Magnetic molecularly imprinted microcapsules derived from Pickering emulsion polymerization and their novel adsorption characteristics for λ-cyhalothrin†
Jianming Pan*a,
Wenjing Zhua,
Xiaohui Daia,
Xuesheng Yanb,
Mengyin Ganc,
Linzi Lic,
Hui Hangc and
Yongsheng Yanc
aSchool of Chemistry and Chemical Engineering, Jiangsu University, Zhenjiang 212013, China. E-mail: zhenjiangpjm@126.com (Jianming Pan)
bSchool of Energy and Power Engineering, Jiangsu University, Zhenjiang 212013, China
cSchool of the Environment, Jiangsu University, Zhenjiang 212013, China
First published on 28th October 2013
Abstract
Magnetic molecularly imprinted microcapsules (MMIMs) were synthesized by Pickering emulsion polymerization. In this work, an oil-in-water Pickering emulsion stabilized by halloysite nanotubes (HNTs) was first established in the presence of a few hydrophilic Fe3O4 nanoparticles as magnetic separation carriers. The imprinting system was fabricated by radical polymerization and subsequent combination with functional and polymeric monomers in the oil phase. The formation mechanism of the Pickering emulsion and MMIMs is discussed in detail, and the as-prepared MMIMs are successfully evaluated as sorbents for the recognition of λ-cyhalothrin. The results demonstrated that the MMIMs exhibited magnetic sensitivity (Ms = 8.45 emu g−1), a hollow structure, a semipermeable external surface and excellent robustness. The batch mode experiments proved that the imprinting effect synchronously improved the adsorption kinetics and equilibrium for the MMIMs. The selective recognition experiments also suggested high affinity and selectivity of MMIMs towards λ-cyhalothrin over fenvalerate and diethyl phthalate.
Introduction
Recently, hollow microspheres with well-defined structures have received increasing interest because of their lower density, larger cavity volume and widespread potential applications in controlled drug release, adsorption of environmental pollutants and chemical reactors for catalysis, etc.1–3 The template method as a versatile approach has been used to design and synthesize the hollow microspheres. To date, numerous templates including hard templates such as mesoporous silica,4 Ag nanocubes,5 polystyrene spheres6 and Au particles,7 and soft templates such as block copolymers,8 Pickering emulsion droplets,9 micelles10 and gas bubbles11 have been extensively employed to prepare hollow structures. Among which, Pickering emulsion droplets that are stabilized by solid nanoparticles have been proved to be an efficient and feasible platform for the synthesis of microcapsules with shells constructed from colloidal particles.12 The resulting microcapsules have semipermeable shells arising from the particle interstices, and the permeability is modulated by changing the particle size and morphology.13,14 Moreover, the structural robustness of the microcapsules can also be reinforced by the chemical crosslinking of the solid nanoparticle surfaces.14 These benefits of such an approach are well established in separation technology where the obtained composite materials possess superior mechanical performance, semipermeable surfaces and lower density.15Molecular imprinting is a facile and versatile technique used for preparing polymers with specific binding sites, namely molecularly imprinted polymers (MIPs). In order to fabricate tailor-made binding sites, molecular interactions between the template molecule and the functional monomer in a self-assembly step are first obtained. Then the self-organised architecture formed is fixed by additional crosslinking polymerization. Subsequently, the removal of the template molecule from the polymer matrix leaves behind binding sites possessing functional groups complementary to the template molecules.16 Inspired by the benefits of molecular imprinting and Pickering emulsion, Ye's group reported several novel and important studies about the imprinted polymer beads derived from Pickering emulsion polymerization.17–19 For example, using water-in-oil Pickering emulsion polymerization, the molecular imprinting of proteins was carried out to prepare spherical hydrogels with pre-designed selectivity for proteins.17 Furthermore, a new interfacial nano and molecular imprinting method based on Pickering emulsion polymerization was also developed.19 These studies adopted spherical SiO2 particles to irreversibly adsorb at the oil and water interface, as opposed to particles of non-spherical shapes. The irreversible adsorption at oil and water interface is commonly caused by capillary energy that is several orders of magnitude higher than the thermal energy.20 However, Lehle etc. demonstrated that for non-spherical colloids at free interfaces capillary interactions appear to be dominant.21 There is, therefore, a clear interest in the synthesis of MIPs by Pickering emulsion polymerization stabilized solely by non-spherical particles. In order to meet the need for a stable emulsion, the spherical SiO2 particles needed to be modified to adjust their surface wettability, which was time consuming and complicated.
In recent years, disk-shaped Laponite clay has been reported to stabilize water-in-oil emulsions or oil-in-water emulsions. For example, Cauvin et al. successfully prepared polymer latex particles via Pickering emulsion polymerization using Laponite RD clay.22 Recently, Isabelle Capron's group fabricated several oil-in-water Pickering emulsions stabilized solely by cellulosic colloidal nanorods of different origins.23 In previous studies, our group adopted attapulgite (ATP) with fiber bundle morphology as the stabilized particles to obtain magnetic/hollow double-shelled imprinted sorbents.24 The ATP particles used in our work had diameters of 300 nm and lengths of 1.0–3.0 μm. Compared with cellulose, Laponite RD and ATP, halloysite nanotubes (HNTs), a naturally occurring clay mineral with large reserves in China, have smaller sizes, open-ended tubular structures and superior surface areas, which make them attractive for the preparation of a more stable Pickering emulsion.25 Moreover, HNTs with a chemical formula of Al2Si2O5(OH)4·nH2O also possess hydrophilic surfaces.26 Thus, HNTs appear to be excellent candidates to produce a stable oil-in-water emulsion in addition to their size and shape versatility. However, the work required to take advantage of HNTs to build a stable emulsion without modification has never been investigated to the best of our knowledge.
To get a rapid separation of chemical species, magnetic nanoparticles such as Fe3O4 have recently attracted considerable attention in Pickering emulsion polymerization. The synthesis of magnetic hollow silica was reported through a Pickering emulsion solely stabilized by cetyltrimethylammonium bromide modified Fe3O4 particles.27 Liu et al. prepared superparamagnetic polyaniline hybrid hollow microspheres with sodium citrate modified Fe3O4 nanoparticles as a template.28 Pickering emulsion polymerization based on Fe3O4 particles can be used for the fabrication of magnetic composites, which can be easily collected and separated by an external magnetic field without additional centrifugation or filtration. However, many pure Fe3O4 particles adsorbed at the oil and water interface may have problems associated with the formation of large aggregates and easy leakage.
Inspired by the studies mentioned above, the molecular imprinting and magnetic separation concepts were introduced into a Pickering emulsion polymerization, and than the magnetic molecularly imprinted microcapsules (MMIMs) were synthesized. In this work, we chose 4-vinylpyridine (4-VP) as the functional monomer to recognize the target pollutant molecule λ-cyhalothrin, HNTs as a stabilizer for the oil-in-water Pickering emulsion and a few hydrophilic Fe3O4 nanoparticles as magnetic separation carriers, which provided imprinted and magnetic microcapsules. The whole process of the synthesis of MMIMs and the recognition of λ-cyhalothrin is presented in Fig. 1. The characterization, adsorption equilibrium, kinetics, and selectivity recognition of the MMIMs were investigated in detail.
Results and discussion
Formation mechanism of Pickering emulsion
In this study, HNT particles were used as the stabilizer and a few hydrophobic Fe3O4 nanoparticles (HFNs) were employed as magnetic carriers. The TEM pictures of the HNTs and HFNs are shown in the ESI (Fig. ES1†). It can be seen that the HNTs were open at the ends and their cross-section was about 100 nm to 120 nm, and the HFNs were sphere-like and had an average size of 10 nm as estimated from TEM. To form a stable Pickering emulsion, HNT, NaCl and HFN particles were dispersed in the water phase, and St, 4-VP, λ-cyhalothrin, and AIBME were dispersed in toluene forming the oil phase. According to a previous study, HNTs had favorable hydrophilicity due to the presence of a large number of hydroxyl groups on the HNTs, and this was not beneficial for favorable partitioning at the interface in competition with the two bulk phases.29 Thus, in order for the HNT building blocks to adsorb at the oil–water interface and to provide emulsion stabilization, NaCl was added to water phase to reduce the electrostatic repulsion resulting from the permanent negative charges on the surface of HNTs in our work.30 While this did not prevent stable emulsion formation, it only affected the level of HNT inclusion into the composite capsules. Photos of the water phase dispersion (a), and Pickering emulsion before (b) and after (c) ultrasonication are shown in Fig. 2. As shown in Fig. 2a, the HNTs were well dispersed in the water phase, while a few black HFNs were not obvious. This fact also illustrates that the effect of a few HFNs for Pickering emulsion could be neglected. The photo of the mixture of the oil and water phase is shown in Fig. 2b, and the transparent oil phase is located at the top of the cloudy water phase dispersion. After ultrasonication and emulsification, the HNT particle-stabilized O/W Pickering emulsion was obtained. | ||
| Fig. 2 Photos of the water phase dispersion (a), and Pickering emulsion before (b) and after (c) ultrasonication. | ||
Fabrication mechanism of MMIMs
In the process of imprinting polymerization, 4-VP as functional monomer was first allowed to form a self-assembled complex with the template molecule λ-cyhalothrin under the driving force of π–π stacking interactions. Then the Pickering emulsion polymerization was implemented in the presence of the polymeric monomer St, functional monomers 4-VP and initiator AIBME. In this step, the self-organized architectures between and 4-VP and λ-cyhalothrin were fixed in the imprinted layer, and the cross-linked polymer network tended towards phase separation in the poor solvent, toluene, driven by the interfacial tension and precipitation at the interface. By elution with water and ethanol, hollow structures were readily formed by the removal of the residual toluene and unreacted 4-VP from the MMIMs. Furthermore, by elution with a mixture of methanol–acetic acid (95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, v/v), the imprinted template molecules were removed. Finally, binding sites with stereochemistry complementary to λ-cyhalothrin were formed in the MMIMs. The optical micrographs of Pickering emulsion (a) and the MMIMs before (b) and after (c) elution are shown in Fig. 3. Fig. 3a shows a typical stable Pickering emulsion that was used to produce the MMIMs. The emulsion droplets were tiled on the glass slide and exhibited a spherical hollow structure without collapse. Moreover, the Pickering emulsion prepared in this work had relatively broad droplet size distributions, and a mean droplet diameter of approximately 80 μm estimated from the optical micrograph. After heating at 65 °C to initiate polymerization, the surface of the spherical MMIMs before elution was rough, which suggested that the HNT particles adsorbed at the oil–water interface were subsequently locked into the external surface of the capsule (Fig. 3b). As a result of this, the HNT building blocks were firmly attached onto the surface of polymer network which enhanced the mechanical properties of the MMIMs.31 It can be clearly seen from Fig. 3c that hollow structures existed in the MMIMs after elution, and the capsule shells were asymmetric and nonuniform with a thickness range of 7.0–20 μm.
5, v/v), the imprinted template molecules were removed. Finally, binding sites with stereochemistry complementary to λ-cyhalothrin were formed in the MMIMs. The optical micrographs of Pickering emulsion (a) and the MMIMs before (b) and after (c) elution are shown in Fig. 3. Fig. 3a shows a typical stable Pickering emulsion that was used to produce the MMIMs. The emulsion droplets were tiled on the glass slide and exhibited a spherical hollow structure without collapse. Moreover, the Pickering emulsion prepared in this work had relatively broad droplet size distributions, and a mean droplet diameter of approximately 80 μm estimated from the optical micrograph. After heating at 65 °C to initiate polymerization, the surface of the spherical MMIMs before elution was rough, which suggested that the HNT particles adsorbed at the oil–water interface were subsequently locked into the external surface of the capsule (Fig. 3b). As a result of this, the HNT building blocks were firmly attached onto the surface of polymer network which enhanced the mechanical properties of the MMIMs.31 It can be clearly seen from Fig. 3c that hollow structures existed in the MMIMs after elution, and the capsule shells were asymmetric and nonuniform with a thickness range of 7.0–20 μm.
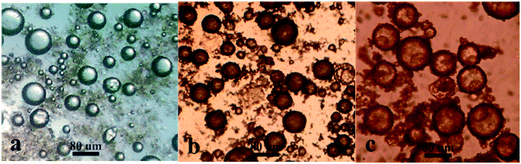 | ||
| Fig. 3 Optical micrographs of Pickering emulsion (a), and MMIMs before (b) and after (c) elution with water and ethanol. | ||
Characterization of MMIMs
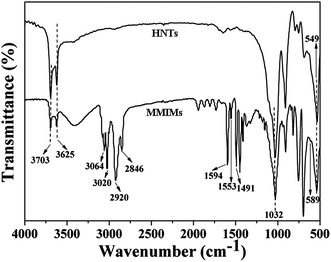
The main functional groups of the predicted structure were further observed with using the corresponding infrared absorption spectra. As shown in Fig. 4, the peaks at 3703 cm−1 and 3625 cm−1 of the HNTs and MMIMs could be assigned to the stretching vibrations of the inner-surface hydroxyl groups, while the band at 1032 cm−1 was attributed to the stretching vibration of Si–O–Si.32 Moreover, the band at 549 cm−1, caused by the deformation vibration of Al–O–Si, further confirmed the existence of HNTs in the MMIMs. The absorption band at 589 cm−1, corresponding to the Fe–O bond for spinel Fe3O4 particles, was observed for the MMIMs. The observed peaks at 1594 cm−1 and 1553 cm−1 in the spectrum, attributed to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching vibrations from the aromatic ring, were due to 4-VP.33 The appearance of absorption bands at 3064 cm−1 and 3020 cm−1, assigned to H–C
C stretching vibrations from the aromatic ring, were due to 4-VP.33 The appearance of absorption bands at 3064 cm−1 and 3020 cm−1, assigned to H–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, also gave direct evidence for the action of 4-VP. Furthermore, other absorption bands due to the aromatic stretching vibrations of the styrene units were also observed around 1491 cm−1, 2920 cm−1 and 2846 cm−1.
C, also gave direct evidence for the action of 4-VP. Furthermore, other absorption bands due to the aromatic stretching vibrations of the styrene units were also observed around 1491 cm−1, 2920 cm−1 and 2846 cm−1.
SEM images of the MMIMs (a), a single broken MMIM (b), and the external (c) and internal (d) surfaces of the MMIMs are shown in Fig. 5. The SEM image of the MMIMs prepared with Pickering emulsion polymerization revealed the formation of an intact spherical structure with a size distribution of 80 ± 10 μm (Fig. 5a). The microspheres were coarse and symmetrical without dimples under the conditions of the SEM analysis, suggesting excellent mechanical stability. The hollow nature of the capsules was visualized using an SEM image of a single broken MMIM in Fig. 5b. As shown in Fig. 5c, the external surface consisted of randomly distributed HNTs which were deposited on the exterior surface of the imprinted polymer shell rather than being embedded within it, indicating that the surface of the HNTs was sufficiently wettable by the dispersed oil phase without further modification. But only a few HNT particles were embedded into the imprinted polymer shell. This observation suggests that the surface wettability of HNTs was different from most modified inorganic colloids, and the incorporation of HNTs into the composite MMIMs was dependent on the addition of NaCl to the aqueous phase as previously described for Laponite clays.24 In Fig. 5d, many crosslinked polymer units arising from the nature of the interfacial phase separation were observed on the internal surface of the MMIMs. Both the external and internal surfaces of the MMIMs possessed denser but semipermeable structures, and surely played an important role in molecular capture by the obtained MMIMs. The EDX spectra of area 1 (d) and area 2 (e) were also measured and are shown in Fig. 5. Peaks for the Fe, O, C, Al, Si and Au elements were observed together for the exterior surface of the MMIMs as shown in Fig. 5e. The Au peak was due to the electrically-conducting material which was deposited by low vacuum sputter coating of the sample, and the Al and Si peaks resulted from the stabilized HNT particles. The low percentage of Fe in the pattern in Fig. 5e indicated the presence of magnetic carrier Fe3O4 on the exterior surface of the MMIMs. Besides Fe, Al, Si and Au peaks, a higher percentage of C and O confirmed the successful synthesis of the imprinted polymer. In Fig. 5f, only the peaks for the C and O elements were observed in the EDX spectrum of area 2. This phenomena is consistent with the results from the SEM image of the internal surface of the MMIMs, in which there were only a few HNTs existing in the polymer layer of the internal surface.
 | ||
| Fig. 5 SEM images of MMIMs (a), a single broken MMIM (b), and the external (c) and internal (d) surfaces of the MMIMs, and EDX spectra of area 1 (e) and area 2 (f). | ||
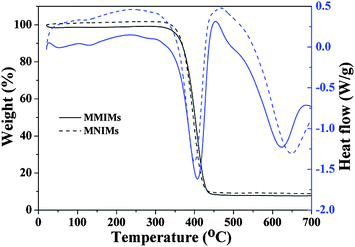
The MMIMs and MNIMs (magnetic non-molecularly imprinted microcapsules) were further characterized by TGA and DTG, and the corresponding curves are shown in Fig. 6. As shown in Fig. 6, the MMIMs and MNIMs cannot be easily decomposed within the initial temperature range (<300 °C), and TGA curves also indicated that both of them decomposed in a single step at temperature >350 °C. Moreover, the HNT content was about 10 wt% for the MMIMs and MNIMs, which was determined by the remaining mass of thermal resistance after full decomposition. The TGA analysis also revealed that the prepared MMIMs included both inorganic and organic composites. The MMIMs and MNIMs had the same decomposition profile and weight loss, and this result confirmed that the MMIMs and MNIMs had similar morphological structures and size distributions.34 Furthermore, two exothermic peaks around 407.5 °C and 400.0 °C for the MMIMs and MNIMs are shown in the DTG curves with obvious weight losses in the TGA curves, which were assigned to the significant decomposition of bulk polymers. However, two wide exothermic peaks around 623.3 °C and 648.5 °C for the MMIMs and MNIMs, respectively, were also observed without corresponding changes in the TGA curves, indicating the phase transition of the magnetic carriers.35
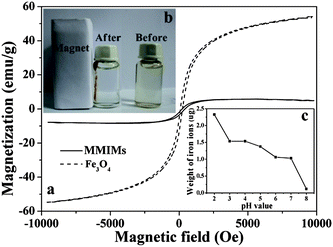
Magnetic properties play an important role for the application of MMIMs in fast extraction and convenient separation. The magnetic hysteresis loops analysis of the MMIMs and as-prepared hydrophobic Fe3O4 particles in this work are shown in Fig. 7a. The hysteresis loops show that there was no magnetic hysteresis, which indicated that both MMIMs and as-prepared hydrophobic Fe3O4 revealed superparamagnetic behavior. The Fe3O4 particles had a saturation magnetization of 54.13 emu g−1, and the MMIMs exhibited a value of 8.45 emu g−1. The significant decrease of the magnetization value can possibly be attributed to the presence of additional imprinted polymer layers and the introduction of a few Fe3O4 particles into the MMIMs. A photograph of the MMIMs suspended in water in the absence and presence of an externally placed magnet is shown in Fig. 7b. The homogeneously dispersed MMIMs could adhere to the inner sides of the wall of the vial when the external magnetic field was applied, and the test solution was clear and transparent. The results strongly suggest that the magnetic MMIMs could be attracted by an external magnetic field effectively, and that the yellow MMIMs are feasible magnetic separation carriers. Moreover, magnetite leakage was crucial to the performance of the sorbent in different pH environments. Thus, the number of Fe(III) ions that were likely to leach from the MMIMs was determined using a graphite furnace atomic absorption spectrophotometer (Fig. 7c). At pH = 8.0, 0.12 μg of leached Fe(III) ions were detected from 50 mg of the MMIMs. Moreover, the weight of the leached Fe(III) ions slightly increased from a pH value 7.0 to 3.0, and then obviously increased at a lower pH of 2.0. At pH = 2.0, the maximum weight of leached Fe(III) ions was only 2.32 μg, suggesting that the MMIMs prevented magnetite leakage successfully. In recent years, silica, carbon and polymers have been used to coat magnetic nanoparticles, and then the preparation of magnetic molecularly imprinted polymer based magnetic composites could overcome the problem of magnetite leakage.36,37 But the step for synthesizing the magnetic composites prior to the imprinting polymerization were time-consuming and complicated. Thus, Pickering emulsion polymerization combined with a few Fe3O4 particles as magnetic carriers may be considered to be a simple and effective way to overcome the limitation of magnetite leakage.
Properties of MMIMs for λ-cyhalothrin adsorption
In practical applications, MMIMs were employed as sorbents for removal of λ-cyhalothrin from ethanol–water (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 v/v) solution, and the functional adsorption behaviour of the MMIMs was investigated in detail. First, the adsorption capacity at different initial concentrations was analyzed, and the adsorption isotherms of the MMIMs and MNIMs were plotted (Fig. 8a). From Fig. 8a, when the equilibrium concentration increased, the equilibrium adsorption capacity (Qe) for λ-cyhalothrin first increased sharply, then increased slightly, and finally reached a maximum, as expected. It could also be seen that the adsorption capacities for λ-cyhalothrin were in the order MMIMs > MNIMs, and this was probably because the MMIMs demonstrated good specificity for the imprinted molecule. In order to obtain the maximum binding capacity of the MMIMs and MNIMs, Scatchard analysis was carried out.38 The Scatchard equation is expressed as following:
50 v/v) solution, and the functional adsorption behaviour of the MMIMs was investigated in detail. First, the adsorption capacity at different initial concentrations was analyzed, and the adsorption isotherms of the MMIMs and MNIMs were plotted (Fig. 8a). From Fig. 8a, when the equilibrium concentration increased, the equilibrium adsorption capacity (Qe) for λ-cyhalothrin first increased sharply, then increased slightly, and finally reached a maximum, as expected. It could also be seen that the adsorption capacities for λ-cyhalothrin were in the order MMIMs > MNIMs, and this was probably because the MMIMs demonstrated good specificity for the imprinted molecule. In order to obtain the maximum binding capacity of the MMIMs and MNIMs, Scatchard analysis was carried out.38 The Scatchard equation is expressed as following:
 | (1) |
 | ||
| Fig. 8 Equilibrium data (a), modeling (b) and ΔQ values (c) for the adsorption of λ-cyhalothrin onto MMIMs and MNIMs. | ||
Modeling of the adsorption of λ-cyhalothrin onto MMIMs and MNIMs by the Scatchard equation is shown in Fig. 8b. As shown, good fitting by the Scatchard equation is observed because the R2 values are 0.9832 and 0.9659 for the MMIMs and MNIMs, respectively. Moreover, the calculated maximum binding capacities of the MMIMs and MNIMs were 39.27 μmol g−1 and 25.74 μmol g−1, respectively. Al-Qodah's group studied the adsorption characteristics of λ-cyhalothrin in aqueous solutions using oil shale ash (OSA) as a sorbent.39 In their work, the maximum binding capacity was 13.0 μmol g−1, which was just one third of that in our work. This result indicates the advantage of our hollow microspheres.
To investigate the imprinting effect, the values of ΔQ were calculated using eqn (2), and the relationship between ΔQ and the initial concentration is shown in Fig. 8c.
| ΔQ = Qe,m − Qe,n | (2) |
In Fig. 8c, ΔQ significantly increased over the concentration range of 0–80 mg L−1, indicating that the empty imprinted sites were gradually occupied by λ-cyhalothrin upon increasing the testing concentration. Then ΔQ was approximately constant over the concentration range of 100–250 mg L−1, and then slightly increased in the concentration range of 300–350 mg L−1. This result implied that the specific binding sites reached saturation when the initial concentration of λ-cyhalothrin was 250 mg L−1, and then non-homogeneous binding sites with low affinity sites may be mainly responsible for the adsorption mechanism when the testing concentration exceeded 250 mg L−1.
As a means to study the effect of the contact time, the adsorption kinetics data and modeling by the pseudo-second-order equation are shown in Fig. 9a and Fig. 9b, respectively, and the half equilibrium times (tA and tB) are also marked in Fig. 9a. The pseudo-second-order equation is shown in eqn (3):40
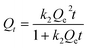 | (3) |
 | ||
| Fig. 9 Adsorption kinetic data (a) and modeling (b) for the adsorption of λ-cyhalothrin onto MMIMs and MNIMs. | ||
The values of the half equilibrium times, tA and tB, were 119.4 min and 108.1 min, and the related QA and QB were 21.62 μmol g−1 and 13.57 μmol g−1, respectively. The results showed that the MMIMs needed more time to reach the adsorption equilibrium than the MNIMs, while their adsorption rate was higher than that of MNIMs, especially in the first half of the adsorption time. This phenomenon further proved that the imprinting effect synchronously improved the properties of the adsorption kinetics and the equilibrium. R2 for the pseudo-second-order kinetic model was 0.9995 and 0.9968 for the MMIMs and MNIMs, respectively, indicating that a chemical process could be the rate-limiting step in the adsorption process.41
The specificity of the binding sites was investigated by studying the selectivity of the MMIM particles. Fenvalerate and diethyl phthalate had structures similar to λ-cyhalothrin and therefore were selected as template analogs (Fig. 10a). The uptake of λ-cyhalothrin, fenvalerate and diethyl phthalate by MMIMs and MNIMs is depicted in Fig. 10b. The differences in the re-binding capacities for the MMIMs or MNIMs can reflect the specific adsorption amounts of λ-cyhalothrin, fenvalerate and diethyl phthalate at the λ-cyhalothrin imprinted sites. ΔQ was also obtained using eqn (2). As evident in Fig. 9, the adsorption capacity of the MMIMs for λ-cyhalothrin was higher than those for fenvalerate and diethyl phthalate, while the adsorption capacities of the MNIMs were low and almost equal for the template and template analog molecules. By calculation, the values of ΔQ were 8.752 μmol g−1, 2.905 μmol g−1 and 2.115 μmol g−1 for λ-cyhalothrin, fenvalerate and diethyl phthalate, respectively. The results suggested that MMIMs were specific to λ-cyhalothrin but non-specific to fenvalerate and diethyl phthalate. Moreover, the external surface of the MMIMs was hydrophilic, resulting from the existence of hydrophilic HNTs, while the internal surface of the MMIMs was hydrophobic due to the use of hydrophobic monomers such as St and 4-VP. Owing to these properties, it was easy to pass the test solution through the semipermeable HNT shell on the external surface of the MMIMs, then λ-cyhalothrin molecules could be easily assembled at the imprinted sites.
 | ||
| Fig. 10 Chemical structures of the template and template analog molecules (a), uptake of λ-cyhalothrin, fenvalerate and diethyl phthalate by MMIMs and MNIMs (b). | ||
Conclusions
A facile, general and highly efficient approach to obtain magnetic molecularly imprinted microcapsules (MMIMs) with both hollow structures and magnetic properties was described, which involved the fabrication of a stable emulsion with tubular HNTs as stabilizers and their subsequent imprinting polymerization allowing the direct noncovalent imprinting of λ-cyhalothrin. Then the obtained MMIMs were successfully evaluated as the sorbent for the recognition of λ-cyhalothrin. The MMIMs possessed the following interesting features: (1) due to the irreversible adsorption of pure HNT particles with their high energy of attachment at the oil–water interface, as-prepared Pickering emulsion exhibited excellent stabilization compared with molecular surfactant or spherical colloid stabilized emulsions. (2) The resulting microcapsules possessed semipermeable shells arising from the non-uniform and tubular HNT interstices, and the structural robustness of the microcapsules can also be reinforced by the chemical crosslinking of the solid HNT surfaces. (3) MMIMs could not only selectively recognize the template molecules in a complex matrix, but also be easily collected and separated by an external magnetic field without additional centrifugation or filtration, which made the separation process easier and faster. (4) Pickering emulsion polymerization combined with a few Fe3O4 particles as magnetic carriers may be considered to be a simple and effective way to overcome the limitation of magnetite leakage. The results from this work could be valuable for future research towards the development of new separation and enrichment methods, especially their wide application for the selective recognition and detection of target molecules.Experimental
Chemicals
Halloysite nanotubes (HNTs) were purchased from Zhengzhou Jinyangguang Chinaware Co. Ltd, (Henan, China). Prior to use, the materials were milled and sieved followed by oven-drying at 373 K for 24 h. Iron(II) chloride tetrahydrate (FeCl2·4H2O), iron(III) chloride hexadecane (FeCl3·6H2O), sodium chloride (NaCl), styrene (St) and toluene, and tetramethylammonium hydroxide solution (v/v, 25%) were obtained from Sinopharm Chemical Reagent Co., Ltd (Shanghai, China). 4-vinylpyridine (4-VP), dimethyl 2,2′-azobis(2-methylpropionate) (AIBME) and diethyl phthalate were all purchased from Aladdin reagent Co., Ltd (Shanghai, China). λ-cyhalothrin and fenvalerate were supplied by Jiangsu Huangma Agrochemicals Co., Ltd.Preparation of hydrophobic Fe3O4 nanoparticles (HFNs)
The synthetic process for hydrophilic Fe3O4 particles followed a modified coprecipitation method,42 as follows: 1.35 g of FeCl3·6H2O and 0.6 g of FeCl2·4H2O were dispersed in 50 mL of H2O under mechanical stirring at 30 °C. After 15 min, 50 mL of sodium hydroxide solution (0.5 mol L−1) was added, and then the mixture was stirred vigorously and purged with nitrogen gas while the temperature was increased to 80 °C. After 30 min of stirring, black nanoparticles were collected by an Nd–Fe–B permanent magnet. After washing three times with doubly distilled water and ethanol, the resulting hydrophilic Fe3O4 nanoparticles were dried at 50 °C under vacuum conditions.Preparation of magnetic molecularly imprinted microcapsules (MMIMs)
First, λ-cyhalothrin (0.4499 g) and 4-VP (0.64 mL) were dispersed in toluene (0.14 mL). This solution was purged with nitrogen gas and sonicated for 5.0 min, and then stored in the dark for 3.0 h, allowing self-assembly of the λ-cyhalothrin and 4-VP. Second, NaCl (0.572 g), the prepared Fe3O4 nanoparticles (0.025 g) and HNT particles (0.30 g) were added to doubly distilled water (25 mL) under mechanical stirring. After 15 min, the obtained water phase was further sonicated using a Branson 450 W sonifier at 70% amplitude for 6.0 min, 30 s in 1.0 min intervals with a 30 seconds rest. Subsequently, an oil phase was formed by adding St (4.0 mL) and AIBME (0.12 mL) into the self-assembled solution. Then, the oil phase was mixed with the water phase, and a stable Pickering emulsion was generated via digital sonication for 6.0 min at 70% amplitude and with a 30 s pause for every minute of sonication. To prevent the increase of the dispersion temperature as a result of the ultrasonication, the Pickering emulsion was continuously cooled with an ice bath. After purging the oxygen with nitrogen gas for 15 min, the temperature of the resulting Pickering emulsion was increased to 65 °C. After 12 h, the obtained powers was washed with water and ethanol three times, and then the template molecules were removed by extensive washing with a mixture of methanol–acetic acid (95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, v/v), until no λ-cyhalothrin leakage was observed. Finally, the prepared MMIMs were dried at 50 °C under vacuum. For comparison, magnetic non-molecularly imprinted microcapsules (MNIMs) were also prepared as a control in parallel but without the addition of λ-cyhalothrin.
5, v/v), until no λ-cyhalothrin leakage was observed. Finally, the prepared MMIMs were dried at 50 °C under vacuum. For comparison, magnetic non-molecularly imprinted microcapsules (MNIMs) were also prepared as a control in parallel but without the addition of λ-cyhalothrin.
Materials characterization
Infrared spectra (4000–400 cm−1) were recorded on a Nicolet NEXUS-470 FTIR apparatus (U.S.A.). The morphology of the MMIMs was observed by scanning electron microscopy (SEM, S-4800). An image of the hydrophilic Fe3O4 particles was obtained by transmission electron microscopy (TEM, JEOL IEM-200CX). The optical micrographs of the Pickering emusion and MH-MIPs were collected by a DMM-330C optical microscope equipped with a high performance digital camera (CAIKON, China). Magnetic measurements were carried out using a VSM (7300, Lakeshore) under a magnetic field of up to 10 kOe. TGA and DSC of samples were performed for powder samples (about 10 mg) using a Diamond TG/DTA instrument (Perkin-Elmer, USA) under a nitrogen atmosphere up to 800 °C with a heating rate of 5.0 °C min−1. A TBS-990 atomic absorption spectrophotometer (Beijing Purkinge General Instrument Co. Ltd, Beijing, China) with a deuterium background correction and a GF990 graphite furnace atomizer system was used. Sonication was performed using a Branson digital 450 W sonifier.Batch mode binding experiments
To evaluate the binding affinity of the MMIMs in a static adsorption experiment, 10 mg of MMIM (or MNIM) particles were weighed and suspended in 10 mL of λ-cyhalothrin in ethanol–water (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 v/v) solution, and then the suspension was shaken in a thermostatically controlled water bath at 25 °C. After the desired time, MMIM (or MNIM) particles were isolated by an external magnetic field, and the solutions were immediately filtered through a millipore cellulose nitrate membrane (pore size was 0.45 mm) to remove suspended particles. The concentration of λ-cyhalothrin in the filtrate was determined by a UV-vis spectrophotometer at 278 nm (UV-2450, Shimadzu, Japan). The amount of captured λ-cyhalothrin was calculated by subtracting the amount of free λ-cyhalothrin from the initial amount added to the mixture.
50 v/v) solution, and then the suspension was shaken in a thermostatically controlled water bath at 25 °C. After the desired time, MMIM (or MNIM) particles were isolated by an external magnetic field, and the solutions were immediately filtered through a millipore cellulose nitrate membrane (pore size was 0.45 mm) to remove suspended particles. The concentration of λ-cyhalothrin in the filtrate was determined by a UV-vis spectrophotometer at 278 nm (UV-2450, Shimadzu, Japan). The amount of captured λ-cyhalothrin was calculated by subtracting the amount of free λ-cyhalothrin from the initial amount added to the mixture.
To measure the specificity of the MMIMs, we incubated 10 mg of the MMIMs (or MNIMs) with 100 mg L−1 of λ-cyhalothrin, fenvalerate and diethyl phthalate in 10 mL of ethanol–water (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 v/v) solution, and then proceeded to the binding experiments. After that, the concentrations of λ-cyhalothrin, fenvalerate and diethyl phthalate in the filtrate were determined by a UV-vis spectrophotometer at 278 nm, 277.5 nm and 275 nm, respectively. The selectivity of the imprinted polymers was determined by comparing the equilibrium adsorption ability to the template λ-cyhalothrin with that of the structural analogs.
50 v/v) solution, and then proceeded to the binding experiments. After that, the concentrations of λ-cyhalothrin, fenvalerate and diethyl phthalate in the filtrate were determined by a UV-vis spectrophotometer at 278 nm, 277.5 nm and 275 nm, respectively. The selectivity of the imprinted polymers was determined by comparing the equilibrium adsorption ability to the template λ-cyhalothrin with that of the structural analogs.
Magnetite leakage studies
Magnetite leakage studies followed the procedure in our previous work.43 Briefly, 50 mg of the MMIMs were placed in test tubes containing 10 mL of deionized water with different pH values (ranging from 2.0 to 8.0) and were shaken by a rotary shaker. After 24 h, the MNIMs were isolated by an external magnetic field, and the amount of the magnetite leached into media was determined by a graphite furnace atomic absorption spectrophotometer.Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (No. 21107037, No. 21176107, No. 21004031), Natural Science Foundation of Jiangsu Province (No. BK2011461, No. BK2011514, No. BK2011459), National Postdoctoral Science Foundation (No. 2013M530240), Postdoctoral Science Foundation funded Project of Jiangsu Province (No. 1202002B) and Programs of Senior Talent Foundation of Jiangsu University (No. 12JDG090).Notes and references
- X. B. Zhao, P. C. Du and P. Liu, Mol. Pharmacol., 2012, 9, 3330 CrossRef CAS PubMed.
- Y. b. Liu, Y. Q. Wang, S. M. Zhou, S. Y. Lou, L. Yuan, T. Gao, X. P. Wu, X. J. Shi and K. Wan, ACS Appl. Mater. Interfaces, 2012, 4, 4913 CAS.
- L. Wei, Y. L. Lei, H. B. Fu and J. N. Yao, ACS Appl. Mater. Interfaces, 2012, 4, 1594 CAS.
- L. You, T. Y. Wang and J. P. Ge, Chem. Eur. J., 2013, 19, 2142 CrossRef CAS PubMed.
- W. Q. Zhang, J. Z. Yang and X. M. Lu, ACS Nano, 2012, 6, 7397 CrossRef CAS PubMed.
- X. N. Li, R. K. Huang, Y. H. Hu, Y. J. Chen, W. J. Liu, R. S. Yuan and Z. H. Li, Inorg. Chem., 2012, 51, 6245 CrossRef CAS PubMed.
- K. L. Young, A. W. Scott, L. L. Hao, S. E. Mirkin, G. L. Liu and C. A. Mirkin, Nano Lett., 2012, 12, 3867 CrossRef CAS PubMed.
- Z. C. Yang, Y. Zhang, J. H. Kong, S. Y. Wong, X. Li and J. Wang, Chem. Mater., 2013, 25, 704 CrossRef CAS.
- J. V. Wijk, J. W. O. Salari, N. Zaquen, J. Meuldijk and B. Klumperman, J. Mater. Chem. B, 2013, 1, 2394 RSC.
- Q. Y. Liu, X. H. Guo, Y. Li and W. J. Shen, Langmuir, 2009, 25, 6425 CrossRef CAS PubMed.
- C. Z. Wu, Y. Xie, L. Y. Lei, S. Q. Hu and C. Z. OuYang, Adv. Mater., 2006, 18, 1727 CrossRef CAS.
- F. A. Lee and A. P. Steven, J. Mater. Chem., 2012, 22, 11235 RSC.
- S. Laib and A. F. Routh, J. Colloid Interface Sci., 2008, 317, 121 CrossRef CAS PubMed.
- Y. Lin, H. Skaff, A. Boeker, A. D. Dinsmore, T. Emrick and T. P. Russell, J. Am. Chem. Soc., 2003, 125, 12690 CrossRef CAS PubMed.
- M. A. Aroon, A. F. Ismail, T. Matsuura and M. M. Montazer-Rahmati, Sep. Purif. Technol., 2010, 75, 229 CrossRef CAS PubMed.
- S. Noee, N. Salimraftar, M. Abdouss and G. Riazi, Polym. Int., 2013, 62, 1711 CrossRef CAS.
- X. T. Shen, T. C. Zhou and L. Ye, Chem. Commun., 2012, 48, 8198 RSC.
- X. T. Shen, C. G. Xu and L. Ye, Soft Matter, 2012, 8, 7169 RSC.
- X. T. Shen and L. Ye, Macromolecules, 2011, 44, 5631 CrossRef CAS PubMed.
- P. Pieranski, Phys. Rev. Lett., 1980, 45, 569 CrossRef CAS.
- H. Lehle, E. Noruzifar and M. Oettel, Eur. Phys. J. E: Soft Matter Biol. Phys., 2008, 26, 151 CrossRef CAS PubMed.
- S. Cauvin, P. J. Colver and S. A. F. Bon, Macromolecules, 2005, 38, 7887 CrossRef CAS.
- I. Kalashnikova, H. Bizot, P. Bertoncini, B. Cathala and I. Capron, Soft Matter, 2013, 9, 952 RSC.
- J. M. Pan, L. Z. Li, H. Hang, R. R. Wu, X. H. Dai, W. D. Shi and Y. S. Yan, Langmuir, 2013, 29, 8170 CrossRef CAS PubMed.
- J. M. Pan, H. Hang, X. H. Dai, J. D. Dai, P. W. Huo and Y. S. Yan, J. Mater. Chem., 2012, 22, 17167 RSC.
- E. Abdullayev, A. Joshi, W. B. Wei, Y. F. Zhao and Y. R. Lvov, ACS Nano, 2012, 6, 7216 CrossRef CAS PubMed.
- W. Wu, S. L. Shen, C. L. Cheng, H. Mengc, K. Guo and J. F. Chen, Mater. Chem. Phys., 2009, 113, 696 CrossRef CAS PubMed.
- L. Zhang, P. Liu and T. M. Wang, Chem. Eng. J., 2011, 171, 711 CrossRef CAS PubMed.
- M. X. Liu, Z. X. Jia, F. Liu, D. M. Jia and B. C. Guo, J. Colloid Interface Sci., 2010, 350, 186–193 CrossRef CAS PubMed.
- D. J. Voorn, W. Ming and A. M. V. Herk, Macromolecules, 2006, 39, 2137 CrossRef CAS.
- L. M. Croll, H. D. H. Stover and A. P. Hitchcock, Macromolecules, 2005, 38, 2903 CrossRef CAS.
- P. Luo, Y. F. Zhao, B. Zhang, J. D. Liu, Y. Yang and J. F. Liu, Water Res., 2010, 44, 489 CrossRef PubMed.
- N. Sahiner and O. Ozay, Colloids Surf., A, 2011, 378, 50 CrossRef CAS PubMed.
- Y. Zhan, R. J. Liu, Y. L. Hu and G. K. Li, Anal. Chem., 2009, 81, 967 CrossRef PubMed.
- R. Gtmez-villacieros, L. Heman, J. Morales and J. L. Tirado, J. Colloid Interface Sci., 1984, 101, 392 CrossRef.
- X. W. Kan, Q. Zhao, D. L. Shao, Z. R. Geng, Z. L. Wang and J. J. Zhu, J. Phys. Chem. B, 2010, 114, 3999 CrossRef CAS PubMed.
- J. M. Pan, W. Hu, Xi. H. Dai, W. Guan, X. H. Zou, X. Wang, P. W. Huo and Y. S. Yan, J. Mater. Chem., 2011, 21, 15741 RSC.
- Z. J. Wu, H. Joo and K. Lee, Chem. Eng. J., 2005, 11, 227 CrossRef PubMed.
- Z. Al-Qodah, A. T. Shawaqfeh and W. K. Lafi, Desalination, 2007, 208, 294–305 CrossRef CAS PubMed.
- Y. S. Ho and G. McKay, Process Biochem., 1999, 34, 451 CrossRef CAS.
- G. Baydemir, M. Andac, N. Bereli, R. Say and A. Denizli, Ind. Eng. Chem. Res., 2007, 46, 2843 CrossRef CAS.
- L. Chen, X. P. Zhang, L. Sun, Y. Xu, Q. L. Zeng, H. Wang, H. Y. Xu, A. M. Yu, H. Q. Zhang and L. Ding, J. Agric. Food Chem., 2009, 57, 10073 CrossRef CAS PubMed.
- J. M. Pan, B. wang, J. D. Dai, X. H. Dai, H. Hang, H. X. Ou and Y. S. Yan, J. Mater. Chem., 2012, 22, 3360 RSC.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c3ra43178a |
| This journal is © The Royal Society of Chemistry 2014 |