Tuned synthesis of doped rare-earth orthovanadates for enhanced luminescence†
Vairapperumal Tamilmani,
Kalarical Janardhanan Sreeram* and
Balachandran Unni Nair
Chemical Laboratory, CSIR-Central Leather Research Institute, Adyar, Chennai, India 600 020. E-mail: kjsreeram@clri.res.in; Fax: +91 44 2491 1589; Tel: +91 44 2441 1630
First published on 5th November 2013
Abstract
Lanthanide orthovanadates of appropriate morphology, polymorph character and size are attractive for various electronic and magnetic applications. Though template free methods exist, there is a preference to facile template based methods for fine tuning of shape and size. The novelty of the methodology reported herein is the use of a biologically acceptable chelate such as catechin (5,7,3′,4′-tetrahydroxyflavan-3-ol for a selective synthesis of tetragonal LaVO4 over the preferred monoclinic phase) in a short duration of time. Optimal conditions ([cat4−]/[La3+] = 1/20; T = 210 °C for 4 h and pH 7) for the synthesis of t-LaVO4 (JCPDS no. 10-705226) with a crystal size of around 16 nm have been established. Our experiments on the role of pH in the formative steps of the complex indicated that catechin serves not only as a polymorph controller but it also aids in shape selection. Under optimal synthesis conditions, a switch over from isotropic to anisotropic structure was brought about by a pH change from 5 to 7. The process methodology has been tuned to ensure that the drawbacks associated with template based methods, viz., the presence of template impurities, have been significantly overcome. From CHN analysis and FTIR measurements, less than 2% carbon from catechin is found to coat the nanoparticle surfaces, which contributed to prevention of aggregation. Extending the work further, Gd3+ doped t-LaVO4 was also synthesized to obtain luminescent and magnetic properties (fluorescence lifetime of 0.2 ms and magnetization of 0.79 emu g−1 at RT), which are possibly ideal for imaging applications, such as for T1 contrast agents for MRI.
Introduction
Unmatched features of 4f intra-shell electrons enable the transformation of rare earth materials into components for electronic and optical devices. Rare earth orthovanadates are reported amongst materials with unusual magnetic, luminescent, electronic and thermal properties, leading to technological advancements in optical devices, laser hosts, catalysts, polarizers etc.1–4 Inherent crystallographic structures of materials play crucial roles in both physical and chemical properties, thus making the phase control of nanoparticles quite important. Lanthanide orthovanadates crystallize in two polymorphs, viz., m- (monoclinic) monazite type and t- (tetragonal) zircon type.5 A wealth of information that exists on the structure and properties of the orthovanadates indicate that with increasing ionic radius, Ln3+ ions show a stronger tendency towards monazite-structured orthovanadate as the thermodynamically stable state.1,6 However, m-LaVO4 is neither a suitable host for luminescent activators7,8 nor a promising catalyst9 because of its ordinary monoclinic structure.Synthesis of t-LaVO4, which is expected to be a promising phosphor host candidate, is a challenge as it is metastable and cannot be obtained by conventional solid-state reactions. Among the enumerated methods, hydrothermal synthesis using water as the reaction medium is highly desirable because it allows to obtain particles with high crystallinity, narrow size distribution and high purity under mild reaction conditions without milling or further calcination treatment.10 Hydrothermal synthesis also represents an environmentally benign and user-friendly approach, which may be considered to be a relatively green chemical alternative of practical significance.11–13 Fan et al., through appropriate dissolution-recrystallization transformation and Ostwald ripening, reported phase and anisotropy control in LaVO4.14,15 The use of templates or organic additives in bringing about phase transformations in LaVO4 has also been in vogue for long, though drawbacks such as the presence of additive impurities in the final product; complicated processes etc., have been reported. Yet another recent development in this area is the use of dopants to induce phase transformation.16
In order to overcome the known and reported drawbacks of the additive based systems and also to achieve newer advantages from their use, we have for the first time explored the use of catechin (5,7,3′,4′-tetrahydroxyflavan-3-ol) as a polymorph selector. Further, doping along with process variable modifications has been carried out to transform the synthesized t-LaVO4, from a material having optical fluorescence imaging applications to a contrast agent for magnetic resonance imaging (MRI).17,18
Catechin belongs to the group of flavonoids with 5 hydroxyl groups and is an excellent chelator of metal ions. It is abundant in the human diet with possible beneficial health effects such as anti-carcinogenic, anti-atherosclerotic, anti-microbial, anti-oxidant, anti-bacterial and anti-viral activities.19–23 Catechin has two different pharmacophores, the catechol moiety in ring B, and the resorcinol group in the ring A, along with the OH group at position 3 in ring C.24 The presence of a large number of hydroxyl groups in the makeup of phenolic compounds is important for binding/chelation.25–27
Results and discussion
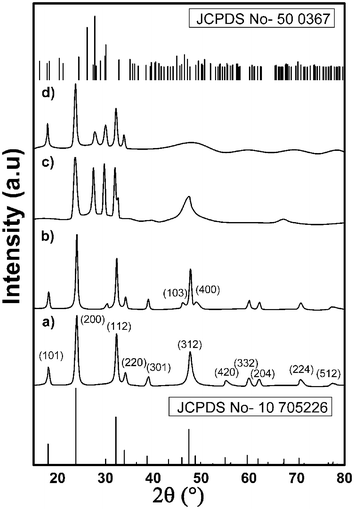
In this work, the use of catechin hydrate for polymorph selection along with size and shape control during the synthesis of t-LaVO4 is reported. The phase purity and structure of the products synthesized under various conditions were confirmed from the XRD patterns. In the absence of a template and employing a temperature of 210 °C for 4 h, the direct hydrothermal treatment of La(NO3)3·6H2O and Na3VO4 resulted in a product (Fig. 1a), whose diffraction peaks were well indexed to m-LaVO4 [JCPDS no. 50-0367; space group (p21/n) ] and cell parameters a = 7.043 Å; b = 7.279 Å; c = 6.721 Å with no trace of other phases. TEM images of the as-obtained m-LaVO4 nanoparticles exhibited a near spherical morphology with a diameter ranging from 20–30 nm as shown in ESI Fig. S1a.†When the crystallinity is low (as seen from the SAED pattern in the inset of ESI Fig. S1a†) and the overall growth rate is slow due to dissolution–reprecipitation process, a sphere shape is favoured, in accordance with the reported literature.28 The HRTEM image (ESI Fig. S1b†) displays an interplanar distance of 0.2857 nm ascribed to the lattice spacing of the (012) crystal plane of m-LaVO4. The crystalline domain calculated by the Scherrer formula from the XRD pattern of (012) planes was 15 ± 2 nm.
In order to understand the role of doping and to synthesize a multifunctional product, Gd3+ doped orthovanadate was also synthesized. ESI Fig. S2† presents the XRD patterns of lanthanum vanadate after doping with Gd3+. The XRD pattern is characteristic of an efficient and homogeneous incorporation of the Gd3+ ions as substituents for the La3+ ions inside the LaVO4 host matrix, similar to earlier reports.29 A further confirmation of uniform doping was obtained from the unit cell volume 304 Å3, which corresponded to 5 mol% doping level of the smaller Gd3+ ion compared to that of La3+ in LaVO4 host matrix with cell volume of 327 Å3 [ionic radius of La3+ = 1.300 Å and Gd3+ = 1.193 Å]. According to Stouwdam et al., a decrease in the unit cell volume is an indication of homogeneously distributed dopant in the host nanoparticles.30,31 The TEM image of Gd3+ doped LaVO4 nanoparticles (ESI Fig. S3†) indicated a near spherical geometry with size in the range of 80–120 nm.
As per the reaction chemistry employed in this study, zircon-type LaVO4 could not be obtained through direct hydrothermal treatment of the mixture of La(NO3)3·6H2O and Na3VO4 solution.
Based on the fact that additives like EDTA, acetic acid or citric acid when introduced into the reaction system mediated the growth of tetragonal LaVO4,32 we explored the possibility of using catechin hydrate to modulate the phase of the LaVO4 products. The choice of catechin hydrate was led by its biocompatibility and previous knowledge on its medicinal applications. XRD patterns of the samples with Na3VO4 as vanadium source and molar ratio of cat4−![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) La3+ varying from 0, 1/40, 1/20, 1/13 and 1/10 are shown in Fig. 1a–e. A pH and catechin concentration dependent change from monoclinic to tetragonal phase was observed. When cat4− was added without adjusting the pH of the solution (pH 9), peaks corresponding to t-LaVO4 appeared alongside other phases. At pH 9 and with increasing amount of catechin hydrate, the formation of the monoclinic phase was restrained.
La3+ varying from 0, 1/40, 1/20, 1/13 and 1/10 are shown in Fig. 1a–e. A pH and catechin concentration dependent change from monoclinic to tetragonal phase was observed. When cat4− was added without adjusting the pH of the solution (pH 9), peaks corresponding to t-LaVO4 appeared alongside other phases. At pH 9 and with increasing amount of catechin hydrate, the formation of the monoclinic phase was restrained.
It is evident from the XRD patterns that three major peaks at 26°, 27° and 30° corresponding to monoclinic phase were altered when changing the lanthanum to catechin ratio. At [cat4−]/[La3+] = 1/20, (200) and (112) peaks corresponding to tetragonal phase are predominant alongside traces of the monoclinic phase. When [cat4−] exceeds above 1/20 to that [La3+], the (200) peak of the synthesized product remained almost constant but the (112) peak decreased to a small extent. This indicates that high concentration of [cat4−] decelerated the tetragonal phase formation, allowing the highly crystallized m-LaVO4 to form. Below a molar ratio of (1/20), a large proportion of peaks of the synthesized product correlated well with the monoclinic phase while a small proportion correlating to t-LaVO4 was formed. From the above observation, it can be deduced that conversion of m-LaVO4 to t-LaVO4, can be achieved at [cat4−]/[La3+] = 1/20. We presume that at lower [cat4−]/[La3+] ratio, the weak co-ordination between La3+ and OH ligands in cat4− is not sufficient for the formation of the tetragonal phase. On the contrary, under higher [cat4−]/[La3+] ratios (i.e. [cat4−]/[La3+] > 1/20), the co-ordination ability of catechin to La3+ is altered due to the heavy competition of the higher number of OH ligands in cat4−.
The above study indicates that a complete elimination of the monoclinic phase was not possible under the reaction conditions employed. Besides concentration of catechin hydrate, other factors such as reaction time, temperature and pH can also direct the formation of t-LaVO4, not only in terms of its polymorph character but also in terms of its shape and size.
Temperature
Diffraction peaks of synthesized LaVO4 (ESI Fig. S4†) could not be indexed to any particular phase of LaVO4, for reaction temperatures below 180 °C. When the temperature reached 180 °C, the formation of t-LaVO4 was observed. At 210 °C, the XRD pattern of the synthesized product could be well indexed to standard tetragonal phase with monoclinic phase as impurity. At temperatures above 210 °C, t-LaVO4 formation was retarded.Duration of treatment
In the presence of catechin hydrate ([cat4−]/[La3+] = 1/20), even at room temperature, the formation of the monoclinic phase was decelerated and traces of other phases appeared (ESI Fig. S5†). The hydrothermal treatment resulted in gradual reduction in the formation of m-LaVO4, with t-LaVO4 becoming dominant. For a treatment duration of 4 h, pure t-LaVO4 was obtained. Treatment durations above 4 h, increased the monoclinic phase to a larger proportion of that of the tetragonal phase.pH
Hydrogen ion concentration of the precursor solution was found to be a crucial factor in structural transformation of LaVO4 nanoparticles. The main effect of tuning the pH value is to modulate the thermodynamics/kinetics of nucleation and growth of the nanoparticle by controlling experimentally the surface free energy.14 Under fixed conditions of [cat4−]/[La3+] = 1/20 at 210 °C for 4 h, the pH value of the final solution was varied, in order to investigate its effect on the phase transformation. The formation of pure t-LaVO4 is favoured only in the pH range of 5 to 7. Fig. 2a provides the XRD pattern of the product obtained at pH = 5 and peaks corresponding to the tetragonal phase were not completely formed under these conditions.TEM micrograph (Fig. 3a) and the corresponding SAED pattern (inset) of the nanoparticles synthesized at pH 5 indicate spherical geometry with crystalline character and a size of ± 25 nm. HRTEM image (Fig. 4a) provides for an interplanar spacing of 0.3704 nm attributed to the (200) plane of t-LaVO4. With a further increase of pH to 7, all diffraction peaks exactly correlated to the standard tetragonal phase with space group (I41/amd (141)) and cell parameters a = b = 7.4578 Å; c = 6.5417 Å. As the pH of synthesis was increased to 7 (Fig. 3b), the morphology turned to platelet shapes, with size around 16 ± 2 nm. HRTEM (Fig. 4b) also confirmed the presence of (200) plane of t-LaVO4. This type of structure is composed of a chain of alternating edge-sharing VO4 tetrahedra and LaO8 triangular dodecahedra parallel to the C axis, which is joined laterally by edge sharing LaO8 dodecahedra (the point symmetry of La3+ is D2d). As the a plane is the most close packed plane, the (200) plane is an energetically favoured plane for LaVO4.33 Beyond this pH, the intensity of correlative XRD peaks of tetragonal phase became weaker and traces of monoclinic peaks are observed.
 | ||
| Fig. 3 TEM image along with SAED pattern (inset) for LaVO4 nanoparticles synthesized at (a) pH 5.0; (b) pH 7.0 (experimental conditions: [cat4−]/[La3+] = 1/20, T = 210 °C, t = 4 h). | ||
 | ||
| Fig. 4 HRTEM image for LaVO4 nanoparticles synthesized at (a) pH 5.0; (b) pH 7.0 (experimental conditions: [cat4−]/[La3+] = 1/20, T = 210 °C, t = 4 h). | ||
The lattice constant calculated was a = b = 7.388 Å; c = 6.476 Å, which closely matched their corresponding standard data, confirming the crystallinity of the tetragonal phase.34 The XRD pattern agreed well with the EDAX data (ESI Fig. S6†). Strong peaks of La3+, V, O, C and a weak peak of Gd3+, indicate the existence of LaVO4:Gd3+. The obtained atomic ratio of La3+![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Gd3+ = 0.92
Gd3+ = 0.92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.06 approached the theoretical value for 5 mol% Gd3+ doped LaVO4. In our system, we presume that the transformation from m- to t-LaVO4 nanoparticle is effective only at pH 7, as evidenced by XRD. Similar to the case of pH < 7, the obtained LaVO4 (at pH 9, 13) also inherited spherical morphology (∼10 nm) as shown in ESI Fig. S7.† SAED pattern, showed a series of clear diffraction rings demonstrating that the nanoparticles synthesized at pH 9 and 13 are polycrystalline in nature. As the pH was increased to 9, an interplanar distance of 0.2222 nm, consistent with the (301) crystal plane of t-LaVO4 was observed (ESI Fig. S7†). However, a further increase in pH to 13 resulted in a mixture of t- and m-LaVO4 as can be seen from the interplanar distances of 0.3704 and 0.3333 nm which can be attributed to the (200) plane of the two forms respectively (ESI Fig. S7†).
0.06 approached the theoretical value for 5 mol% Gd3+ doped LaVO4. In our system, we presume that the transformation from m- to t-LaVO4 nanoparticle is effective only at pH 7, as evidenced by XRD. Similar to the case of pH < 7, the obtained LaVO4 (at pH 9, 13) also inherited spherical morphology (∼10 nm) as shown in ESI Fig. S7.† SAED pattern, showed a series of clear diffraction rings demonstrating that the nanoparticles synthesized at pH 9 and 13 are polycrystalline in nature. As the pH was increased to 9, an interplanar distance of 0.2222 nm, consistent with the (301) crystal plane of t-LaVO4 was observed (ESI Fig. S7†). However, a further increase in pH to 13 resulted in a mixture of t- and m-LaVO4 as can be seen from the interplanar distances of 0.3704 and 0.3333 nm which can be attributed to the (200) plane of the two forms respectively (ESI Fig. S7†).
Gd3+ doped LaVO4 was synthesised using catechin hydrate at optimised hydrothermal treatment to understand the role of doping on morphology at different pH conditions. TEM images of Gd3+ doped LaVO4 nanoparticles, synthesized at pH 5 and 7 are presented in Fig. 5. A change in pH from 5 to 7 indicates a shift from spherical to spindle-like morphology, thereby indicating that the role of cat4− is not limited to only phase regulation but also to a shape and size controller, more specifically under the influence of experimental conditions such as pH and temperature. A further increase in pH to 9 resulted in anisotropic structures (ESI Fig. S8†).
 | ||
| Fig. 5 TEM image Gd3+ doped LaVO4 nanoparticles synthesized at (a) pH 5.0; (b) pH 7.0 (experimental conditions: [cat4−]/[La3+] = 1/20, T = 210 °C, t = 4 h). | ||
DLS measurements carried out on samples synthesized in the presence and absence of catechin hydrate indicated an increase in the polydispersity index (0.170 → 0.668) and hydrodynamic diameter (177 nm → 566 nm). Such a change could be attributed to the presence of a varied morphology and size of particles, when LaVO4 was synthesized in the absence of catechin hydrate (ESI Fig. S9†). It is quite interesting to note that only at pH 7 the pure tetragonal phase was formed, which is distinct from other organic additives. Thus cat4− serve as a selective sacrificial template as evidenced by FT-IR spectra (ESI Fig. S10†).
A strong band at 794 cm−1 for m-LaVO4 and 802 cm−1 for t-LaVO4, corresponding to the characteristic peak of V–O (from the VO43− group) has been observed. The red shift is justified by the phase transition from monoclinic to tetragonal which is in good agreement with the XRD results.
The doublet peak around 1417 cm−1 and 1628 cm−1 are related to the localized vibration of the VO4 group. The absorption band around 3400 cm−1 can be ascribed to the O–H stretching and bending vibration of water. As the samples were prepared as aqueous solutions, the surface of the particles will be covered inevitably with the absorbed water molecules. FT-IR also has been used to confirm the absence of organic ligands on the nanoparticle surface. In our system, cat4− ligand plays an important role in the selective synthesis of LaVO4. Peaks corresponding to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, CH2 group around 1000–1500 cm−1 in the catechin molecule were not observed in tetragonal phase, confirming that the unbound catechin hydrate has been predominantly removed by dialysis. On the basis of the above analysis, cat4− can be considered as only assisting the selective formation of the crystallographic phase rather than the expected passivation of the polyphenol on the nanoparticle surface.
C, CH2 group around 1000–1500 cm−1 in the catechin molecule were not observed in tetragonal phase, confirming that the unbound catechin hydrate has been predominantly removed by dialysis. On the basis of the above analysis, cat4− can be considered as only assisting the selective formation of the crystallographic phase rather than the expected passivation of the polyphenol on the nanoparticle surface.
It is well-known that formation of a stable colloidal solution can be attributed to equilibrium built between van der Waals’ force and the steric repulsion from the capping ligand.35,36 Zeta potential values reveal details about the surface charge of the synthesized metal nanoparticles.37 It should be noted that the solution of t- and m-LaVO4 nanoparticles showed a zeta potential of −20.1 ± 0.05 mV and 1.91 ± 0.1 mV respectively when measured immediately after preparation. This ultimately explains that there must be a significant change of functional groups on the t-LaVO4 nanoparticle surface. The negative zeta potential can be attributed to the unbound oxygen groups of catechin moiety left on the surface of tetragonal LaVO4 nanoparticles. Zeta potential analysis indicates that the point of zero charge (PZC) is at pH = 3.0 for t-LaVO4, while the m-LaVO4 phase has the same at pH = 3.28 (ESI Fig. S11†). This confirms that the structural difference had a small effect on the zeta potential. While FTIR results do not indicate any presence of catechin on the surface of the nanoparticles, an additive induced change in the aggregation behaviour has been observed from zeta potential measurements. In order to understand the presence/absence of catechin on the nanoparticle surface, a CHN analysis was performed on the LaVO4 synthesized at pH 5 and 9. The results indicated a very small percentage of carbon (1–2%) and minor amounts of hydrogen (∼0.3%) in the samples. The presence of such minor quantities of carbonaceous material should be considered as the cause for the observed change in zeta potential of the samples.
A plausible formation mechanism is suggested based on the above experimental results which show that the m-LaVO4 can be obtained without catechin hydrate. On the other hand, catechin hydrate plays an important role in the formation of zircon-type LaVO4. Catechin hydrate acts as a chelating ligand through the interaction between La3+ and OH groups. While the –OH groups in -3′, -4′ in ring B and 5, 7 in ring A are involved in metal chelation due to their electron delocalization, absence of conjugation and steric hindrance resists the interaction of metal ion to hydroxyl groups in 3 position,25 as represented in Scheme 1.
According to the LaMer-mechanism, the reaction can be divided into three phases: (a) the concentration of La3+ increases gradually and eventually exceeds solubility, (b) the concentration of La3+ reaches critical limit of super saturation and rapid nucleation occurs and nucleation burst results in a sudden decrease of La3+ concentration, finally, (c) nuclei grow slowly as the reaction solution depletes in reactants. Since growth is usually thermodynamically favoured over nucleation, nanoparticles can be grown in a monodisperse manner when the second phase can be limited to a short period of time by suitable choice of reactant concentration and temperature. Initially, cat4− reacts with La3+ ions to form the weakly coordinated [La3+⋯cat4−] complex (CP-1), thereby decreasing the free La3+ ions in solution. In the next step, [La3+⋯cat4−] is attacked by VO43−, forming [VO43−⋯La3+⋯cat4−] complex (CP-2), as depicted in Scheme 2. Under hydrothermal conditions, cat4− is readily cleaved from CP-2, leaving t-LaVO4. Our studies indicate that catechin hydrate occupies one of these sites, resulting in the formation of the tetragonal phase, a condition similar to that of EDTA.38 For the conversion of m-LaVO4 to t-LaVO4, a critical molar ratio of ([cat4−]/[La3+] = 1/20) should be satisfied at the initial stage. Crystallographic phase transformation in solution usually operates through a dissolution–recrystallization process to minimize the surface energy of the system.14 Our XRD results shows that hydrothermal temperature (210 °C for 4 h) is beneficial for the transformation from the m- to t-LaVO4, by speeding up the dissolution–recrystallization process. This process requires a careful adjustment of the pH value to the range of 5–7, to ensure tetragonal phase formation with no secondary phase. Furthermore pure t-LaVO4 is favoured only within the pH range of 5 to 7. The higher the pH values of the solution, an increase in the oxidation state of catechin hydrate is observed.39 In basic aqueous media for pH > 7, the catechol moiety in the catechin hydrate is oxidized to the quinonoid form,39 thereby retarding the formation of t-LaVO4. In this case, a mixture of m- and t-LaVO4 is obtained. In acidic and neutral aqueous media, the co-ordination ability of La3+ to OH ligands in cat4− is strengthened by the Hard Soft Acid Base (HSAB) theory.
Photoluminescence enhancement with phase tailoring
In our experiments, at a [cat4−]![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [La3+] of 1/20 the formed t-LaVO4 is expected to possess superior properties and is expected to be a promising phosphor host candidate. m- and t-LaVO4:Gd3+ (5%) was obtained by hydrothermal method with cat4− as chelating ligand. Changes to the luminescent properties with varying structure of nanoparticles was monitored in the UV-Vis region of the spectra. Fig. 6 shows the absorption and emission spectra of aqueous colloid Gd3+ doped LaVO4, excited at 267 nm. The absorption spectra (Fig. 6a) of LaVO4:Gd3+ (5%) consists of a strong absorption band with maxima at 267 nm. The strong absorption is due to charge transfer from the oxygen ligands to the central vanadium metal inside the VO43− groups, while no lines in the longer wavelength region were observed. From the view point of molecular orbital theory, this corresponds to transitions from the 1A2 (1T1) ground state to 1A1 (1E) and 1E (1T2) excited states of the VO43− ion,40 which in turn indicates that the excitation of Gd3+ ions is mainly through the VO43− groups. A small shift of the Charge Transfer Band (CTB) could be observed for the t-LaVO4:Gd3+ and the variation is proportional to the V–O distance. The longer the V–O bond, the longer is the wavelength of the CTB. Hence, the small red shift observed for t-LaVO4:Gd3+ indicates that the average V–O bond is somewhat longer in t-LaVO4:Gd3+ sample than their monoclinic ones.41 Absorption around 220 nm observed in the case of t-LaVO4 synthesised using catechin template (Fig. 6a) originates from the A ring of catechin moiety present as an encapsulant on the t-LaVO4 nanoparticle.24
[La3+] of 1/20 the formed t-LaVO4 is expected to possess superior properties and is expected to be a promising phosphor host candidate. m- and t-LaVO4:Gd3+ (5%) was obtained by hydrothermal method with cat4− as chelating ligand. Changes to the luminescent properties with varying structure of nanoparticles was monitored in the UV-Vis region of the spectra. Fig. 6 shows the absorption and emission spectra of aqueous colloid Gd3+ doped LaVO4, excited at 267 nm. The absorption spectra (Fig. 6a) of LaVO4:Gd3+ (5%) consists of a strong absorption band with maxima at 267 nm. The strong absorption is due to charge transfer from the oxygen ligands to the central vanadium metal inside the VO43− groups, while no lines in the longer wavelength region were observed. From the view point of molecular orbital theory, this corresponds to transitions from the 1A2 (1T1) ground state to 1A1 (1E) and 1E (1T2) excited states of the VO43− ion,40 which in turn indicates that the excitation of Gd3+ ions is mainly through the VO43− groups. A small shift of the Charge Transfer Band (CTB) could be observed for the t-LaVO4:Gd3+ and the variation is proportional to the V–O distance. The longer the V–O bond, the longer is the wavelength of the CTB. Hence, the small red shift observed for t-LaVO4:Gd3+ indicates that the average V–O bond is somewhat longer in t-LaVO4:Gd3+ sample than their monoclinic ones.41 Absorption around 220 nm observed in the case of t-LaVO4 synthesised using catechin template (Fig. 6a) originates from the A ring of catechin moiety present as an encapsulant on the t-LaVO4 nanoparticle.24
The emission spectrum (Fig. 6b) of Gd3+ doped LaVO4 under excitation of 267 nm at room temperature exhibits a weak emission line of Gd3+ at 334 nm (6P → 8S)42 and three major groups of emission lines between 360 and 520 nm which can be attributed to the transitions of VO43− groups.14 However, an obvious emission from the vanadate group can also be detected, attributed to the fact that the energy transfer from the vanadate group to Gd3+ is not complete.43 It is seen that both monoclinic and tetragonal phases showed similar emission lines but with different intensities. The differences in degree of emission enhancement exist because of the host lattice environment. The whole excitation and emission process of LaVO4:Gd3+ under UV excitation include three major steps (i) absorption of UV light by VO43− groups (ii) transfer of the excited energy to Gd3+ ions after a thermally activated energy migration through the vanadate sub lattice and (iii) de-excitation of excited Gd3+ ions, producing strong blue emissions. A LaVO4 concentration dependant spectral profile was observed (ESI Fig. S12†). Contrary to the reports available,44 when broad emission peaks are observed for VO43−, our studies indicated that in the concentration range investigated (absorbance >4), the VO43− peaks observed around 360 nm to 520 nm were sharp. This change could be attributed to the molecular level clusters formed in the presence of catechin and/or dopant, similar to other reports.45 In order to confirm that the spectral features observed are not from the catechin encapsulant, the excitation spectra were also monitored. From ESI Fig. S12,† it is clear that the emission occurs only from the VO4 groups. The enhanced luminescence from Gd3+ doped t-LaVO4 as against the m-LaVO4 was confirmed from the relative quantum yield measurements, wherein the tetragonal phase exhibited twice the quantum yield of the monoclinic phase.
The kinetic curves in Fig. 7 are representative for the emission of Gd3+ (334 nm) and were taken to exhibit the decay property of LaVO4 host lattices.
 | ||
| Fig. 7 The luminescence decay curves for Gd3+ doped LaVO4 nanoparticles at 334 nm (experimental conditions: [cat4−]/[La3+] = 1/20, T = 210 °C, t = 4 h, pH = 7). | ||
The decay curves can be well fitted into a single exponential function as I = I0e−(t/τ), in which τ is the decay lifetime. The lifetime of Gd3+ in LaVO4 host lattices was determined to be 0.2 ms. It should be mentioned that the lifetime values of Gd3+ in LaVO4 nanoparticles in the millisecond range indicates that there might be an energy transfer between excited states. Owing to the long-time of the excited states, the excited lanthanide ion may sequentially absorb a second photon of suitable energy at comparatively low excitation densities and reach an ever-higher excited state,46 indicating its suitability as luminescent material as well, possibly in the presence of other dopants. It should be pointed out that one of the advantages of rare-earth luminescent nanoparticles as biological labels is their long fluorescence lifetime and stable photoluminescence features.
Magnetism
The magnetization of m-LaVO4:Gd3+ prepared by a co-precipitation method in the absence of cat4− was 0.60675 emu g−1 at RT, which decreased to 0.79 emu g−1 at RT for t-LaVO4:Gd3+ (ESI Fig. S13†). cat4− co-ordinates to La through hydrogen bonds, acting as a stabilizer to prevent the aggregation of the nanoparticles. Smaller and monodisperse LaVO4:Gd3+ obtained leads to the reduction of magnetization. In Fig. 8, magnetization recorded for t-LaVO4:Gd3+ at 25 K and 100 K did not exhibit hysteresis but displayed a unique sign of paramagnetic behavior. The saturation magnetization (Ms) of t-LaVO4:Gd3+ at 25 K (1.3216 emu g−1) was greater than the Ms at 100 K (0.37581 emu g−1). The paramagnetic property of Gd3+ ions arises from seven unpaired inner 4f electrons, which are closely bound to the nucleus and effectively shielded by the outer closed shell electrons 5s25p6 from the crystal field. The magnetic moment associated with the Gd3+ ions are all localized and non-interacting, giving rise to paramagnetic behaviour. As an added advantage, the use of paramagnetic Gd3+ dopant ions provides the nanoparticles with a second imaging capability, such as T1 MRI contrast agent.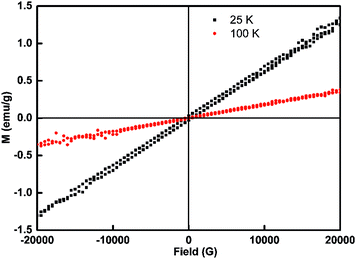 | ||
| Fig. 8 Magnetization curves of t-LaVO4:Gd3+ nanoparticles at 25 K and 100 K (experimental conditions: [cat4−]/[La3+] = 1/20, T = 210 °C, t = 4 h, pH = 7). | ||
Experimental section
Materials and methods
Catechin hydrate, lanthanum(III) nitrate hexahydrate (puriss. p.a., ≥99.0%), gadolinium(III) nitrate hexahydrate (99.9% trace metals basis), sodium orthovanadate (99.98% trace metals basis) were purchased from Sigma-Aldrich, USA and used without further purification. Sodium hydroxide and nitric acid were received from Rankem, India. Ethanol was purchased from Hayman speciality products.Synthesis of LaVO4 nanoparticles
In a typical procedure, 1.54 mmol of catechin hydrate was dissolved in 10 mL Millipore water, to which 0.0031 mol of La(NO3)3·6H2O aqueous solution was mixed under vigorous stirring for 20 min. To the well-stirred solution, 0.0032 mol of Na3VO4 solution was added, resulting in a milky precipitate and the final volume of the mixture was kept as 80 mL. The pH of the resulting mixture was adjusted to 5, 7, 9 and 13 by the addition of 1 M HNO3 or NaOH solution. After stirring for 30 min, the un-coordinated catechin hydrate was removed by dialyzing the mixture against double distilled water using dialysis tubing cellulose membrane (avg. flat width 33 mm, 1.1 inch) for one day. A Teflon vessel containing the dialyzed suspension was loosely capped and autoclaved at 210 °C for 4 h. After the autoclave was cooled to room temperature naturally, the precipitate was separated by centrifugation, washed thrice with Millipore water and absolute ethanol, and then vacuum-dried at 80 °C.Synthesis of Gd3+ LaVO4 nanoparticles
Gd3+ ion-doped LaVO4 nanoparticles was prepared by introducing an appropriate amount of Gd(NO3)3·6H2O to the precursor mixture as described above. The same hydrothermal treatment was conducted. Pure t-LaVO4:Gd3+(5%) nanoparticles were prepared from optimised conditions.Phase purity and crystal structure of the obtained samples were examined by X-ray diffraction (XRD) using a Rigaku Miniflex II desktop X-ray diffractometer equipped with Cu Kα radiation (λ = 1.540562 Å). A scan rate of 4° min−1 in the 2θ range from 10–80° was employed. The operation voltage and current were maintained at 30 kV and 15 mA, respectively. Morphology and size of the as-synthesized doped LaVO4 nanoparticles was characterized by Tecnai 10 Philips TEM at an accelerating voltage of 200 kV. HRTEM and electron diffraction patterns of nanoparticles were obtained from on a TECHNAI G2 transmission electron microscope operating at an accelerating voltage of 250 kV. The samples for these measurements were dispersed in Millipore water under sonication in a ultrasonic pool for 10 minutes. Ultraviolet-Visible (UV-Vis) absorption spectra of the colloidal solutions of the same concentration were measured in a Shimadzu UV-1800 spectrophotometer. Fluorescence spectra were recorded on a Cary Eclipse (Varian Inc.) using Xe flash lamp at room temperature. The luminescence decay curves were obtained from Laser Flash Photolysis Spectrometer (Spectra-Physics LAB-150) excited at 266 nm. Fluorescence quantum yields were obtained by using proflavine in water (pH = 7) as a standard (0.34). FT-IR spectra were obtained using ABB MB3000 Fourier-transform infrared spectrophotometer using the KBr pellet technique. Energy-dispersive X-ray spectroscopy (EDAX) measurements were carried out using a High-resolution scanning electron microscope (FEI QUANTA FEG 200). Magnetic properties were studied by vibrating sample magnetometer (VSM, Lakeshore model 7410) with fields up to 2 T at room temperature, 25 K and 100 K. Zeta potential and particle size measurements were carried out using Zetasizer Nano ZS (Malvern instruments) by Dynamic Light Scattering (DLS) through Non-Invasive Back Scattering (NIBS) method. Elemental analysis (determination of C, H) was performed by means of CHNS analyzer (Vario microcube, M/S Elementer).
Conclusions
In this paper the use of catechin hydrate, as a chelating agent for the phase selective synthesis of LaVO4 has been demonstrated for the first time. The process has been found to occur in three steps, namely the formation of [La3+⋯cat4−] complex, followed by the attack of vanadate resulting in [VO43−⋯La3+⋯cat4−] complex, and then the elimination of catechin hydrate. By this process, cat4− serves as a sacrificial ligand, with little or no influence on the final product as confirmed by FT-IR and CHN analysis. Temperature, time, pH, molar ratio of cat4− to La3+ were found critical and the optimal condition were found to be [cat4−]/[La3+] = 1/20 at 210 °C for 4 h with a pH 7. Catechin hydrate also plays a major role in size as well as shape selection, in association with experimental variables such as pH and temperature. The synthesized t-LaVO4 when doped with Gd3+ has been demonstrated to be paramagnetic and fluorescence intensity of product was much higher to that of m-LaVO4. Based on the longer lifetime of the excited state as well as the paramagnetic character, applications of these nanoparticles in areas such as luminescent materials and T1 MRI contrast agent for imaging purposes is promising.Acknowledgements
The authors acknowledge the funding support under the Indo-Mexico (DST-CONACYT) joint collaboration program between CSIR-CLRI and UANL Mexico. VT thanks the DST-Inspire, Govt. of India for the INSPIRE fellowship. CSIR-CLRI Communication number 1017.Notes and references
- B. C. Chakoumakos, M. M. Abraham and L. A. Boatner, J. Solid State Chem., 1994, 109, 197–202 CrossRef CAS.
- L. Sun, Y. Zhang, J. Zhang, C. Yan, C. Liao and Y. Lu, Solid State Commun., 2002, 124, 35–38 CrossRef CAS.
- J. R. O'Connor, Appl. Phys. Lett., 1966, 9, 407–409 CrossRef CAS PubMed.
- A. Rapaport, O. Moteau, M. Bass, L. A. Boatner and C. Deka, J. Opt. Soc. Am. B, 1999, 16, 911–916 CrossRef CAS.
- Z. Xu, C. Li, Z. Hou, C. Peng and J. Lin, CrystEngComm, 2011, 13, 474–482 RSC.
- Y. Oka, T. Yao and N. Yamamoto, J. Solid State Chem., 2000, 152, 486–491 CrossRef CAS.
- F. C. Palilla, A. K. Levine and M. Rinkevics, J. Electrochem. Soc., 1965, 112, 776–779 CrossRef CAS PubMed.
- U. Rambabu, D. P. Amalnerkar and B. B. K. Buddhudu, Mater. Res. Bull., 2000, 35, 929–936 CrossRef CAS.
- C.-J. Jia, L.-D. Sun, F. Luo, X.-C. Jiang, L.-H. Wei and C.-H. Yan, Appl. Phys. Lett., 2004, 84, 5305–5307 CrossRef CAS PubMed.
- M. Yoshimura and K. Byrappa, J. Mater. Sci., 2007, 43, 2085–2103 CrossRef PubMed.
- Y. Mao and S. S. Wong, J. Am. Chem. Soc., 2006, 128, 8217–8226 CrossRef CAS PubMed.
- R.-Q. Song, A.-W. Xu and S.-H. Yu, J. Am. Chem. Soc., 2007, 129, 4152–4153 CrossRef CAS PubMed.
- N. Zhao and L. Qi, Adv. Mater., 2006, 18, 359–362 CrossRef CAS.
- W. Fan, Y. Bu, X. Song, S. Sun and X. Zhao, Cryst. Growth Des., 2007, 7, 2361–2366 CAS.
- W. Fan, X. Song, Y. Bu, S. Sun and X. Zhao, J. Phys. Chem. B, 2006, 110, 23247–23254 CrossRef CAS PubMed.
- F. Wang, Y. Han, C. S. Lim, Y. Lu, J. Wang, J. Xu, H. Chen, C. Zhang, M. Hong and X. Liu, Nature, 2010, 463, 1061–1065 CrossRef CAS PubMed.
- P. H. Kuo, E. Kanal, A. K. Abu-Alfa and S. E. Cowper, Radiology, 2007, 242, 647 CrossRef PubMed.
- P. Caravan, Chem. Soc. Rev., 2006, 35, 512–523 RSC.
- B. Frei and J. V. Higdon, J. Nutr., 2003, 133, 3275S–3284S CAS.
- K. Mabe, M. Yamada, I. Oguni and T. Takahashi, Antimicrob. Agents Chemother., 1999, 43, 1788–1791 CAS.
- M. D. Navarro-Martínez, E. Navarro-Perán, J. Cabezas-Herrera, J. Ruiz-Gómez, F. García-Cánovas and J. N. Rodríguez-López, Antimicrob. Agents Chemother., 2005, 49, 2914–2920 CrossRef PubMed.
- C. A. Rice-Evans, N. J. Miller and G. Paganga, Free Radical Biol. Med., 1996, 20, 933–956 CrossRef CAS.
- J.-M. Song, K.-H. Lee and B.-L. Seong, Antiviral Res., 2005, 68, 66–74 CrossRef CAS PubMed.
- M. S. Maoela, O. A. Arotiba, P. G. L. Baker, W. T. Mabusela, N. Jahed, E. A. Songa and E. I. Iwuoha, Int. J. Electrochem. Sci., 2009, 4, 1497 CAS.
- S. Khokhar and R. K. Owusu Apenten, Food Chem., 2003, 81, 133–140 CrossRef CAS.
- J. F. Moran, R. V. Klucas, R. J. Grayer, J. Abian and M. Becana, Free Radical Biol. Med., 1997, 22, 861–870 CrossRef CAS.
- S. A. B. E. Van Acker, D.-j. Van Den Berg, M. N. J. L. Tromp, D. H. Griffioen, W. P. Van Bennekom, W. J. F. Van Der Vijgh and A. Bast, Free Radical Biol. Med., 1996, 20, 331–342 CrossRef CAS.
- J. Yang, Z. Quan, D. Kong, X. Liu and J. Lin, Cryst. Growth Des., 2007, 7, 730–735 CAS.
- A. Huignard, T. Gacoin and J.-P. Boilot, Chem. Mater., 2000, 12, 1090–1094 CrossRef CAS.
- J. W. Stouwdam, M. Raudsepp and F. C. J. M. van Veggel, Langmuir, 2005, 21, 7003–7008 CrossRef CAS PubMed.
- V. Sudarsan, F. C. J. M. van Veggel, R. A. Herring and M. Raudsepp, J. Mater. Chem., 2005, 15, 1332–1342 RSC.
- C. J. Jia, L. D. Sun, L. P. You, X. C. Jiang, F. Luo, Y. C. Pang and C. H. Yan, J. Phys. Chem. B, 2005, 109, 3284–3290 CrossRef CAS PubMed.
- W. Wang, Z. Cheng, P. Yang, Z. Hou, C. Li, G. Li, Y. Dai and J. Lin, Adv. Funct. Mater., 2011, 21, 456–463 CrossRef CAS.
- X. Li, S. Gai, C. Li, D. Wang, N. Niu, F. He and P. Yang, Inorg. Chem., 2012, 51, 3963–3971 CrossRef CAS PubMed.
- Z. Quan, Z. Wang, P. Yang, J. Lin and J. Fang, Inorg. Chem., 2007, 46, 1354–1360 CrossRef CAS PubMed.
- Y. Li, X. Li, C. Yang and Y. Li, J. Phys. Chem. B, 2004, 108, 16002–16011 CrossRef CAS.
- S. P. Dubey, M. Lahtinen and M. Sillanpää, Process Biochem., 2010, 45, 1065–1071 CrossRef CAS PubMed.
- C. J. Jia, L. D. Sun, L. P. You, X. C. Jiang, F. Luo, Y. C. Pang and C. H. Yan, J. Phys. Chem. B, 2005, 109, 3284–3290 CrossRef CAS PubMed.
- K.-M. Bark, J.-E. Yeom, J.-I. Yang, I.-J. Yang, C.-H. Park and H.-R. Park, Bull. Korean Chem. Soc., 2011, 32, 3443–3447 CrossRef CAS.
- C. Hsu and R. C. Powell, J. Lumin., 1975, 10, 273–293 CrossRef CAS.
- L. Zhang, M. Yin, H. You, M. Yang, Y. Song and Y. Huang, Inorg. Chem., 2011, 50, 10608–10613 CrossRef CAS PubMed.
- Z. Xu, C. Li, D. Yang, W. Wang, X. Kang, M. Shang and J. Lin, Phys. Chem. Chem. Phys., 2010, 12, 11315–11324 RSC.
- C. Yu, M. Yu, C. Li, C. Zhang, P. Yang and J. Lin, Cryst. Growth Des., 2009, 9, 783–791 CAS.
- F. Wang, X. Xue and X. Liu, Angew. Chem., Int. Ed., 2008, 47, 906–909 CrossRef CAS PubMed.
- Y. Lin and D. M. Hanson, J. Phys. Chem., 1987, 91, 2279–2282 CrossRef CAS.
- M. Haase and H. Schäfer, Angew. Chem., Int. Ed., 2011, 50, 5808–5829 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Additional results relevant to tetragonal LaVO4 nanoparticles synthesis, XRD patterns at different hydrothermal temperature and time, EDAX data, TEM images, DLS measurements, FT-IR spectra, zeta potential measurements, fluorescence integrated intensity at different absorbance, magnetization curves at room temperature. See DOI: 10.1039/c3ra44979c |
| This journal is © The Royal Society of Chemistry 2014 |