Origin of enhanced thermoelectric properties of doped CrSi2†
Tribhuwan Pandey and
Abhishek K. Singh*
Materials Research Centre, Indian Institute of Science, Bangalore 560012, India. E-mail: abhishek@mrc.iisc.ernet.in
First published on 23rd October 2013
Abstract
Using first principles density functional theory, we report for CrSi2, a linear relationship between thermodynamic charge state transition levels of defects and maxima of thermopower Tm, thus proposing a unique way of tuning thermoelectric properties. We show for doped CrSi2 that the peak of thermopower occurs at the temperature which corresponds to the position of the defect transition level. Therefore, by modifying the defect transition level, a thermoelectric material with a given operational temperature can be designed.
The efficiency of a thermoelectric material to convert heat to electrical energy is quantified in terms of the dimensionless parameter called figure of merit (ZT), which is defined as ZT = S2σT/κ, where S, σ, and κ are thermopower (also known as the Seebeck coefficient), electrical, and thermal conductivity, respectively. A high value of ZT requires thermoelectric materials with high thermopower, low thermal and high electrical conductivities. In the last decade, enormous efforts have been made to design new thermoelectric materials with high ZT value.1–7 In particular, thermoelectric materials like bismuth telluride,8 filled skutterudites,6,9 clathrates,10–12 lead telluride1,13,14 and bismuth selenide are studied very extensively because of their relatively better value of ZT (1–1.3)15 at high temperatures. Recently, doping and alloying of lead chalcogenides has made it possible to achieve ZT values as high as two.1,15 Although promising, these chalcogenides suffer from the scarcity of tellurium and selenium as well as the high toxicity of lead. Hence, there is a need for eco-friendly thermoelectric materials with high ZT.
Recently, the transition-metal disilicides (TMSi2) have been considered as better alternatives because of the abundance of the constituent elements, large electrical conductivity, high temperature stability, and corrosion resistance.16 Among them, chromium disilicide CrSi2 has attracted attention due to its semiconducting nature17 and resistance to oxidation at very high temperatures.18 While as-grown CrSi2 is generally a p-type semiconductor,19–22 recently n-type doping has also been achieved.23–25 Furthermore, CrSi2 exhibits good electrical conductivity (σ = 105 S m−1) and thermopower (S = 100 μV K−1 at 300 K). However, due to large thermal conductivity (κ = 10 W m−1 K−1),26 it suffers from a low figure of merit.19
It has been shown that transport properties of the thermoelectric materials in general and CrSi2 in particular can be changed significantly by doping.27–29 Essentially doping leads to an increase in carrier concentration as well as a decrease in the lattice part of the thermal conductivity. As a consequence of these effects, doping can improve the ZT, but the temperature at which ZT peaks could be very much undesirable. The temperature dependence of ZT and thermopower follow the same trend. The thermopower of a material reaches a peak value at a particular temperature, which essentially determines the maximum value of ZT and the operational temperature. So far, there has been no systematic way of controlling the operational temperature of thermoelectric materials. Here, we show that doping can also be employed to control the peak thermopower and hence, the operational temperature of CrSi2. We find for CrSi2, a one-to-one correspondence between defect transition level and the temperature at which the thermopower peaks. We demonstrate that modifying the position of the defect transition level can tune the transport properties as well as the operational temperature of n, p, and co-doped CrSi2, systematically. The temperature at which thermopower peaks depends linearly on the position of charge transition level of impurity.
So far, the effect of doping on the thermoelectric properties of CrSi2 has been studied within the rigid band approximation (RBA).30 RBA works on the assumption that on doping the host band structure does not change. RBA works reasonably well for the dopants, where the defect levels are in resonance with the host valence band maxima or conduction band minima. However, for dopants which affect the band structure as well as the effective potential of the host materials, RBA can overestimate31 the thermopower. Furthermore the RBA does not give any insight about what kind of dopants one should choose for efficient performance. Therefore, to search for an optimal dopant, it is necessary to model a more realistic situation. In the current study, CrSi2 is explicitly doped and its transport properties are calculated and compared with equivalent RBA results.
Structural optimization, total energy and electronic structure calculations were performed using first principles density functional theory. The ionic cores are described by all-electron projector augmented wave potentials32,33 and the Perdew–Burke–Ernzerhof34 generalized gradient approximation (GGA) to the electronic exchange and correlation as implemented in the Vienna Ab Initio Simulation Package (VASP).32,33,35 The unit cell was optimized using the conjugate gradient scheme until the forces on every atom were ≤0.005 eV Å−1. The Brillouin zone was sampled by a well converged 12 × 12 × 8 Monkhorst–Pack k-mesh.36 The optimized lattice parameters a = b = 4.37 Å, c = 6.37 Å and band gap = 0.37 eV are in good agreement with the experimental values.19
Concentration of a defect in a material depends on its formation energy. A large formation energy implies that the defects will have lower concentration and vice versa. The formation energy Ef of a defect or impurity X in a charge state q is defined as37
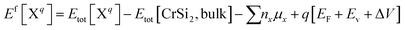 | (1) |
The formation energy will depend on the chemical potential of the host atoms as well as of the dopants. The chemical potentials of Cr and Si are referenced to the calculated total energies of bulk Cr [Etot (Cr)] and Si [½Etot (Si)], respectively. They are subjected to upper bounds of μCr ≤ 0 (Cr-rich limit) and μSi ≤ 0 (Si-rich limit) and must satisfy the stability condition of CrSi2: μCr+ 2μSi = ΔHf(CrSi2), where ΔHf(CrSi2) is the formation enthalpy of CrSi2. We obtain ΔHf(CrSi2) = −1.10 eV, which matches well with the experimental value of −1.20 eV.40 Depending on the growth conditions μCr and μSi can vary in the range ΔHf ≤ μCr ≤ 0, and ½ΔHf ≤ μSi ≤ 0, respectively. All the details regarding reference materials, formation energy and defect transition level along with optimized lattice parameters are documented in the ESI, Tables ST-1 to ST-3.†
The electronic bands closer to the Fermi level in CrSi2 originate from Cr d-states and Si s-, and p-states.19 This implies that the substitutions at both Cr and Si sites can change the transport properties significantly.19 To understand the thermodynamics of doping in bulk CrSi2, the formation energies and transition levels of various dopants at different sites of bulk Cr1−xAxSi2−yBy, where A = V, Mn, Fe and B = Al, P have been calculated. The formation energies of the substituted impurities, both at the Si and Cr is shown in Fig. 1.
The charge transition levels show the value of electron chemical potential at which a defect changes its charge state. A shallow defect will have transition levels very close to the valence band maxima (VBM) or conduction band minima (CBM). A schematic diagram of defect transition level across the band gap for different dopants is shown in Fig. S1.† From Fig. 1 and S1† it is clear that the AlSi, VCr are shallow acceptors (p-type doping) and PSi, FeCr and MnCr are shallow donors (n-type doping). These shallow defects in the CrSi2 will provide significant concentration of charge carriers.
The effect of defect transition level via carrier concentration on thermopower can be described by the Mott formula.41,42 The Mott formula describes the dependence of S on the energy derivative of the electrical conductivity σ(E) = n(E)eμ(E) calculated at the Fermi level EF.
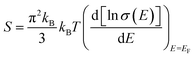 | (2) |
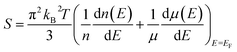 | (3) |
To verify this, we estimated the effect of shallow defects on the transport properties such as electrical conductivity as well as thermopower of CrSi2 using Boltzmann transport theory.43 In this theory, the motion of an electron is treated semi-classically, and its group velocity in a specific band is given by
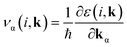 | (4) |
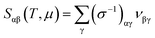 | (5) |
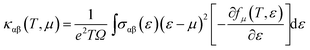 | (6) |
The p-type doping by AlSi, VCr leads to a significant decrease in the thermopower of CrSi2 as shown in Fig. 2. The thermopower increases with the increase of temperature, until a maximum is achieved. This temperature at which thermopower peaks (Tm) corresponds to the defect transition level. As soon as the temperature crosses the transition level of the corresponding defect, the carrier concentration increases suddenly, which leads to a peak in the thermopower. This is in agreement with the Mott formula, where S depends linearly on the change in carrier concentration with energy. Subsequent to that due to bipolar effects the thermopower starts to decrease. For example, the transition levels of Al and V are around 900 K and 800 K, respectively and their thermopower also peaks at the same temperature as shown in the left inset of Fig. 2.
In order to test the performance of the RBA, we calculated the thermopower of the CrSi2 with same carrier concentration obtained by explicit doping cases and plotted in Fig. 3. The details of our RBA calculation are given in the ESI.† The thermopower obtained by explicit doping, with the RBA and by experiments are shown in Table 1. Our calculations show that thermopower achieved by explicit doping is in very good agreement with experiment whereas, RBA overestimates the thermopower as shown in Table 1. RBA neglects the perturbation to the band structure caused by the doping, which leads to overestimated values of thermopower.
 | ||
| Fig. 3 Variation of thermopower with temperature using RBA at doping level of 0.12 and 0.24 electrons and holes per unit cell equivalent to the carrier concentration of explicitly doped cases. | ||
| System | x | TDTL (K) | Experiments | Thermopower (S) μV K−1 | |||
|---|---|---|---|---|---|---|---|
| Explicit doping (ED) | RBA | ||||||
| Sexp | SED | Tm (K) | SRBA | Tm (K) | |||
| CrSi2−xAlx | 0.04 | 900 | 160 (ref. 21) | 151 | 900 | 180 | 950 |
| Cr1−xVxSi2 | 0.04 | 800 | 155 (ref. 20) | 146 | 800 | 180 | 950 |
| CrSi2−xPx | 0.04 | 750 | N/A | −130 | 750 | −225 | 800 |
| Cr1−xMnxSi2 | 0.04 | 900 | N/A | −205 | 900 | −225 | 800 |
| Cr1−xFexSi2 | 0.08 | 800 | N/A | −150 | 800 | −190 | 900 |
For n-type doping by PSi, FeCr, and MnCr, the corresponding thermopower as a function of temperature is shown in Fig. 2. Here also the thermopower follows the same trend as discussed above for p-type doping. Fe and P are shallower donors compared with Mn, and therefore, have lower thermopower. Like the p-type doped system, there is an excellent match between Tm and the defect transition level (Fig. 1). The right inset of Fig. 2 shows a linear variation of the peak temperature of the thermopower Tm with the defect transition level of the dopants. There is a one-to-one correspondence between the position of the defect transition levels and the thermopower. From this analysis, the defect transition levels have been identified as an important parameter to design a thermoelectric material. For n-type doped CrSi2 also RBA overestimates the thermopower as shown in Table 1. At temperatures higher than Tm, the concentration of minority carriers becomes considerable and starts to contribute to the electrical conduction. The resulting thermopower is a sum of relative contributions from both types of carriers weighted by their respective electrical conductivity. The thermopower has opposite sign for majority and minority carriers hence the average thermopower starts to decrease.46 In Table ST-2† we present a head to head comparison of activation temperature of defect transition level and peak thermopower, and as can be seen there is excellent agreement between activation temperature and peak temperature of thermopower for all the cases investigated in the present study.
Next, we study the co-substitution at both Cr and Si sites and its effect on the transport properties. Mn(V) and Al are substituted at Cr and Si sites, respectively. The choice of dopants is made based on recent experimental work,29 where the significant gain in thermopower was observed. This gives rise to p–p (Cr1−xVxSi2−yAly) and n–p (Cr1−xMnxSi2−xAly) type of doping, respectively. In the case of p–p type Cr1−xVxSi2−xAly, the carrier concentration is much higher than the individual Al or V doped case. This leads to a decrease in the thermopower by 30 μV K−1. Importantly, the thermopower in the case of n–p doping also remains positive over the investigated temperature range, as shown in Fig. 2. This result implies that the holes are the majority carriers for the n–p system. This can be understood by the position of the transition levels of Al and Mn from the band edges as shown in Fig. S1.† The transition level of Al is much closer to the VBM compared to the transition level of Mn from the CBM, therefore there will be more holes than electrons in this system. Due to compensation, the overall carrier concentration decreases. The value of the thermopower lies between that of Al- and Mn-doped CrSi2. The thermopower is increased by 30 μV K−1 to 170 μV K−1 with a peak around 500 K. Therefore, the relative defect transition levels can once again help in choosing the right type of co-substitution in designing a thermoelectric material. Our results indicate that higher thermopower can be achieved in n-type than p-type doped CrSi2. The thermopower for n-type 4% Mn doped CrSi2 is −205 μV K−1, which is the highest among all the doped systems studied in the present work. For P and Fe doped systems, the maximum thermopower achieved is −130, and −150 μV K−1, respectively.
In conclusion, using first principles density functional theory, we have shown the important role played by defect transition levels in controlling the thermoelectric properties of doped CrSi2. A linear relationship between defect transition level and the maxima of the thermopower has been reported, which can be employed to enhance the performance of thermoelectric materials. We demonstrate a one-to-one correspondence between the defect transition level and the temperature at which the thermopower peaks. This can be utilized in designing thermoelectric materials for a given operational temperature range. The defect transition level emerges as the parameter which can be varied by doping and should tune the thermoelectric properties of other classes of materials as well.
Acknowledgements
The authors acknowledge Prof. A. M. Umarji for sharing experimental results on doped CrSi2 and useful discussions. The authors also acknowledge financial support from ADA under NPMASS and DST Nanomission. We thankfully acknowledge the Supercomputer Education and Research Centre, and Materials Research Centre, Indian Institute of Science for computing facilities.References
- Y. Pei, X. shi, A. LaLonde, H. Wang, L. Chen and G. J. Snyder, Nature, 2011, 473, 66–69 CrossRef CAS PubMed.
- G. J. Snyder, E. Nishibori, T. Caillat and B. B. Iversen, Nature, 2007, 3, 458–463 Search PubMed.
- H. Ohta, S. Kim, Y. Mune, T. Mizoguchi, K. Nomura, S. Ohta, T. Nomura, Y. Nakanishi, Y. Ikuhara, M. Hirano, H. Hosono and K. Koumoto, Nature, 2007, 6, 129–134 CrossRef CAS PubMed.
- F. J. DiSalvo, Science, 1999, 285, 703–706 CrossRef CAS.
- G. D. Mahan, J. Appl. Phys., 1989, 65, 1578–1583 CrossRef PubMed.
- B. C. Sales, D. Mandrus and R. K. Williams, Science, 1996, 272, 1325–1328 CAS.
- K. F. Hsu, S. Loo, F. Guo, W. Chen, J. S. Dyck, C. Uher, T. Hogan, E. K. Polychroniadis and M. G. Kanatzidis, Science, 2004, 303, 818–821 CrossRef CAS PubMed.
- Y. Q. Cao, X. B. Zhao, T. J. Zhu, X. B. Zhang and J. P. Tu, Appl. Phys. Lett., 2008, 92, 143106 CrossRef PubMed.
- B. Chen, J.-H. Xu, C. Uher, D. T. Morelli, G. P. Meisner, J.-P. Fleurial, T. Caillat and A. Borshchevsky, Phys. Rev. B: Condens. Matter Mater. Phys., 1997, 55, 1476–1480 CrossRef CAS.
- E. S. Toberer, A. F. May and G. J. Snyder, Chem. Mater., 2010, 22, 624–634 CrossRef CAS.
- A. Bentien, M. Christensen, J. D. Bryan, A. Sanchez, S. Paschen, F. Steglich, G. D. Stucky and B. B. Iversen, Phys. Rev. B: Condens. Matter Mater. Phys., 2004, 69, 045107 CrossRef.
- G. S. Nolas, J. L. Cohn, G. A. Slack and S. B. Schujman, Appl. Phys. Lett., 1998, 73, 178–180 CrossRef CAS PubMed.
- J. P. Heremans, V. Jovovic, E. S. Toberer, A. Saramat, K. Kurosaki, A. Charoenphakdee, S. Yamanaka and G. J. Snyder, Science, 2008, 321, 554–557 CrossRef CAS PubMed.
- G. J. Snyder and E. S. Toberer, Nat. Mater., 2011, 7, 105–114 CrossRef PubMed.
- D. Parker and D. J. Singh, J. Appl. Phys., 2010, 108, 083712 CrossRef PubMed.
- V. Cronin, Thermoelectric Properties of Silicides, in CRC Handbook of Thermoelectrics, CRC Press, 1995 Search PubMed.
- I. Nishida, J. Mater. Sci., 1972, 7, 1119–1124 CrossRef CAS.
- J. Ma, Y. Gu, L. Shi, L. Chen, Z. Yang and Y. Qian, J. Alloys Compd., 2004, 376, 176–179 CrossRef CAS PubMed.
- T. Dasgupta, J. Etourneau, B. Chevalier, S. F. Matar and A. M. Umarji, J. Appl. Phys., 2008, 103, 113516 CrossRef PubMed.
- Z. Pan, L. Zhang and J. Wu, Scr. Mater., 2007, 56, 257–260 CrossRef CAS PubMed.
- Z. Pan, L. Zhang and J. Wu, Scr. Mater., 2007, 56, 245–248 CrossRef CAS PubMed.
- T. Dasgupta and A. Umarji, J. Alloys Compd., 2008, 461, 292–297 CrossRef CAS PubMed.
- H. N. Zhu, K. Y. Gao and B. X. Liu, J. Phys. D: Appl. Phys., 2000, 33, L49 CrossRef CAS.
- H. Zhu and B. Liu, Appl. Surf. Sci., 2000, 161, 240–248 CrossRef CAS.
- I. Nishida and T. Sakata, J. Phys. Chem. Solids, 1978, 39, 499–505 CrossRef CAS.
- L. F. Mattheiss, Phys. Rev. B: Condens. Matter Mater. Phys., 1991, 43, 12549–12555 CrossRef CAS.
- M. Komabayashi, K.-i. Hijikata and S. Ido, Jpn. J. Appl. Phys., 1991, 30, 331–334 CrossRef CAS.
- Q. Zhang, H. Wang, Q. Zhang, W. Liu, B. Yu, H. Wang, D. Wang, G. Ni, G. Chen and Z. Ren, Nano Lett., 2012, 12, 2324–2330 CrossRef CAS PubMed.
- S. Perumal, S. Gorsse, U. Ail, B. Chevalier, R. Decourt and A. Umarji, J. Mater. Sci., 2012, 48, 227–231 CrossRef.
- D. Parker and D. J. Singh, New J. Phys., 2012, 14, 033045–033056 CrossRef.
- M.-S. Lee and S. D. Mahanti, Phys. Rev. B: Condens. Matter Mater. Phys., 2012, 85, 165149 CrossRef.
- P. E. Blöchl, Phys. Rev. B: Condens. Matter Mater. Phys., 1994, 50, 17953–17979 CrossRef.
- G. Kresse and D. Joubert, Phys. Rev. B: Condens. Matter Mater. Phys., 1999, 59, 1758–1775 CrossRef CAS.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1997, 78, 1396 CrossRef CAS.
- G. Kresse and J. Furthmller, Comput. Mater. Sci., 1996, 6, 15–50 CrossRef CAS.
- H. J. Monkhorst and J. D. Pack, Phys. Rev. B: Solid State, 1976, 13, 5188–5192 CrossRef.
- C. G. V. de Walle and J. Neugebauer, J. Appl. Phys., 2004, 95, 3851–3879 CrossRef PubMed.
- C. Freysoldt, J. Neugebauer and C. G. Van de Walle, Phys. Rev. Lett., 2009, 102, 016402 CrossRef.
- C. Freysoldt, J. Neugebauer and C. G. Van de Walle, Phys. Status Solidi B, 2011, 248, 1067–1076 CrossRef CAS.
- J. M. Andrews and J. C. Phillips, Phys. Rev. Lett., 1975, 35, 56–59 CrossRef CAS.
- M. Jonson and G. D. Mahan, Phys. Rev. B: Condens. Matter Mater. Phys., 1980, 21, 4223–4229 CrossRef CAS.
- G. D. Mahan and J. O. Sofo, Proc. Natl. Acad. Sci. U. S. A., 1996, 93, 7436–7439 CrossRef CAS.
- J. M. Ziman, Principles of the Theory of Solids, Cambridge University Press, Cambridge, 1964 Search PubMed.
- L. L. Boyer, Phys. Rev. B: Condens. Matter Mater. Phys., 1979, 19, 2824–2830 CrossRef CAS.
- G. K. Madsen and D. J. Singh, Comput. Phys. Commun., 2006, 175, 67–71 CrossRef CAS PubMed.
- C. Wood, Rep. Prog. Phys., 1988, 51, 459 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c3ra44822c |
| This journal is © The Royal Society of Chemistry 2014 |


