Design of dialkyl surfactants from nitrilotriacetic acid as head group
Juan V. Trilloa,
Francisco Meijide*a,
José Vázquez Tatoa,
Aida Jovera,
Victor H. Sotob,
Santiago de Frutosa and
Luciano Galantinic
aDepartamento de Química Física, Facultad de Ciencias, Universidad de Santiago de Compostela, Avda. Alfonso X El Sabio s/n, 27002 Lugo, Spain
bEscuela de Química, Centro de Investigación en Electroquímica y Energía Química (CELEQ), Universidad de Costa Rica, San José, Costa Rica
cDipartimento di Chimica, Università di Roma “Sapienza”, P. le A. Moro 5, 00185 Roma, Italy
First published on 20th December 2013
Abstract
New double-chain surfactants can be designed in which nitrilotriacetic acid plays the role of glycerol in phospholipids. The synthetic methodology is applied to the synthesis of (2-(bis(2-dodecylamino)-2-oxoethyl)amino)acetic acid. This is a symmetric dimeric surfactant but the methodology allows the synthesis of asymmetric double-chain surfactants as well. The Krafft temperature is 16–18 °C. The critical aggregation concentration of the new surfactant (4.76 × 10−5 mol dm−3 in NaOH 0.01 M and 25 °C; surface tension measurements) is two hundred times lower than that for sodium 2-dodecanamidoacetate, which can be considered its monoalkyl surfactant analog, indicating the enhanced surfactant properties resulting from the presence of two hydrophobic alkyl chains. The shape of the aggregates was investigated by transmission electron microscopy. At 40 °C, the main structure present in solution corresponds to vesicles and, from the size distribution of their radii, values of (0.34 ± 0.06)kT and 56.7 ± 2.6 nm were obtained for the effective bending elasticity constant and the spontaneous radius of curvature of vesicles, respectively.
Introduction
During the last few years dimeric surfactants (gemini,1 hybrid2 and phospholipids) have attracted considerable interest because they are characterized by critical micelle concentrations that are one to two orders of magnitude lower than those for the corresponding monomeric surfactants. Dimeric surfactants are also much more efficient than monomeric in decreasing the surface tension of water.1 Some zwitterionic gemini surfactants, with phosphate and ammonium as ionic groups,3 exhibit extraordinary properties and structures forming gels, micelles, vesicles, and coacervates. In the coacervate region, someone resembles sponges. As a result of the screening of many surfactant derivatives, it is possible to predict confidently which compounds will self-assemble into a sponge morphology.Phosphatidylcholine is a fat derivative of glycerol carrying a saturated hydrophobic palmitic acid, an unsaturated hydrophobic oleic acid, and a phosphate group forming an ester with choline. Consequently a zwitterionic surfactant is formed. We have to notice the different nature of the two fatty chains. Phosphatidylcholine is usually the most abundant phospholipid in animal and plants, and it is a key building block of membrane bilayers. Generally, as it is well-known, these phospholipids have a strong tendency to self-aggregate in water forming bilayers as in vesicles. Length and nature of linked chains can be modified to obtain different families of compounds. Two common examples are dipalmitoylphosphatidylcholine (DPPC, the major constituent of the pulmonary surfactant) and dioleoylphosphatidylcholine (DOPC). It has been observed a slight decrease in the average area per lipid as well as a linear increase in the bilayer thickness with increasing chain lengths.4 Positive correlation between most probable vesicle size and acyl chain lengths have also been noticed.5 Many other variants are possible as well, sphingomyelins being known examples.
On the other hand, a series of glycerol-based double- or triple-chain surfactants with two ionic (either sulfonate, sulfate or carboxylate) groups have been prepared by Zhu et al.6 The sulfate and carboxylate derivatives have higher water solubilities than the corresponding sulfonate ones, and the triple-chain surfactants show excellent surface-active properties, such as micelle forming and ability to lower the surface tension, compared with the corresponding single- and double-chain surfactants.
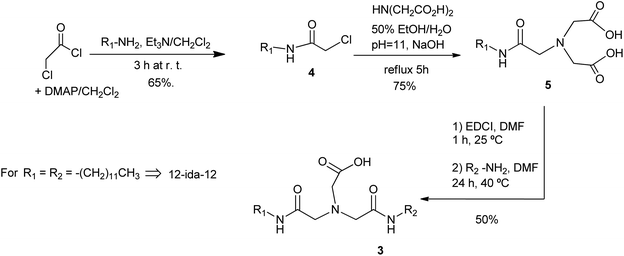
The structure of nitrilotriacetic acid 1 (Fig. 1) resembles the structure of glycerol since both compounds present a central atom from which three organic functions emerge. In glycerol the central atom is a carbon one and hydroxy groups are the organic functions, while a nitrogen atom and carboxylic acids are the characteristics of 1.
 | ||
| Fig. 1 General structure of surfactants 2 from nitrilotriacetic acid 1. Structure of (2-(bis(2-dodecylamino)-2-oxoethyl)amino)acetate (12-ida-12, 3). | ||
Having all previous information in mind, new derivatives can be designed in which nitrilotriacetic acid plays the core role of glycerol in phospholipids. With appropriate substituents, new surfactants with structure 2 may be obtained. For that purpose, in 2, at least one of the R groups (say R1) must have a hydrophobic nature (as, for instance, a long alkyl chain). The nature of the second R group (say R2) allows to obtain symmetric (R1 = R2) or asymmetric (R1 ≠ R2) surfactants which could emulate the characteristics of phospholipids mentioned above. However, as we will comment on below, this structure analysis does not mean that the synthesis of the new surfactants starts from 1.
In this paper, we probe the correctness of this approach for designing new surfactants, starting with the symmetric compound (2-(bis(2-dodecylamino)-2-oxoethyl)amino)acetate (compound 3 with R1 = R2 = C12H25). We will use the acronym 12-ida-12 (“ida” corresponds to IminoDiAcetic acid used in the synthesis). The behavior of this compound in water has been studied by means of surface tension, fluorescence of pyrene as a probe, transmission electron microscopy (TEM), and light scattering techniques. The compound presented here is an example of molecular biomimetics as its design is approached from the knowledge of structure and properties of natural phospholipids.
Results and discussion
The synthesis of 12-ida-12 was performed according to the three steps procedure outlined in Scheme 1. Amide 4 (R1 = C12H25) was prepared as described by Puigmartí-Luis et al.7 The ida function 5 was introduced by means of nucleophilic substitution of the α-chloride with iminodiacetic acid according to the method published by van Wyk et al.8 In situ closure of the iminodiacetic acid 5 to the anhydride with 3-ethyl-1-[3-(dimethylamino)propyl]carbodiimide hydrochloride (EDCI), followed by treatment with the R2NH2 amine (R2 = R1)9 afforded the diamide 12-ida-12 (3).The 12-ida-12 surfactant was characterized by measurement of its Krafft temperature, Tk,10 and critical aggregation concentration, cac, as the aggregation of a surfactant has to be studied above its Tk and its cac. The measurements were performed in NaOH (0.01 M, pH = 12) aqueous solutions. This high pH value, which ensures complete ionization of the carboxylic acid, was chosen to prevent possible effects observed for some carboxylic surfactant systems at lower pH values. Although there should not be much difference in carboxyl group deprotonation at basic pH values, it is known that myristic acid crystallizes as 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture with its sodium salt at pH 9.8 and as pure sodium salt upon addition of sodium chloride, but gives long fibers (curds) of a carboxylate monohydrate at pH 11.5. According to Träger et al.11 the curvature of the myristate bilayer is enforced by hydration, not by repulsive forces between anions. It is also well known the effect of pH on the aggregation behavior of some bile salts in water, even forming gels at pH values close to neutrality.12 In these systems hydrogen ions have the tendency to be adsorbed onto bile salt micelles as they form,13 and acidic species in micelles and at the interface are present in a more appreciable extent than in the bulk aqueous solution.14 Thus micelles of natural bile salts are mixed micelles formed by species with a different degree of ionization.14,15 It seems that hydrogen ions are adsorbed more weakly onto smaller aggregates than onto larger ones.13
1 mixture with its sodium salt at pH 9.8 and as pure sodium salt upon addition of sodium chloride, but gives long fibers (curds) of a carboxylate monohydrate at pH 11.5. According to Träger et al.11 the curvature of the myristate bilayer is enforced by hydration, not by repulsive forces between anions. It is also well known the effect of pH on the aggregation behavior of some bile salts in water, even forming gels at pH values close to neutrality.12 In these systems hydrogen ions have the tendency to be adsorbed onto bile salt micelles as they form,13 and acidic species in micelles and at the interface are present in a more appreciable extent than in the bulk aqueous solution.14 Thus micelles of natural bile salts are mixed micelles formed by species with a different degree of ionization.14,15 It seems that hydrogen ions are adsorbed more weakly onto smaller aggregates than onto larger ones.13
For measuring Tk, a solution of the surfactant (2.5 × 10−4 mol dm−3) was slowly cooled (approx. at 1 °C h−1) till 3 °C and then heated (at the same rate). During the cooling process a sharp change in the scattered light16 (accompanied by a visual clarity/translucence change)17 was recorded at 16 °C. When heating the translucent/clear change was observed at 18 °C. This narrow temperature range is taken as Tk.
Surface tension measurements were carried out at 25 °C, and after observing the effect of temperature and ageing of samples on the type of aggregates (see below TEM images) such measurements were also performed at 40 °C.
Fig. 2 shows the dependence of the surface tension of the solution with the surfactant concentration. The typical profile for a surfactant in aqueous solution is observed and from the breaking point of γ vs. ln[12-ida-12] plot, the critical aggregation concentration and the surface tension at cac, γcac, were determined. Values are recompiled in Table 1.
 | ||
| Fig. 2 Dependence of the surface tension of water solutions (NaOH 0.01 M) of 12-ida-12 with its concentration at 25 °C. | ||
| Compound | Temp. (°C) | cac (mol dm−3) | γcac (mN m−1) | 106 Γ (mol m−2) | ao (Å2) | Ref. | |
|---|---|---|---|---|---|---|---|
| 12-ida-12 | 25 | 4.76 × 10−5 | 40.2 | 3.36 | 49 | This work | |
| Freshly prepared | 40 | 5.2 × 10−5 | 31.7 | 4.49 | 37 | ||
| After 4 days | 3.3 × 10−5 | 32.1 | 5.81 | 29 | |||
| Sodium 2-dodecanamidoacetate | 25 | 8.3 × 10−3–0.014 | 39 | 63 | 18 and 19 | ||
| Sodium laurate | 25 | 0.016–0.026 | 36.8 | 46.9 | 20–24 | ||
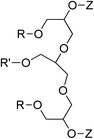 |
R = C10H21; R′ = CH3, Z = (CH2)3SO3Na | 20 | 8.1 × 10−5 | 36.0 | 6 | ||
| R = R′ = C10H21, Z = (CH2)3SO3Na | 1.4 × 10−5 | 28.0 | |||||
| R = R′ = C10H21, Z = SO3Na | 9 × 10−6 | 27.0 | |||||
| R = R′ = C10H21, Z = CH2CO2Na | 4.0 × 10−5 | 29.0 | |||||
| 3C10NAc-Tz | 25 | 1.1 × 10−5 | 28.6 | 4.84 | 34 | 25 | |
| 3C8taAm | 9.2 × 10−7 | 39.9 | 1.84 | 91 | 26 | ||
| 3C10taAm | 8.34 × 10−6 | 33.3 | 4.88 | 34 | |||
| 3C12taAm | 6.41 × 10−6 | 36.9 | 4.42 | 38 | |||
The value obtained for cac at 25 °C (temperature at which most values have been published) is approximately two hundred times lower than the cmc of the N-dodecyl derivative of glycine (2-dodecanamidoacetate) for which cmc values of 8.3 × 10−3–0.014 mol dm−3 have been published.18,19 This surfactant has an amide group as linkage between the hydrophobic and hydrophilic moieties and is probably the closer monoalkyl surfactant analog of 12-ida-12 found in the literature. The comparison is also advantageous with sodium laurate, for which cmc values higher than those for 2-dodecanamidoacetate have been published by Mori and Matsumura and others.20–24 The comparison between these three surfactants suggests that the lower cac value for 12-ida-12 may be ascribed to the presence of the two alkyl chains and not to the presence of the amide groups.
The cac value is comparable to those obtained by Zhu et al.6 for the series of glycerol-based double- or triple-chain surfactants with two ionic (sulfonate, sulfate, and carboxylate) groups, and by Yoshimura and Esumi25 for a ring-type trimeric surfactant (3C10NAc-Tz) of 1,3,5-tris[1-(carboxyalkyl)amino]triazine with three lauryl chains. Other anionic trimeric surfactants show even lower cmc values. All of them are recompiled in Table 1. However the comparison is not straightforward as in these trimeric surfactants the ratio [number of alkyl chains/number of ionic groups] is one, but it is two for 12-ida-12. The reduction of cmc of trimeric surfactants respect to the corresponding monomeric and dimeric surfactants has been also found for cationic triquaternary ammonium surfactants by Zana et al.27 Guo et al.28 have obtained three trimeric quaternary ammonium surfactants having adamantane cores with cmc values in the range 0.5–2 × 10−3 mol dm−3.
TEM images were collected at 25 °C in freshly prepared samples and after ageing for several days. Fig. 3 shows two typical TEM images at initial time and after four days.
 | ||
| Fig. 3 TEM images of a 9.2 × 10−4 mol dm−3 surfactant solution (in NaOH 0.01 M) for freshly prepared samples (top) and after ageing for four days at 25 °C. | ||
These images confirm the existence of an aggregation process as small dark objects, having some tendency to associate in larger ones, are evident. However, no differences can be noticed between samples of different ageing time.
However, examples in which the temperature effect on aggregation processes and on the morphology of the aggregates is highly important are well known.29,30 In this system, increasing the temperature to 40 °C induced a different growth process, and vesicle like structures are evident (A in Fig. 4), some of them having a large size (B). At this temperature we have noticed that the number of vesicle structures increases with time, but no significative differences are observed after four days. Some open vesicles (C) and associated particles (D) are also observed.
Consequently, surface tension measurements were also carried at 40 °C at zero time and after four days (Fig. 5). The cac is more clearly defined after four days and a decrease in its value (from 5.2 × 10−5 to 3.3 × 10−5 mol dm−3) is also observed. This may be due to a different arrangement of the molecules at the interface as the ao values suggest (see below).
 | ||
| Fig. 5 Dependence of the surface tension of water solutions (NaOH 0.01 M) of 12-ida-12 with its concentration at 40 °C for freshly prepared samples (□) and after ageing them for four days (■). | ||
The cac for 12-ida-12 was also estimated by means of steady state fluorescence measurements in the presence of a probe (pyrene) at 40 °C. The dependence of the ratio of intensities of the first (373 nm) and third (384 nm) vibronic peaks of pyrene, I1/I3, is related to the polarity of the microenvironment where pyrene is located. The dependence of I1/I3 with the concentration of the surfactant is plotted in Fig. 6. A typical sigmoid profile is obtained with an inflection point at 4.64 × 10−5 mol dm−3. This value corresponds to the cac of the surfactant and compares well with the value obtained by surface tension measurements in freshly prepared solutions. At high surfactant concentrations I1/I3 reaches a plateau value around to 1.0. This limiting value is lower than those for sodium laurate (I1/I3 ≈ 1.1)21 and for 3C10NAc-Tz (I1/I3 ≈ 1.2).25 For vesicles, values ranging from 0.9 (ref. 31 and 32) to 1.67 (ref. 33) have been published. It is accepted that in these aggregates pyrene is solubilized in the lipophilic core of the bilayer.34 Furthermore, significative differences in I1/I3 values are observed for different structures (as micelles, vesicles or tubes) originated by the same or similar surfactants. One example is the formation of micelles by lauryl sulfonate betaine (LSB) (I1/I3 = 1.19) and bis(2-ethylhexyl) sulfosuccinate (AOT) (I1/I3 = 0.83), and vesicles formed by a mixture LSB–AOT: 3/7 (I1/I3 = 0.96).31 Another example is the case of sodium deoxycholate35 which forms small aggregates at high pH (I1/I3 = 0.70, a value which is very close to the one for the very unpolar cyclohexane),35 and the sodium salts of p-tert-butylphenyl amide of cholic and ursodeoxycholic acids which form tubes (I1/I3 = 1.19 and 1.1, respectively).30,36 A third example corresponds to N-(2-hydroxydodecyl)-L-alanine which forms bilayer structures (I1/I3 = 1.65–1.27) and N-(n-dodecyl)-L-alanine which form micelles (I1/I3 = 1.059). The value mentioned above for 12-ida-12 is compatible with less closed structures than micelles, as vesicles.
The fluorescence spectrum of pyrene (Fig. 6) also shows a broad band around 480 nm, generally attributed to the fluorescence emission of excimers,35,37 as the energy of the excited state of excimers is lower than for an excited monomer. Excimers are formed when an excited-state monomer interacts with a close enough ground-state monomer forming an excited dimer. In this band the vibrational structure observed in monomers is lost. The low intensity of this band does not change with time.
The surface behavior may be analyzed in terms of the adsorption Gibbs equation, which relates the change in the equilibrium surface tension with changes in the chemical potentials of all of the solutes at constant temperature. From this isotherm, the surface excess concentration, Γ, is determined by eqn (1).
 | (1) |
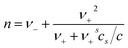 | (2) |
This equation shows that the prefactor n in the Gibbs equation depends on the concentration and stoichiometric coefficients of the surfactant (c, ν−, and ν+) and also on both the stoichiometry (ν+s) and the concentration, cs, of an added inert electrolyte. When the concentration of electrolyte is in a large excess with respect to the surfactant concentration, the prefactor becomes n = ν− that is, n = 1. Once Γ is known, the surface area per molecule can be determined by the eqn (3),
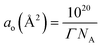 | (3) |
The value obtained at 25 °C for ao (=49 Å2) is close to the value determined for sodium laurate by Mori and Matsumura20 and lower than the one for sodium 2-dodecanamidoacetate.19 Menger et al.39 have recently questioned the validity of the Gibbs equation to determine the surface area per molecule at the air/water interface. According to this criticism, the Gibbs analysis overestimates the area per molecule at saturation. However such an overestimation is hardly applicable to the present case. The cross section of one hydrocarbon chain,40 Ac ≈ 20 Å2 matches well with the area occupied by each molecule of long alkyl chain alcohols in the monolayer. For instance, for 1octadecanol a value of 23 ± 0.1 Å2 per molecule has been measured.41 This is an expected agreement because of the alcohol molecules are highly ordered at the interface. For a double tail surfactant the cross section of two alkyl chains will correspondingly be 40–42 Å2.42,43 Such a value is a minimum one for ao because of steric constraints between the alkyl chains of adjacent molecules in the monolayer. The small difference between the experimental ao and 2Ac may be due to the electrostatic ion–ion repulsions of ionic headgroups. This agreement suggests that a monolayer at the air/water interface is formed.
For monoalkyl ionic surfactants as sodium laurate and oleate,20–24,44 values around 47 Å2 have been published while for phospholipids the accepted values are in the range 60–67 Å2.45–47 These values are consistent with the surface area per lipid head group in egg phosphatidylcholine vesicles as values of 74 Å2 and 61 Å2 are obtained for outer and inner lipids, respectively.48
The ao value observed at 40 °C in freshly prepared samples is slightly lower than 2Ac and, in our opinion, previous comments are still applicable. As cac, ao is time dependent and after four days the value is 29 Å2, not far from Ac. This probably means that a surfactant bilayer is now formed at the interface. This interpretation would be in agreement with Rosen et al.49 who have proposed the formation of multilayer structures to explain low ao values measured for some gemini surfactants. Tsubone et al.50 have also accepted the formation of multilayer films. Similar comments would probably apply to 3C10NAc-Tz and to 3CntaAm (with three carboxylate headgroups and three hydrocarbon chains; n = 10, 12) since the geometry and charge of their head group would suggest a higher value for ao than the observed one.26
A recount of the size of the vesicles (A type; total number = 231) observed at 40 °C gives a mean value for the radius of ![[R with combining macron]](https://www.rsc.org/images/entities/i_char_0052_0304.gif) = 48 ± 12 nm, and the distribution of Fig. 7, where f is the fraction of vesicles with sizes in a given interval.
= 48 ± 12 nm, and the distribution of Fig. 7, where f is the fraction of vesicles with sizes in a given interval.
 | ||
| Fig. 7 Histogram corresponding to the radii distribution of vesicles of the surfactant 12-ida-12 in the experimental conditions of Fig. 4. f is the fraction of vesicles with sizes in a given interval. The superimposed continuous line is the best fit of eqn (4) to it. | ||
By applying the thermodynamic theory of aggregation from Israelachvili51 and the Helfrich elasticity theory for membranes,52 Denkov et al.53 have deduced eqn (4) for vesicles, which represents the size distribution of the vesicles in the sample.
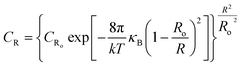 | (4) |
In this equation, CR and CRo are the molar concentrations of aggregates of size R and Ro, respectively, Ro is the spontaneous radius of curvature of vesicles, κB is the effective bending elasticity constant, and kT is the thermal energy.
By fitting this equation to data of Fig. 6 by a non-linear optimization procedure, the effective bending elasticity constant of vesicles and the spontaneous radius of curvature can be deduced. Values of κB = (0.34 ± 0.06)kT and Ro = 56.7 ± 2.6 nm are obtained. Fig. 7 shows a good result for the fit.
κB is very close to the one obtained for vesicles formed by of a p-tertbutylphenyl amide derivative of cholic acid, (0.36 ± 0.06)kT,30 and for cetyltrimethyl-ammonium bromide/sodium octyl sulfonate catanionic vesicles, (0.7 ± 0.2)kT.54 However, most of the published values for bilayer vesicles of lipids are within the range (1–20)kT.53,55–58 Under a low polydispersity condition, eqn (4) can be simplified by expanding its right side around Ro, allowing a quick estimation of Ro and κB from the average radius of vesicles and its standard deviation (see Denkov et al.53 for details). This represents an extraordinary simplicity for the analysis of experimental results since only the knowledge of the normal distributions of the vesicle radius is required. However, a consequence of the low value for the effective bending constant is that the vesicles have a rather broad size distribution and this simplified analysis is not advisable. Since κB < kBT the vesicles are stabilized by thermal fluctuations (rather than by the spontaneous curvature), and their size distribution is rather broad.59
Above its cmc, sodium laurate forms spherical micelles with radii lower than 2 nm (fully compatible with the length of the alkyl chain) and an average aggregation number N = 89.24 The aggregation number of 3C10NAc-Tz at the cmc was found to be about N = 580 ± 20,25 while from dynamic light scattering, DLS, measurements, two main populations with hydrodynamic diameters of 41 ± 9 and 338 ± 167 nm were observed at 4.7 × 10−5 mol dm−3 of 3C10-NAc-Tz. For this compound Yoshimura and Esumi accept that the trimeric surfactants are more likely to form micelles in the bulk solutions. The aggregation numbers of 3C8taAm, 3C10taAm, and 3C12taAm at the cmc were about 10, 82, and 63, respectively.26
From the values of ![[R with combining macron]](https://www.rsc.org/images/entities/i_char_0052_0304.gif) for the vesicles and the surface area per molecule ao, the aggregation number of vesicles formed by 12-ida-12 can be roughly estimated as 1.8 × 104.
for the vesicles and the surface area per molecule ao, the aggregation number of vesicles formed by 12-ida-12 can be roughly estimated as 1.8 × 104.
The size of the aggregates in aqueous solution was also investigated by DLS measurements. The results shown in Fig. 8 correspond to a sample with a concentration 6.0 × 10−4 mol dm−3 after four days, and shows the volume weighted distribution after analysis of the intensity correlation function by the NLLS method at a scattering angle of 90°. This analysis gives a more meaningful representation of size polydispersity than cumulant methods.60
 | ||
| Fig. 8 Size distribution of aggregates from dynamic light scattering of 12-ida-12 at 40 °C after four days at a concentration of 6.0 × 10−4 mol dm−3 in aqueous solution and [NaOH] = 0.01 M. | ||
The most significant populations are in the ranges 81.6–164.8 and 333.1–673.2 nm, with maxima at 97.2 and 397.1 nm, respectively, for the hydrodynamic diameter of particles. Dimensions of the lower population compares favorably with diameters from TEM images of type A vesicles. The larger size population, which represents a lower proportion, can correspond to large type B vesicles, open vesicles (C) and associated particles (D). The limits of the accuracy of DLS analysis have been commented on by Viseu et al.60
Conclusions
It has been proved that new surfactants can be designed from nitrilotriacetic acid, playing a similar core role to glycerol in phospholipids. The proposed synthetic route allows the synthesis of symmetric or asymmetric dialkyl chain surfactants. The methodology is illustrated with the compound 12-ida-12. For its synthesis dodecyl amine has been used as the carrier of the alkyl chains. TEM images strongly suggest that above a critical aggregation concentration, the surfactant form vesicles.Experimental
Synthesis
Step 1:7 Chloroacetyl chloride (55 mmol, 4.4 ml) and N,N-dimethyl-4-aminopyridine, DMAP, (0.1 mg) were dissolved in dried CH2Cl2 (50 ml) under N2 atmosphere. The mixture was cooled to 0 °C and dodecylamine (50 mmol, 9.3 g) with triethylamine (8.4 ml) in dried CH2Cl2 (10 ml) was slowly (1 h) added dropwise. The resulting suspension was stirred for 3 h at room temperature. The solvent residue was filtered and the filtrate was washed with CH2Cl2. The organic phase was washed with aqueous HCl (1 M, 3 × 15 ml). The resulting organic phase was dried with MgSO4, filtered and concentrated in vacuo. The crude product was purified by silica gel column chromatography eluting with ethyl acetate![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) hexane (6
hexane (6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4). The resulting white solid 4 was dried in vacuo at 50 °C overnight. Yield: 65%.
4). The resulting white solid 4 was dried in vacuo at 50 °C overnight. Yield: 65%.
1H NMR (300 MHz, d6-DMSO, δ/ppm): 6.63 (s, 1H, NH); 3.97 (s, 2H, ClCH2–); 3.23 (q, J = 6.9 Hz, 2H, NHCH2–); 1.54–1.27 (m, 20H, –NHCH2(CH2)10CH3); 0.83 (t, J = 6.4 Hz, 3H, –CH3). 13C NMR (75 MHz d6-DMSO, δ/ppm): 163.4 (CONH–); 43.5 (CH2Cl); 39.8 (–NHCH2–); 31.9; 29.3; 29.2; 29.3; 29.0; 28.9; 26.5; 22.8; 14.6 (–(CH2)10CH3).
Step 2:8 4 (R1 = C12H25) (5.85 mmol, 1.45 g) and iminodiacetic acid (11.7 mmol, 1.55 g) in 50% aqueous ethanol (80 ml) at pH = 11 (adjusted with NaOH solution) were kept at reflux temperature for 5 h. The ethanol was removed in vacuo and the aqueous phase was extracted with ether (3 × 20 ml). The pH of the aqueous phase was adjusted to 2 with aqueous HCl to precipitate the product. The solid product 5 was washed with water till neutral pH and finally dried in vacuo at 50 °C overnight. Yield: 75%.
1H NMR (300 MHz, d6-DMSO, δ/ppm): 7.87 (t, J = 5.8 Hz, 1H, –NH); 3.42 (s, 4H, –CH2COOH); 3.28 (s, 2H, -NCH2CO); 3.08 (q, J = 6.9 Hz, 2H, –NHCH2CH2); 1.39–1.16 (m, 20H, –NHCH2(CH2)10CH3); 0.85 (t, J = 6.5 Hz, 3H, –CH3). 13C NMR (75 MHz d6-DMSO, δ/ppm): 173.1 (COOH); 171.0 (CONH); 58.9 (N–CH2CO–); 56.3 (N–CH2–COOH); 39.1 (–NHCH2CH2–); 31.9; 29.6; 29.3; 29.3; 27.0; 22.7; 14.5 (–(CH2)10CH3).
Step 3:9 A solution of 5 (0.75 mmol, 0.27 g) in dried DMF (8 ml) was treated with EDCI, (0.79 mmol, 0.15 g) at 25 °C. The mixture was stirred at room temperature under N2 for 1 h before dodecylamine (0.79 mmol, 0.15 g) was added, and the solution was stirred at for 24 h at 40 °C. The reaction mixture was poured into 10% aqueous HCl (50 ml). The filtrate was washed with 10% HCl (50 ml) and with water till neutral pH. The solid was dried in vacuo at 45 °C and recrystallized from hot ethanol. A white solid was obtained (3, 12-ida-12). Yield: 50%.
1H NMR (300 MHz, d6-DMSO, δ/ppm): 8.08 (t, 2H J = 5.8 Hz, –NH–); 3.36 (s, 2H, –CH2COOH); 3.23 (s, 4H, –NCH2CO–); 3.05 (q, 4H, J = 6.9 Hz, –NHCH2CH2–); 1.39–1.24 (m, 40H, NHCH2(CH2)10CH3); 0.85 (t, 6H, J = 6.6 Hz, –CH3). 13C NMR (75 MHz d6-DMSO, δ/ppm): 173.1 (COOH); 170.7 (–CONH–); 58.9 (–COCH2N); 56.4 (–NCH2–COOH); 39.1 (–CH2CH2NH–); 31.9; 29.8; 29.7; 29.6; 29.4; 29.3; 27.0; 22.7; 14.4 (–(CH2)10CH3). FT-IR (![[small nu, Greek, tilde]](https://www.rsc.org/images/entities/char_e0e1.gif) /cm−1): 3256.7, 3334.6 (NH and OH), 2912.9, 2847.5 (CH), 1697.0 (C
/cm−1): 3256.7, 3334.6 (NH and OH), 2912.9, 2847.5 (CH), 1697.0 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, acid), 1663.4 (C
O, acid), 1663.4 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, amide), 1566.8 (NH, amide).
O, amide), 1566.8 (NH, amide).
Mass spectrum (ESI-FIA-TOF): m/z calculated for C30H59N3O4 525.81, found 526.45. Elemental analysis: Calculated for C30H59N3O4: N= 7.99%, C = 68.53%, H = 11.31%; found: N = 8.30%, C = 68.08%, H = 11.40%. M.p. 91.2–92.0 °C.
Equipment
Surface tension measurements were carried out in a KRUSS model K-10-ST tensiometer equipped with a Wilhelmy plate. Temperature was kept constant by recirculating water from a PolyScience 9100 thermostat. The Wilhelmy plate method was used. Solutions were prepared according to the step-by-step dilution-extraction method,61 with a total volume of 10 ml and dilution steps of 3 ml. A complete set of measurements was performed in less than two hours. In order to keep a pH constant value, the dilution solvent was a 0.01 M NaOH aqueous solution. Temperature was kept constant either at 25 °C or 40 °C.Steady-state fluorescence measurements, using pyrene (Sigma) as a probe, were recorded on a Cary Eclipse (excitation wavelength 336 nm, excitation slit 2.5 nm, emission slit 2.5 nm). Solutions containing pyrene were prepared according to the method described by Zana and Guveli.62 Temperature was kept constant at 40 °C.
TEM images were obtained at room temperature in a JEOL JEM-1011, operated at 80 kV, equipped with a MegaView III camera. For sample preparation, a drop of the solution (kept at 40 °C for most experiments) was deposited onto a carbon-coated copper grid and left for a few minutes to allow complete grid permeation. After that, the grid was deposited on filter paper to absorb the drop excess and measured immediately.
A Brookhaven instrument constituted by a BI-2030AT digital correlator with 136 channels and a BI-200SM goniometer was used for dynamic (DLS) light scattering measurements. The light source was a Melles Grio He–Ne laser operating at 632 nm.
In the DLS experiments the intensity–intensity autocorrelation function was measured at a particular q value, and related to the normalized electric field autocorrelation function g1(q,τ) by the Siegert relation. The autocorrelation function was analyzed by a NLLS algorithm (from Brookhaven software). From the so called apparent diffusion coefficient, Dapp, an apparent hydrodynamic radius Rapp can be calculated by the Stokes-Einstein equation, Rapp = kBT/6πηDapp, where kB is the Boltzmann constant, T is the absolute temperature and η is the solvent viscosity.63
Dust was eliminated by filtering the samples through Albet PTFE filters with a pore size of 0.45 μm at room temperature, the volume of the sample being about 5.0 ml. The temperature of the samples was kept constant within 0.5 °C by a circulating water bath. The samples were placed in the cell for at least 30 min prior to measurement to allow for thermal equilibration.
NMR experiments were performed in a Varian Mercury 300 instrument. The solvent used was d6-DMSO.
FT-IR spectrum was performed in an ABB Bomen model MB 102 instrument. Solid powdered samples were placed on a GOLDEN-GATE accessory for solid samples.
Mass spectra were performed in a Bruker Microtof equipment. A Flash EA TCD Thermo Finnigan elemental model 1112 analyzer with thermal conductivity detector was used for elemental analysis of 12-ida-12. The melting points were determined using a Gallenkamp capillary melting point apparatus (model MPD 350 BM 2.5; SANYO Gallenkamp PLC).
OriginPro 7 (OriginLab Corporation) was used for fitting all experimental data to the appropriate equations.
Acknowledgements
The authors thank the Ministerio de Ciencia y Tecnología, Spain (Project MAT2010-19440) for financial support. J. V. Trillo also thanks for a scholarship.Notes and references
- R. Zana, Adv. Colloid Interface Sci., 2002, 97, 205–253 CrossRef CAS.
- M. Álvarez Alcalde, A. Jover, F. Meijide, L. Galantini, N. V. Pavel, Á. Antelo and J. Vázquez Tato, Langmuir, 2009, 25, 9037–9044 CrossRef PubMed.
- F. M. Menger, Proc. Natl. Acad. Sci. U. S. A., 2002, 99, 4819–4822 CrossRef PubMed.
- P. S. Niemelä, M. T. Hyvönen and I. Vattulainen, Biophys. J., 2006, 90, 851–863 CrossRef PubMed.
- T. J. Racey, M. A. Singer, L. Finegold and P. Rochon, Chem. Phys. Lipids, 1989, 49, 271–288 CrossRef CAS.
- Y.-P. Zhu, A. Masuyama, Y. Kirito, M. Okahara and M. J. Rosen, J. Am. Oil Chem. Soc., 1992, 69, 626–632 CrossRef CAS.
- J. Puigmartí-Luis, A. Minoia, Á. Pérez del Pino, G. Ujaque, C. Rovira, A. Lledós, R. Lazzaroni and D. B. Amabilino, Chem. – Eur. J., 2006, 12, 9161–9175 CrossRef PubMed.
- A. J. Van Wyk, P. J. Fourie, P. Bekker, S. Wood, K. Smith, T. Gerber and J. Du Preez, Curr. Appl. Radiopharmacol., Proc. Int. Symp. Radiopharmacol., 4th, 1986, 27–34 CAS.
- S. Cheng, D. D. Comer, J. P. Williams, P. L. Myers and D. L. Boger, J. Am. Chem. Soc., 1996, 118, 2567–2573 CrossRef CAS.
- D. H. Everett, Manual of Symbols and Terminology for Physicochemical Quantities and Units, IUPAC, BUTTERWORTHS, LONDON, 1971 Search PubMed.
- O. Träger, S. Sowade, C. Böttcher and J.-H. Fuhrhop, J. Am. Chem. Soc., 1997, 119, 9120–9124 CrossRef.
- A. Jover, F. Meijide, E. Rodríguez Nuñez and J. Vázquez Tato, Langmuir, 2002, 18, 987–991 CrossRef CAS.
- M. Swanson-Vethamuthu, M. Almgren, P. Hansson and J. Zhao, Langmuir, 1996, 12, 2186–2189 CrossRef CAS.
- M. C. Carey, Hepatology, 1984, 4, 66S CrossRef CAS.
- H. Igimi and M. C. Carey, J. Lipid Res., 1980, 21, 72 CAS.
- R. Klein, M. Kellermeier, D. Touraud, E. Müller and W. Kunz, J. Colloid Interface Sci., 2013, 392, 274–280 CrossRef CAS PubMed.
- D. A. Jaeger, Y. Wang and R. L. Pennington, Langmuir, 2002, 18, 9259–9266 CrossRef CAS.
- W. M. Linfield, J. Am. Oil Chem. Soc., 1978, 55, 87–92 CrossRef CAS.
- R. Bordes, J. Tropsch and K. Holmberg, J. Colloid Interface Sci., 2009, 338, 529–536 CrossRef CAS PubMed.
- K. Mori and S. Matsumura, J. Oleo Sci., 2012, 61, 609–620 CrossRef CAS.
- R. Eising, A. C. Felippe and J. B. Domingos, J. Dispersion Sci. Technol., 2012, 33, 75–82 CrossRef CAS.
- P. S. Sales, R. H. de Rossi and M. A. Fernandez, Chemosphere, 2011, 84, 1700–1707 CrossRef CAS PubMed.
- R. Eising, S. Mores, I. C. Bellettini, A. C. Felippe, A. G. Dal-Bo and D. Zanette, Quim. Nova, 2008, 31, 2065–2070 CrossRef CAS PubMed.
- A. Rodriguez-Pulido, A. Casado, M. Muñoz-Ubeda, E. Junquera and E. Aicart, Langmuir, 2010, 26, 9378–9385 CrossRef CAS PubMed.
- T. Yoshimura and K. Esumi, Langmuir, 2003, 19, 3535–3538 CrossRef CAS.
- T. Yoshimura, N. Kimura, E. Onitsuka, H. Shosenji and K. Esumi, J. Surfactants Deterg., 2004, 7, 67–74 CrossRef CAS.
- R. Zana, H. P. Levy, D. Papoutsi and G. Beinert, Langmuir, 1995, 11, 3694–3698 CrossRef CAS.
- H. Zhu, J. Guo, C. Yang, S. Liu, Y. Cui and X. Zhong, Synth. Commun., 2013, 43, 1161–1167 CrossRef CAS.
- S. Polarz, C. Bährle, S. Landsmann and A. Klaiber, Angew. Chem., Int. Ed., 2013, 52, 1–7 CrossRef PubMed.
- V. H. Soto Tellini, A. Jover, F. Meijide, J. Vázquez Tato, L. Galantini and N. V. Pavel, Adv. Mater., 2007, 19, 1752–1756 CrossRef.
- L. Zhai, J. Zhang, Q. Shi, W. Chen and M. Zhao, J. Colloid Interface Sci., 2005, 284, 698–703 CrossRef CAS PubMed.
- L. Zhai, X. Lu, W. Chen, C. Hu and L. Zheng, Colloids Surf., A, 2004, 236, 1–5 CrossRef CAS PubMed.
- S. Roy and J. Dey, Langmuir, 2005, 21, 10362–10369 CrossRef CAS PubMed.
- E. Brueckner, P. Sonntag and H. Rehage, Langmuir, 2001, 17, 2308–2311 CrossRef CAS.
- A. Jover, F. Meijide, E. Rodríguez Núñez, J. Vázquez Tato, M. Mosquera and F. Rodríguez Prieto, Langmuir, 1996, 12, 1789–1793 CrossRef CAS.
- F. Meijide, J. V. Trillo, S. de Frutos, L. Galantini, N. V. Pavel, V. H. Soto, A. Jover and J. Vázquez Tato, Steroids, 2012, 77, 1205–1211 CrossRef CAS PubMed.
- D. Sahoo, V. Narayanaswami, C. M. Kay and R. O. Ryan, Biochemistry, 2000, 39, 6594–6601 CrossRef CAS PubMed.
- A. J. Prosser and E. I. Franses, Colloids Surf., A, 2001, 178, 1–40 CrossRef CAS.
- F. M. Menger, L. Shi and S. A. A. Rizvi, J. Am. Chem. Soc., 2009, 131, 10380–10381 CrossRef CAS PubMed.
- K. Okuyama, Y. Soboi, N. Iijima, K. Hirabayashi, T. Kunitake and T. Kajiyama, Bull. Chem. Soc. Jpn., 1988, 61, 1485–1490 CrossRef CAS.
- E. L. Prime, D. N. H. Tran, M. Plazzer, D. Sunartio, A. H. M. Leung, G. Yiapanis, S. Baoukina, I. Yarovsky, G. G. Qiao and D. H. Solomon, Colloids Surf., A, 2012, 415, 47–58 CrossRef CAS PubMed.
- C. Tanford, The Hydrophobic Effect: Formation of Micelles and Biological Membranes, Wiley, New York, 1980 Search PubMed.
- R. Nagarajan, Langmuir, 2002, 18, 31–38 CrossRef CAS.
- W. D. Harkins and H. Zollman, J. Am. Chem. Soc., 1926, 48, 69–80 CrossRef CAS.
- N. Kucerka, J. F. Nagle, J. N. Sachs, S. E. Feller, J. Pencer, A. Jackson and J. Katsaras, Biophys. J., 2008, 95, 2356–2367 CrossRef CAS PubMed.
- S. H. White, R. E. Jacobs and G. I. King, Biophys. J., 1987, 52, 663–665 CrossRef CAS.
- N. Kucerka, J. Gallova, D. Uhrikova, P. Balgavy, M. Bulacu, S.-J. Marrink and J. Katsaras, Biophys. J., 2009, 97, 1926–1932 CrossRef CAS PubMed.
- C. Huang and J. T. Mason, Proc. Natl. Acad. Sci. U. S. A., 1978, 75, 308–310 CrossRef CAS.
- M. J. Rosen, J. H. Mathias and L. Davenport, Langmuir, 1999, 15, 7340–7346 CrossRef CAS.
- K. Tsubone, T. Ogawa and K. Mimura, J. Surfactants Deterg., 2003, 6, 39–46 CrossRef CAS PubMed.
- J. Israelachvili, Intermolecular and Surface Forces, Santa Barbara, 3rd edn, 2011 Search PubMed.
- W. Helfrich, Z. Naturforsch. C, 1973, 28, 693–703 CAS.
- N. D. Denkov, H. Yoshimura, T. Kouyama, J. Walz and K. Nagayama, Biophys. J., 1998, 74, 1409–1420 CrossRef CAS.
- H. T. Jung, B. Coldren, J. A. Zasadzinski, D. J. Iampietro and E. W. Kaler, Proc. Natl. Acad. Sci. U. S. A., 2001, 98, 1353–1357 CrossRef CAS.
- E. Evans, V. Heinrich, F. Ludwig and W. Rawicz, Biophys. J., 2003, 85, 2342–2350 CrossRef CAS.
- W. Rawicz, K. C. Olbrich, T. McIntosh, D. Needham and E. Evans, Biophys. J., 2000, 79, 328–339 CrossRef CAS.
- S. Dieluweit, A. Csiszar, W. Rubner, J. Fleischhauer, S. Houben and R. Merkel, Langmuir, 2010, 26, 11041–11049 CrossRef CAS PubMed.
- G. Popescu, T. Ikeda, K. Goda, C. A. Best-Popescu, M. Laposata, S. Manley, R. R. Dasari, K. Badizadegan and M. S. Feld, Phys. Rev. Lett., 2006, 97, 218101 CrossRef.
- B. Coldren, R. Van Zanten, M. J. Mackel, J. A. Zasadzinski and H.-T. Jung, Langmuir, 2003, 19, 5632–5639 CrossRef CAS.
- M. I. Viseu, K. Edwards, C. S. Campos and S. M. B. Costa, Langmuir, 2000, 16, 2105–2114 CrossRef CAS.
- A. Jover, F. Meijide, V. Mosquera and J. Vázquez Tato, J. Chem. Educ., 1990, 67, 530 CrossRef CAS.
- R. Zana and D. Güveli, J. Phys. Chem., 1985, 89, 1687–1690 CrossRef CAS.
- An Introduction to Dynamic Light Scattering by Macromolecules, ed. K. S. Schmitz, Academic Press, Boston, 1990 Search PubMed.
| This journal is © The Royal Society of Chemistry 2014 |