Abstract
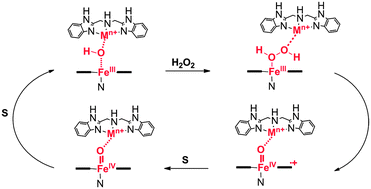
Inspired by catalytic sites of cytochrome c oxidase (CcO) and nitric oxide reductase (NOR), a new series of dinuclear heme–non-heme complexes is described. The complexes are derived from the association between an iron(III)–protoporphyrin IX containing a covalently attached Gly-L-His-OMe residue to one propionic acid substituent (HMGH) and a metal complex with a tridentate amino-bis(benzimidazole) (BBH) ligand, mimicking the tris-histidine coordination of FeB and CuB in NOR and CcO, respectively. Besides the coordination of FeIII and CuII with the BBH ligand, we also explored the role of “non-biomimetic” metals, such as CoII, MnII, or ZnII, in order to establish the priority among the ancillary metal ions in cooperating with the ferric heme and promoting its catalytic activity in oxidation reactions. pH-spectrophotometric titrations show that the presence of the non-heme metal decreases the pKa of water-bound to hemin (pKa = 8.4 ± 0.1), with a larger effect with iron(III), copper(II) and zinc(II) complexes (pKa of 6.4 ± 0.1, 6.0 ± 0.1 and 6.5 ± 0.1, respectively), which suggests that an interaction with the non-heme metal center takes place also at a micromolar range. NMR spectra indicate that the interaction between hemin and the non-heme center is not strong enough to convert the high spin configuration of FeIII–heme to low spin as observed for CcO and NOR enzymes. The dinuclear complex enhances the peroxidase-like activity of heme in kinetic studies performed at pH 5.5, 7.0 (using 3-(4-hydroxyphenyl)-propanoic acid as the substrate) and 9.0 (using o-phenylenediamine). In particular, the stronger effects are observed with FeIII, CuII, and CoII complexes, which increase the turnover rates of hemin throughout the pH range analyzed. At neutral and basic pH the KM value decreases up to one fourth indicating a positive cooperation between HMGH and [M(BBH)]n+ in binding the substrates. Moreover, the presence of the non-heme center facilitates the binding of H2O2 and formation of high valent ˙PFeIV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O species. These data show that interaction between the two metal centers occurs with heme in several oxidation states.
O species. These data show that interaction between the two metal centers occurs with heme in several oxidation states.
- This article is part of the themed collection: In honour of Bernard Meunier

 Please wait while we load your content...
Please wait while we load your content...