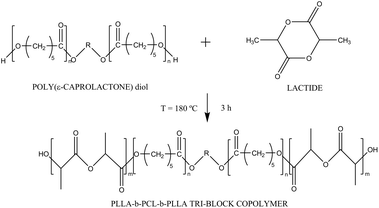
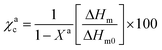Synthesis of PLLA-b-PCL-b-PLLA linear tri-block copolymers and their corresponding poly(ester-urethane)s: effect of the molecular weight on their crystallisation and mechanical properties
Ivan Navarro-Baenaa,
Angel Marcos-Fernándezb,
Alberto Fernández-Torresb,
José M. Kennyab and
Laura Peponi*b
aUniversity of Perugia, Strada di Pentima 4, Terni, Italia
bInstitute of Polymer Science and Technology (ICTP-CSIC), Calle Juán de la Cierva 3, Madrid, Spain. E-mail: lpeponi@ictp.csic.es
First published on 16th January 2014
Abstract
With the general objective to design polymer based materials with specific thermal and mechanical properties, a systematic study on the crystallinity and the mechanical properties of synthesized linear tri-block copolymers based on poly(L-lactic acid) and poly(ε-caprolactone) and of their corresponding poly(ester-urethane)s has been performed. In particular, eleven tri-block copolymers were synthesized varying both the molecular weight of the blocks as well as the relative content of each block in the copolymer, focusing the attention on the relationship between their chemical compositions and their tailored final properties in order to tune them taking into account their possible applications (i.e. as shape memory materials). From them, eleven poly(ester-urethane)s were synthesized by condensation with hexamethylene diisocyanate (HDI). The chemical composition of the synthesized polymers was studied and correlated with their thermal and crystalline properties obtained by both dynamic scanning calorimetry (DSC) and small angle X-ray diffraction (SAXS) experiments. The relationship between their crystalline structure, thermal and mechanical properties with the molecular weight as well as with the relative content of each comonomer in the copolymers and the amount of HDI in the poly(ester-urethane) was analysed. The results obtained demonstrate that these bio tri-block copolymers and the corresponding poly(ester-urethane)s can be tailored with interesting crystalline and mechanical properties. Future applications as shape memory systems are thus envisaged.
Introduction
The development and use of biopolymers have increased in recent years due to their versatility and their unique properties well-adaptable to the innovative applications requested in bio-medicine and other advanced fields. In this context, aliphatic polyesters such as poly(L-lactic acid) (PLLA), poly(ε-caprolactone) (PCL) or poly(glycolic acid) (PGA) are interesting polymers for several bio-applications.1–4 These polymers are biocompatible and they degrade through hydrolytic scission of the ester groups, thus being suitable for biomedical applications such as scaffolds for tissue engineering, drug delivery systems or sutures among others.5–7 In the last years copolymers and blends of these homopolymers have been studied in order to design new materials with specific properties in which their synergetic effect could be exploited.8–11 At this regard, it is important to consider not only their synergetic interaction but also how this interaction can influence their final properties in order to design specific materials with tailored specific properties.PLLA, obtainable from renewable sources, is a rigid polymer having a glass transition temperature (Tg) around 60 °C. It melts around 180 °C and shows good mechanical properties. Nowadays PLLA is mainly used in packaging applications,12 replacing polymers derived from non-renewable sources but its main drawback is its brittleness.8 On the other hand, PCL is a flexible polymer with a melting temperature (Tm) around 60 °C and a Tg at −60 °C. In order to enhance their mechanical, thermal or biodegradability properties, the preparation of synergistic blends and different di-, tri-, multi-block and random copolymers based on PCL and PLLA have been reported recently on the scientific literature.13–18 In particular, Castillo et al.13 and Peponi et al.14 reported the influence of the composition of di-block copolymers of PCL and PLLA on their thermal properties. In those research works, the melting temperature and the crystallinity of each block were related to the chain length. Furthermore Fernandez et al.15 reported the synthesis of statistical copolymers of PCL and PLLA, and they found variations on thermal as well as on mechanical properties related to the chemical composition. Cohn et al.16 reported thermoplastic elastomers based on PCL and PLLA with a block structure which showed a wide range of values in terms of mechanical properties and biodegradability depending on their chemical composition. Vilay et al.17 studied blends of PCL and PLLA and reported the poor miscibility of the homopolymers. In their research they improved the miscibility of the system using a tri-block copolymer based on poly(ethylene oxide) and poly(propylene oxide) as compatibilizer. Lu et al.18 studied the effect of the composition on the shape memory behaviour of statistical copolymers based on PCL and PLLA. The shape memory behaviour was investigated in terms of shape recovery rate and recovery stress. They found that the addition of PCL at low concentrations enhanced the shape memory parameters at different strain rates. Also Peponi et al.19 synthesized a poly(ester-urethane) based on PCL-PLLA block copolymers with shape memory behaviour with a transition temperature of about 40 °C for deformations up to 350%.
Different aspects evidence the advantages of tri-block copolymer over polymer blends. For example, tri-block copolymers can be synthesized with short molecular weight of the blocks, and the corresponding poly(ester-urethane)s will have good mechanical properties,19 whereas polymer blends prepared from commercial materials will use homopolymers with high molecular weight because low molecular weight homopolymers will have poor mechanical properties. Also, the tri-block structure has the possibility to tune the thermal properties by changing the ratio of the blocks and the length of the blocks with the interaction between both blocks playing an important role,13,17 whereas in polymer blends, due to the immiscibility of the homopolymers, the thermal properties will be basically the thermal properties of the starting homopolymers. Finally, the presence of both blocks in the same chain does not allow the migration of one block, whereas in polymer blends, the immiscibility of the blocks can produce, if one homopolymer has sufficiently low molecular weight, the exudation of this homopolymer with time, producing a significant change in properties.20
Furthermore, linear block copolymers play an advantage over the use of random copolymers. Random copolymers will produce amorphous materials21,22 unless it is so highly enriched in one of the monomers that the sequences of this monomer will be long enough to crystallize, but to reach this critical length the content in this monomer will be so high that it will be more properly a modified or plastified homopolymer. With linear block copolymers, the critical length for the crystallization of the individual blocks can be reached even at relatively low contents of one of the monomers.14
In the research reported here the attention has been focused on the synthesis and the characterization of PLLA-b-PCL-b-PLLA tri-block copolymers and of their corresponding poly(ester-urethane)s, analysing the effects of their molecular weight and of their composition on the crystallisation behaviour of the synthesized polymers. The mechanical properties of the synthesized poly(ester-urethane)s were also investigated and related to their crystalline structure. This correlation is very important due to the necessity to design new materials with specific properties, where not only the ratio between the different blocks is relevant but also their thermo-mechanical response, such as in designing materials with shape memory behaviour.19 Eleven tri-block copolymers based on PCL and PLLA have been synthesized, varying both the ratio of the two blocks and the molecular weight of each block. After that, eleven poly(ester-urethane)s have been synthesized through the reaction of these copolymers with hexamethylene diisocyanate (HDI). The molecular weight of the tri-blocks was studied with nuclear magnetic resonance (1H NMR) and matrix-assisted laser desorption/ionization time-of-flight (MALDI-TOF) experiments. To confirm the reaction of the hydroxyl groups of the tri-block copolymers with the isocyanate groups of the HDI, 1H NMR and Fourier transform infrared spectroscopy (FT-IR) measurements were performed. The amorphous/crystalline nature of all polymers was studied by Small Angle X-ray Scattering (SAXS) experiments and Differential Scanning Calorimetry (DSC). The mechanical properties were investigated using standard tensile tests.
Experimental
Materials
L-Lactide (L-LA), HDI, stannous octoate (Sn(Oct)2), chloroform, methanol and dichloroethane were purchased from Sigma Aldrich (Spain). Three different commercial PCL diols, with different nominal molecular weights (2000, 4000 and 8000 g mol−1), donated by Perstorp (Sweden), were used. All the materials employed in this work were used as received without further purification.Synthesis of PLLA-b-PCL-b-PLLA tri-block copolymers
Eleven different tri-block copolymers were synthesized by ring opening polymerization of L-lactide by the hydroxyl-end groups of the PCL diol in the presence of Sn(Oct)2 as catalyst (Scheme 1). The amount of L-lactide and PCL diol was calculated in order to obtain 5 grams of final product; Sn(Oct)2 was added in 0.1 wt% respect to the L-lactide monomer. The reaction was carried out in bulk, in a 15 ml round bottom flask immersed in an oil bath at 180 °C for 3 h. After that, when the temperature decreased, the mixture was dissolved in chloroform and precipitated in cold methanol. The white precipitate was filtered off and dried under vacuum to extract all residual solvents. The synthesized tri-block copolymers are named with the prefix T-. Their composition in terms of block molecular weight and weight ratio between the blocks are reported in Table 1.| Sample | CL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LAa LAa |
CL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LAb LAb |
Mn,PCLc (g mol−1) | Mn triblockb (g mol−1) | Mn triblockd (g mol−1) |
|---|---|---|---|---|---|
| a Feed weight ratio.b Calculated by 1H NMR using CL/LA ratio.c Determined from the hydroxyl value.d Calculated by 1H NMR using LA end groups. | |||||
| T1 | 85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 08 08 |
8049 | 8735 | 8840 |
| T2 | 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 30 |
89![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 11 11 |
2054 | 2306 | 2562 |
| T3 | 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 30 |
84![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16 16 |
4021 | 4808 | 4937 |
| T4 | 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 30 |
77![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 23 23 |
8049 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 393 393 |
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 286 286 |
| T5 | 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 50 |
73![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 37 37 |
2054 | 2903 | 3152 |
| T6 | 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 50 |
54![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 46 46 |
4021 | 7375 | 7266 |
| T7 | 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 50 |
54![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 46 46 |
8049 | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 022 022 |
13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 670 670 |
| T8 | 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 70 70 |
34![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 66 66 |
2054 | 6056 | 5756 |
| T9 | 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 70 70 |
33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 67 67 |
8049 | 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 608 608 |
19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 410 410 |
| T10 | 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 70 70 |
32![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 68 68 |
4021 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 502 502 |
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 316 316 |
| T11 | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85 85 |
18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 82 82 |
8049 | 45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 375 375 |
28![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 335 335 |
Synthesis of poly(ester-urethane)s based on PCL-PLLA
Starting from the synthesized tri-block copolymers, a polycondensation reaction with hexamethylene diisocyanate (HDI) at 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio was carried out taking advantage of the presence of the hydroxyl groups at the ends of the tri-blocks chains. This reaction linked the tri-blocks through urethane bonds thus forming a poly(ester-urethane) and was performed in a 25 ml round bottom flask using dichloroethane as solvent and Sn(Oct)2 as catalyst, at 80 °C for 5 hours. The initial molar concentration of the tri-blocks was around 0.1 mol l−1. Once the reaction finished, the product was diluted with chloroform and the solution casted onto a leveled glass. The films were dried at ambient conditions for 12 hours and then under vacuum at room temperature for at least 24 hours to remove the residual solvent. The obtained poly(ester-urethane)s are named with the prefix PU-, following the same order of the corresponding previously synthesized tri-block materials.
1 molar ratio was carried out taking advantage of the presence of the hydroxyl groups at the ends of the tri-blocks chains. This reaction linked the tri-blocks through urethane bonds thus forming a poly(ester-urethane) and was performed in a 25 ml round bottom flask using dichloroethane as solvent and Sn(Oct)2 as catalyst, at 80 °C for 5 hours. The initial molar concentration of the tri-blocks was around 0.1 mol l−1. Once the reaction finished, the product was diluted with chloroform and the solution casted onto a leveled glass. The films were dried at ambient conditions for 12 hours and then under vacuum at room temperature for at least 24 hours to remove the residual solvent. The obtained poly(ester-urethane)s are named with the prefix PU-, following the same order of the corresponding previously synthesized tri-block materials.
Characterization
Solution 1H NMR spectra were recorded at room temperature in a Varian Unity Plus 400 instrument using deuterated chloroform (CDCl3) as solvent. Spectra were referenced to the residual solvent protons at 7.26 ppm.Fourier transform infrared (FT-IR) spectra were performed in a Perkin Elmer Spectrum One FT-IR spectrometer equipped with an ATR accessory. Spectra were taken in the range 650–4000 cm−1 with a resolution of 1 cm−1 and an accumulation of 16 scans.
Differential scanning calorimetry (DSC) analysis was performed in a Mettler Toledo DSC822e instrument. Samples of about 10 mg were sealed in aluminum pans. Thermal cycles consisted of two heating scans (from 0 to 200 °C and from −90 to 200 °C) and a cooling scan (from 200 to −90 °C) in between them, at a heating/cooling rate of 10 °C min−1 under nitrogen purge. The values of Tm were obtained as the maximum of the endothermic peak and the enthalpy of melting (ΔHm) was calculated as the integral of the area below the peaks. Data reported here were taken from the second heating scan.
SAXS measurements of both tri-block copolymers and poly(ester-urethane)s were taken at beamline BM16 at the European Synchrotron Radiation Facility (Grenoble, France). Tri-blocks were melted and sealed in DSC aluminum pans, placed within a Linkam hot stage and heated at 10 °C min−1 while the SAXS spectra were recorded. Poly(ester-urethane) samples were wrapped in aluminum foils without any thermal treatments before carrying out the SAXS experiments. Temperature calibration gave a difference of approximately 7 °C between the temperature reading at the hot stage display and the real temperature at the sample. Detector calibration was performed with silver behenate (AgC22H43O2). The long period L was calculated as 2π/q, where q is the scattering vector.
Tensile properties were measured in a MTS Synergie 200 testing machine equipped with a 100 N load cell. Type 3 dumbbell test specimens (according to ISO 37) were cut from film. A crosshead speed of 200 mm min−1 was used. Strain was measured from crosshead separation and referred to 10 mm initial length. Five samples were tested for each poly(ester-urethane).
Results and discussion
Synthesis of tri-block copolymers
Eleven different PLLA-b-PCL-b-PLLA tri-block copolymers have been synthesized varying both the relative content as well as the molecular weight of each block in the copolymer. Three different commercial PCL diols were selected having nominal molecular weights of about 2000, 4000 and 8000 g mol−1. The real molecular weights of these materials were calculated from the hydroxyl value provided in the data sheet and are reported in Table 1. The theoretical PCL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PLLA weight ratios selected were 15
PLLA weight ratios selected were 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85, 30
85, 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 70, 50
70, 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50, 70
50, 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 and 85
30 and 85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15. All synthesized copolymers were analysed by 1H NMR spectroscopy. The real weight ratios as well as the molecular weights calculated by 1H NMR analysis for the eleven tri-block copolymers synthesized are summarized in Table 1.
15. All synthesized copolymers were analysed by 1H NMR spectroscopy. The real weight ratios as well as the molecular weights calculated by 1H NMR analysis for the eleven tri-block copolymers synthesized are summarized in Table 1.
The analysis of 1H NMR spectra was performed taking into account the work of Kasperczyk.23 In Fig. 1 the spectrum for T6 is shown (for the other tri-block copolymers the spectra are not reported, but they were similar to the one reported in Fig. 1). The multiplet at 5.05–5.25 ppm and the multiplet at 4.35 ppm correspond to the methine proton f of polymerized lactide and to the methine proton of lactide end group f′ respectively. The signal of un-reacted L-lactide in the final products, peak at 5.03 ppm, was less than 1 wt% for all samples. The triplet at 4.05 ppm corresponds to protons a and it is related to CL units linked to another CL unit and to the CL units linked to the LA units. The triplet at 2.30 corresponds to the protons e of CL linked to another CL unit. The other signals correspond to CL protons b, d and c at 1.66, 1.63 and 1.37 ppm respectively and the multiplet at 1.57 ppm corresponds to the LA methyl proton g, while the peak at 1.49 ppm corresponds to the end group of LA methyl proton g′. Two main aspects can be underlined: signals of residual solvents were not present and no signal of CL terminal groups appeared, at about 3.74 ppm, so the desired molecular structure with PCL midblock (PLLA-b-PCL-b-PLLA) was confirmed.
In order to determine the molecular weight of the copolymers two methods have been used. In the first one, the ratio between the signals for the PCL and the PLLA units is used. The selected signals for calculation are the protons f+f′ for the PLLA units and the protons a for the PCL units. In the second method, the ratio between the signal for PLLA ester units f and the signal for PLLA end group, f′ has been used. The molecular weight for the tri-blocks calculated by both methods is reported in Table 1. It can be observed that the agreement in the values calculated by both methods is good, with a difference of approximately 10% or less except for the longer copolymers (above 10.000 g mol−1). For the latter, the larger difference can be attributed to a higher error in the calculation of f′ signal when the molecular weight increases. For this reason, the first method was considered as more precise, and the molecular weight for the copolymer was taken as the value calculated from the ratio between PCL and PLLA signals. The molecular weight distribution was investigated by MALDI-TOF and GPC experiments (data not-shown). The values of polydispersity of the different synthesized copolymers ranged from 1.2 to 1.5.
The main characteristic peaks corresponding to the PCL and the PLLA blocks have been studied by FT-IR analysis. In general, the intensity of the peaks is related to the polymer composition; however, in our case it is difficult to quantify the chemical composition with this technique, thus FT-IR analysis is qualitative. In Fig. 2 the carbonyl stretching region of the FT-IR spectra for the synthesized tri-block copolymers is represented. This figure is divided into three graphs depending on the nominal molecular weight of the starting PCL diol, that is 2000, 4000, 8000 g mol−1. Moreover, the percentages of PCL are reported in the graphs in order to facilitate the lecture of the diagrams. The more intense peaks in the carbonyl stretching region of 1800–1650 cm−1 correspond to both ester units (at about 1756 cm−1 for PLLA and 1721 cm−1 for PCL). The peaks in the region of 600–1200 cm−1 correspond to characteristic vibration modes of PCL and PLLA as it is detailed in literature.24,25 The variation on the peak intensities with the polymer composition is evidenced. The polymers with high content of PCL, such as T1, T2 and T3, corresponding to the first line of each diagram, respectively, show a strong peak at 1721 cm−1, while for the polymers having intermediate compositions, this peak appears with less intensity and the peak at 1756 cm−1 corresponding to the carbonyl stretching of PLLA increases. Finally, for the polymers with higher content of PLLA, (line on the top) the peak at 1756 cm−1 appears more intense.
 | ||
| Fig. 2 Carbonyl stretching FT-IR spectra of tri-block copolymers: (a) synthesized from PCL2000, (b) synthesized from PCL4000 and (c) synthesized from PCL8000. | ||
Synthesis of poly(ester-urethane)s
Starting from the synthesized tri-block copolymers with hydroxyl groups at the end of the chain, a polycondensation reaction using HDI at 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar concentration was carried out. In this manner the PLLA blocks were linked through urethane bonds. The formation of the urethane bonds was confirmed by FT-IR analysis. In Fig. 3, two examples of FT-IR spectra corresponding to a tri-block copolymer (T6) and its corresponding poly(ester-urethane), are shown. The characteristic peaks for PCL and PLLA can be seen, and for the poly(ester-urethane), new peaks at about 1534 cm−1 due to the amide II band of the urethane bond and around 3500 cm−1 due to the NH stretching of the urethane group, appear.26 The presence of these new peaks confirms the reaction between the hydroxyl end groups of tri-block copolymers and the isocyanate groups of HDI.19 Another way to confirm urethane formation is by 1H NMR. So, in Fig. 4 the 1H NMR spectrum for PU6 is reported. Thus, the presence of a new peak at 3.15 ppm confirms the formation of urethane groups.17,27 Moreover the signal corresponding to the PLLA end groups (peak at 4.35 ppm) disappears almost completely, confirming that the condensation reaction between the isocyanate groups of HDI and the hydroxyl groups of the tri-block copolymer proceeded correctly.
1 molar concentration was carried out. In this manner the PLLA blocks were linked through urethane bonds. The formation of the urethane bonds was confirmed by FT-IR analysis. In Fig. 3, two examples of FT-IR spectra corresponding to a tri-block copolymer (T6) and its corresponding poly(ester-urethane), are shown. The characteristic peaks for PCL and PLLA can be seen, and for the poly(ester-urethane), new peaks at about 1534 cm−1 due to the amide II band of the urethane bond and around 3500 cm−1 due to the NH stretching of the urethane group, appear.26 The presence of these new peaks confirms the reaction between the hydroxyl end groups of tri-block copolymers and the isocyanate groups of HDI.19 Another way to confirm urethane formation is by 1H NMR. So, in Fig. 4 the 1H NMR spectrum for PU6 is reported. Thus, the presence of a new peak at 3.15 ppm confirms the formation of urethane groups.17,27 Moreover the signal corresponding to the PLLA end groups (peak at 4.35 ppm) disappears almost completely, confirming that the condensation reaction between the isocyanate groups of HDI and the hydroxyl groups of the tri-block copolymer proceeded correctly.
Crystallisation analysis
The thermal behaviour of the tri-block copolymers as well as their crystalline nature have been studied by DSC. The DSC thermograms for the synthesized tri-block copolymers are reported in Fig. 5. | ||
| Fig. 5 DSC thermograms of tri-block copolymers (a: tri-blocks synthesized from the PCL2000; b: tri-blocks synthesized from PCL4000; c: tri-blocks synthesized from PCL8000). | ||
In order to better understand the effects of the molecular weight on the thermal properties of these materials, the results for the tri-blocks synthesized from the PCL2000 are reported in Fig. 5a; while those starting from PCL4000 are reported in Fig. 5b and those from PCL8000 are reported in Fig. 5c. Moreover, the theoretical composition of each material is indicated above each thermogram represented in these figures.
The values of Tm, Tg and Xc obtained from the DSC experiments are summarized in Table 2 for each material.
| Sample | PCL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PLLA PLLA |
Mn,PCL (g mol−1) | Mn,PLLAtot (g mol−1) | Tm,PCL (°C) | Xc,PCL (%) | Tm,PLLA (°C) | Xc,PLLA (%) | Tg,PCL (°C) |
|---|---|---|---|---|---|---|---|---|
| T1 | 92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 08 08 |
8049 | 686 | 54 | 44 | — | 0 | −54 |
| T2 | 89![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 11 11 |
2054 | 252 | 48 | 50 | — | 0 | −53 |
| T3 | 84![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16 16 |
4021 | 787 | 46 | 50 | — | 0 | −40 |
| T4 | 77![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 23 23 |
8049 | 2344 | 55 | 44 | 142 | 49 | −56 |
| T5 | 73![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :27 :27 |
2054 | 774 | 44 | 43 | — | 0 | −13 |
| T6 | 54![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 46 46 |
4021 | 3354 | 43 | 9 | 126 | 47 | −58 |
| T7 | 54![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 46 46 |
8049 | 6973 | 51 | 32 | 156 | 44 | −59 |
| T8 | 34![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 66 66 |
2054 | 4002 | — | 0 | 129 | 50 | −45 |
| T9 | 33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 67 67 |
8049 | 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 559 559 |
34 | 7 | 162 | 59 | −49 |
| T10 | 32![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 68 68 |
4021 | 8481 | — | 0 | 149 | 54 | −49 |
| T11 | 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 82 82 |
8049 | 37![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 326 326 |
— | 0 | 166 | 57 | — |
The degree of crystallization for each block has been calculated according to the following equation:
For each material χac represents the degree of crystallinity of the component a, ΔHm is the melting enthalpy, ΔHm0 is the melting enthalpy for a 100% crystalline material and Xa is the fraction of the component a in the sample. The value taken for ΔHm0 of PLLA was 93 KJ mol−1,28 while for PCL was 148 KJ mol−1.29
It is evident that the melting temperatures as well as the enthalpies of melting are strongly influenced by the composition of the copolymer. The endothermic peaks at temperatures close to 40 °C correspond to the melting of PCL blocks while those at higher temperature correspond to the melting of PLLA. On the other hand, the exothermal peaks that appear in some of the DSC scans between 60 °C and 80 °C correspond to the cold crystallisation of PLLA. In the samples with high content of PLLA, such as 70 and 85 wt%, two endothermic peaks, indicating a double melting point of the PLLA-block are presented. The appearance of this phenomenon has also been reported in the literature for isothermal and dynamical crystallisation of copolymers of PCL-PLLA. Castillo et al.13 attributed this phenomenon, observed in di-block copolymers of PCL-PLLA, to a reorganization process during the scan, due to the presence of PCL. Sarasua et al. attributed the double peak of different synthesized PLLA homopolymers to the annealing occurring during the DSC scans where the small crystals had time to recrystallize a few degrees above and to remelt.30 It has also been reported that the presence of different peaks in the melting of PLLA could be induced by changes in the crystallisation behaviour induced during processing. For instance, Sonseca et al.31 observed a similar crystallisation behaviour induced by the alignment of the electrospun fibres in PLLA fibre mats.
The influence of the molecular weight of each block on the Tm and on the degree of crystallinity for the tri-blocks is reported in Fig. 6. It is clear that the crystallisation of the PCL-block is affected by the presence of crystalline PLLA-block (white circles) and of the amorphous PLLA (black squares). In particular, Fig. 6a represents the Tm of PCL and Fig. 6b the degree of crystallinity of PCL. In tri-blocks with amorphous PLLA-block (Fig. 6a) the melting temperature of the PCL-block slightly increases with the increase in the molecular weight of the PCL block. And, when PLLA blocks crystallizes, the restrictions imposed by the presence of the PLLA crystals reduces the ability of the PCL blocks to crystallize, and the resulting crystals are smaller and less perfect, resulting in a lower melting point of the PCL crystals. These restrictions increased with the length and the content of the PLLA block, as it can be seen in Table 2 (entries T4, T7 and T9) for the tri-blocks based on PCL8000, until the PCL crystallinity is completely suppressed (entry T11). Fig. 6b shows the degree of crystallinity of the PCL-block. The copolymers with amorphous PLLA (black squares) show a degree of crystallinity around 50%, as already found for PCL-PLLA di-blocks with amorphous PLLA.14 On the other hand, when PLLA-block is crystalline (white circles), the degree of crystallinity of the PCL-block is strongly reduced for all the compositions analysed and in particular, the degree of crystallinity of the PCL-block, at the same molecular weight, decreased with increasing the length of the PLLA blocks, until complete suppression. At similar PLLA block length, the decrease was higher at lower molecular weight of the PCL block (see entries T7 and T10 in Table 2). A significant difference is found respect to the crystallisation of PCL in di-block copolymers, as reported before by our research group.14 For di-block copolymers, when the PLLA block was crystalline, PCL started to crystallize for PCL molecular weight higher than 2000 g mol−1 and the crystalline content increased exponentially until it reached a plateau at 6500 g mol−1 with a value of approximately 50% crystallinity irrespective of the PLLA content. Only at intermediate concentrations (PCL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PLA 50
PLA 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 by weight) PCL crystallized substantially without a strong restriction promoted by the presence of PLLA crystals. For the tri-blocks synthesized here, the length and the content of PLLA-block strongly influence the PCL crystallinity. Thus, for the PCL8000 series, when PLLA content is increased, PCL crystallisation is reduced until complete suppression (entries T4, T7, T9 and T10) in parallel to the decrease found in the melting point of the crystals.
50 by weight) PCL crystallized substantially without a strong restriction promoted by the presence of PLLA crystals. For the tri-blocks synthesized here, the length and the content of PLLA-block strongly influence the PCL crystallinity. Thus, for the PCL8000 series, when PLLA content is increased, PCL crystallisation is reduced until complete suppression (entries T4, T7, T9 and T10) in parallel to the decrease found in the melting point of the crystals.
 | ||
| Fig. 6 Melting temperature (Tm) and degree of crystallinity (Xc) of tri-block copolymers vs. molecular weight of PCL (a and b) and PLLA (c and d) respectively. | ||
PLLA melting temperature and degree of crystallisation are represented in Fig. 6c and d respectively. PLLA crystallinity starts at 2340 g mol−1 and reaches a plateau very rapidly. Coherent with the crystallisation behaviour, the PLLA melting point increases with the PLLA block length until a plateau is reached above 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1. This behaviour is similar to the one found for di-block copolymers.14 For di-block copolymers, PLLA crystallisation started at a PLLA molecular weight of 964 g mol−1 and the crystallisation was not influenced by the presence of PCL reaching a plateau at approximately 2500 g mol−1. Moreover, for di-block copolymers, the PLLA melting point was also dependent on the PLLA molecular weight but maximum melting point was reached at about 7500 g mol−1. In the case of the tri-block copolymers synthesized in this work, it has to be taken into account considered that the total PLLA molecular weight is equally distributed in the two blocks linked to the middle PCL-block, and therefore the melting point almost levels at approximately 8500 g mol−1 of the PLLA blocks whereas crystallisation starts at 1170 g mol−1 and a plateau at approximately 3000 g mol−1, which are in good agreements with the values found for di-block copolymers. In addition, the values for Tm and maximum crystallinity for PLLA in the tri-blocks, approximately 165 °C and 55%, respectively, are practically the same values achieved in the di-blocks composition. These similarities demonstrate that PLLA blocks in tri-block copolymers have the same ability to crystallize that in di-block copolymers and that the main parameter that influences the PLLA crystallisation is its block length.14
000 g mol−1. This behaviour is similar to the one found for di-block copolymers.14 For di-block copolymers, PLLA crystallisation started at a PLLA molecular weight of 964 g mol−1 and the crystallisation was not influenced by the presence of PCL reaching a plateau at approximately 2500 g mol−1. Moreover, for di-block copolymers, the PLLA melting point was also dependent on the PLLA molecular weight but maximum melting point was reached at about 7500 g mol−1. In the case of the tri-block copolymers synthesized in this work, it has to be taken into account considered that the total PLLA molecular weight is equally distributed in the two blocks linked to the middle PCL-block, and therefore the melting point almost levels at approximately 8500 g mol−1 of the PLLA blocks whereas crystallisation starts at 1170 g mol−1 and a plateau at approximately 3000 g mol−1, which are in good agreements with the values found for di-block copolymers. In addition, the values for Tm and maximum crystallinity for PLLA in the tri-blocks, approximately 165 °C and 55%, respectively, are practically the same values achieved in the di-blocks composition. These similarities demonstrate that PLLA blocks in tri-block copolymers have the same ability to crystallize that in di-block copolymers and that the main parameter that influences the PLLA crystallisation is its block length.14
The effects of the block composition on the glass transition temperature were also analysed by DSC. As reported in literature, PCL and PLLA form immiscible or partially miscible systems.10,13,29 Typically, the Tg of pure PCL is around −60 °C while for pure PLLA is around 60 °C, depending on their molecular weight and tacticity.20
The Tg of the PCL-blocks are reported in Fig. 7. The values were divided in three different sets: tri-blocks with amorphous PCL-block (black stars), those with crystalline PCL-block and amorphous PLLA-block (black squares) and those with both crystalline blocks (white circles). Tri-blocks with both crystalline blocks show a Tg of the PCL-block around −60 °C, which increases slightly with the PCL-block content. In the case of tri-blocks with amorphous PLLA-blocks and crystalline PCL-block (black squares) a strong influence of the composition of the copolymers has been found. This effect is due to the plasticizing effect of PLLA with short chain length. This fact indicates a strong interaction of the amorphous PLLA and PCL chains promoting a partial miscibility between the blocks. This effect appears also in copolymers with crystalline PLLA-blocks and amorphous PCL-blocks (black stars). The depression of Tm (Fig. 6) of each block and the changes on Tg (Fig. 7) reveal that there is a partial miscibility of the two blocks in these copolymers. The glass transition of the PLLA-block is difficult to measure from DSC thermograms because it is in the range of the Tm of the PCL-block.32
In Fig. 8 the thermograms corresponding to the second heating scan of the poly(ester-urethane)s synthesized from the tri-block copolymers are reported.
Striking differences on the melting temperatures as well as on the degree of crystallinity compared with the initial tri-block copolymers have been obtained on the poly(ester-urethane)s as shown in Table 3, where the amounts of HDI in the poly(ester-urethane)s are reported in order to easily understand the effects of the urethane bonds on the thermal properties. As shown in Fig. 8, the poly(ester-urethane)s are less crystalline than the initial corresponding tri-block copolymers. Furthermore, in this case there is a cold crystallisation process in the second heating scan indicating that the polymer chains could not crystallize during the cooling step at the cooling rate used. The exothermal peaks at about 0 °C correspond to the cold crystallisation of PCL while those between 60 °C and 80 °C correspond to the cold crystallisation of the PLLA. The appearance of these crystallisation peaks is due to the inability of the chains to crystallize during cooling at the cooling rates used as a consequence of the restrictions imposed to the tri-block chains by the urethane linkages and the increase in the molecular weight respect to the starting tri-blocks.
| Sample | PCL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PLLA PLLA |
Mn,PCL (g mol−1) | Mn,PLLAtot (g mol−1) | HDI (%) | Tm,PCL (°C) | Xc,PCL (%) | Tm,PLLA (°C) | Xc,PLLA (%) | Tg,PCL (°C) |
|---|---|---|---|---|---|---|---|---|---|
| PU1 | 92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 08 08 |
8049 | 686 | 1.89 | 53 | 46 | — | 0 | −56 |
| PU2 | 89![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 11 11 |
2054 | 252 | 6.80 | 45 | 29 | — | 0 | −51 |
| PU3 | 84![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16 16 |
4021 | 787 | 3.38 | 48 | 39 | — | 0 | −42 |
| PU4 | 77![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 23 23 |
8049 | 2344 | 1.59 | 52 | 39 | 137 | 24 | −44 |
| PU5 | 73![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 27 27 |
2054 | 774 | 5.61 | 41 | 0.5 | — | 0 | −44 |
| PU6 | 54![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 46 46 |
4021 | 3354 | 2.23 | 45 | 2 | 118 | 0 | −35 |
| PU7 | 54![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 46 46 |
8049 | 6973 | 1.11 | 51 | 25 | 152 | 38 | −54 |
| PU8 | 34![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 66 66 |
2054 | 4002 | 2.70 | — | 0 | 125 | 0.5 | −45 |
| PU9 | 33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 67 67 |
8049 | 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 559 559 |
0.68 | — | 0 | 160 | 13 | −60 |
| PU10 | 32![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 68 68 |
4021 | 8481 | 1.33 | — | 0 | 144 | 0.5 | −49 |
| PU11 | 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 82 82 |
8049 | 37![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 326 326 |
0.37 | — | 0 | 164 | 15 | −54 |
Whereas in the tri-blocks, the PLLA blocks are at the chain ends and are free to crystallize, in the poly(ester-urethane), with a segmented structure, the PLLA blocks are now part of a longer chain that restricts the movements of the individual blocks. In addition, the urethane groups can form intermolecular and intramolecular hydrogen bonds that restrict further the movement of the chains.
Fig. 9 reports the values of the Tm and the degree of crystallinity for both blocks in the synthesized poly(ester-urethane)s as a function of the molecular weight of each block. PCL melting point is barely reduced in the PUs based on tri-blocks of PCL8000 and PCL4000 (Fig. 9a), but it was significantly reduced in PUs based on tri-blocks of PCL2000. Changes in PCL crystallinity are more notorious, and, when PUs with the same PCL length are compared, PCL crystallinity is increasingly reduced when the PLLA content in the tri-block is increased (see for example entries PU1, PU4 and PU7 in Table 3). This reduction is also dependent on the PCL block length, with a drastic reduction for PCL2000 (compare T5 in Table 2 with PU5 in Table 3), moderate reduction for PCL8000 (compare T7 in Table 2 with PU7 in Table 3), and intermediate reduction for PCL4000 (compare T3 in Table 2 with PU3 in Table 3). Thus, the multi-block formation by the reaction of the tri-blocks with a diisocyanate reduced the PCL crystallinity, with increased effects at higher PLLA content and at shorter PCL length.
 | ||
| Fig. 9 Melting temperature (Tm) and degree of crystallinity (Xc) of poly(ester-urethanes) vs. molecular weight of PCL (a and b) and PLLA (c and d) blocks respectively. | ||
Similar tendencies were found for PLLA blocks. The PLLA melting point is slightly reduced (Fig. 9c) by poly(ester-urethane) formation but it follows the same tendency than for the tri-block precursors. However, the PLLA crystallinity is strongly affected in the poly(ester-urethane)s (Fig. 9d). PLLA crystallinity in PUs based on PCL2000 and PCL4000 is negligible and only PUs based on PCL8000 crystallize significantly, indicating an important different behaviour with respect to the corresponding tri-block copolymers. Moreover, analysing the PUs behaviour, (Fig. 9, Table 3) a maximum in the PCL crystallinity has been reached at the smaller PLLA molecular weight. This fact can be due to the higher concentration of the urethane bonds at smaller PLLA molecular weight. However the degrees of crystallinity shown by poly(ester-urethane)s are lower than the values obtained for the corresponding tri-blocks due to mobility restrictions imposed by the urethane linkages and the increase in the molecular weight. When PU4 (based on PCL8000) and PU6 (based on PCL4000) are compared, PU6 has longer PLLA blocks than PU4, and therefore a higher crystallinity was expected while, indeed, PU6 is almost amorphous. However, if the total molecular weight of the tri-block is evaluated, PU6 is shorter than PU4 (7375 g mol−1 vs. 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 393 g mol−1) and therefore, urethane groups concentration in PU6 is higher than in PU4, leading to a higher concentration of hydrogen bonds and a stronger restriction of the chain mobility. These facts can be confirmed by further evaluation of the values of degree of crystallinity as well as of wt% of HDI reported in the Table 3. In fact, PU4 presents 1.59 wt% HDI and a degree of crystallinity of 24%. PU7, with a 1.11 wt% HDI shows a degree of crystallinity of 38% and PU9 with a 0.68 wt% HDI present a degree of crystallinity of 13%. So, it is possible to conclude that the crystallisation restrictions must be considered as determined by complex combined effects of the presence of the urethane groups and of the corresponding hydrogen bonds.
393 g mol−1) and therefore, urethane groups concentration in PU6 is higher than in PU4, leading to a higher concentration of hydrogen bonds and a stronger restriction of the chain mobility. These facts can be confirmed by further evaluation of the values of degree of crystallinity as well as of wt% of HDI reported in the Table 3. In fact, PU4 presents 1.59 wt% HDI and a degree of crystallinity of 24%. PU7, with a 1.11 wt% HDI shows a degree of crystallinity of 38% and PU9 with a 0.68 wt% HDI present a degree of crystallinity of 13%. So, it is possible to conclude that the crystallisation restrictions must be considered as determined by complex combined effects of the presence of the urethane groups and of the corresponding hydrogen bonds.
Finally, the values of the Tg of the synthesized poly(ester-urethane)s are reported in Fig. 10. They can be grouped in three sets depending on the crystalline/amorphous nature of both blocks. In general the Tg values are slightly higher for the poly(ester-urethane)s compared with their initial corresponding tri-blocks. This fact confirms the contribution of the urethane and hydrogen bonds to the stiffness of the synthesized materials compared to the initial tri-blocks. Moreover, taken into account the melting temperature of PCL and PLLA blocks for the tri-blocks and for the corresponding crystalline poly(ester-urethane)s, it should be noted that the transition temperatures of the poly(ester-urethane)s are shifted with respect to the tri-blocks. For example T6 shows a TmPCL of 43 °C and a TmPLLA of 126 °C and a Tg of −58 °C. Its corresponding U6 shows a TmPCL of 45 °C and a TmPLLA of 118 °C and a Tg of −35 °C. That is, the transition temperatures are closer in the PUs than in the corresponding three block copolymers. Although the difference in the melting temperatures is relatively small for both the PCL and the PLLA blocks, it probably indicates smaller and more imperfect crystals formed during the crystallization of the PUs. The change in the Tg of PCL towards higher values can be attributed to the higher miscibility of the blocks in the PU respect to the tri-block copolymer. Same behaviour is found for the couple of materials T4/PU4 and T7/PU7.
SAXS characterization
The Small Angle X-ray Scattering (SAXS) experiments confirm the existence of a phase-separated morphology in the tri-block copolymers and in the corresponding poly(ester-urethane)s and allow for the determination of the Tm of both PCL and PLLA blocks. From SAXS experiments, two parameters, the long period (L) that defines the mean distance between the phases and the relative Invariant (Q′) that is related to the extent of phase separation, are obtained. L is calculated using the equation 2π/q, where q is the scattering vector corresponding to a maximum in intensity, and Q′ is calculated as the value of the integral below the curve Iq2 vs. q.The SAXS spectra are represented in terms of Iq2 vs. q, because this representation is more efficient to detect changes than the representation of I vs. q. The calculated value for L from the Iq2 vs. q representation is different from the calculated value from the I vs. q representation, but it is correct when the morphology of the phase-separated system is lamellar, as it is the case for PCL and PLLA.33,34 In Fig. 11 the scattering curves at different temperatures of three selected samples are represented: T2 with only the PCL-block crystalline, T7 with both blocks able to crystallize and T11 with only the PLLA-block crystalline. The first one shows a peak at room temperature that disappears when the temperature is increased and PCL blocks melt. After PCL melting, no scattering is obtained, indicating that the copolymer forms a homogeneous melt. For T7, the maximum of the scattering peak changes in shape at 60 °C, corresponding to the Tm of PCL, in fact, the peak becomes sharper with increasing temperature indicating that there is a lower dispersity on the size of the remaining PLLA crystals. At higher temperature, when the melting of PLLA is reached, the scattering peak disappears completely leaving a homogeneous melt. For the T11, a change in q and in Q′ is found when the temperature reached 60 °C. This phenomenon cannot be due to PCL melting but is related to a cold crystallisation process of PLLA which, in agreement with the DSC curves, taking place at this temperature, Fig. 9. Also in this case, at higher temperature, when the melting of PLLA is reached, the scattering peak disappears completely leaving a homogeneous melt. All the tri-blocks synthesized with only PCL crystalline behave as the copolymer T2; those with both blocks crystalline behave as the copolymer T7 and those with only PLLA crystalline behave as the copolymer T11. The behaviour for these tri-block copolymers was similar to that found for di-block copolymers.14
Similar SAXS experiments were performed for the synthesized poly(ester-urethane)s. The same changes in q and Q′ were found when melting of PCL and PLLA was reached. Moreover, also in this case, when PLLA blocks melted, no scattering was detected, showing that also the poly(ester-urethane)s form a homogeneous melt. Furthermore, for all the materials studied here no peaks for nanostructuration of the blocks in more ordered morphologies at long distances have been detected from SAXS analysis, as well as from TEM images (data not shown). In Fig. 12 the spectra of all the synthesized tri-block copolymers and their poly(ester-urethane)s at two different temperatures, 30 °C and 70 °C, are represented. In the case of tri-block copolymers at 30 °C, Fig. 12a, all of them are crystalline and show a broad peak. The position of the peak is around 0.5 nm−1 for all copolymers except for the copolymers with high content of PLLA, where the position is around 0.6 nm−1. T11 shows the peak at lower position, indicating that the polymer cannot crystallize during the sample preparation. However at 70 °C, Fig. 12b, the peak of T11 moves to higher values, as shown in Fig. 11, due to the cold crystallisation effect. On the other hand the polymers with a PLLA amorphous block do not show the peak at 70 °C indicating that the system is completely molten and homogenous at this temperature. Finally, for the tri-block copolymers with both crystalline blocks at this temperature the peak shifts to lower values of q in all cases.
 | ||
| Fig. 12 SAXS curves of tri-block copolymers at: (a) 30 °C and (b) 70 °C and poly(ester-urethane)s: (c) 30 °C and (d) 70 °C. | ||
Differences between tri-block copolymers and poly(ester-urethane)s are revealed by SAXS experiments, as reported in Fig. 12c and d. It is worth to note that the spectrum at 70 °C of the sample PU5 with amorphous PLLA blocks, shows no peaks, while the initial tri-block, T5, is crystalline.
In order to better visualize these differences the values of the long period for the tri-blocks as well as for the poly(ester-urethane)s are summarized in Table 4, at two different temperatures, such as 30 °C and 70 °C, that is, below and above the melting temperature of PCL. Table 4 shows that the long period L in the tri-blocks having both crystalline blocks, increases when PCL is in the molten state. Only for T11, the long period value decreases with increasing temperature, thus indicating the cold crystallisation phenomenon as observed in the second cycle of DSC. However, comparing T11 with PU11, it is worth to note that in PU11 this behaviour is not observed, and the long period is the same at 30 °C and at 70 °C. This difference can be attributed to the different sample processing conditions, because the tri-blocks have been molten and cooled at room temperature in an aluminum pan, while the poly(ester-urethane)s have been wrapped in aluminum foil without any thermal treatments before SAXS spectra were recorded. For the tri-blocks, L ranges from 9.1 to 21.7 nm at 30 °C and from 11.5 to 21.5 nm at 70 °C. The same range of values has been found for di-block copolymers.14 For the poly(ester-urethane)s, L ranges from 14.5 to 24.2 nm at 30 °C and from 15.2 to 28.8 nm at 70 °C, and thus comparing the tri-blocks with their corresponding PUs, the latter present higher values. No trend in L values with PCL length or PLLA length was found neither for the tri-blocks nor for the poly(ester-urethane)s.
| Sample | PCL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PLLA PLLA |
Long period (tri-block) | Long period (poly(ester-urethane)) | ||
|---|---|---|---|---|---|
| T = 30 °C | T = 70 °C | T = 30 °C | T = 70 °C | ||
| 1 | 92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 08 08 |
13.6 | — | 15.5 | — |
| 2 | 89![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 11 11 |
12.9 | — | 18.0 | — |
| 3 | 84![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16 16 |
13.4 | — | 21.0 | — |
| 4 | 77![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 23 23 |
12.3 | 21.5 | 14.5 | 28.8 |
| 5 | 73![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 27 27 |
14.6 | — | 24.2 | — |
| 6 | 54![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 46 46 |
13.4 | 14.7 | 17.5 | 17.8 |
| 7 | 54![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 46 46 |
18.3 | 15.5 | 14.5 | 21.5 |
| 8 | 34![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 66 66 |
10.1 | 11.5 | 15.0 | 15.2 |
| 9 | 33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 67 67 |
11.1 | 16.3 | 17.5 | 17.5 |
| 10 | 32![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 68 68 |
9.1 | 14.1 | 16.0 | 16.0 |
| 11 | 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 82 82 |
21.7 | 14.9 | 17.3 | 17.3 |
The variation of the long period L and invariant Q′ with temperature and the thermograms obtained by differential scanning calorimetry for two representative materials are compared in Fig. 13a and b. In particular, Fig. 13a corresponds to the sample T11. In this case it is possible to confirm the cold crystallisation process of this sample by three ways: the exothermic peak from DSC, the change in the invariant and the change in the long period, which reveals the formation of new crystals reducing the separation between them. The melting behaviour of PLLA is similar comparing the results obtained by using both techniques: the endothermic peak of DSC appears at the same range of temperature at which the long period growth to infinity and the invariant Q′ starts to reduce until the scattering disappearance indicating that at the end, the blocks are mixed in a homogeneous melt. Fig. 13b shows the poly(ester-urethane) PU4 with both crystalline PCL and PLLA blocks. In this sample when the PCL melts, the long period reaches higher values due to the increase of the distance between crystals. After the melting of PCL, q values show a reduction until the PLLA begins to melt, and then the long period starts to increase and the relative invariant Q′ starts to decrease until the scattering peak disappears, indicating that a homogeneous melt is formed. Finally, it can be observed that for both materials there is a good agreement between the crystallisation data obtained by the two different characterization techniques applied (DSC and SAXS).
 | ||
| Fig. 13 Comparison of DSC data and SAXS data for: (a) tri-block T11 and (b) poly(ester-urethane) PU4. | ||
Mechanical properties
Tensile tests of all synthesized poly(ester-urethane)s were performed except for the sample PU11 due to its high fragility. The stress–strain curves obtained for all the samples analysed are reported in Fig. 14, divided in three graphs as a function of the nominal molecular weight of the PCL block. In these figures the curves with the dashed lines correspond to the polymers with PLLA-blocks completely amorphous. The values of the elastic modulus, elongation at break and maximum stress are summarized in Table 5. Regarding Fig. 14 results, the polymers show the typical curve for semi-crystalline polymers characterized by yielding and ductility. In general, the PCL block influences the ductile behaviour while the crystalline PLLA block influences the brittle behaviour. In particular, the copolymers characterized by the presence of amorphous PLLA-blocks (PU2, PU3 and PU5) and those having the values of PLLA-block crystallinity range from 0% to 1% in Table 3 (PU6, PU8 and PU10), show the highest elongation at break. Increasing the PLLA content, the poly(ester-urethane)s become stiffer and brittle, due to the presence of PLLA crystals, showing lower values of the elongation at break (PU4, PU6, PU7 and PU9). On the other hand the poly(ester-urethane)s reported in Fig. 14c, which have been synthesized using the PCL diol of 8000 g mol−1, have the highest elastic moduli values but the elongation at break is reduced and these polymers are more brittle than the others. Indeed, we expected that the behaviour of poly(ester-urethane)s rich in PCL and with the PCL8000 (PU1 and PU4) were similar to PU2, PU3 or PU5. So, the results obtained on the mechanical properties indicate that these poly(ester-urethanes)s did not reach a large molecular weight in the polycondensation reaction. The main reason for these low molecular weight obtained can be attributed to the difficulty to define the precise stoichiometric ratio for the polycondensation reaction, as a consequence of the high error in the calculation of the molecular weight of the initial tri-block copolymer by 1H NMR which is not accurate for the computation of high molecular weights.| Sample | PCL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PLLA PLLA |
Elastic modulus (MPa) | Maximun stress (MPa) | Elongation at break (%) |
|---|---|---|---|---|
| PU1 | 92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 08 08 |
210 ± 30 | 14 ± 1 | 10.0 ± 1.5 |
| PU2 | 89![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 11 11 |
116 ± 12 | 29 ± 5 | 1631 ± 127 |
| PU3 | 84![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16 16 |
93 ± 4 | 49 ± 4 | 2020 ± 80 |
| PU4 | 77![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 23 23 |
230 ± 10 | 17.5 ± 0.1 | 25 ± 4 |
| PU5 | 73![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 27 27 |
123 ± 5 | 8.7 ± 1.8 | 800 ± 200 |
| PU6 | 54![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 46 46 |
29 ± 2 | 16.3 ± 0.6 | 1360 ± 32 |
| PU7 | 54![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 46 46 |
203 ± 9 | 14.6 ± 0.2 | 30 ± 5 |
| PU8 | 34![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 66 66 |
105 ± 2 | 12.8 ± 0.4 | 537 ± 25 |
| PU9 | 33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 67 67 |
183 ± 7 | 14.6 ± 0.6 | 400 ± 200 |
| PU10 | 32![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 68 68 |
110 ± 20 | 11.0 ± 1.5 | 70 ± 30 |
| PU11 | 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 82 82 |
— | — | — |
Conclusions
Eleven linear tri-block copolymers, PLLA-b-PCL-b-PLLA, have been synthesized via ring opening polymerization of L-lactide with three different PCL diols, varying the molecular weight of the blocks as well as the relative content of each block. 1H NMR proved that the desired tri-block structure was obtained and allowed for the determination of the molecular weight and the composition of the tri-blocks. From these tri-blocks, poly(ester-urethane)s were synthesized using HDI. The crystallisation behaviour of the tri-block copolymers and the poly(ester-urethane)s was studied by DSC and SAXS experiments.In the tri-block copolymers the crystallinity of the PCL is strongly influenced by the presence and the nature of PLLA. In particular the presence of PLLA crystals is related to a reduction of the crystal size as well as the degree of crystallinity of the PCL blocks. The effect of the presence of PLLA crystals on the PCL crystallinity is stronger in tri-blocks than for PCL-PLLA di-blocks. The crystallinity of PLLA on the tri-blocks is not influenced by the presence of PCL depending only on the molecular weight of the PLLA block, with no significant difference with PCL-PLLA di-blocks.
Poly(ester-urethane)s showed less degree of crystallinity of both blocks than the initial tri-block precursors due to the restrictions imposed by the multi-block segmented structure of the final poly(ester-urethane). The crystallisation restrictions must be considered as determined by complex combined effects based on the presence of the urethane groups and on the corresponding hydrogen bonds. Moreover, the shift in the Tm values of both crystalline blocks towards closer values in the poly(ester-urethane)s can be attributed to the higher miscibility of the blocks in the PU with respect to the tri-block copolymer. In addition, the values of Tg for the PCL block are slightly higher for the poly(ester-urethane)s compared with their initial corresponding tri-blocks, as expected, thus confirming the contribution of the urethane and hydrogen bonds to the stiffness of the PUs.
SAXS experiments confirmed the phase separated structure of the tri-blocks and the poly(ester-urethane)s. Through the variation of the long period and the relative invariant with temperature, the melting processes of the two blocks has been studied and compared with the results obtained by DSC, showing good agreement obtained with both techniques. In all cases, when all crystals were melted, the copolymers formed a homogeneous melt.
The mechanical properties are well related with the crystalline nature of the poly(ester-urethane)s. In fact, PUs with the amorphous PLLA show the higher elongations at break. Increasing the crystallinity of PLLA-block the materials are more brittle and reach higher values of the elastic modulus. However, the difficulties with the stoichiometry in the synthesis procedure for the poly(ester-urethane)s obtained from the higher molecular weight tri-block copolymers, can affect dramatically the final molecular weight of the polymer and, consequently, its mechanical properties.
The results of this study demonstrated that the PLLA-b-PCL-b-PLLA tri-block copolymers and their corresponding poly(ester-urethane)s studied here can be tailored with interesting thermal and mechanical properties and are potential candidates for shape memory materials.
Acknowledgements
We are indebted to the Spanish Ministry of Science and Innovation (MICINN) for their economic support (MAT2010-21494-C03-03). LP acknowledges also, the support of a JAEdoc grant from CSIC cofinanced by EU.Notes and references
- H. Seyednejad, A. H. Ghassemi, C. F. van Nostrum, T. Vermonden and W. E. Hennink, J. Controlled Release, 2011, 152, 168 CrossRef CAS PubMed.
- R. Langer, Nature, 1998, 392, 5 CAS.
- B. Jeong, Y. H. Bae, D. S. Lee and S. W. Kim, Nature, 1997, 388, 860 CrossRef CAS PubMed.
- B. M. Chamberlain, B. A. Jazdzewski, M. Pink, M. A. Hillmyer and W. B. Tolman, Macromolecules, 2000, 33, 3970 CrossRef CAS.
- C. Wischke, A. T. Neffe, S. Steuer and A. Lendlein, J. Controlled Release, 2009, 138, 243 CrossRef CAS PubMed.
- S. Neuss, I. Blomenkamp, R. Stainforth, D. Boltersdorf, M. Jansen, N. Butz, A. Perez-Bouza and R. Knuchel, Biomaterials, 2009, 30, 1697 CrossRef CAS PubMed.
- A. Lendlein and R. Langer, Science, 2002, 296, 1673 CrossRef PubMed.
- R. Auras, B. Harte and S. Selke, J. Appl. Polym. Sci., 2004, 92, 1790 CrossRef CAS.
- D. W. Grijpma and A. J. Pennings, Polym. Bull., 1991, 25, 335 CrossRef CAS.
- G. Maglio, A. Migliozzi and R. Palumbo, Polymer, 2003, 44, 369 CrossRef CAS.
- O. Jeon, S. H. Lee, S. H. Kim, Y. M. Lee and Y. H. Kim, Macromolecules, 2003, 36, 5585 CrossRef CAS.
- G. Kale, R. Auras and S. P. Singh, J. Polym. Environ., 2006, 14, 317 CrossRef CAS PubMed.
- R. V. Castillo, A. J. Müller, J. M. Raquez and P. Dubois, Macromolecules, 2010, 43, 4149 CrossRef CAS.
- L. Peponi, I. Navarro-Baena, J. E. Baez, J. M. Kenny and A. Marcos-Fernandez, Polymer, 2012, 53, 4561 CrossRef CAS PubMed.
- J. Fernandez, A. Etxeberria and J. R. Sarasua, J. Mech. Behav. Biomed. Mater., 2012, 9, 100 CrossRef CAS PubMed.
- D. Cohn and A. F. Salomon, Biomaterials, 2005, 26, 2297 CrossRef CAS PubMed.
- V. Vilay, M. Mariatti, Z. Ahmad, K. Pasomsouk and M. Todo, Mater. Sci. Eng., A, 2010, 527, 6930 CrossRef PubMed.
- X. L. Lu, Z. J. Sun, W. Cai and Z. Y. Gao, J. Mater. Sci.: Mater. Med., 2008, 19, 395 CrossRef CAS PubMed.
- L. Peponi, I. Navarro-Baena, A. Sonseca, E. Gimenez, A. Marcos-Fernandez and J. M. Kenny, Eur. Polym. J., 2013, 49, 893 CrossRef CAS PubMed.
- L. L. Zhang, C. D. Xiong and X. M. Deng, J. Appl. Polym. Sci., 1995, 56, 103 CrossRef CAS.
- L. Peponi, A. Marcos-Fernandez and J. M. Kenny, Nanoscale Res. Lett., 2012, 7, 1 CrossRef PubMed.
- J. Fernández, A. Larrañaga, A. Etxeberría and J. R. Sarasua, Polym. Degrad. Stab., 2013, 98, 481 CrossRef PubMed.
- M. Bero, J. Kasperczyk and G. Adamus, Macromol. Chem. Phys., 1993, 194, 907 CrossRef CAS.
- G. Kister, G. Cassanas and M. Vert, Polymer, 1998, 39, 267 CrossRef CAS.
- T. Elzein, M. Nasser-Eddine, C. Delaite, S. Bistac and P. Dumas, J. Colloid Interface Sci., 2004, 273, 381 CrossRef CAS PubMed.
- J. E. Baéz, D. Ramírez, J. L. Valentín and A. Marcos-Fernandez, Macromolecules, 2012, 45, 6966 CrossRef.
- J. Tuominen, J. Kylma and J. Seppala, Polymer, 2002, 43, 3 CrossRef CAS.
- Y. Kobori, T. Iwata, Y. Doi and H. Abe, Biomacromolecules, 2004, 5, 530 CrossRef CAS PubMed.
- D. W. Van Krevelen, in Properties of polymers, Elsevier, Amsterdam, 3rd edn, 1990, ch. 5 Search PubMed.
- J. R. Sarasua, R. E. Prud'homme, M. Wisniewski, A. Le Borgne and N. Spassky, Macromolecules, 1998, 31, 3895 CrossRef CAS.
- A. Sonseca, L. Peponi, O. Sahuquillo, J. M. Kenny and E. Giménez, Polym. Degrad. Stab., 2012, 97, 2052 CrossRef CAS PubMed.
- J. Ahmed, J. X. Zhang, Z. Song and S. K. Varshney, J. Therm. Anal. Calorim., 2009, 95, 957 CrossRef CAS.
- R. V. Castillo and A. J. Müller, Prog. Polym. Sci., 2009, 34, 516 CrossRef CAS PubMed.
- I. W. Hamley, P. Parras, V. Castelletto, R. V. Castillo, A. J. Müller, E. Pollet, P. Dubois and C. M. Martin, Macromol. Chem. Phys., 2006, 207, 941 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2014 |