor was increased. An increase in Zn doping concentration led to a decrease in the number of cubes and an increase in the number of microflowers (Fig. 1b–f). When the In![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Zn molar ratio was 7
Zn molar ratio was 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (S5, Fig. 1e), lots of thin sheets were organized to form regular flower-like structures, consisting of the maximum number of layers with a size of 1–3.5 μm. Also, fewer cubes with a length of 200 nm were observed in the sample. Upon further increasing the Zn doping concentration (S6), the size of the microflowers continued to increase to 2–5 μm, while a decrease in the number of layers in the microflowers was observed. Fig. 1g demonstrates the XRD patterns of the crystalline phase of precursor before calcination. All the diffraction peaks marked by a star can be indexed to the cubic In(OH)3 structure, with space group Ia
1 (S5, Fig. 1e), lots of thin sheets were organized to form regular flower-like structures, consisting of the maximum number of layers with a size of 1–3.5 μm. Also, fewer cubes with a length of 200 nm were observed in the sample. Upon further increasing the Zn doping concentration (S6), the size of the microflowers continued to increase to 2–5 μm, while a decrease in the number of layers in the microflowers was observed. Fig. 1g demonstrates the XRD patterns of the crystalline phase of precursor before calcination. All the diffraction peaks marked by a star can be indexed to the cubic In(OH)3 structure, with space group Ia![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) (204) (no. 85-1338), and all the diffraction peaks marked by a circle can be indexed to the orthorhombic InOOH structure with space group Pnnm (58) (no. 73-1592). Obviously, the cubic In(OH)3 (bcc-In(OH)3) was transformed into a mixture of bcc-In(OH)3 and orthorhombic InOOH (o-InOOH) due to the Zn doping, and an increased Zn concentration gave rise to an enhancement in the o-InOOH diffraction peak intensity and a suppression in the intensity of the bcc-In(OH)3 diffraction peaks.
(204) (no. 85-1338), and all the diffraction peaks marked by a circle can be indexed to the orthorhombic InOOH structure with space group Pnnm (58) (no. 73-1592). Obviously, the cubic In(OH)3 (bcc-In(OH)3) was transformed into a mixture of bcc-In(OH)3 and orthorhombic InOOH (o-InOOH) due to the Zn doping, and an increased Zn concentration gave rise to an enhancement in the o-InOOH diffraction peak intensity and a suppression in the intensity of the bcc-In(OH)3 diffraction peaks.
 | ||
| Fig. 1 SEM images of the (a) S1, (b) S2, (c) S3, (d) S4, (e) S5 and (f) S6 precursors before calcination, and (g) their XRD patterns. | ||
The thermal decomposition behavior of the precursor was examined by thermogravimetric (TG) analysis, as shown in Fig. 2. A total weight loss of 47.98 wt% and two weight loss steps were observed in the temperature range from 50 to 600 °C. The first weight loss step of ∼6.02 wt% from 50 to 200 °C is attributed to the removal of adsorbed water and ethylene glycol (boiling point: 197.8 °C) in the bcc-In(OH)3 and o-InOOH products. As the temperature continued to increase, a significant weight loss of ∼41.96 wt% was observed in the range of 200–600 °C, which is assigned to the decomposition of bcc-In(OH)3 and o-InOOH and the removal of glycerol (boiling point: 290 °C).
Fig. 3a shows the XRD patterns of the as-obtained hydrothermal products after calcination at 500 °C for 2 h. All the diffraction peaks marked by a star can be indexed to a cubic lattice (space group Ia![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) (206)) of In2O3 according to the Joint Committee on Powder Diffraction Standards (JCPDS) data card no. 06-0416. All the diffraction peaks marked by a circle can be readily indexed to a rhombohedral lattice (space group R
(206)) of In2O3 according to the Joint Committee on Powder Diffraction Standards (JCPDS) data card no. 06-0416. All the diffraction peaks marked by a circle can be readily indexed to a rhombohedral lattice (space group R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) c (167)) of In2O3 according to the JCPDS data card no. 22-0336. It is interesting that the pure bcc-In2O3 was transformed into a mixture of bcc-In2O3 and rh-In2O3 when it was doped with Zn. With increasing Zn concentration, the diffraction peaks for rh-In2O3 increase, while the peaks for bcc-In2O3 decrease. The cell parameters for bcc-In2O3 and rh-In2O3 and the grain sizes of the crystallites determined by the Scherrer formula are listed in Table 1. As can be seen, the calculated lattice constants compare well with the literature values of a = b = c = 10.118 Å (bcc-In2O3, JCPDS 06-0416), and a = b = 5.487 Å, c = 14.510 Å (rh-In2O3, JCPDS 22-0336).
c (167)) of In2O3 according to the JCPDS data card no. 22-0336. It is interesting that the pure bcc-In2O3 was transformed into a mixture of bcc-In2O3 and rh-In2O3 when it was doped with Zn. With increasing Zn concentration, the diffraction peaks for rh-In2O3 increase, while the peaks for bcc-In2O3 decrease. The cell parameters for bcc-In2O3 and rh-In2O3 and the grain sizes of the crystallites determined by the Scherrer formula are listed in Table 1. As can be seen, the calculated lattice constants compare well with the literature values of a = b = c = 10.118 Å (bcc-In2O3, JCPDS 06-0416), and a = b = 5.487 Å, c = 14.510 Å (rh-In2O3, JCPDS 22-0336).
| Sample | bcc-In2O3 cell parameters | rh-In2O3 cell parameters | D (nm) | Theoretical In![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Zn molar ratio Zn molar ratio |
Actual Zn content (wt%) | |
|---|---|---|---|---|---|---|
| a = b = c (Å) | a = b (Å) | c (Å) | ||||
| S1 | 10.1181 | — | — | 14.2 | — | — |
| S2 | 10.1182 | 5.4084 | 14.3369 | 16.4 | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
0.67 |
| S3 | 10.1193 | 5.4568 | 14.4113 | 15.9 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
0.92 |
| S4 | 10.1184 | 5.4232 | 14.3478 | 14.7 | 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
1.21 |
| S5 | 10.1176 | 5.4302 | 14.3754 | 14.1 | 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
1.69 |
| S6 | 10.1187 | 5.4061 | 14.3419 | 17.2 | 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
2.13 |
According to the three Gaussian profile fitting of the curves,35 the intensities were determined for the bcc-In2O3 (400), bcc-In2O3 (440), and rh-In2O3 (012), rh-In2O3 (110) peaks. The relative phase concentration of the bcc-In2O3 phase with respect to the rh-In2O3 phase was estimated from the profile fitting. The fractions of bcc-In2O3 (bcc) and rh-In2O3 (rh) phases were determined using the relations:
| rh = (1 − bcc) |
As expected, the bcc-In2O3 phase fraction decreased with the decrease in In![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Zn molar ratio, as shown in Fig. 3b. Only the bcc-In2O3 phase was present in the pure In2O3 structure, as shown in sample S1 with the In
Zn molar ratio, as shown in Fig. 3b. Only the bcc-In2O3 phase was present in the pure In2O3 structure, as shown in sample S1 with the In![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Zn molar ratio of 1
Zn molar ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0, while the coexistence of bcc- and rh-In2O3 phases appeared when the materials was doped with Zn. When the In
0, while the coexistence of bcc- and rh-In2O3 phases appeared when the materials was doped with Zn. When the In![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Zn molar ratio was 15
Zn molar ratio was 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, the fraction of bcc-In2O3 was 53.1%; almost half of the bcc-In2O3 was transformed into the rh-In2O3 phase, indicating the presence of a mixture of bcc- and rh-In2O3. When the In
1, the fraction of bcc-In2O3 was 53.1%; almost half of the bcc-In2O3 was transformed into the rh-In2O3 phase, indicating the presence of a mixture of bcc- and rh-In2O3. When the In![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Zn molar ratio was 5
Zn molar ratio was 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, the bcc-In2O3 fraction was only 29.9%, while the fraction of rh-In2O3 was 70.1%. The degree of bcc-In2O3 remained unchanged throughout the two-phase region, only the relative amounts of the bcc-In2O3 and rh-In2O3 phases changed.
1, the bcc-In2O3 fraction was only 29.9%, while the fraction of rh-In2O3 was 70.1%. The degree of bcc-In2O3 remained unchanged throughout the two-phase region, only the relative amounts of the bcc-In2O3 and rh-In2O3 phases changed.
The concentrations of Zn element in the Zn-doped In2O3 structures were determined using ICP-MS, and the results are shown in Table 1. The results indicated that the Zn content (wt%) was very low for all the samples doped with different amounts of Zn, and the Zn content increased with the decrease in In![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Zn molar ratio.
Zn molar ratio.
Fig. 4a–f show the SEM images of the pure bcc-In2O3 and the mixture of bcc- and rh-In2O3 structures after calcination at 500 °C for 2 h. They indicate that Zn doping plays an important role in controlling the phase transformation and the morphology of In2O3. As shown in Fig. 4a, pure bcc-In2O3 consists of cubes with a size of 100–500 nm. Upon the introduction of Zn, the morphology of In2O3 changed into a mixture of cubes and microflowers, and the size of the In2O3 particles increased. With the increase of the amount of Zn doping, the number of cubes decreased while the number of microflowers increased, as shown in Fig. 4b–f. As depicted in Fig. 4b, very few microflowers were present in S2, with a size of 1–2.5 μm. Upon further increasing the Zn doping concentration (S3–S6), the amount of cubes decreased sharply, while most of the In2O3 sample consisted of microflowers. When the In![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Zn molar ratio was 7
Zn molar ratio was 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (S5), as shown in Fig. 4e, a mass of thin sheets was organized into the most regular three-dimensional flower-like structures, with the largest number of layers of all the samples. The length of thin sheets was 0.4–1 μm, the width was 5–10 nm, and the size of the microflowers was 1–3.5 μm. Meanwhile, there existed very few 200 nm length cubes. On further increasing the Zn doping amount (S6), 70.1% of the bcc-In2O3 was transformed into rh-In2O3. As can been seen from Fig. 4f, this sample contained the smallest number of cubes, the size of the microflowers increased further to 2–5 μm, and the number of layers in the microflowers was decreased, which may destroy the morphology and affect the NO2 sensing performance of In2O3.
1 (S5), as shown in Fig. 4e, a mass of thin sheets was organized into the most regular three-dimensional flower-like structures, with the largest number of layers of all the samples. The length of thin sheets was 0.4–1 μm, the width was 5–10 nm, and the size of the microflowers was 1–3.5 μm. Meanwhile, there existed very few 200 nm length cubes. On further increasing the Zn doping amount (S6), 70.1% of the bcc-In2O3 was transformed into rh-In2O3. As can been seen from Fig. 4f, this sample contained the smallest number of cubes, the size of the microflowers increased further to 2–5 μm, and the number of layers in the microflowers was decreased, which may destroy the morphology and affect the NO2 sensing performance of In2O3.
 | ||
| Fig. 4 SEM images of the structures after calcination at 500 °C for 2 h: (a) S1, (b) S2, (c) S3, (d) S4, (e) S5 and (f) S6 (inset: high-magnification SEM images of S1–S6). | ||
To give further insight into the morphology and structure of Zn-doped flower-like In2O3, TEM and HRTEM images and EDX spectroscopy, along with the SAED pattern, were obtained. Fig. 5a shows the TEM image of the Zn-doped flower-like In2O3 structure of S5. It shows that the size of the microflower is 2 μm, which is consistent with the value estimated from the SEM image (Fig. 4e). The corresponding HRTEM image (Fig. 5b) exhibits well-defined lattice fringes, and two kinds of lattice spacing can be observed. The spacing of 0.506 nm corresponds to the (200) planes of bcc-In2O3, while the lattice spacing of 0.275 nm corresponds to the inter-planar distance of the (110) planes of rh-In2O3.
 | ||
| Fig. 5 (a) TEM and (b) HRTEM images and (c) the EDX spectrum of the Zn-doped flower-like In2O3 (S5); SAED patterns taken from the corresponding areas marked (d) A1 and (e) A2. | ||
The average grain sizes of all samples were calculated by the Scherrer formula, giving sizes of 14.2, 16.4, 15.9, 14.7, 14.1, 17.2 nm for S1, S2, S3, S4, S5 and S6, respectively (Table 1). Apparently, the evolution of the grain sizes of the S1–S6 samples doesn't follow a regular trend, and the values fluctuate within the narrow range of 14.1–17.2 nm. Comparing those values with the sizes estimated from the SEM and TEM images, the pure In2O3 and the Zn-doped In2O3 structures are consistent with the small nanoparticles calculated from XRD data, by means of a mechanism of “oriented attachment”.36 Therefore, the small change in grain size has little effect on the increment of particle size.
The EDX spectroscopy (Fig. 5c) shows that the Zn-doped flower-like In2O3 samples are elementally composed of In, Zn and O. The atomic ratio for In, Zn and O in S5 calculated from the EDX analysis was In![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Zn
Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) O = 61.92
O = 61.92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.88
1.88![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 36.21 (atomic ratio). Fig. 5d and e show the SAED patterns taken from the corresponding marked areas of A1 and A2, respectively. Fig. 5d indicates that the Zn-doped flower-like In2O3 grows along the [200] direction for bcc-In2O3, while Fig. 5e demonstrates that the crystals grew along the [110] direction for rh-In2O3, which is consistent with the values estimated from the HRTEM image (Fig. 5b).
36.21 (atomic ratio). Fig. 5d and e show the SAED patterns taken from the corresponding marked areas of A1 and A2, respectively. Fig. 5d indicates that the Zn-doped flower-like In2O3 grows along the [200] direction for bcc-In2O3, while Fig. 5e demonstrates that the crystals grew along the [110] direction for rh-In2O3, which is consistent with the values estimated from the HRTEM image (Fig. 5b).
It is clear that pure bcc-In(OH)3 and the mixture of bcc-In(OH)3 and o-InOOH were obtained in the hydrothermal process (Fig. 1g). Pure bcc-In(OH)3 was obtained for the undoped products, while the mixture of bcc-In(OH)3 and o-InOOH was obtained with Zn doping. Mixtures of glycerol and ethylene glycol were used as the solvents, and the reaction mechanism is given below:
| In3+ + 3OH− → bcc-In(OH)3 | (1) |
| In3+ + 3OH− → o-InOOH + 2H2O | (2) |
After calcination at 500 °C for 2 h, pure bcc-In2O3 and the mixture of bcc- and rh-In2O3 were obtained, where bcc-In2O3 comes from the decomposition of the bcc-In(OH)3 precursor and rh-In2O3 was obtained after the annealing of the o-InOOH precursor:
| 2bcc-In(OH)3 → bcc-In2O3 + 3H2O↑ | (3) |
| 2o-InOOH → rh-In2O3 + H2O↑ | (4) |
Inspection of the SEM images in Fig. 1a–f and Fig. 4 indicates that the products before and after calcination had similar morphologies. Therefore, the calcination should not affect the phase transformation of In2O3, and the transformation occurred during the hydrothermal process. Also, the TG measurements suggest that the phase transformation occurs completely before calcination, and the Zn doping had a substantial effect on the phase transformation of In2O3. Thus, no intermediate species were produced.
From both a thermodynamic and a kinetic point of view, rh-In2O3, one of the so-called metastable states with high energy, will irreversibly transform to stable states (bcc-In2O3) when sufficient energy is available, the occurrence of which has been investigated for several decades.37 From the energy relations, all shifts between the individual polymorphs depend on the minimum free energy and abide by Gibbs’ phase rule. Under certain physical and chemical conditions, if the change of a crystal structure reduces the free energy of the system, the polymorphic transformation is inevitable.38 X. Feng et al.39 reported the role of ZnCl2 in transforming a cubic to a rhombohedral phase, in which Zn acted as a mineralizer for the induced rh-In2O3 phase. As we know, the crystal plane of the bcc-In2O3 phase grows along the vertical direction. The Zn dopants can adsorb on the surface of the In2O3 crystallites and change the vertical growth rate in the adsorbed crystal plane of the bcc-In2O3. The crystal phase transition of In2O3 is promoted by Zn entering into the lattice of bcc-In2O3, which causes the displacive transformation and morphotropy effect. Some of the bcc-In2O3 phase was transformed into the rh-In2O3 phase, and two kinds of crystallites of In2O3 (bcc-In2O3 and rh-In2O3) coexisted in the Zn-doped In2O3 samples. With increase of Zn doping concentration, more and more bcc-In2O3 phase was transformed into the rh-In2O3 phase, so the ratio of the rh-In2O3 phase increased. The effect of Zn was very similar to the role of SnCl4, NaCl, and NH4Cl reported by Cheng et al., in which the product was a mixture of rutile and anatase phase TiO2 in the absence of additives, and a pure rutile phase in the presence of additives.40
Determination of specific surface area and porosity
Fig. 6a and b and Fig. S1a–d, ESI† show the N2 adsorption–desorption isotherms of the pure In2O3 and Zn-doped In2O3. According to the IUPAC classification, the similar N2 adsorption–desorption isotherms of the six samples can be classified as type IV isotherms, with a hysteresis loop where the desorption required definitively higher energy than did the adsorption. The isotherms of the Zn-doped flower-like In2O3 (S5) show a hysteresis loop at a relatively high pressure, indicating the largest surface area. The amount of N2 adsorbed was higher for S5 (371.2 cm3 g−1) than S1 (146.3 cm3 g−1), S2 (179.9 cm3 g−1), S3 (205.1 cm3 g−1), S4 (254.7 cm3 g−1), or S6 (295.5 cm3 g−1). | ||
| Fig. 6 N2 adsorption–desorption curves of (a) pure In2O3 (S1) and (b) the Zn-doped flower-like In2O3 (S5). | ||
Formation mechanism
We propose a formation process for the Zn-doped flower-like In2O3. Firstly, the nucleation of In(OH)3 began. The boiling point of glycerol is the highest among the solvents used. Under the hydrothermal conditions, glycerol will remain in a liquid state, while the others are in the gas state. The liquid glycerol may form hydrogen bonds with InOOH which may favor the stabilization of InOOH.31 The glycerol–ethylene glycol mixed solvent may also coordinate to the In(OH)3 crystals, hindering the growth of certain surfaces. As the final hydrothermal product, In2O3 was simply enclosed by {001} faces because these faces have the slowest growth rate and the lowest surface energy. The cubic shape is consistent with the cubic crystal structure of In2O3. In the In2O3 cubic structure, the {001} family of planes contain three equivalent planes, (100), (010), and (001), which are perpendicular to the three directions [100], [010], and [001], respectively. The In2O3 nanocrystallites grow in all three directions at an equal speed.41 Consequently, the cubic morphology of the product enclosed with crystal faces of {001} is obtained (S1 in Fig. 4a).The doping with Zn could affect the morphology or coordination structure of the growth unit, which favoured transition coordination cubes of the In3+ ions (cube-like bcc-In2O3), resulting in the formation of sheet-like rh-In2O3. With increasing Zn doping concentration, more and more of the bcc-In2O3 phase was transformed into the rh-In2O3 phase, the amount of cube-like bcc-In2O3 decreased, and more and more sheet-like rh-In2O3 was formed. Then, the sheet-like rh-In2O3 self-assembled by oriented attachment to aggregate into the flower-like structures. The Zn dopant has different anisotropic and crystal planes to the In2O3, and those planes with different surface energies have a strong tendency to organize and form Zn-doped flower-like structures (S5) with more layers of sheets to reduce their surface energy. It is well known that a larger ionic radius induces a higher diffusion barrier, so the diffusion coefficient is lower with a bigger radius.42 Because of the larger size of the indium ions (In3+: 80 pm, Zn2+: 74 pm), the diffusion of zinc ions through the In2O3–Zn interface is faster than that of indium ions to the In2O3–Zn interface, and the gradual inward diffusion of zinc ions leads to an increase in the overall size of the microflowers. Furthermore, as the bcc-In2O3 phase is still present, the morphology of In2O3 exhibits a mixture of cubes and microflowers, as shown in Fig. 4b–f.
Gas sensing properties
Fig. 7 displays the plots of response versus gas concentration when sensors based on the samples with different amounts of Zn were exposed to NO2 at concentrations ranging from 5 to 100 ppm at 300 °C. The results show that for each sensor the response increased with an increase of the concentration of NO2. The appropriate addition of Zn would be beneficial to the improvement of the gas sensing properties, but excessive addition may reduce the available adsorption sites and worsen the gas sensing properties. The results reveal that the Zn-doped flower-like In2O3 (S5) sensor has the highest response, which can reach 27.4 ± 2.5, 165.2 ± 15.9, 427.5 ± 41.7, 821.7 ± 78.9, 1151.1 ± 108.6 for 5, 10, 30, 50, and 100 ppm NO2, respectively. Therefore, the following study of the selectivity was focused on the S5 sensor.The single point surface area was clearly the largest for S5 (46.41 m2 g−1) compared to the other samples. The increase in surface area for S5 is due to its well-organized 3D structural features, which was evidenced by the SEM and TEM images. With the support of the 3D In2O3 structures, the BET specific surface became larger. The larger the surface area, the easier the mass transport of NO2 in the material. So, the Zn-doped flower-like In2O3 (S5) may possess excellent gas sensing characteristics.
In order to optimize the proper working temperature of the sensor, parallel experiments were carried out in the range of 80–300 °C. Fig. 8a shows the relationship between the resistance and the operating temperature of the Zn-doped flower-like In2O3 (S5) sensor. The resistance in air decreased with an increase of temperature in the range of 80–300 °C, while the resistance in an NO2 atmosphere increased, and the relative resistance difference value increased. Moreover, it can be seen from Fig. 8b that the response increased with the operating temperature; when the operating temperature was 300 °C, the response was 821.7 ± 78.9 for 50 ppm NO2. Therefore, we chose 300 °C as the operating temperature for subsequent detection experiments of the Zn-doped flower-like In2O3.
Fig. 9a illustrates the typical response recovery characteristics of the sensor based on the Zn-doped flower-like In2O3 (S5) to NO2, with concentrations of 5, 10, 30, 50, and 100 ppm at 300 °C. The sensor exhibited an excellent response when exposed to various NO2 concentrations. When exposed to 5 ppm NO2, the response was 27.4 ± 2.5, indicating that a high gas response can be achieved in detecting low concentrations of NO2 using the Zn-doped flower-like In2O3 as a sensing material. With an increase in the concentration of NO2, the response increased. Furthermore, the sensor showed a quick response and short recovery time. When exposed to 50 ppm NO2, the response and recovery times (defined as the time required to reach 90% of the final equilibrium value) were 2 s and 6 s, respectively, as shown in Fig. 9b.
 | ||
| Fig. 9 Gas response of the sensor based on the Zn-doped flower-like In2O3 (S5) exposed to NO2 at (a) concentrations ranging from 5 to 100 ppm at 300 °C and (b) 50 ppm. | ||
To investigate the selectivity of the sensor, the Zn-doped flower-like In2O3 (S5) sensor was tested at 50 ppm NO2 and the same concentration of other gases, including toluene, acetone, ammonia, Cl2, H2S, formaldehyde and gasoline. The results are shown in Fig. 10. As can be seen, the gas response to NO2 is significantly higher than that for the other tested gases, with a magnitude about 14–500 times greater for 50 ppm NO2 than that for other tested gases under the same concentration. According to the experimental results, the Zn-doped flower-like In2O3 (S5) sensor can selectively detect NO2 with the interference of other gases.
 | ||
| Fig. 10 Selectivity of the Zn-doped flower-like In2O3 (S5) sensor to NO2 with a concentration of 50 ppm at 300 °C. | ||
Fig. 11 displays the stability of the Zn-doped flower-like In2O3 (S5) sensor to NO2 with a concentration of 50 ppm at 300 °C. The results show that the response decreased over time, but the response was still very high even after 30 days. So the Zn-doped flower-like In2O3 (S5) sensor is an excellent gas sensor material.
Gas sensing mechanism
Based on the above results, the Zn-doped flower-like In2O3 sensor shows improved sensing properties compared with a pure In2O3 sensor. Herein, we propose an analogous model for the Zn-doped flower-like In2O3. First, oxygen species were adsorbed on the surface of the particles in the air, and then were ionized into oxygen ions (O−, O2− and O2−) by capturing free electrons from the particles, thus leading to the formation of a thick space charge layer and an increase of surface band bending. NO2 is a typical oxidation gas with electron affinity of 2.30 eV, which is higher than the electron affinity of O2 (0.44 eV),43 so when the Zn-doped flower-like In2O3 sensor was exposed to NO2 gas, NO2 could not only capture the electrons from the conduction band due to its higher electrophilic property but also reacted with the adsorbed oxygen species leading to the formation of adsorbed NO2−(ads). The mechanism can be explained by several chemical reactions, which are shown below:44–46| O2(gas) → O2(ads) | (5) |
| O2(ads) + e− → O2−(ads) | (6) |
| O2−(ads) + e− → 2O−(ads) | (7) |
| NO2(gas) + e− → NO2−(ads) | (8) |
| NO2(gas) + O2−(ads) + 2e− → NO2−(ads) + 2O−(ads) | (9) |
The above reactions decrease the carrier concentration and electron mobility on the sensor surface, which led to the increase of depletion layer width accompanied by an increase in resistance. The electron transfer between In2O3 and Zn also led to the formation of an accumulation layer on the surface of Zn-doped flower-like In2O3. On the other hand, the trapped electrons were released to the Zn-doped flower-like In2O3 by NO2 after the supply of NO2 was stopped, leading to a decrease of the resistance.
A probable explanation for the enhanced sensing properties of the Zn-doped In2O3 is related to the defects resulting from the Zn doping. The doping resulted in some defects, e.g., oxygen vacancies, metal interstitials and surface defects,47,48 and these defects would play a significant role in the enhanced sensing ability for NO2 gas. The defects in the Zn-doped and pure In2O3 samples can be detected from the PL spectrum (Fig. 12a). Some PL peaks (420 nm, 445 nm, 450 nm, 468 nm and 485 nm) associated with different types of defects can be observed. As compared to those in previous reports of In2O3,32,41,49 a new peak at 445 nm was observed in the Zn-doped sample, which is consistent with that observed in the Zn-doped In2O3 nanowires prepared by N. Singh et al.32 This peak can be attributed to the neutral and ionized oxygen vacancies (Vo, 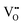 and
and 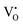 ) and metal vacancies
) and metal vacancies 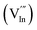 due to the Zn doping.47 Lots of defects can act as preferential adsorption sites for gas molecules.50 Due to the chemical potential gradient between Zn-doped In2O3 and NO2, the electrons migrated from Zn-doped In2O3 to NO2. When NO2 was adsorbed on the surface of the Zn-doped In2O3, the electrons transfer from a higher chemical potential gradient to a lower chemical potential gradient at equilibrium. The chemical potential gradient between the Zn-doped In2O3 and the adsorbed NO2 is higher than that between the undoped In2O3 and the adsorbed NO2. This results in the migration of more electrons when at equilibrium, resulting in an increase in the gas sensing performance of the Zn-doped In2O3.
due to the Zn doping.47 Lots of defects can act as preferential adsorption sites for gas molecules.50 Due to the chemical potential gradient between Zn-doped In2O3 and NO2, the electrons migrated from Zn-doped In2O3 to NO2. When NO2 was adsorbed on the surface of the Zn-doped In2O3, the electrons transfer from a higher chemical potential gradient to a lower chemical potential gradient at equilibrium. The chemical potential gradient between the Zn-doped In2O3 and the adsorbed NO2 is higher than that between the undoped In2O3 and the adsorbed NO2. This results in the migration of more electrons when at equilibrium, resulting in an increase in the gas sensing performance of the Zn-doped In2O3.
 | ||
| Fig. 12 (a) PL spectrum of the pure In2O3 (S1) and the Zn-doped flower-like In2O3 (S5) at room temperature. (b) AC impedance spectroscopy of S1, S2, S3, S4, S5 (insert) and S6 based sensors. | ||
The enhanced sensing performance of the Zn-doped flower-like In2O3 also can be ascribed to its large BET surface area. With the increasing Zn doping amount, the pure bcc-In2O3 was transformed into the mixture of bcc- and rh-In2O3, which may destroy the morphology of In2O3, and the surface area was changed accordingly. As the Zn-doped flower-like In2O3 (S5) has the largest BET surface area of 46.41 m2 g−1 (while for S1 it was only 11.90 m2 g−1), the sensor could absorb more NO2, thus the resistance increase and the resistance decrease became more noticeable, which can enhance its sensing performance. The large BET surface area increased the accessible surface area and facilitated the mass transport of gas in the material, which is favorable for their application in fields such as catalysis and sensing.
In order to clearly observe the dielectric response of the pure bcc-In2O3 and the coexistence of the bcc- and rh-In2O3, AC impedance spectra of the In2O3–Zn sensors with different amounts of Zn doping in the frequency range of 100 Hz to 10 MHz at 300 °C (50 ppm NO2) are shown in Fig. 12b. Upon the introduction of Zn, the diameter of the semicircle of AC impedance spectroscopy was enlarged, and the impedance also increased. The AC impedance spectrum of the sensor based on S5 shows the largest semicircle (the insert in Fig. 12b), which is far larger than the values of the sensors based on S1, S2, S3, S4, and S6. This is mainly due to the larger surface area of the S5 sample, which led to the largest resistance. With further increase of Zn doping concentration, the surface area decreased and the resistance declined. This agrees well with the SEM images, N2 adsorption–desorption curves and the BET surface area values. More details of the enhancing effect of the Zn-doped flower-like In2O3 on sensing properties need further investigation.
Conclusions
A facile hydrothermal route for the phase transformation of In2O3, in which pure bcc-In2O3 was transformed into a mixture of bcc- and rh-In2O3, was discussed. The molar ratio of In![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Zn profoundly affected the morphologies of the In2O3–Zn composites, and Zn-doped flower-like In2O3 structures were obtained when the molar ratio of In
Zn profoundly affected the morphologies of the In2O3–Zn composites, and Zn-doped flower-like In2O3 structures were obtained when the molar ratio of In![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Zn was 7
Zn was 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (S5). A possible growth mechanism of the Zn-doped flower-like In2O3 structures has been proposed. The Zn-doped flower-like In2O3 (S5) sensor exhibited the highest response of 27.4 ± 2.5 for 5 ppm NO2 at 300 °C. This is mainly due to the large specific surface area (utility function), the broad necks between the individual architectures (transducer function) and the defects created by Zn doping of the Zn-doped flower-like In2O3 structures. Moreover, the sensor also showed quick response–recovery behavior, and excellent selectivity and stability. Therefore, it is expected that this facile route to prepare the Zn-doped flower-like In2O3 structures will be an ideal candidate for applications in NO2 sensors.
1 (S5). A possible growth mechanism of the Zn-doped flower-like In2O3 structures has been proposed. The Zn-doped flower-like In2O3 (S5) sensor exhibited the highest response of 27.4 ± 2.5 for 5 ppm NO2 at 300 °C. This is mainly due to the large specific surface area (utility function), the broad necks between the individual architectures (transducer function) and the defects created by Zn doping of the Zn-doped flower-like In2O3 structures. Moreover, the sensor also showed quick response–recovery behavior, and excellent selectivity and stability. Therefore, it is expected that this facile route to prepare the Zn-doped flower-like In2O3 structures will be an ideal candidate for applications in NO2 sensors.
Acknowledgements
This work was supported by the National Natural Science Foundation (51172187), the SPDRF (20116102130002, 20116102120016) and 111 Program (B08040) of MOE, the Xi'an Science and Technology Foundation (CX12174, XBCL-1-08), the Shaanxi Province Science Foundation (2013KW12-02), and the NPU Fundamental Research Foundation (NPU-FRF-JC201232) of China.Notes and references
- S. S. Farvid, T. Wang and P. V. Radovanovic, Colloidal gallium indium oxide nanocrystals: a multifunctional light-emitting phosphor broadly tunable by alloy composition, J. Am. Chem. Soc., 2011, 133, 6711–6719 CrossRef CAS PubMed.
- X. Q. Wang, M. F. Zhang, J. J. Liu, T. Luo and Y. T. Qian, Shape- and phase-controlled synthesis of In2O3 with various morphologies and their gas-sensing properties, Sens. Actuators, B, 2009, 137, 103–110 CrossRef.
- H. X. Yang, L. Liu, H. Liang, J. J. Wei and Y. Z. Yang, Phase-controlled synthesis of monodispersed porous In2O3 nanospheres via an organic acid-assisted hydrothermal process, CrystEngComm, 2011, 13, 5011–5016 RSC.
- Y. G. Park, K. H. Seo, J. H. Lee, J. J. Kim, S. H. Cho, C. J. O'Conner and J. S. Lee, Phase transformation behavior of nanocrystalline ITO powders during heat-treatment: oxygen partial pressure effect, J. Electroceram., 2004, 13, 851–855 CrossRef CAS.
- W. H. Zhang and W. D. Zhang, Synthesis and optical properties of nanosheet-based rh-In2O3 microflowers by triethylene glycol-mediated solvothermal process, J. Phys. Chem. Solids, 2013, 74, 1271–1274 CrossRef CAS.
- P. D. C. King, T. D. Veal, F. Fuchs, C. Y. Wang, D. J. Payne, A. Bourlange, H. Zhang, G. R. Bell, V. Cimalla, O. Ambacher, R. G. Egdell, F. Bechstedt and C. F. McConville, Band gap, electronic structure and surface electron accumulation of cubic and rhombohedral In2O3, Phys. Rev. B: Condens. Matter Mater. Phys., 2009, 79, 205211 CrossRef.
- Z. B. Zhou, R. Q. Cui, Q. J. Pang, Y. D. Wang, F. Y. Meng, T. T. Sun, Z. M. Ding and X. B. Yu, Preparation of indium tin oxide films and doped tin oxide films by an ultrasonic spray CVD process, Appl. Surf. Sci., 2001, 172, 245–252 CrossRef CAS.
- J. T. Han, Y. H. Huang, X. J. Wu, C. L. Wu, W. Wei, B. Peng, W. Huang and J. B. Goodenough, Tunable synthesis of bismuth ferrites with various morphologies, Adv. Mater., 2006, 18, 2145–2148 CrossRef CAS.
- A. Murali, A. Barve, V. J. Leppert, S. H. Risbud, I. M. Kennedy and H. W. Lee, Synthesis and characterization of indium oxide nanoparticles, Nano Lett., 2001, 1, 287–289 CrossRef CAS.
- J. F. Yin and H. Q. Cao, Synthesis and photocatalytic activity of single-crystalline hollow rh-In2O3 nanocrystals, Inorg. Chem., 2012, 51, 6529–6536 CrossRef CAS PubMed.
- C. S. Lee, Il. D. Kim and J. H. Lee, Selective and sensitive detection of trimethylamine using ZnO–In2O3 composite nanofibers, Sens. Actuators, B, 2013, 181, 463–470 CrossRef CAS.
- H. H. Jiang, L. C. Zhao, L. G. Gai, L. Ma, Y. Ma and M. Li, Hierarchical rh-In2O3 crystals derived from InOOH counterparts and their sensitivity to ammonia gas, CrystEngComm, 2013, 15, 7003–7009 RSC.
- S. J. Kim, I. S. Hwang, C. W. Na, Il. D. Kim, Y. C. Kang and J. H. Lee, Ultrasensitive and selective C2H5OH sensors using rh-loaded In2O3 hollow spheres, J. Mater. Chem., 2011, 21, 18560–18567 RSC.
- L. N. Han, D. J. Wang, J. B. Cui, L. P. Chen, T. F. Jiang and Y. H. Lin, Study on formaldehyde gas-sensing of In2O3-sensitized ZnO nanoflowers under visible light irradiation at room temperature, J. Mater. Chem., 2012, 22, 12915–12920 RSC.
- Y. S. Li, J. Xu, J. F. Chao, D. Chen, S. X. Ouyang, J. H. Ye and G. Z. Shen, High-aspect-ratio single-crystalline porous In2O3 nanobelts with enhanced gas sensing properties, J. Mater. Chem., 2011, 21, 12852–12857 RSC.
- M. Pashchanka, R. C. Hoffmann, A. Gurlo and J. J. Schneider, Molecular based, chimie douce approach to 0D and 1D indium oxide nanostructures: evaluation of their sensing properties towards CO and H2, J. Mater. Chem., 2010, 20, 8311–8319 RSC.
- Y. C. Her, C. K. Chiang, S. T. Jean and S. Li, Huang, Self-catalytic growth of hierarchical In2O3 nanostructures on SnO2 nanowires and their CO sensing properties, CrystEngComm, 2012, 14, 1296–1300 RSC.
- M. Epifani, E. Comini, J. Arbiol, R. Diaz, N. Sergent, T. Pagnier, P. Siciliano, G. Faglia and J. R. Morante, Chemical synthesis of In2O3 nanocrystals and their application in highly performing ozone-sensing devices, Sens. Actuators, B, 2008, 130, 483–487 CrossRef CAS.
- K. Yao, D. Caruntu, S. Wozny, R. Huang, Y. H. Ikuhara, B. B. Cao, C. J. O'Connor and W. L. Zhou, Towards one key to one lock: catalyst modified indium oxide nanoparticle thin film sensor array for selective gas detection, J. Mater. Chem., 2012, 22, 7308–7313 RSC.
- P. Li, Y. Cai and H. Q. Fan, Porous thin sheet-based α-Fe2O3-doped In2O3 structures: hydrothermal synthesis and enhanced Cl2 sensing performance, RSC Adv., 2013, 3, 22239–22245 RSC.
- T. Wagner, C. D. Kohl, C. Malagù, N. Donato, M. Latino, G. Neri and M. Tiemann, UV light-enhanced NO2 sensing by mesoporous In2O3: interpretation of results by a new sensing model, Sens. Actuators, B, 2013, 187, 488–494 CrossRef CAS.
- W. H. Zhang and W. D. Zhang, Biomolecule-assisted synthesis and gas-sensing properties of porous nanosheet-based corundum In2O3 microflowers, J. Solid State Chem., 2012, 186, 29–35 CrossRef CAS.
- C. H. Lee, M. Kim, T. Kim, A. Kim, J. Paek, J. W. Lee, S. Y. Choi, K. Kim, J. B. Park and K. Lee, Ambient pressure syntheses of size-controlled corundum-type In2O3 nanocubes, J. Am. Chem. Soc., 2006, 128, 9326–9327 CrossRef CAS PubMed.
- B. Wang, X. Jin and Z. B. Ouyang, Synthesis, characterization and cathodoluminescence of self-assembled 1D ZnO/In2O3 nano-heterostructures, CrystEngComm, 2012, 14, 6888–6903 RSC.
- Z. Y. He, Z. H. Chen, Y. G. Li, Q. H. Zhang and H. Z. Wang, Molar ratio of In to urea directed formation of In2O3 hierarchical structures: cubes and nanorod-flowers, CrystEngComm, 2011, 13, 2557–2565 RSC.
- W. Cho, Y. H. Lee, H. J. Lee and M. Oh, Systematic transformation of coordination polymer particles to hollow and non-hollow In2O3 with pre-defined morphology, Chem. Commun., 2009, 31, 4756–4758 RSC.
- H. Y. Lai and C. H. Chen, Highly sensitive room-temperature CO gas sensors: Pt and Pd nanoparticle-decorated In2O3 flower-like nanobundles, J. Mater. Chem., 2012, 22, 13204–13208 RSC.
- Z. Y. Sun, A. Kumbhar, K. Sun, Q. S. Liu and J. Y. Fang, One-pot synthesis of reverse type-I In2O3@In2S3 core–shell nanoparticles, Chem. Commun., 2008, 16, 1920–1922 RSC.
- S. J. Luo, D. N. Yang, J. L. Zhuang and K. M. Ng, Synthesis and characterization of nearly monodisperse deltoidal icositetrahedral In2O3 nanocrystals via one-pot pyrolysis reaction, CrystEngComm, 2013, 15, 8065–8068 RSC.
- L. A. Mîinea and D. M. Hoffman, Low pressure chemical vapor deposition of fluorine-doped indium oxide films from an indium alkoxide complex, J. Mater. Chem., 2000, 10, 2392–2395 RSC.
- H. Dong, Z. Chen, L. Sun, L. Zhou, Y. Ling, C. Yu, H. H. Tan, C. Jagadish and X. Shen, Nanosheets-based rhombohedral In2O3 3D hierarchical microspheres: synthesis, growth mechanism, and optical properties, J. Phys. Chem. C, 2009, 113, 10511–10516 CAS.
- N. Singh, C. Y. Yan and P. S. Lee, Room temperature CO gas sensing using Zn-doped In2O3 single nanowire field effect transistors, Sens. Actuators, B, 2010, 150, 19–24 CrossRef CAS.
- H. X. Yang, S. P. Wang and Y. Z. Yang, Zn-doped In2O3 nanostructures: preparation, structure and gas-sensing properties, CrystEngComm, 2012, 14, 1135–1142 RSC.
- P. Li, H. Q. Fan and Y. Cai, In2O3/SnO2 heterojunction microstructures: Facile room temperature solid-state synthesis and enhanced Cl2 sensing performance, Sens. Actuators, B, 2013, 185, 110–116 CrossRef CAS.
- M. L. Zhao, G. S. Li, J. Zheng, L. P. Li and L. S. Yang, Fabrication of assembled-spheres YVO4:(Ln3+, Bi3+) towards optically tunable emission, CrystEngComm, 2012, 14, 2062–2070 RSC.
- H. Q. Fan and H. E. Kim, Perovskite stabilization and electromechanical properties of polycrystalline lead zinc niobate-lead zirconate titanate, J. Appl. Phys., 2002, 91, 317–322 CrossRef CAS.
- C. N. R. Rao, Modern Aspects of Solid State Chemistry, Plenum Press, New York, 1970, pp. 351–353 Search PubMed.
- R. S. Zhao, Q. J. Bian and Q. C. Ling, Crystallography and mineralogy, Higher Education Press, Beijing, 2004, pp. 169–170 Search PubMed.
- X. Feng, W. W. Yang, Q. L. Tang, Y. Zhang, G. L. Wang, W. Shi, J. H. Fang, J. Zhou, L. Y. Shi and W. Yan, ZnCl2 salt as both molten medium and mineralizer in ambient pressure synthesis of metastable corundum-type In2O3 nanocrystals, Powder Technol., 2012, 221, 164–167 CrossRef CAS.
- H. M. Cheng, J. M. Ma, Z. G. Zhao and L. M. Qi, Hydrothermal preparation of uniform nanosize rutile and anatase particles, Chem. Mater., 1995, 7, 663–671 CrossRef CAS.
- J. Yang, C. X. Li, Z. W. Quan, D. Y. Kong, X. M. Zhang, P. P. Yang and J. Lin, One-step aqueous solvothermal synthesis of In2O3 nanocrystals, Cryst. Growth Des., 2008, 8, 695–699 CAS.
- A. M. Asaduzzaman, F. Y. Wang and G. Schreckenbach, Quantum-chemical study of the diffusion of Hg(0, I, II) into the ice(Ih), J. Phys. Chem. C, 2012, 116, 5151–5154 CAS.
- Y. Itagaki, M. Mori, Y. Hosoya, H. Aono and Y. Sadaoka, O3 and NO2 sensing properties of SmFe1−xCoxO3 perovskite oxides, Sens. Actuators, B, 2007, 122, 315–320 CrossRef CAS.
- H. Kim, S. An, C. Jin and C. Lee, Structure and NO2 gas sensing properties of SnO2-core/In2O3-shell nanobelts, Curr. Appl. Phys., 2012, 12, 1125–1130 CrossRef.
- B. J. Melde and B. J. Johnson, Mesoporous materials in sensing: morphology and functionality at the meso-interface, Anal. Bioanal. Chem., 2010, 398, 1565–1573 CrossRef CAS PubMed.
- J. Zhao, T. L. Yang, Y. P. Liu, Z. Y. Wang, X. W. Li, Y. F. Sun, Y. Du, Y. C. Li and G. Y. Lu, Enhancement of NO2 gas sensing response based on ordered mesoporous Fe-doped In2O3, Sens. Actuators, B, 2014, 191, 806–812 CrossRef CAS.
- S. Y. Li, C. Y. Lee, P. Lin and T. Y. Tseng, Low temperature synthesized Sn doped indium oxide nanowires, Nanotechnology, 2005, 16, 451–457 CrossRef CAS.
- M. Kumar, V. N. Singh, F. Singh, K. V. Lakshmi, B. R. Mehta and J. P. Singh, On the origin of photoluminescence in indium oxide octahedron structures, App. Phys. Lett., 2008, 92, 171907/1–171907/3 CAS.
- S. W. Shu, D. B. Yun, Y. Wang, F. Wang, Z. R. Wang and W. Zhong, Thermal-induced phase transition and assembly of hexagonal metastable In2O3 nanocrystals: A new approach to In2O3 functional materials, J. Cryst. Growth, 2010, 312, 3111–3116 CrossRef CAS.
- M. W. Ahn, K. S. Park, J. H. Heo, J. G. Park, D. W. Kim, K. J. Choi, J. H. Lee and S. H. Hong, Gas sensing properties of defect controlled ZnO-nanowire gas sensor, Appl. Phys. Lett., 2008, 93, 263103/1–263103/3 CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c3ra47467d |
| This journal is © The Royal Society of Chemistry 2014 |