Hyperbranched polyisobutylenes for self-healing polymers†
Abstract
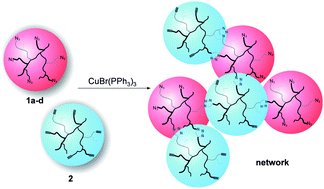
Hyperbranched azide- and alkyne-functionalized poly(isobutylene)s (PIBs, Mn = 25 200–35 400 g mol−1) suitable for crosslinking at room temperature via the copper-catalyzed alkyne–azide “click” cycloaddition reaction (CuAAC) are reported, aiming at the design of improved crosslinking reactions for novel self-healing materials. Based on the low glass transition temperature (Tg) the high molecular mobility of the hyperbranched PIBs was linked with an efficient crosslinking chemistry while introducing reactive endgroups into the polymer and tuning molecular architecture via living carbocationic polymerization (LCCP). Hyperbranched PIBs were prepared by inimer-type LCCP followed by direct end quenching of living chain ends with either 3-(bromopropoxy)benzene (BPB) or trimethyl(3-phenoxy-1-propynyl)silane (TMPPS). Optimized quenching time conditions (6 hours quenching with BPB and 20 hours quenching with TMPPS) yielded the fully bromine- and trimethylsilyl-protected alkyne-functionalized hyperbranched PIBs with up to ∼9 endgroups, subsequently being converted into the respective fully azide- and alkyne-functionalized polymers. “Click” reactions between the azide and the alkyne group of hyperbranched PIBs linked the two spherical polymers resulting in fast gelation times of 30 to 50 minutes as investigated via melt rheology. Materials with high network densities were obtained due to the high amount of reactive endgroups and the architecture of the applied polymers. Thus, the concept of crosslinking hyperbranched azide- and alkyne-functionalized PIBs via “click” chemistry at room temperature towards self-healing polymers could be successfully proven.

 Please wait while we load your content...
Please wait while we load your content...