Catalytic functionalization of tertiary alcohols to fully substituted carbon centres
Abstract
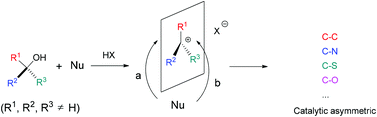
The catalytic nucleophilic substitution of tertiary alcohols using carbon or heteroatom based nucleophiles is a versatile methodology for the efficient, diverse and atom economical construction of fully substituted carbon centres, including both quaternary carbons and heteroatom substituted tetrasubstituted carbons, which only produces water as the by-product. This review summarizes the recent progress in this field, including the catalytic asymmetric studies and their application in the natural product synthesis, briefly discusses the reaction mechanism and challenges, and outlines synthetic opportunities that are still open.

 Please wait while we load your content...
Please wait while we load your content...