Glycine-modified polyamidoamine dendrimers: synthesis and structural characterization using nuclear magnetic resonance, ion-mobility mass spectrometry and capillary electrophoresis†
Emma-Dune
Leriche
a,
Carlos
Afonso
a,
Catherine M.
Lange
a,
Martin C.
Grossel
b,
Lina
Truong
a,
Gael
Coadou
a,
Hassan
Oulyadi
a and
Corinne
Loutelier-Bourhis
*a
aNormandie Université, COBRA, UMR6014 and FR3038, Université de Rouen; INSA de Rouen; CNRS, IRCOF, 1 rue Tesnière, 76821 Mont-Saint-Aignan Cedex, France. E-mail: corinne.loutelier@univ-rouen.fr; Fax: +33(0)2 3552 2441; Tel: +33(0)2 3552 2924
bUniversity of Southampton, Chemistry Department, Highfield, Southampton, Hants SO17 1BJ, UK. E-mail: M.C.Grossel@soton.ac.ukc; Fax: +41(0) 23 8059 3781; Tel: +41(0) 23 8059 3153
First published on 31st October 2013
Abstract
We present here the preparation and the structural characterization of first-generation ammonia-cored polyamidoamine (PAMAM) dendrimers modified with glycine residues. Chemical modification of the dendrimer was done to increase the biocompatibility of these compounds, known to be effective delivery agents for drugs or genes. Fully modified PAMAM [Gly6G1(N)] on the one hand and partially modified [GlynG1(N), with n = 0 to 6)] on the other hand were obtained depending on the experimental conditions. The resulting modified PAMAM dendrimers have to be cautiously characterized to understand and interpret their physico-chemical and biochemical properties as well as to control their chemical design. The structural characterization was carried out using ion mobility spectrometry-mass spectrometry (IM-MS), multistage tandem mass spectrometry (MSn), accurate mass measurements by high resolution–mass spectrometry (HR-MS), two dimensional nuclear magnetic resonance (NMR) and capillary electrophoresis (CE). Characteristic fragmentation patterns for these compounds were obtained from ESI/MSn (with n = 2 to 4) experiments. IM-MS and CE analysis showed that a single component was mainly obtained for the complete grafting experimental conditions while a distribution of oligomers was produced for partially grafted products. The physical separation of GlynG1(N) oligomer ions was achieved in the gas phase (IM-MS) as well as in the condensed phase (CE). Besides, the collision cross sections (CCS) were estimated by IM-MS and compared to theoretical values. Then, the glycine grafting yield for GlynG1(N) PAMAM was determined by both NMR and IM-MS experiments.
Introduction
PAMAM dendrimers are a class of synthetic polycationic polymers showing a unique spatial structure of globular shape. They are composed of three architectural elements: a core, repeating units and terminal functional groups.1 The reiteration of monomer addition yields dendrimers of higher generation. The size, shape and surface functionalities of these compounds are well-controlled and their synthesis yields narrow molecular weight distributions, if prepared carefully. It has been reported that high generations of PAMAM dendrimers can form complexes with DNA molecules offering the possibility to use such dendrimers as non-viral transfection systems for gene therapy.2 However, the cationic surface of PAMAMs, appropriate for binding chemical entities, induces cytotoxicity limiting their biomedical applications. Therefore, various chemical modifications of the PAMAM surface have been carried out to both decrease the toxic effects and increase transfection efficiency as drug and gene carrier.3,4 Thus, the grafting of various amino acid residues at PAMAM chain ends to form heterobifunctional dendrimers5 and reduced surface positive charge density was envisaged;6 phenylalanine- and arginine-grafted-PAMAM dendrimers showed lower cytotoxity and higher gene delivery activity than unmodified PAMAMs or the commercial transfection agent polyethylenimine (PEI).6,7 The biological activities of these modified PAMAMs have been widely evaluated and some analytical studies using gel electrophoresis, gel permeation, NMR and HPLC analyses permitted to study distributions of modified PAMAM.4,6–8 Thus, G5 PAMAM bioconjugates as well as dendrimer nanoparticule-ligands distributions could be quantitatively assessed.8 On the other hand, unmodified small generation PAMAM dendrimers have been extensively studied by infra-red, ultra-violet/visible, nuclear magnetic resonance (NMR) and mass spectrometry (MS) techniques.9–13 In particular, MS appeared to be useful for the full-characterization of small generation PAMAM dendrimers using either electrospray (ESI) or matrix assisted laser desorption ionization (MALDI) techniques.10–13To understand the molecular mechanisms involved in DNA/modified-PAMAM complexes formation, the study of non covalent interactions implicated in such dendriplexes formation can be envisaged on small dendrimer models. Thus, even if small generations of PAMAM are not expected to be applied in biomedical applications, we intend to design amino acid-modified G1 PAMAM as a good starting point for optimization and monitoring of the chemical design as well as for analytical methodology implementation.
Thus, in the present study, we propose to chemically modify ammonia-cored PAMAM G1(N) dendrimers with glycine residues and to present the structural characterization of the resulting modified-PAMAMs. Glycine was chosen as a model amino acid to optimize and control the surface chemical modification. Moreover, the glycine can be considered as a potential spacer for further conjugation with functional groups such as drugs and targeting moieties. The amino acid grafting step was optimized to yield fully surface-modified PAMAM G1(N) [Gly6G1(N)] in the one hand, and partially modified PAMAM G1(N) [GlynG1(N), n = 0 to 6] in the other hand.
A thorough structural characterization of our modified PAMAM dendrimers involving mono- and two-dimensional NMR analyses, multi-stage mass spectrometry, accurate mass measurements, ion-mobility coupled to mass spectrometry (IM-MS) and capillary electrophoresis (CE) is presented. CE and IM-MS were proposed as alternative techniques for cationic PAMAM separation, appending literature data.13,14 Moreover, IM-MS is well-known to present the advantage to permit in a single run the separation and the identification of ionic species in the millisecond time-scale from direct infusion of samples (a prior LC separation being not necessarily required). It is the first time to our knowledge that such comprehensive structural analysis is reported.
Results and discussion
1. PAMAM synthesis, chemical modification and deprotection
The PAMAM G1(N) was synthesized according to Tomalia's divergent approach,15 based on combination of acrylate Michael addition and amidation chemistry. PAMAM G1(N) was then conjugated with tert-butoxycarbonyl protected glycine (N-Boc-glycine) in order to obtain, in a first step, N-BocGly6G1(N) in the one hand and N-BocGlynG1(N) in the other hand (step (a) of Scheme 1). The conjugation of the primary amine groups of PAMAM surface with N-Boc-glycine involved O-(benzotriazol-1-yl)-N,N,N′,N′- tetramethyluronium hexafluoro-phosphate (HBTU) and 1-hydroxybenzotriazole hydrate (HOBt hydrate) as coupling agents to activate the carboxylic acid groups of the protected glycine.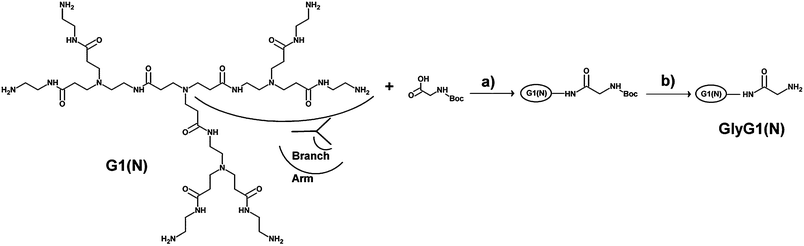 | ||
| Scheme 1 Synthesis of glycine modified PAMAM GlynG1(N). Reagents: (a) HBTU, HOBt, DIPEA in DMF/H2O (b) H3PO4 in TFE. | ||
In the case of N-BocGly6G1(N), a slight excess of N-Boc-glycine was necessary to obtain fully modified PAMAM.
In the other hand, it appeared necessary to adjust and optimize the HBTU/HOBt coupling method to obtain a distribution of partially modified N-Boc-GlynG1(N) (with n = 0 to 6) since the sole diminution of N-Boc-glycine/PAMAM ratio only afforded a mixture of both unmodified and fully modified PAMAM (data not shown). The new grafting strategy involved the prior activation of N-Boc-glycine with HBTU/HOBt coupling agents followed by the slow dropwise introduction of activated N-Boc-glycine to a solution of PAMAM G1(N) in order to avoid high concentration of the activated amino acid in the reaction medium. Under such conditions, a distribution of differently modified PAMAM could be obtained. Note that, in the case of N-BocGly1G1(N), N-BocGly5G1(N) and N-BocGly6G1(N) PAMAM dendrimers, a single regioisomeric structure can be formed while the other partially modified samples (Gly2G1(N) to Gly4G1(N)) can yield two different isomers depending on the position of the grafted glycine residues (onto the branches of the same arm or on different arms) (Scheme 1S of the ESI†).
The second and final step of modified PAMAM preparation consisted in N-Boc groups deprotection (Step (b) of Scheme 1).
The classical approach uses trifluoroacetic acid (TFA, pKa 0.3). However, such approach induced the hydrolysis of the amide linkage of our modified PAMAM dendrimers, even when mild conditions were used, with consecutive losses of grafted glycine residues. Thus, we developed a new strategy of deprotection inspired by the approach of Li et al.16,17 These authors have reported the use of aqueous phosphoric acid (85% Wt, pKa1 2.15) as an alternative reagent for N-Boc groups deprotection. In our case, the use of H3PO4 in 2,2,2-trifluoroethanol (TFE) afforded the Gly6G1(N) and GlynG1(N) PAMAM dendrimers in quantitative yields.
The resulting partially and totally modified PAMAM were then structurally characterized using NMR, ESI-MSn, IM-MS and CE techniques after dialysis.
2. Structural characterization of glycine modified-PAMAM dendrimers
Characteristic spin systems were first identified in each structure from 1H–1H COSY and 1H–13C HMQC NMR spectra. Then, they were assigned to specific locations in the structure by the observation of multiple bond 1H–13C heteronuclear correlation (HMBC) between resonances of sequentially adjacent spin systems (Fig. 2B). This methodology permitted to obtain the complete 1H NMR signal assignments for Gly6G1(N) (Fig. 1C).
As previously mentioned, the proton signals between 2.50 and 3.50 ppm were assigned to the different methylene groups of the PAMAM G1(N) moiety while the signal at 3.73 ppm corresponded to the methylene (H-17) protons of the grafted glycine. Indeed, this signal was unambiguously identified by the HMBC experiment which showed the 1H–13C correlation between glycine and dendrimer moieties providing evidence for the grafting of the amino acid residues onto the PAMAM surface; the carbonyl carbon C-16 of the glycine substituent shows three bonds 1H–13C correlation with the methylene protons H-14 of the dendrimer moiety in addition to the expected 1H–13C two bonds correlation with the methylene protons H-17 of the glycine residue (dotted lines in Fig. 2B).
In addition, the 1H NMR data allowed the determination of the average number of glycine residues attached on the PAMAM G1(N) surface. The integration ratio of the H-17 protons at 3.73 ppm to that of the summed integral for the protons in the 2.35–3.50 ppm region indicated 100% conversion of amino groups meaning that six molecules of glycine were grafted to the six amino groups of the G1(N) molecule, as expected for Gly6G1(N).
The structure of GlynG1(N) was elucidated from the combination of COSY, HMQC and HMBC experiments (data not shown), as afore mentioned, and by comparison with NMR data previously acquired for both the unmodified and fully modified PAMAM G1(N) samples. Thus, the methylene protons (H-17) of the attached glycine were observed at 3.56 ppm while the cluster of signals between 2.35 and 3.50 ppm correspond to the methylene protons of PAMAM branching units (Fig. 1B). Comparison of the Fig. 1B with both Fig. 1A and C provide evidence that the partially modified dendrimers contained a mixture of unreacted and modified branch termini. Indeed, the different signals corresponding to protons of both the unmodified and modified branches were detected in Fig. 1B, the H-13 and H-14 methylene protons of the modified branches appeared at δ 3.29 and 3.32 ppm, respectively, whilst the methylene protons of the unmodified end branches H-13′ and H-14′ respectively were at δ 3.46 and 3.10 ppm. Chemical shift differences were also observed for the methylene protons H-9, H-9′ and H-10, H-10′.
It is important to note that by comparing the three 1H NMR spectra, strong changes in the signals characteristic for each system were observed. Indeed, from Fig. 1A to C, one can notice an increase of the downfield shifts and a broadening of the peaks for the whole spin systems. These changes were due to pH variations, as we established by studying the chemical shifts of unmodified PAMAM with the decrease of sample pH (Figure 1S of the ESI†). Such 1H NMR chemical shift changes had also been reported for model bases during titration with acids.20 The pH decrease between the unmodified, partially and totally modified PAMAM samples could easily be explained by the use of phosphoric acid during the deprotection step.
The average number of grafted glycine was estimated from 1H NMR data as previously described; 25% conversion of the amino groups was deduced from the integration ratio of the protons at 3.56 ppm to that of the summed protons within the 2.35–3.50 ppm region (Figure S2 of the ESI†).
| Compound | Elemental compositionsa | m/z exp. | m/z calcdb | Error (ppm) | Ion |
|---|---|---|---|---|---|
| a Elemental compositions of the neutral species. b Monoisotopic values calculated with the Waters MassLynx software. | |||||
| G1(N) | C45H93N19O9 | 544.8614 | 544.8594 | 3.6 | [M + 2Na]2+ |
| Gly1G1(N) | C47H96N20O10 | 573.3701 | 573.3707 | −1.0 | [M + 2Na]2+ |
| Gly2G1(N) | C49H99N21O11 | 601.8814 | 601.8814 | 0.0 | [M + 2Na]2+ |
| Gly3G1(N) | C51H102N22O12 | 630.3920 | 630.3922 | −0.3 | [M + 2Na]2+ |
| Gly4G1(N) | C53H105N23O13 | 658.9033 | 658.9029 | 0.6 | [M + 2Na]2+ |
| Gly5G1(N) | C55H108N24O14 | 687.4142 | 687.4136 | 0.9 | [M + 2Na]2+ |
| Gly6G1(N) | C57H111N25O15 | 715.9253 | 715.9243 | 1.4 | [M + 2Na]2+ |
 | ||
| Fig. 4 ESI-MS2 mass spectra of [M + 2Na]2+ ion of (A) Gly6G1(N) (m/z 715.92) and (B) Gly3G1(N) (m/z 630.15). These data were acquired using the SYNAPT G2 mass spectrometer. | ||
Abundant [M + 2Na]2+ and [M + 3Na]3+ multiply charged ions of fully modified PAMAM Gly6G1(N) were respectively observed at m/z 715.92 and m/z 484.94 in the ESI mass spectrum (Fig. 3A). Their assignments were confirmed by accurate mass measurements (Table 1). Additional low-intensity ions were also detected at m/z 628.86 corresponding to the [M + 2Na]2+ ion of a defective modified structure since the Tomalia synthesis is known to yield structural defects.1 Thus, the m/z 628.86 corresponded to the cyclic defect with one missing ethylenediamine molecule (60 Da) and four grafted glycine residues (C51H197N21O13, m/z calculated: 628.8685; found: 628.8690 for [M + 2Na]2+ion). Multi-stage mass spectrometry carried out on the doubly-charged precursor ion [M + 2Na]2+ of Gly6G1(N) yielded product ions arising from neutral losses of modified branches (171 Da) and of a fully modified arm (456 Da) (Fig. 4A for MS2 data), in accordance with the systematic fragmentation pattern previously described for the unmodified PAMAM.11–13
The well-established fragmentation pathway involved retro-Michael rearrangements leading to successive losses of branches and arm(s). Such dissociation processes yielded the singly-charged product ions at m/z 1237.74, m/z 1066.64, m/z 952.60, m/z 781.47 and m/z 479.26 as well as doubly-charged product ions at m/z 630.37 and m/z 544.82 (Fig. 4A). Note that the ion at m/z 479.26 corresponds to the sodiated fully modified arm. In a similar way, the product ions at m/z 194.08 and m/z 308.17 correspond to sodiated modified branches containing one and two branch unit(s), respectively. These fragmentation pathways were confirmed by ESI-MS3 and MS4 experiments selecting the different product ions detected in Fig. 4A (data not shown).
The ESI mass spectrum of the partially modified PAMAMs showed a main distribution corresponding to the doubly-charged ions [M + 2Na]2+ of GlynG1(N) with n = 0 to 6 (Fig. 3B). The accurate mass measurements of these ions were consistent with the elemental composition of the various partially substituted GlynG1(N) derivatives (Table 1). Thus, the ESI mass spectrum displayed the different individual modified species and showed that partial surface-functionalisation had been achieved, the most abundant species corresponding to the grafting of one, two and three glycine residues. Minor distributions were also detected and corresponded to either [M + Na + H]2+ (♦) and [M + 2H]2+ (■) doubly-charged ions of GlynG1(N) or to defective structures [M + 2H]2+ ions (★) (Fig. 3B) for which elemental compositions were also determined by accurate mass measurements (data not shown).
ESI/MS2 experiments were performed selecting the doubly charged ion [M + 2Na]2+ of each partially modified PAMAM as the precursor ion. The fragmentation processes were analogous to those previously mentioned for the doubly charged ion of Gly6G1(N) although, in the case of partially modified PAMAM, neutral losses of either modified or unmodified branch(es) or arm(s) could be possible depending of the number of glycine residues grafted onto the PAMAM surface. Thus, the [M + 2Na]2+ ion (m/z 630.15) of partially modified PAMAM Gly3G1(N) dissociated via a retro-Michael rearrangement with the neutral losses of a modified branch (171 Da), an unmodified branch (114 Da), an unmodified arm (342 Da) and/or modified-arm(s) with one or two grafted glycine units (399 or 456 Da) (Fig. 4B).
The loss of 285 Da corresponding to a moiety containing two branches with only one grafted glycine (two branches + one glycine) could also occur consecutively to the elimination of one branch, modified or not. As previously observed in the case of Gly6G1(N), some product ions corresponding to the sodiated modified branch (two branches and one glycine) moiety and modified arms with one or two Gly were detected at m/z 194.08, 308.17, 422.24 and 479.26, respectively. The sodiated unmodified two-branches and an unmodified arm were detected at m/z 251.14 and 365.22, respectively. The presence of product ions at m/z 479.26 and 365.22 respectively indicated that two glycine residues were grafted onto the same arm while one arm remained unmodified. Such result is consistent with the presence of a Gly3G1(N) PAMAM isomer, which presents a totally modified arm, an unmodified arm and a third half-modified arm since, as previously mentioned in the chemical modification section, the grafting of three glycine residues could yield two different isomers for Gly3G1(N) PAMAM (Scheme S1 of the ESI†). The assumption of this structural isomer presenting two grafted glycine residues onto one arm and one glycine onto another arm was subsequently verified through MS3 and MS4 experiments which permitted determination of the relationship between the different product ions. In fact, the whole fragmentation pathways reported in the first generation product ion spectrum presented in Fig. 4B were deduced from several multi-stage mass spectrometry experiments. Therefore, the presence of m/z 781 and 895 in the MS3 spectrum “630.1 → 1237.8 → 2nd generation product ions” is consistent with the supposed structure because m/z 781.5 and 895.6 arose from the m/z 1237.8 intermediate ion with neutral losses of a totally modified arm (456 Da) and an unmodified arm (342 Da), respectively (Scheme S1-A of the ESI†). Moreover, the MS4 spectrum “630.1 → 1237.8 → 781.5 → 3rd generation product ions” showed m/z 439.2 and 382.2 that arose from m/z 781.5 with elimination of a half-modified arm (399 Da) and an unmodified arm (342 Da), respectively (Scheme S1-A of the ESI†). Therefore, unmodified, half-modified and totally modified arms constituted this isomer.
The presence of a second isomer of Gly3G1(N), formed during glycine grafting and carrying three half-modified arms (Scheme S1–B of the ESI†) was also evidenced because the “630.1 → 1237.8 → 2nd generation product ions” spectrum also showed an ion at m/z 838.5 ion which further dissociated into the m/z 439 ion as confirmed by the MS4 experiment “630.1 → 1237.8 → 838.5 → 3rd generation product ions” as well as the MS3 experiment “630.1 → 838.5 → 2nd generation product ions”. Both m/z 838 and m/z 439 arose from elimination of a half-modified arm indicating that three half-modified arms constituted this second isomer of Gly3G1(N) formed during glycine grafting (Figure S3 of the ESI†).
In the same way, the presence of two isomers for Gly2- and Gly4- G1(N), according to the distribution of the glycine residues onto the arms, was demonstrated through specific and appropriate MS3 and MS4 experiments.
Therefore, the charge density was expected to decrease when the number (n) of grafted glycine units increased, and consequently the electromigration time should increase.
The electropherogram acquired after injection of the partially modified PAMAM G1(N) showed the distribution of the differently grafted GlynG1(N) (Fig. 5A). Another injection of this sample spiked with unmodified PAMAM G1(N) showed an increase of the first peak (noted a in Fig. 5) therefore assigned to unmodified PAMAM G1(N). In consequence, the peaks (b) to (g) could be assigned to GlynG1(N) with n = 1 to 6, respectively, in agreement with the expected electromigration order. However, under our conditions, the CE separation was insufficient to resolve individual species impeding peak integration and the estimation of the level of amino group conversion.
The two-dimensional ion mobility vs. m/z correlation data of the partially modified PAMAMs showed three groups of multi-charged ions distributed on two main diagonals (Fig. 6). Doubly-charged ions ([M + 2Na]2+, [M + H + Na]2+ and [M + 2H]2+ ions) were aligned on a first diagonal whilst triply- and quadruply-charged ions were fitted with a second diagonal. Thus, doubly-charged species were well-separated from other multiply-charged ions of GlynG1(N) as also evidenced on the ion mobility spectrum (Fig. 5C). Focusing on the drift time distributions of doubly-charged ions which were selectively extracted, seven peaks could be differentiated (inset of Fig. 5C): these peaks labeled (a) to (g) could be respectively assigned to G1(N), Gly1G1(N), Gly2G1(N), Gly3G1(N), Gly4G1(N), Gly5G1(N) and Gly6G1(N) thanks to the orthogonal information of the m/z ratio. Then, comparing IM to CE data, identical migrating orders were obtained using both gas and condensate phase electromigration.
However, the IM analysis was in the millisecond time scale while the CE separation was performed in the minute time scale. Moreover, since our CE apparatus was not coupled to mass spectrometry, the separation of individual modified dendrimers was only achieved using the IM-MS approach involving the combination of orthogonal IM and MS data. It was therefore possible to estimate the average number of grafted glycine units by integrating the peaks extracted from the ion mobility spectrum of Fig. 5C; the ion mobility peak areas corresponding to multi-charged ions were determined. The peak areas of the different multi-charged ions were summed after application of correction factors because discrepancies were evidenced in the response factors of the different modified PAMAMs depending on the number of grafted glycine residues; indeed, the ion mobility spectrum of an equimolar solution of unmodified G1(N) and totally modified Gly6G1(N) showed that Gly6G1(N) had higher peak intensities (by a factor of 5). After application of the correction factors calculated according to the method outlined in the experimental section, the percentage of each modified PAMAM could be determined from these integration data (they are related to the “% of areas” column of the Table S1 of the ESI†). Thus, the percentages from major species to minor species are: Gly1G1(N) (31%), native G1(N) (28%), Gly2G1(N) (15%), Gly3G1(N) (11%), Gly4G1(N) (8%), Gly5G1(N) (5%) and Gly6G1(N) (3%). Such quantitative information regarding the distribution of partially grafted PAMAM could therefore be provided by the integration of ion mobility peaks from IM-MS data. Moreover, the average number of grafted glycine residues was consecutively estimated to be near 28% (Table S1 of the ESI†). This result was in the same order of magnitude to that estimated by 1H NMR. The difference between MS and NMR values could be explained by possible drawbacks in each analytical technique, i.e. discrepancies in MS response factors between the different modified-PAMAMs and possible bias in the 1H NMR integrations because of the presence of defective structures. The IM-MS analysis could also afford information concerning the three-dimensional structure of the PAMAM ions, in particular their dimensions in the gas phase14 that could help the understanding of the bioactive conformation.21 Indeed, collision cross sections (CCS) and consequently the radii (according to the spherical ion model) could be estimated from calibration curves obtained using reference ions having well-described collision cross-sections, such as the protonated molecules of the polyalanine peptides. Thus, the experimental CCS of doubly-charged modified PAMAMs ([M + 2H]2+ in the particular case of IMS analysis) were determined and compared to theoretical CCS calculated using trajectory method (TM) model22 as well as CCS estimated from molecular weight (g mol−1) using the spherical ion model (SIM) according to Ruotolo et al.23 (Table 2).
| G1(N) | Gly1G1(N) | Gly3G1(N) | Gly6G1(N) | |
|---|---|---|---|---|
| a Theoretical collision cross-sections (Ω) calculated with the TM model.21 b Collision cross-section determined using the equation Ω = 2453 × M2/3 for GlynG1(N) according to Ruotolo et al.22 c Experimental collision cross-section measured in triplicate for doubly charged ions of PAMAM dendrimers. | ||||
| Calculated Ωaverage (Å2) using the TM modela | 328 | 335 | 343 | 373 |
| Ω (Å2) (SIM)b (Ω = 2453 × M2/3) | 252 | 261 | 288 | 337 |
| Ω experimental (Å2) (TWIM)c | 240 | 245 | 261 | 282 |
As expected the calculated as well as the measured CCS increased with the number of grafted glycine units. Moreover, a linear relationship could be observed between the experimental CCS of the GlynG1(N) PAMAMs and the number of grafted glycine residues (Fig. 7). The measured CCS were however smaller than both the TM and the SIM values. That could be explained by the formation of more compact structures in the gas phase than in solutions.24,25 These results showed that PAMAMs exhibited, as a general trend, compact structures from unmodified to fully modified forms which is consistent with the spherical nature of these compounds known to present limited extensibility.26 However, to obtain more accurate cross section values, the use of calibration standards that most closely resemble our analytes should be envisaged, as previously reported by Salbo et al.27
Conclusions
The synthesis of a series of fully- as well as partially-substituted PAMAM G1(N) dendrimers functionalized with glycine residues was achieved using different and separate optimized grafting reactions. Indeed, two different experimental conditions were necessary for the coupling step. The N-Boc deprotection step was also adjusted for the particular case of these modified dendrimers because the usual procedure induced hydrolysis of the grafted PAMAM. The full structural characterization of the series of surface-modified PAMAMs was performed using the combination of MS and NMR techniques. The glycine grafting yield as determined by one-dimensional 1H NMR was approximately 25% in the case of partially modified PAMAM G1(N) while it was found to be 100% in the case of Gly6G1(N). Effective grafting of glycine onto the dendrimer surface was verified by both two-dimensional NMR and MS experiments. The distribution of partially modified PAMAMs was evidenced by MS, IM-MS and CE analyses. Elemental composition of each synthesized dendrimer was determined by accurate mass measurements. Mass spectrometry permitted the detection of individual ionic species of each modified GlynG1(N) which were submitted to multi-stage MSn experiments. Thus, appropriate MS2, MS3 and MS4 experiments allowed the structural identification of each GlynG1(N) dendrimers, the differentiation and the identification of two isomeric structures for Gly2-,Gly3- and Gly4G1(N) and the presence of only one isomer in the case of Gly1G1(N), Gly5G1(N) and Gly6G1(N).Both CE and IM-MS displayed separation of the differently modified dendrimers and consequently showed the distribution in a complementary way to the sole MS approach. In the case of IM-MS, the glycine grafting yield was estimated to be nearly 28%.
This work constitutes a model for the synthesis and the structural characterization of glycine-modified PAMAMs and represents a new approach for the construction of a library of amino acid-modified PAMAM dendrimers with ammonia cores and subsequently the extension of the data to EDA-cored surface-modified PAMAMs. Moreover, the glycine attached to the dendrimer surface can be further used as a spacer to conjugate functional groups such as drugs and targeting moieties.
Experimental section
Materials
Methyl acrylate, ethylenediamine (EDA), N-(tert-butoxycarbonyl)glycine (Boc-gly-OH), O-(benzotriazol-1-yl)-N,N,N′,N′-tetramethyluronium hexafluorophosphate (HBTU), 1-hydroxybenzotriazole hydrate (HOBt hydrate), N,N-dimethylformamide (DMF), N,N-diisopropylethylamine (DIPEA), phosphoric acid (H3PO4), toluene, dichloromethane (DMC), and HPLC grade methanol were purchased from sigma-aldrich (Saint-Quentin-Fallavier, France). Acetonitrile (ACN), 2,2,2-trifluoroethanol (TFE), acide phosphorique (H3PO4), Float-A-Lyzer G2 dialyzer (0.5 à 1kDa), ammonium acetate and LC-MS grade acetonitrile (ACN) were purchased from fisher scientific (Illkirch, France). Deionised water (18 MΩ) was obtained from a Milli-Q apparatus (Millipore, Bedford, MA, USA).Synthesis of ammonia core PAMAM G1(N)
The PAMAM G1(N) was prepared according to Tomalia's divergent synthesis.14 A solution of methyl acrylate (2.14 equiv. per dendrimer amine end group) in methanol was added dropwise to a stirred solution of ammonia in methanol. The reaction mixture was stirred at room temperature in dark for three days then concentrated in vacuo to yield the half-generation PAMAM G-0.5. In a second step, to a solution of ethylene diamine (EDA) (10 equiv. per dendrimer ester end group) in methanol was directly added a solution of half-generation PAMAM G-0.5 in methanol then stirred for six days at room temperature giving the full generation PAMAM G0. Preceding two-steps were repeated using the full generation PAMAM G0 followed by a repetition azeotropic distillation [toluene 100 mL was added to the product dissolved in methanol (10 mL)]. For generations 0.5, the crude product was purified by column chromatography (Rf: 0.40, 12% MeOH in DCM, silica 60). The reiteration of this two-steps procedure gives the ammonia-cored PAMAM G1(N) (yield 97%). ESI-HRMS: m/z calculated and measured for C45H93N19O9 ([M + H]+) were 1044.7482 and: 1044.7487, respectively. 1H NMR (D2O). δ = 2.40 (18H, m, H-3 and H10′), 2.60 (6H, t, H7), 2.69 (12H, t, H9′), 2.76 (6H, t, H-2), 2.79 (12H, t, H-14′), 3.21 (12H, t, H-13′), 3.26 (6H, t, H-6).Preparation of Gly6G1(N)
The fully modified PAMAM-G1(N) was prepared in DMF/water (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 v/v) by mixing 0.95 equiv. of PAMAM G1(N), 6 equiv. of N-Boc-Gly-OH, 6.6 equiv. of HBTU, HOBt·H2O and 24 equiv. of DIPEA for one night at room temperature. The product was dialyzed (Mw cutoff = 1000 Da) for 6 days against deionized water. The dialysed sample was then lyophilized to afford viscous orange oil. The N-Boc protecting group was removed in TFE by adding 17 equiv. of aqueous H3PO4 (85% Wt). The reaction mixture was stirred at room temperature in dark for 12 min (a precipitate was formed almost instantaneously). The reaction mixture was then diluted in H2O and the pH was adjusted to ≈7 with ammonium acetate (1 M). The product was dialyzed for 6 days against deionized water and lyophilized to give a yellow solid (yield 70%). The ESI-HRMS data are reported in Table 1 of the Results and Discussion section. 1H NMR (D2O). δ = 2.59 (12H, t, H-10), 2.64 (6H, br. t, H-3), 3.03 (6H, br. t, H-7), 3.17 (18H, br. t, H-2 and H-9), 3.28 (12H, m, H-13), 3.31 (12H, m, H-14), 3.46 (6H, br. t, H-6), 3.73(12H, s, H-17). 13C NMR. δ = 30.6 (C-3 and C-10), 35.1 (C-6), 38.4 (C-13 and C-14), 40.7 (C-17), 49.4(C-2 and C-9), 52 (C-7),167.5 (C-16), 173.2 (C-4 and C-11).
2 v/v) by mixing 0.95 equiv. of PAMAM G1(N), 6 equiv. of N-Boc-Gly-OH, 6.6 equiv. of HBTU, HOBt·H2O and 24 equiv. of DIPEA for one night at room temperature. The product was dialyzed (Mw cutoff = 1000 Da) for 6 days against deionized water. The dialysed sample was then lyophilized to afford viscous orange oil. The N-Boc protecting group was removed in TFE by adding 17 equiv. of aqueous H3PO4 (85% Wt). The reaction mixture was stirred at room temperature in dark for 12 min (a precipitate was formed almost instantaneously). The reaction mixture was then diluted in H2O and the pH was adjusted to ≈7 with ammonium acetate (1 M). The product was dialyzed for 6 days against deionized water and lyophilized to give a yellow solid (yield 70%). The ESI-HRMS data are reported in Table 1 of the Results and Discussion section. 1H NMR (D2O). δ = 2.59 (12H, t, H-10), 2.64 (6H, br. t, H-3), 3.03 (6H, br. t, H-7), 3.17 (18H, br. t, H-2 and H-9), 3.28 (12H, m, H-13), 3.31 (12H, m, H-14), 3.46 (6H, br. t, H-6), 3.73(12H, s, H-17). 13C NMR. δ = 30.6 (C-3 and C-10), 35.1 (C-6), 38.4 (C-13 and C-14), 40.7 (C-17), 49.4(C-2 and C-9), 52 (C-7),167.5 (C-16), 173.2 (C-4 and C-11).
Preparation of GlynG1(N)
A solution containing 3 equiv. of N-Boc-Gly-OH, 3.3 equiv. of HBTU, HOBt·H2O and 12 equiv. of DIPEA in 5 mL of DMF was prepared (the flask was immersed in an ice and water bath) and stirred for 40 minutes. This solution was added dropwise over 30 minutes to a solution of PAMAM G1(N) (0.95 equiv.) in DMF/water (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 v/v) at 40 °C. The whole was then allowed to stir at 40 °C for 4 hours. Then, purification and deprotection of the N-Boc protecting group (as above) to give yellow solid of partially modified PAMAM G1 (N) with a distribution of attached glycine from 0 up to 6 (yield 68%). The ESI-HRMS data are reported in Table 1 of the Results and Discussion section. 1H NMR (D2O). δ = 2.41 (10H, m, H-10′), 2.46 (8H, m, H-3 and H-10), 2.63 (6H, m, H-7), 2.82 (18H, m, H-2, H-9 and H9′), 3.10 (8H, t, H-14′), 3.29 (6H, m, H-6 and H-13), 3.32 (8H, m, H-14), 3.46 (8H, t, H-13′), 2.94 (3.5H, Br.s, H-17). 13C NMR. δ = 32.38 (C-3, C-10 and C-10′), 36.73 (C-6 and C-13), 38.73 (C-14), 38.98 (C-14′), 41.79 (C-17), 48.84 (C-2, C-9 and C-9′), 51.39 (C-7), 171.2.5 (C-16), 175.0 (C-4 and C-11).
2 v/v) at 40 °C. The whole was then allowed to stir at 40 °C for 4 hours. Then, purification and deprotection of the N-Boc protecting group (as above) to give yellow solid of partially modified PAMAM G1 (N) with a distribution of attached glycine from 0 up to 6 (yield 68%). The ESI-HRMS data are reported in Table 1 of the Results and Discussion section. 1H NMR (D2O). δ = 2.41 (10H, m, H-10′), 2.46 (8H, m, H-3 and H-10), 2.63 (6H, m, H-7), 2.82 (18H, m, H-2, H-9 and H9′), 3.10 (8H, t, H-14′), 3.29 (6H, m, H-6 and H-13), 3.32 (8H, m, H-14), 3.46 (8H, t, H-13′), 2.94 (3.5H, Br.s, H-17). 13C NMR. δ = 32.38 (C-3, C-10 and C-10′), 36.73 (C-6 and C-13), 38.73 (C-14), 38.98 (C-14′), 41.79 (C-17), 48.84 (C-2, C-9 and C-9′), 51.39 (C-7), 171.2.5 (C-16), 175.0 (C-4 and C-11).
The average number of grafted glycine was measured by 1H-NMR using the 1H proton integration method. The 1H-NMR spectrum of GlynG1(N) showed the summed protons within the 2.35–3.50 ppm region (72 H) corresponded to methylene of PAMAM G1(N) dendrimer. The proton signal at 3.56 ppm (2.94H) corresponded to the methylene of glycine residue. The integration ratio of the methylene protons in glycine residue and all the methylene protons of G1(N) moiety was used to calculate the grafting ratio; an average of 1.5 grafted glycine was therefore estimated (Figure S2 of the ESI†).
Nuclear magnetic resonance spectroscopy (NMR)
1H, 13C, COSY, HMBC and HMQC NMR spectra of PAMAM G1(N), GlynG1(N) and Gly6G1(N) were obtained in D2O using a Bruker Avance III 600 MHz spectrometer equipped with a 5 mm CPTXi cryoprobe.Mass spectrometry (MS) and ion mobility spectrometry (IM)
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 v/v) with a concentration of 2 ng μL−1. Polyalanine peptide mixture was prepared to a concentration of 10 ng μL−1 in 1% formic acid, acetonitrile and H2O mixture (50
50 v/v) with a concentration of 2 ng μL−1. Polyalanine peptide mixture was prepared to a concentration of 10 ng μL−1 in 1% formic acid, acetonitrile and H2O mixture (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 v/v).
50 v/v).
For the ESI parameters the capillary and end plate voltages respectively set to −4.0 kV and −3.5 kV in positive ion mode. The skimmer voltage was set to 40 V and the injection low mass cut off (LMCO, corresponding to the “trap drive” parameter) value was m/z 69. The nebulizer gas (N2) pressure, drying gas (N2) flow rate and drying gas temperature were 10 psi, 5.0 L min−1 and 300 °C, respectively. Helium pressure in the ion trap was 9.6 × 10−6 mbar. Spectra were acquired in the m/z 200–1300 range, using the ‘Standard-Enhanced’ mode (scan rate of 8000 m/z per second). The number of ions entering the ion trap was automatically adjusted by controlling the accumulation time with the ion charge control (ICC) mode (target 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000) with a maximum accumulation time of 50 ms. The values of spectra averages and rolling average were 5 and 2 respectively. ESI-MSn experiments were carried out by collision induced dissociation (CID) using a resonant excitation frequency with amplitudes from 0.29 to 1.0 Vp−p, helium as the collision gas, isolation width of 1 m/z unit for the precursor ions and of 2 m/z unit for the intermediate ions. Sample solutions were infused into the source at a flow-rate of 3 μL min−1 by means of a syringe pump (Cole-Palmer, Vernon Hills, Illinois, USA).
000) with a maximum accumulation time of 50 ms. The values of spectra averages and rolling average were 5 and 2 respectively. ESI-MSn experiments were carried out by collision induced dissociation (CID) using a resonant excitation frequency with amplitudes from 0.29 to 1.0 Vp−p, helium as the collision gas, isolation width of 1 m/z unit for the precursor ions and of 2 m/z unit for the intermediate ions. Sample solutions were infused into the source at a flow-rate of 3 μL min−1 by means of a syringe pump (Cole-Palmer, Vernon Hills, Illinois, USA).
Q-TOF-MS2 experiments involved (i) selection of the [M + 2Na]2+ precursor ion with the quadrupole mass analyzer within a 1 m/z unit window (ii) fragmentation in the trap collision cell (first ion guide of the Triwave), collision energy; 38 eV for Gly3G1(N) and 40 eV for Gly6G1(N).
Elemental compositions of protonated and sodiated multicharged ions (MS mode) were proposed from the accurate mass measurements and considering the isotope predictive filtering value (i-FIT™) given for each possible elemental composition.
The IMS conditions were optimized as followed: gas flow (N2); 120 mL min−1, wave height; 37.3 V and wave velocities 800 m s−1. IM-MS analyses were performed in triplicate. For the three measurements, strictly identical drift times were observed for doubly charged ions of PAMAM dendrimers. That could be explained by the well-described performance of IMS technique which gives highly reproducible data.
The peak integration was performed from extracted ion mobility spectrum of each multi-charged ion. Before summing the peak areas, correction factors were estimated for each multi-charged ion from the analysis of an equimolar solution of G1(N) and Gly6G1(N). The extracted ion mobility spectra of the G1(N)/Gly6G1(N) mixture showed that doubly-, triply- and quadruply-charged ions of Gly6G1(N) had higher peak intensities than the corresponding multi-charged ions of G1(N). A decrease of about 80% was observed from Gly6G1(N) to G1(N) signals. Thus, we estimated that the diminution of response factor (X) per ungrafted end group could be calculated according to the formulae (X)6 ≈ 20%. Thus, X was estimated to be about 75–80%. The correction factors were systematically applied on the peak areas. Then, the corrected peak areas were summed for each modified PAMAM and the corresponding area percentage were calculated. The percentage of grafted glycine was determined by multiplying the area percentage with the ratio of grafted glycine which is equal to n/6 with n = number of grafted glycine. Finally, the total percentage of grafted glycine was calculated by summing all the individual percentage of grafted glycine (Table S1 of the ESI†).
Acknowledgements
This work has been partially supported by INSA Rouen, Rouen University, CNRS, EFRD (N°31708), the IS:CE-Chem project and Interreg IV A France -(Channel)- England Programme and Labex SynOrg (ANR-11-LABX-0029). They are gratefully thanked for their financial support.Notes and references
- J. M. J. Fréchet and D. A. Tomalia, Dendrimers and Other Dendritic Polymers, Wiley Series in Polymer Science, John Wiley & Sons, Ltd, Chichester, UK, 2001, p. 1 Search PubMed.
- K. Fant, E. K. Esbjörner, P. Lincoln and B. Nordén, Biochemistry, 2008, 47, 1732–1740 CrossRef CAS PubMed.
- R. Jevprasesphant, J. Penny, R. Jalal, D. Attwood, N. B. McKeown and A. D'Emanuele, Int. J. pharm., 2003, 252, 263–266 CrossRef CAS.
- K. Fant, E. K. Esbjörner, A. Jenkins, M. C. Grossel, P. Lincoln and B. Nordén, Mol. Pharmaceutics, 2010, 7, 1734–1746 CrossRef CAS PubMed.
- R. S. Navath, A. R. Menjoge, B. Wang, R. Romero, S. Kannan and R. M. Kannan, Biomacromolecules, 2010, 11, 1544–1563 CrossRef CAS PubMed.
- X. Wang, Y. He, J. Wu, C. Gao and Y. Xu, Biomacromolecules, 2010, 11, 245–251 CrossRef CAS PubMed.
- H. Y. Nam, H. J. Hahn, K. Nam, W. H. Choi, Y. Jeong, D.-E. Kim and J. S. Park, Int. J. pharm., 2008, 363, 199–205 CrossRef CAS PubMed.
- D. Mullen, M. Fang, K. Desai, J. R. Baker, J. B. G. Orr and M. M. B. Holl, ACS Nano, 2010, 4, 657–670 CrossRef CAS PubMed.
- A.-M. Caminade, R. Laurent and J. P. Majoral, Adv. Drug Delivery Rev., 2005, 57, 2130–2146 CrossRef CAS PubMed.
- J. Peterson, V. Allikmaa, J. Subbi, T. Pehk and M. Lopp, Eur. Polym. J., 2003, 39, 33–42 CrossRef CAS.
- M. He and S. A. McLuckey, Rapid Commun. Mass Spectrom., 2004, 18, 960–972 CrossRef CAS PubMed.
- T. J. Vincent, R. Dole and C. M. Lange, Rapid Commun. Mass Spectrom., 2008, 22, 363–372 CrossRef CAS PubMed.
- E.-D. Leriche, F. Maire, M. C. Grossel, C. M. Lange and C. Loutelier-Bourhis, Rapid Commun. Mass Spectrom., 2012, 26, 1718–1724 CrossRef CAS PubMed.
- F. Maire, G. Coadou, L. Cravello and C. M. Lange, J. Am. Soc. Mass Spectrom., 2013, 24, 238–248 CrossRef CAS PubMed.
- D. A. Tomalia, H. Baker, J. Dewald and M. Hall, Polym. J., 1985, 17, 117–132 CrossRef CAS.
- B. Li, M. Berliner, R. Buzon, C. K. Chiu, S. T. Colgan, T. Kaneko, N. Keene, W. Kissel, T. Le, K. R. Leeman, B. Marquez, R. Morris, L. Newell, S. Wunderwald, M. Witt, J. Weaver and Z. Zhang, J. Org. Chem., 2006, 71, 9045–9050 CrossRef CAS PubMed.
- B. Li, R. Bemish, R. A. Buzon, C. K. Chiu, S. T. Colgan, W. Kissel, T. Le, K. R. Leeman, L. Newell and J. Roth, Tetrahedron Lett., 2003, 44, 8113–8115 CrossRef CAS PubMed.
- K. Nagayama, A. Kumar, K. Wuthrich and R. R. Ernst, J. Magn. Reson., 1980, 40, 321–334 CAS.
- A. Bax and M. F. Summers, J. Am. Chem. Soc., 1986, 108, 2093–2094 CrossRef CAS.
- H. Kim, C. R. Babu and D. J. Burgess, Int. J. Pharm., 2013, 448, 123–131 CrossRef CAS PubMed.
- C. Lapthorn, F. Pullen and B. Z. Chowdhry, Mass Spectrom. Rev., 2013, 32, 43–71 CrossRef CAS PubMed.
- M. F. Mesleh, J. M. Hunter, A. A. Shvartsburg, G. C. Schatz and M. F. Jarrold, J. Phys. Chem., 1996, 100, 16082–16086 CrossRef CAS.
- B. T. Ruotolo, J. L. P. Benesch, A. M. Sandercock, S.-J. Hyung and C. V. Robinson, Nat. Protoc., 2008, 3, 1139–1152 CrossRef CAS PubMed.
- C. A. Scarff, K. Thalassinos, G. R. Hilton and J. H. Scrivens, Rapid Commun. Mass Spectrom., 2008, 22, 3297–3304 CrossRef CAS PubMed.
- T. W. Knapman, J. T. Berryman, I. Campuzano, S. A. Harris and A. E. Ashcroft, Int. J. Mass Spectrom., 2010, 298, 17–23 CrossRef CAS PubMed.
- E. N. Govorun, K. B. Zeldovich and A. R. Khokhlov, Macromol. Theory Simul., 2003, 12, 705–713 CrossRef CAS.
- R. Salbo, M. F. Bush, H. Naver, I. Campuzano, C. V. Robinson, I. Peterson, T. Jorgensen and K. F. Haselmann, Rapid Commun. Mass Spectrom., 2012, 26, 1181–1193 CrossRef CAS PubMed.
- K. Giles, S. D. Pringle, K. R. Worthington, D. Little, J. L. Wildgoose and R. H. Bateman, Rapid Commun. Mass Spectrom., 2004, 18, 2401–2414 CrossRef CAS PubMed.
- S. E. Slade, K. Thalassinos, R. H. Bateman, M. T. Bowers and J. H. Scrivens, Int. J. Mass Spectrom., 2007, 261, 1–12 CrossRef PubMed.
- A. T. Hagler, The Peptides: Analysis, Synthesis, Biology, ed. S. Udenfriend, J. Meienhofer and V. J. Hruby, Academic Press, Orlando, FL, 1985, vol. 7, p. 213 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c3ra43939a |
| This journal is © The Royal Society of Chemistry 2014 |






