Plasma-assisted electrochemical exfoliation of graphite for rapid production of graphene sheets†
Abstract
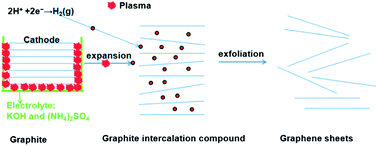
We describe a new and highly efficient plasma-assisted electrochemical exfoliation method, involving a plasma-generated graphite cathode and a graphite anode, for the production of graphene sheets from electrodes in a basic electrolyte solution in a short reaction time.

 Please wait while we load your content...
Please wait while we load your content...