DOI:
10.1039/C3RA46549G
(Paper)
RSC Adv., 2014,
4, 10607-10613
Origin of the synergistic effect between polysaccharide and thiourea towards adsorption and corrosion inhibition for mild steel in sulphuric acid
Received
10th November 2013
, Accepted 18th December 2013
First published on 20th December 2013
Abstract
The origin of the synergistic effect between polysaccharides and thiourea towards adsorption on mild steel in 0.5 M H2SO4 and their effect on corrosion inhibition has been studied. Two different polysaccharides, gum arabic and agar agar are used for this purpose. Results obtained from potentiodynamic polarization and electrochemical impedance spectroscopic studies reveal that thiourea imparts a profound synergistic effect towards the corrosion inhibition of mild steel in H2SO4 by the polysaccharides. The polysaccharide–thiourea system acts essentially as a mixed type corrosion inhibitor and the phenomenon of adsorption mechanism on metal surface is proposed as physisorption. Detailed FTIR studies of the surface adsorbed layers of inhibitors have been done to elucidate the origin of the synergistic effect towards the coadsorption and subsequent corrosion inhibition of the metal.
Introduction
Growing environmental concern and enforcement of strict environmental regulations have compelled the researchers to focus on the development of eco-friendly and biodegradable corrosion inhibitors. In this regard a flurry of work is now being undertaken by various research groups involving plant extracts, drugs, bio-polymeric molecules, amino acids, medicinal products, etc.1–9 Among bio-polymers, polysaccharides have added advantages as corrosion inhibitors by being water soluble, easily available and of low cost.4,10–14 Chemically modified polysaccharides have been reported to impart greater corrosion inhibitory action over the parent polysaccharides.15–17 Apart from chemical modification, halides and surfactants, together with the polysaccharides, provide better results due to a synergistic effect.18,19 In this endeavor, we report the synergistic effect of thiourea (TU) on the corrosion inhibition of mild steel by gum arabic (GA) and agar agar (AA) in 0.5 M H2SO4 solution. GA is a water soluble anionic polymer comprising a complex mixture of polysaccharides (major fraction) and glucoproteins (minor fraction). Its polysaccharide component consists of L-arabinose, D-galactose, L-rhamnose, D-glucuronic acid (found in nature as magnesium, potassium, and calcium salts).20,21 The anticorrosive behavior of GA for various metals in different corrosive conditions is observed, primarily due to the adsorption of GA on the metal surface through the carboxylate group of the glucuronic acid present in GA.11–14,18 For AA, the main backbone is assigned to be a linear polymer structure consisting of alternating 1,3-linked-D-galactopyranose and 1,4-linked 3,6-anhydro-L-galactopyranose units. The neutral form of these polymers is called agarose and it is held responsible for the high gelation property of agar.22 However, the gelation behavior varies with the degree of substitution of the 1,3-linked-D-galactopyranose units by sulphate esters or other groups.23 AA has been established as a potential corrosion inhibitor for aluminium alloys in alkaline media, as well as for mild steel in acid solution.24–26 TU and substituted thioureas have long been known for their excellent corrosion inhibitory action on mild steel in acidic media.27,28 It is the sulfur atom of TU which gets adsorbed on the metal surface and thereby decreases both the cathodic and anodic reactions.27,28
Reports on the synergistic effect produced by thiourea, or its derivatives, towards the inhibition of corrosion by other inhibitors is very limited and the origin of such synergism has not been addressed at all.29 In addition, it is reported that TU exists mostly in a protonated form in acidic media.30 Thus it would be interesting to investigate the effect of any possible electrostatic interactions of the protonated TU with anionic GA or the hydroxyl group of AA towards simultaneous adsorption on the metal surface, which may impart synergism towards the inhibition of corrosion of mild steel in 0.5 M H2SO4.
Experimental
Materials
Test specimens were cut from a commercially available mild steel rod (wt% composition: 0.22 C, 0.31 Si, 0.60 Mn, 0.04 P, 0.06 S and the remainder iron) and the cross-sectional surface was ground with different grade emery papers (400, 600, 800, 1200 and 1600), rinsed with acetone, washed thoroughly with doubly distilled water and used as the working electrode. GA, AA and TU were obtained from Merck India and used without any further purification.
Electrochemical measurements
Potentiodynamic polarization and electrochemical impedance measurements were done using a conventional three-electrode system (model: Gill AC, ACM Instruments, UK) consisting of a mild steel working electrode (WE) with an exposed area of 0.25 cm2, platinum as the counter electrode and a saturated calomel electrode (SCE) as the reference. The potential sweep rate for the potentiodynamic polarization curves was 1 mV s−1. Before polarization measurements, the WE was kept in the test solution for 30 minutes and the open circuit potential (OCP) was then monitored for another 10 minutes for confirmation of a steady state. Corrosion current density (icorr) was determined from the intercept of extrapolated cathodic and anodic Tafel lines at the corrosion potential (Ecorr). Electrochemical impedance measurements were performed in the frequency range 10 mHz to 100 kHz with an a.c. amplitude of ±10 mV (rms) at the rest potential. All the experiments have been carried out at a room temperature of around 30 °C.
Surface analysis
A scanning electron microscope (SEM, S-3000N, Hitachi) was used to study the surface morphology of the metal surface. Before the measurement, test coupons were exposed to 0.5 M H2SO4 solutions with different concentrations of the inhibitors for four hours, washed with distilled water and then kept in a vacuum desiccator. The surface of the dried specimen was scratched with a knife and the resultant powder was used for FTIR studies (KBr pellet method, Thermo Nicolet, model iS10).
Results and discussion
Polarization measurements
Potentiodynamic polarization curves for the mild steel in 0.5 M H2SO4 solution containing 10 mM TU, 1000 ppm GA, 1000 ppm AA and their mixture with 10 mM TU are shown in Fig. 1. Values of the electrochemical corrosion parameters, such as the corrosion potential (Ecorr), cathodic Tafel slope (bc), anodic Tafel slope (ba), and corrosion current density (icorr) for different concentrations of GA, AA, TU and both the GA–TU and AA–TU mixed systems are given in Table 1.
 |
| | Fig. 1 Potentiodynamic polarization curves for mild steel in 0.5 M H2SO4 in the presence of (a) 1000 ppm GA, (b) 1000 ppm AA, (c) 10 mM TU, (d) 1000 ppm GA–10 mM TU and (e) 1000 ppm AA–10 mM TU. | |
Table 1 Data from the potentiodynamic polarization studies for mild steel in 0.5 M H2SO4 in various inhibitor systems
| System |
Ecorr (mV per SCE) |
icorr (mA cm−2) |
ba (mV dec−1) |
bc (mV dec−1) |
η%P |
s |
| Blank |
517 |
1.7 |
93 |
114 |
— |
— |
| 10 mM thiourea |
525 |
0.3 |
81 |
122 |
82 |
— |
| |
| Gum arabic |
| 100 ppm |
488 |
1.1 |
90 |
120 |
35 |
— |
| 250 ppm |
487 |
0.9 |
80 |
123 |
47 |
— |
| 500 ppm |
491 |
0.82 |
86 |
109 |
52 |
— |
| 750 ppm |
479 |
0.79 |
91 |
106 |
54 |
— |
| 1000 ppm |
481 |
0.71 |
78 |
115 |
58 |
— |
| |
| 10 mM thiourea + gum arabic |
| 100 ppm |
524 |
0.19 |
88 |
113 |
88 |
1.33 |
| 250 ppm |
520 |
0.15 |
82 |
101 |
91 |
1.42 |
| 500 ppm |
518 |
0.13 |
72 |
101 |
92 |
1.46 |
| 750 ppm |
518 |
0.1 |
88 |
116 |
94 |
1.45 |
| 1000 ppm |
515 |
0.09 |
66 |
105 |
95 |
1.48 |
| |
| Agar agar |
| 100 ppm |
483 |
0.98 |
66 |
105 |
42 |
— |
| 250 ppm |
477 |
0.86 |
76 |
105 |
49 |
— |
| 500 ppm |
493 |
0.63 |
73 |
107 |
63 |
— |
| 750 ppm |
496 |
0.47 |
67 |
106 |
72 |
— |
| 1000 ppm |
488 |
0.42 |
75 |
105 |
75 |
— |
| |
| 10 mM thiourea + agar agar |
| 100 ppm |
534 |
0.3 |
87 |
117 |
82 |
1.51 |
| 250 ppm |
521 |
0.13 |
77 |
112 |
92 |
1.58 |
| 500 ppm |
527 |
0.11 |
67 |
106 |
93 |
1.41 |
| 750 ppm |
515 |
0.08 |
70 |
110 |
95 |
1.62 |
| 1000 ppm |
516 |
0.07 |
64 |
88 |
96 |
1.64 |
The inhibition efficiency (η%P) values are calculated from the following equation:
| |
 | (1) |
where,
icorr and
icorr(inh) are the values of the corrosion current densities of uninhibited and inhibited specimens, respectively. It can be seen from the table that GA and AA exhibit low inhibition efficiency, but in the presence of thiourea, a substantial increase in the inhibition efficiency is observed. With respect to the uninhibited sample, the corrosion potential is seen to shift towards a negative direction in the presence of TU, indicating a decrease in the rate of the cathodic hydrogen evolution reaction (cathodic inhibitor). Polysaccharides, on the contrary, shift the corrosion potential towards a noble direction and thus act as anodic inhibitors. In the polysaccharide–TU mixed system, the corrosion potential remains in the negative direction at low concentrations of polysaccharides, which gradually changes towards the value of the uninhibited sample with an increase in the polysaccharide concentration. The results indicate that the polysaccharide–TU system acts as a cathodic inhibitor at lower polysaccharide concentrations, while in the presence of higher concentrations of polysaccharides, it behaves as a mixed type inhibitor, suppressing both the cathodic and anodic reactions.
Electrochemical impedance measurements (EIS)
The corrosion behaviour of mild steel in 0.5 M H2SO4 solution and in the presence of various concentrations of inhibitors is investigated by EIS. Nyquist plots for the mild steel in 0.5 M H2SO4 solution containing 1000 ppm GA, 1000 ppm AA, 10 mM TU and their mixture are shown in Fig. 2. The complex plane plots obtained in the presence of various concentrations of inhibitors show only one time constant corresponding to one capacitive loop. The diameter of the capacitive loop increases with an increase in inhibitor concentration, indicating an increased charge transfer resistance at the metal–electrolyte interface.31–35 Closer observations of these plots also reveal that the capacitive loops are depressed with their centre under the real axis, which may be related to the surface heterogeneity due to the microscopic roughness of the electrode surface and inhibitor adsorption on it.31–35 Accordingly, we have fitted the observed Nyquist plots with an equivalent circuit containing a parallel combination of a charge transfer resistance-constant phase element (Rct-CPE) as depicted in Fig. 3. Rs corresponds to the electrolyte resistance.
 |
| | Fig. 2 Nyquist plots for mild steel in 0.5 M H2SO4 in the presence of (a) 1000 ppm GA, (b) 1000 ppm AA, (c) 10 mM TU, (d) 1000 ppm GA–10 mM TU and (e) 1000 ppm AA–10 mM TU. | |
 |
| | Fig. 3 Equivalent circuit used to fit the impedance data shown in Fig. 2. | |
The constant phase element (CPE) is related to the double layer capacity (Cdl) and its impedance is given by:31–35
where,
Q is a proportionality coefficient,
i is the imaginary number,
ω is the angular frequency, and
n is a measure of surface irregularity. For whole numbers of
n = 1, 0, −1, CPE is reduced to the classical lumped element capacitor (
C), resistance (
R), and inductance (
L), respectively. The fitted parameters using the above model are presented in
Table 2.
Table 2 Impedance parameters for the corrosion of mild steel in 0.5 M H2SO4 in various inhibitor systems
| System |
Rct (Ω cm2) |
Q (μΩ−1 sn cm−2) |
n |
Cdl (μF cm−2) |
η%Z |
s |
| Blank |
3.7 |
740 |
0.83 |
221 |
— |
— |
| 10 mM thiourea |
51 |
109 |
0.88 |
54 |
93 |
— |
| |
| Gum arabic |
| 100 ppm |
8.5 |
463 |
0.84 |
161 |
56 |
— |
| 250 ppm |
11 |
327 |
0.85 |
121 |
66 |
— |
| 500 ppm |
12 |
293 |
0.82 |
85 |
69 |
— |
| 750 ppm |
12.5 |
250 |
0.83 |
77 |
70 |
— |
| 1000 ppm |
13 |
235 |
0.83 |
72 |
71 |
— |
| |
| 10 mM thiourea + gum arabic |
| 100 ppm |
85 |
99 |
0.85 |
43 |
96 |
1.56 |
| 250 ppm |
95 |
92 |
0.85 |
40 |
96 |
1.66 |
| 500 ppm |
108 |
81 |
0.86 |
37 |
96.5 |
1.68 |
| 750 ppm |
157 |
71 |
0.86 |
34 |
97.6 |
1.68 |
| 1000 ppm |
163 |
57 |
0.87 |
28 |
97.7 |
1.69 |
| |
| Agar agar |
| 100 ppm |
7.2 |
572 |
0.85 |
217 |
48 |
— |
| 250 ppm |
11.3 |
457 |
0.84 |
167 |
67 |
— |
| 500 ppm |
18.6 |
388 |
0.85 |
162 |
80 |
— |
| 750 ppm |
22 |
288 |
0.85 |
118 |
83 |
— |
| 1000 ppm |
24 |
154 |
0.86 |
62 |
84.5 |
— |
| |
| 10 mM thiourea + agar agar |
| 100 ppm |
53.5 |
89 |
0.9 |
49 |
93 |
1.52 |
| 250 ppm |
121 |
81 |
0.9 |
48 |
97 |
1.65 |
| 500 ppm |
139 |
77 |
0.9 |
46 |
97 |
1.78 |
| 750 ppm |
154 |
76 |
0.89 |
44 |
97.6 |
1.81 |
| 1000 ppm |
173 |
71 |
0.89 |
41 |
97.8 |
1.82 |
To obtain a direct correlation between the charge-transfer resistance (Rct) and the double layer capacitance (Cdl), the latter has been recalculated using the following equation:34,35
and the values are shown in
Table 2. It is seen that with an increase in the concentration of GA and AA in their TU mixed system, the
Rct values gradually increase with a concomitant decrease in
Cdl values. This reflects a gradual increase in the thickness of the adsorbed inhibitor layer with inhibitor concentration. The
Rct values for both the mixed system are found to be several times greater than the sum of the corresponding values when the inhibitors are used separately. This is a clear indication of the existence of a positive interaction towards co-adsorption in the polysaccharide–TU mixed systems on the metal surface.
The inhibition efficiencies, η%Z, in terms of Rct are calculated through the following equation:
| |
 | (4) |
where,
Rct and
R0ct are the values of the charge transfer resistance observed in the presence and absence of an inhibitor. The inhibition efficiencies,
η%Z, are seen to increase considerably in the mixed inhibitor systems (
Table 2) showing the same trend as those obtained from the polarisation measurements (
Table 1).
Synergism parameter
To determine the extent of synergism between GA–TU and AA–TU towards adsorption on the metal surface and the subsequent inhibition of corrosion, a synergism parameter (s), as originally proposed by Aramaki and Hackerman36 and used subsequently by many authors,18,37 has been calculated using the following equation:| | |
s = [1 − (η1 + η2)]/(1 − η1+2)
| (5) |
where, η1 is the inhibition efficiency of TU, η2 is that of the polysaccharide and η1+2 is the measured inhibition efficiency for the polysaccharide in combination with TU. Normally, s < 1 indicates an antagonistic effect, s = 1 means no interaction, whereas s > 1 represents a synergistic interaction between the two inhibitors towards adsorption on the metal surface. Using the inhibition efficiency values from the polarization measurements and the EIS experiment, synergistic parameters have been calculated (Tables 1 and 2). Values of s for all concentrations of the polysaccharides studied are greater than unity, reflecting a strong synergism between the polysaccharide–TU towards co-adsorption on the mild steel surface in 0.5 M H2SO4. It is noteworthy that in 1 M HCl, Rct values of the polysaccharide–TU mixed systems do not vary much from the total Rct values for TU and the polysaccharide (Fig. 4). This rules out the existence of any significant synergistic effect between the polysaccharide–TU towards inhibition of corrosion for mild steel in HCl.
 |
| | Fig. 4 Nyquist plots for mild steel in 1 N HCl in the presence of (a) 500 ppm GA, (b) 500 ppm AA, (c) 10 mM TU, (d) 500 ppm GA–10 mM TU and (e) 500 ppm AA–10 mM TU. | |
Adsorption isotherm
To elucidate the nature of the interaction involving the polysaccharide–TU mixed inhibitor systems with the mild steel surface, we have assessed several types of adsorption isotherms. Excellent fitting of the experimental values was obtained following the simplest Langmuir monolayer adsorption model where equivalency of all the adsorption sites are considered (correlation coefficient, R2 = 0.999). According to this model, the degree of surface coverage θ (θ = η%Z/100) is related to the concentration of the inhibitor (C) as per the equation:where, Kads is the adsorption constant. The plot of C/θ with C at 30 °C (Fig. 5) yields a straight line with a slope value of 0.999 and 0.983 for the GA–TU and AA–TU mixed systems, respectively. From the values of the adsorption constant, Kads, the standard free energy ΔG0ads of adsorption for both the systems are determined using the following equation:| |
ΔG0ads = −RT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln(1 × 106Kads) ln(1 × 106Kads)
| (7) |
where, 1 × 106 is the concentration of water molecules expressed in mg L−1, R is the universal gas constant and T is the temperature. From the above equation, ΔG0ads is calculated as −25.36 kJ mol−1 and −24.38 kJ mol−1 for the GA–TU and AA–TU systems, respectively at 303 K (30 °C). The negative value of ΔG0ads is indicative of the spontaneous adsorption of the inhibitor system on the mild steel surface. The observed range of free energy values suggests that the adsorption is electrostatic in nature, i.e., the physisorption provides a major contribution towards the whole adsorption process.38
 |
| | Fig. 5 Langmuir adsorption plot for mild steel in 0.5 M H2SO4 in the presence of 1000 ppm GA–10 mM TU (-■-) and 1000 ppm AA–10 mM TU (-○-). | |
Surface analysis
Scanning electron micrographs (SEM) of the surface of the mild steel immersed in 0.5 M H2SO4, with and without the inhibitors are shown in Fig. 6(a–d). The mild steel surface immersed in 0.5 M H2SO4 shows a very rough surface owing to severe uniform corrosion. TU is seen to provide a perforated adsorbed layer. However, in the presence of mixed inhibitors, a compact adsorbed layer on the metal surface is observed, which acts as a barrier between the metal surface and the corrosive media, and thereby reduces the rate of corrosion.
 |
| | Fig. 6 SEM images of mild steel after immersion in 0.5 M H2SO4 with (a) no inhibitor, (b) 10 mM TU (c) 1000 ppm GA–10 mM TU and (d) 1000 ppm AA–10 mM TU. | |
To get some insight into any possible interactions between the polysaccharides (GA, AA) and TU in acidic medium, which can play an important role for the synergism towards adsorption on the mild steel sample, FTIR spectra of pure GA, AA, TU, polysaccharides adsorbed on the metal surface, TU adsorbed on the metal surface and the GA–TU and AA–TU mixed systems adsorbed on the metal sample have been recorded and the relative intensities of the major vibrational bands are compared (Fig. 7). For TU, the bands at 1413 cm−1 and 730 cm−1 are assigned to the stretching vibration of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S, whereas the bands around 3100–3400 cm−1 are due to N–H stretching.39,40 Bands at 1470 cm−1 and 630 cm−1 are assignable to different modes of NCN vibrations.39,40 In the TU–steel complex, the intensity of the 1413 cm−1 band is decreased and shifts to 1380 cm−1, the N–H stretching bands, on the contrary, are seen to be shifted towards higher wavenumbers. The band at 730 cm−1 shifts to a lower wavenumber and overlaps with that of the NCN vibration to give a broad band at 675 cm−1. This clearly indicates that the complex is formed through the S atom of TU.39 For pure GA, the band at 1626 cm−1 is assigned to the stretching vibrations of the C
S, whereas the bands around 3100–3400 cm−1 are due to N–H stretching.39,40 Bands at 1470 cm−1 and 630 cm−1 are assignable to different modes of NCN vibrations.39,40 In the TU–steel complex, the intensity of the 1413 cm−1 band is decreased and shifts to 1380 cm−1, the N–H stretching bands, on the contrary, are seen to be shifted towards higher wavenumbers. The band at 730 cm−1 shifts to a lower wavenumber and overlaps with that of the NCN vibration to give a broad band at 675 cm−1. This clearly indicates that the complex is formed through the S atom of TU.39 For pure GA, the band at 1626 cm−1 is assigned to the stretching vibrations of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond of the carboxylate group and the stretching vibration of the C–O bond is located at 1070 cm−1 and 1424 cm−1.14 On adsorption of GA on steel, all these bands are observed with reduced relative intensity and with a shift to a lower frequency for the vibrational modes of the C–O bond (1424 cm−1 band shifts to 1380 cm−1) suggesting bonding of GA with the oxide surface of steel through the carboxylate group.14 In the GA–TU–steel complex, a relatively broad peak centered at 1350 cm−1 is seen to develop, which may be interpreted as discussed below.
O bond of the carboxylate group and the stretching vibration of the C–O bond is located at 1070 cm−1 and 1424 cm−1.14 On adsorption of GA on steel, all these bands are observed with reduced relative intensity and with a shift to a lower frequency for the vibrational modes of the C–O bond (1424 cm−1 band shifts to 1380 cm−1) suggesting bonding of GA with the oxide surface of steel through the carboxylate group.14 In the GA–TU–steel complex, a relatively broad peak centered at 1350 cm−1 is seen to develop, which may be interpreted as discussed below.
 |
| | Fig. 7 FT-IR spectra of (a) TU, (b) TU–steel complex, (c) GA, (d) GA–steel complex, (e) GA–TU–steel complex, (f) AA, (g) AA–steel complex and (h) AA–TU–steel complex. | |
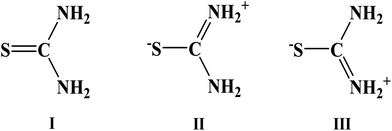
Among the three canonical forms of TU, as shown below, contribution from II and III increases when TU is bonded to the metal surface through its sulfur atom.41 Complete protonation of the NH2 group may also occur in an acidic solution, as used in the present study.30 This cationic or protonated NH2 group of TU interacts electrostatically with the carboxylate group of GA which will increase the contributions from the canonical forms II and III even further. It is already reported that in comparison to HCl medium, a greater number of GA molecules remain free and dispersed in H2SO4 medium.14 Due to GA–TU adduct formation, most of the GA molecules are now activated to get adsorbed on the metal surface, which will in turn help to make the TU–steel bond stronger. This cooperative adsorption of TU and GA is responsible for the observed synergism towards inhibition of corrosion of mild steel in H2SO4. A similar type of cooperative adsorption of TU and ClO4− on iron electrodes in both acidic and neutral media is already documented.30,41 As a result of such interaction, the 1380 cm−1 bands observed both for the GA–steel and TU–steel complexes overlap with each other, and shift further towards a lower wavenumber at 1350 cm−1 in the GA–TU–steel complex.
AA adsorbs on the metal surface mainly through its hydroxyl groups, as evident from a prominent shift of the –OH stretching vibration band from 3440 cm−1 in its pure state,42,43 to 3395 cm−1 in its adsorbed state. The 1380 cm−1 band for the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S stretching vibration in the TU–steel complex is seen to be shifted to a lower wavenumber, and is overlapped with the 1370 cm−1 band of AA belonging to C–C bending,43 resulting in a relatively broad band at around 1345 cm−1 in the AA–TU–steel complex. This suggests that the cationic or protonated NH2 of TU interacts with the hydroxyl group of AA (ion–dipole interaction), which subsequently results in a further decrease in C
S stretching vibration in the TU–steel complex is seen to be shifted to a lower wavenumber, and is overlapped with the 1370 cm−1 band of AA belonging to C–C bending,43 resulting in a relatively broad band at around 1345 cm−1 in the AA–TU–steel complex. This suggests that the cationic or protonated NH2 of TU interacts with the hydroxyl group of AA (ion–dipole interaction), which subsequently results in a further decrease in C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S bond order by forming a stronger TU–steel complex formation. Such ion–dipole interaction is reported to act during the adsorptive removal of cationic methylene blue by agar in acidic medium.42
S bond order by forming a stronger TU–steel complex formation. Such ion–dipole interaction is reported to act during the adsorptive removal of cationic methylene blue by agar in acidic medium.42
Conclusions
Potentiodynamic polarization (Tafel extrapolation) and an EIS method have established the existence of a strong synergism between polysaccharide–TU towards their adsorption on a mild steel surface in H2SO4 medium and the subsequent inhibition of corrosion.
From potentiodynamic polarization studies, it is observed that at lower polysaccharide concentrations, the polysaccharide–TU predominantly retards the cathodic reduction reaction, while at higher polysaccharide concentrations it acts as a mixed inhibitor.
The calculated standard free energy of adsorption indicates the physical adsorption of the polysaccharide–TU system on the metal surface.
FTIR studies reveal that TU gets adsorbed onto the metal surface through the S atom, whereas the carboxylate group present in GA and the hydroxyls of AA are found to be mostly responsible for adsorption. The cationic or protonated NH2 group of TU interacts with the carboxylate group of the non-adsorbed GA molecules (ion–ion interactions) and the hydroxyl group of AA (ion–dipolar interaction). This helps to activate a greater number of polysaccharide molecules to be adsorbed on the metal surface, which in turn results in a stronger metal–TU bond. Such cooperative adsorption of polysaccharide–TU onto the steel surface seems to be responsible for the observed synergistic effect towards inhibition of mild steel corrosion in H2SO4 medium.
Acknowledgements
PI thanks the Department of Science and Technology, Govt of India for supporting a research project under the Fast Track Scheme for Young Scientists (no. SR/FT/CS-110/2010, dt. 20.09.2011).
References
- P. B. Raja and M. G. Sethuraman, Mater. Lett., 2008, 62, 113 CrossRef CAS PubMed.
- D. Kesavan, M. Gopiraman and N. Sulochana, Chem. Sci. Rev. Lett., 2012, 1, 1 Search PubMed.
- G. Gece, Corros. Sci., 2011, 53, 3873 CrossRef CAS PubMed.
- S. A. Umoren, Open Corros. J., 2009, 2, 175 CrossRef CAS.
- K. P. Vinod Kumar, M. S. N. Pillai and G. R. Thusnavis, J. Mater. Sci. Technol., 2011, 27, 1143 Search PubMed.
- G. M. A. El-Hafez and W. A. Badawy, Electrochim. Acta, 2013, 108, 860 CrossRef PubMed.
- A. R. H. Zadeh, I. Danaee and Md. H. Maddahy, J. Mater. Sci. Technol., 2013, 29, 884 Search PubMed.
- J.-J. Fu, S.-N. Li, Y. Wang, X.-D. Liu and L.-D. Lu, J. Mater. Sci., 2011, 46, 3550 CrossRef CAS.
- J. Fu, H. S. Zang, Y. Wang, S. N. Li, T. Chen and X. D. Liu, Ind. Eng. Chem. Res., 2012, 51, 6377 CrossRef CAS.
- M. Abdallah, Port. Electrochim. Acta, 2004, 22, 161 CrossRef CAS.
- S. A. Umoren, I. B. Obot, E. E. Ebenso, P. C. Okafor, O. Ogbobe and E. E. Oguzie, Anti-Corros. Methods Mater., 2006, 53, 277 CrossRef CAS.
- S. A. Umeron, Cellulose, 2008, 15, 751 CrossRef.
- S. A. Umeron, Port. Electrochim. Acta, 2009, 27, 565 CrossRef.
- M. A. Abu-Dalo, A. A. Othman and N. A. F. Al-Rawashdeh, Int. J. Electrochem. Sci., 2012, 7, 9303 CAS.
- S. Banerjee, V. Srivastava and M. M. Singh, Corros. Sci., 2012, 59, 35 CrossRef CAS PubMed.
- S. Cheng, S. Chen, T. Liu, X. Chang and Y. Yin, Mater. Lett., 2007, 61, 3276 CrossRef CAS PubMed.
- A. M. Fekry and R. R. Mohamed, Electrochim. Acta, 2010, 55, 1933 CrossRef CAS PubMed.
- S. A. Umoren, O. Ogbobe, I. O. Igwe and E. E. Ebenso, Corros. Sci., 2008, 50, 1998 CrossRef CAS PubMed.
- M. Mobin and M. Alam Khan, J. Dispersion Sci. Technol., 2013, 34, 1496 CrossRef CAS.
- H. A. Swenson, H. M. Kaustinen, O. A. Kaustinen and N. S. Thompson, J. Polym. Sci., Part A-2, 1968, 6, 1593 CrossRef CAS.
- Y. Dror, Y. Cohen and R. Yerushalmi-Rozen, J. Polym. Sci., 2006, 44, 3265 CrossRef CAS.
- M. Sekkal, J.-P. Huvenne, P. Legrand, B. Sombret, J.-C. Mollet, A. M. Givernaud and M.-C. Verdus, Microchim. Acta, 1993, 112, 1 CrossRef CAS.
- S. Arnott, A. Fullmer, W. E. Scott, J. C. M. Dea, R. Moorhouse and D. A. Rees, J. Mol. Biol., 1974, 90, 269 CrossRef CAS.
- J. D. Talati and R. M. Modi, Anti-Corros. Methods Mater., 1976, 23, 6 CrossRef CAS.
- A. N. Mukherji, I. Singh and V. A. Altekar, Br. Corros. J., 1975, 10, 155 CrossRef CAS PubMed.
- W. A. Siddiqui, V. M. Chaubey and M. S. Ahmad, Mater. Sci. Res. India, 2007, 4, 175 CAS.
- M. Metikoš-Huković and R. Babić, J. Appl. Electrochem., 1996, 26, 443 CrossRef.
- R. T. Loto, C. A. Loto and A. P. I. Popoola, J. Mater.
Environ. Sci., 2012, 3, 885 CAS.
- D. Gopi, N. Bhuvaneswaran, S. Rajeswarai and K. Ramadas, Anti-Corros. Methods Mater., 2000, 47, 332 CrossRef CAS.
- Z. Q. Tian, Y. Z. Lian and M. Fleischmann, Electrochim. Acta, 1990, 35, 879 CrossRef CAS.
- T. Pajkossy, Solid State Ionics, 2005, 176, 1997 CrossRef CAS PubMed.
- S. John and A. Joseph, RSC Adv., 2012, 2, 9944 RSC.
- J.-B. Jorcin, M. E. Orazem, N. Pébère and B. Tribollet, Electrochim. Acta, 2006, 51, 1473 CrossRef CAS PubMed.
- X. Wu, H. Ma, S. Chen, Z. Xu and A. Sui, J. Electrochem. Soc., 1999, 146, 1847 CrossRef CAS PubMed.
- M. Lebrini, F. Robert and C. Roos, Int. J. Electrochem. Sci., 2010, 5, 1698 CAS.
- K. Aramaki and N. Hackerman, J. Electrochem. Soc., 1969, 116, 568 CrossRef CAS PubMed.
- S. A. Umoren, Y. Li and F. H. Wang, Corros. Sci., 2010, 52, 1777 CrossRef CAS PubMed.
- F. Bentiss, M. Lebrini and M. Lagrenee, Corros. Sci., 2005, 47, 2915 CrossRef CAS PubMed.
- S. Selvakumar, S. M. Ravi Kumar, G. P. Joseph, K. Rajarajan, J. Madhavan, S. A. Rajasekar and P. Sagayaraj, Mater. Chem. Phys., 2007, 103, 153 CrossRef CAS PubMed.
- G. B. Aitken, J. L. Duncan and G. P. McQuillan, J. Chem. Soc. A, 1971, 2695 RSC.
- P. Cao, J. Yao, B. Ren, R. Gu and Z. Tian, J. Phys. Chem. B, 2002, 106, 10150 CrossRef CAS.
- B. Samiey and F. Ashoori, Chem. Cent. J., 2012, 6, 14 CrossRef CAS PubMed.
- M. F. Nazarudin, A. A. Shamsuri and M. N. Shamsudin, Int. J. Pure Appl. Sci. Technol., 2011, 3, 35 CAS.
|
| This journal is © The Royal Society of Chemistry 2014 |
Click here to see how this site uses Cookies. View our privacy policy here. 





![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln(1 × 106Kads)
ln(1 × 106Kads)


![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S, whereas the bands around 3100–3400 cm−1 are due to N–H stretching.39,40 Bands at 1470 cm−1 and 630 cm−1 are assignable to different modes of NCN vibrations.39,40 In the TU–steel complex, the intensity of the 1413 cm−1 band is decreased and shifts to 1380 cm−1, the N–H stretching bands, on the contrary, are seen to be shifted towards higher wavenumbers. The band at 730 cm−1 shifts to a lower wavenumber and overlaps with that of the NCN vibration to give a broad band at 675 cm−1. This clearly indicates that the complex is formed through the S atom of TU.39 For pure GA, the band at 1626 cm−1 is assigned to the stretching vibrations of the C
S, whereas the bands around 3100–3400 cm−1 are due to N–H stretching.39,40 Bands at 1470 cm−1 and 630 cm−1 are assignable to different modes of NCN vibrations.39,40 In the TU–steel complex, the intensity of the 1413 cm−1 band is decreased and shifts to 1380 cm−1, the N–H stretching bands, on the contrary, are seen to be shifted towards higher wavenumbers. The band at 730 cm−1 shifts to a lower wavenumber and overlaps with that of the NCN vibration to give a broad band at 675 cm−1. This clearly indicates that the complex is formed through the S atom of TU.39 For pure GA, the band at 1626 cm−1 is assigned to the stretching vibrations of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond of the carboxylate group and the stretching vibration of the C–O bond is located at 1070 cm−1 and 1424 cm−1.14 On adsorption of GA on steel, all these bands are observed with reduced relative intensity and with a shift to a lower frequency for the vibrational modes of the C–O bond (1424 cm−1 band shifts to 1380 cm−1) suggesting bonding of GA with the oxide surface of steel through the carboxylate group.14 In the GA–TU–steel complex, a relatively broad peak centered at 1350 cm−1 is seen to develop, which may be interpreted as discussed below.
O bond of the carboxylate group and the stretching vibration of the C–O bond is located at 1070 cm−1 and 1424 cm−1.14 On adsorption of GA on steel, all these bands are observed with reduced relative intensity and with a shift to a lower frequency for the vibrational modes of the C–O bond (1424 cm−1 band shifts to 1380 cm−1) suggesting bonding of GA with the oxide surface of steel through the carboxylate group.14 In the GA–TU–steel complex, a relatively broad peak centered at 1350 cm−1 is seen to develop, which may be interpreted as discussed below.
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S stretching vibration in the TU–steel complex is seen to be shifted to a lower wavenumber, and is overlapped with the 1370 cm−1 band of AA belonging to C–C bending,43 resulting in a relatively broad band at around 1345 cm−1 in the AA–TU–steel complex. This suggests that the cationic or protonated NH2 of TU interacts with the hydroxyl group of AA (ion–dipole interaction), which subsequently results in a further decrease in C
S stretching vibration in the TU–steel complex is seen to be shifted to a lower wavenumber, and is overlapped with the 1370 cm−1 band of AA belonging to C–C bending,43 resulting in a relatively broad band at around 1345 cm−1 in the AA–TU–steel complex. This suggests that the cationic or protonated NH2 of TU interacts with the hydroxyl group of AA (ion–dipole interaction), which subsequently results in a further decrease in C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S bond order by forming a stronger TU–steel complex formation. Such ion–dipole interaction is reported to act during the adsorptive removal of cationic methylene blue by agar in acidic medium.42
S bond order by forming a stronger TU–steel complex formation. Such ion–dipole interaction is reported to act during the adsorptive removal of cationic methylene blue by agar in acidic medium.42